Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Journal of the South African Veterinary Association
On-line version ISSN 2224-9435
Print version ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.82 n.4 Pretoria Dec. 2011
ARTICLE ARTIKEL
Pharmacokinetics of ceftazidime administered to lactating and non-lactating goats
R RuleI,*; S VillagraI; P BarrenaII; R LacchiniII; F J ReynaldiIII
IIDIP - Institute of Paediatric Research and Development 'Dr. Fernando Viteri' (La Plata Children's Hospital - Scientific Research Commission of the Province of Buenos Aires, CICPBA), Calle 63 Nro. 216 (1900), La Plata. Argentina
IIDepartment of Animal Production, Faculty of Agricultural and Forestry Science, University of La Plata, Argentina
IIICCT CONICET La Plata
ABSTRACT
The aim of this work was to determine the pharmacokinetics of intravenous (iv) and intramuscular (im) ceftazidime administered to lactating (LTG; n = 6) and non-lactating (NLTG; n = 6) healthy Creole goats in 2 trials (T1 and T2). During T1 and T2, goats randomly received a single dose of im or iv ceftazidime (10 mg/kg). Serum concentration of iv ceftazidime in NLTG and LTG goats is best described by 2 and 3 compartment models, respectively. The pharmacokinetic parameters of iv and im ceftazidime administered to LTG and NLTG showed statistically significant differences (P < 0.05) in the constants (λz,T1 vs T2 [iv] 0.5 ± 0.1 vs 0.3 ± 0.1 /h; T1 vs T2 [im] 0.5 ± 0.2 vs 0.3 ± 0.1 /h) and in the mean times (t1/2,T1 vs T 2 [iv] 1.6 ± 0.3 vs 2.3 ±0.6 h;T 1 vs T 2 [im] 1.6 ± 0.7 vs 2.6 ± 0.9 h) of elimination. The bioavailability of ceftazidime in LTG and NLTG was 113.0 ± 17.8 and 96.0 ± 18.0 %, respectively. Ceftazidime concentration in milk at 2 h was: iv = 1.9 ± 0.2 and im = 2.4 ± 0.5 µg/m; the penetration in milk was iv = 18.3 ± 13.5 and im = 14.3 ± 10.6 %. Ninety-six hours after iv and im administration, residues of the drug were not found in milk. In conclusion, ceftazidime, when administered to goats, showed high concentration times in serum, good penetration into milk and a bioavailability that makes it suitable to be used by the im route.
Keywords: ceftazidime, goat, milk, pharmacokinetics.
INTRODUCTION
Ceftazidime (6R, 7R)-7-[(Z)-2-(2-aminothiazol-4-yl)-2-(2-carboxyprop-2-yloxyimino)acetamido]-3-(pyridinium-1-ylmethyl)ceph-3-em-4-carboxylate is a 3rdgeneration cephalosporin developed in 198012. Like the other β-lactam antibiotics, this cephalosporin inhibits peptidoglycan synthesis and produces bacterial lysis12.
Ceftazidime is a cephalosporin active against Escherichia coli, Citrobacter diversus, C. freundii, Enterobacter aerogenes, E. agglomerans, Klebsiella spp. including K. pneumoniae, Proteus spp., Serratia marcescens, Salmonella spp., Shigella spp. and Pseudomonas aeruginosa6. In general, the minimal inhibitory concentration (MIC) value for the mentioned microorganisms is < 4 µg/m 12,22. The MIC90 values of ceftazidime determined for E. coli, Salmonella spp., Pasteurella multocida and P. haemolytica isolates ranged from less than 0.01 to 0.1 µg/ml 21.
Studies carried out using β-lactams in animals support the concept that the time during which the free drug concentration exceeds the MIC (T > MIC) must be 40-60 % of the inter-dose interval in order to assure the success of the therapeutic efficacy of the cephalosporins23,24.T> MIC is mainly determined by the terminal half-life, which is itself a hybrid process involving plasma clearance and drug distribution. In brief, it is suggested that maintaining the concentration of β-lactam antibiotics above the MICs for infecting organisms is beneficial for the treatment of Gram-negative and -positive bacterial infections.
Ceftazidime was used to treat dairy cows with clinical signs of inflammation in 1 mammary gland quarter and presence of 1 or more pathogenic microorganisms in that quarter (3 intramammary doses of 200 mg/quarter/12 h)17. Thirty dairy cows in full milk production were studied; 10 cows were healthy and microbiologically negative and the other 20 cows had mastitis. The 10 healthy cows were administered cefotaxime (5) and ceftazidime (5) via the intra-mammary route (3 doses of 200 mg each at 12 h intervals). Similarly, in the group of mastitic animals, 10 cows were administered ceftazidime and the other 10 received cefotaxime. The values for ceftazidime concentrations in milk over time at 1 h and 12 h post-administration of the 1st dose of antibiotics in healthy and mastitic cows were high (1028.2 ± 723.1 and 30.5 ± 25.7 and 966.8 ± 278.0 and 131.3 ± 34.9 µg/ml, respectively). The authors observed that ceftazidime administered by intramammary infusion to healthy and mastitic cows was distributed from treated to untreated quarters and the t1/2 and rate constant in milk were similar17. The drug had good distribution from treated to untreated quarters and the presence of residues was observed, so that milk destined for human consumption had to be discarded for at least 72 h17. It has been observed that low protein binding resulted in ceftazidime penetration into the mammary gland. Binding sites vary according to the animal species, ranging from 20 to 30 % in mice and rats12, 10 % in humans3 and 17 % in rabbits1. On the other hand, serum bactericidal activity of the drug was found to be more closely related to unbound rather than total antibiotic concentrations and that only the unbound drug was microbiologically active9. Subcutaneous or intramuscular (im) administration of ceftazidime to dogs (25 mg/kg body weight) was effective against P. aeruginosa11. Doses of 50 mg/kg body weight/12 h have been recommended for the treatment of bacterial infections in rabbits.
There is practically no information on the pharmacokinetics of ceftazidime administered parenterally to goats.
The aim of this study was to determine the pharmacokinetic profile of ceftazidime administered intravenously (iv) and im to lactating and non-lactating goats.
MATERIALS AND METHODS
Animals
Lactating (LTG, n = 6) and non-lactating (NLTG, n = 6) Creole goats with a body weight ranging from 35 to 45 kg were used. A crossover design (2 × 2) was used in 2 trials (T1 and T2).
The animals were maintained and handled in accordance with the NIH guide for the care and use of of laboratory animals4. The research protocol was approved by the Commission of Scientific Research of Buenos Aires Province, Argentina (CIC-PBA).
Trial 1
Clinically healthy animals in their 1st to 3rd lactations, weighing 30-45 kg (range), in milk production (range 500 to 750 ml/ day) and milked every 12 h, received a single dose of 10 mg/kg of ceftazidime randomly by the im or iv route at the start of their 3rd week in lactation. Blood samples were collected from the jugular vein into tubes without anti-coagulant at 0.08, 0.17, 0.25, 0.5, 1.0, 2.0, 3.0, 4.0, 5.0, 6.0, 8.0 and 12 h post-administration of the antibiotic. Milk samples were collected (approximately 0.5 ml each) starting at 0.25 h and following the blood sampling strategy. Milk samples were collected at 24, 36, 48, 60, 72, 84, 96, 108 and 120 h post-administration of ceftazidime to determine the presence of residues of the drug.
Two weeks later, the same animals received ceftazidime in equal doses and by the same routes, but the latter were inverted, i.e. animals that had previously received the antibiotic iv received it im and vice versa.
Blood and milk sampling was carried out in the same way and at the same intervals after the initial administration of the drug.
Trial 2
Clinically healthy NLTG (n = 6) received ceftazidime randomly administered either by iv or im routes, in equal doses and manner as in T1. The blood sampling was carried out in the same way and at the same intervals as in T1.
Processing and preservation of samples
Blood samples were allowed to clot and then centrifuged at 3000 g for 15 min in order to separate the serum. All the samples (serum and milk) were stored in individual, sterile containers at -18 ºC until further analysis.
Quantification of the antibiotic
The concentrations of ceftazidime in serum and milk were determined by means of the microbiological assay technique, using Geobacillus stearothermophilus ATCC 12.980 as the test organism8. Each sample was assayed for ceftazidime by an agar diffusion bioassay with Mueller Hinton Agar (Lab. Britania, Argentina) seeded with the Geobacillus stearothermophilus and allowed to solidify on 23 × 28 cm glass plates. Duplicate 25-µl standards were then placed in 6 mm wells cut into the seeded agar. The ceftazidime standards were prepared in goat sera and milk at 20, 10, 1, 0.5, 0.4, 0.3, 0.2, 0.1, 0.15, 0.125, 0.1, 0.075, 0.05 µg/ml concentrations. After incubation of the assay plates for 6 to8hat 64 ºC, the zone of inhibition around each well was measured and standard curves were prepared.
Sampling before administration of the antibiotic showed no bacterial inhibition. The correlation coefficient for the standard curves prepared for all the experiments was greater than 0.98. Intra-and inter-assay coefficients of variation were lower than 8 %. Sensitivity and quantification limits of the assay for serum and milk were 0.2 and 0.125 and 0.4 and 0.3 µg/ml (range, 0.15-0.2 and 0.1-0.125) respectively.
The determination of detectable residues of the antibiotic in milk was performed by means of a biological test (Delvotest SP M Gist-Brocades, Food Ingredients). The sensitivity of Delvotest SP for ceftazidime ranges from 0.03 to 0.015 µg/ml. The Delvotest SP is a qualitative test used by producers, veterinarians and the dairy industry to determine the presence of antibiotic residues in milk.
Pharmacokinetic analysis and statistics
Pharmacokinetic analyses were carried out with WinNonlin (V.4.1 Pharsight Corp., Cary, NC) using standard compartmental methods. Akaike's information criterion (AIC) was used to determine which compartmental model best adapted to the data set26. The pharmacokinetic parameters calculated included hybrid constants (C1,C2, Cz, λ1, λ2 and λz) and were used to calculate the rate constants from the central to the peripheral compartment (k12) and vice versa (k21), the rate constants from the central to a deep compartment (k13) and vice versa (k31) and the rate constants of elimination (k10)5. The maximum milk concentration (Cmax) and the time to achieve Cmax (tmax) were directly observed in the data obtained for each individual animal.
The area under the curve (AUC) was calculated by the trapezoidal method2.
The absorption (t1/2a), rapid distribution (t1/2λ1), slower distribution (t1/2λ2) and elimination (t1/2) half-lives, the apparent volume of the central compartment (Vc), the volume of distribution at steady state (Vss) and the body clearance (CL) were calculated by standard procedures5.
The percentage of bioavailability (F) and penetration or milk passage (P) of ceftazidime were calculated by using the following equation14:
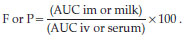
The incidence of the physiological state of the animals (LTG vs NLTG) above pharmacokinetics parameters was analysed by means of a 2-way analysis of variance test (Statgraphic Plus 7.0). A P value less than 0.05 was considered significant.
The time above the minimum inhibitory concentration (T > MIC) was determined by means of the following formula:
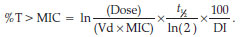
ln is the natural logarithm, Vd the volume of distribution, t1/2 the elimination half-life (h) and DI the dosing interval (h)24.
RESULTS
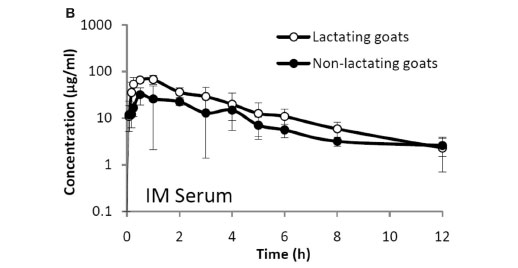
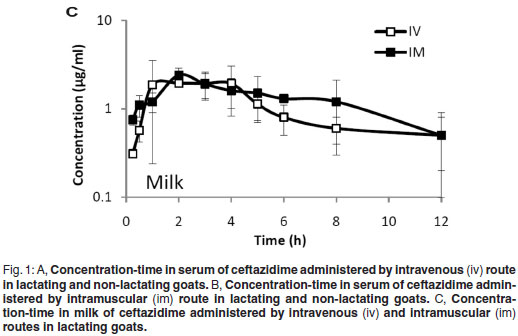
The mean concentrations (±SD) in serum and milk (LTG) and in serum (NLTG) of ceftazidime administered iv and im are shown in Fig. 1A, B, C.
The means of the pharmacokinetic parameters (±SD) are given in Table 1. The pharmacokinetic parameters in serum show statistically significant differences (P < 0.05) between the constants of elimination (T1(iv) 0.5 ± 0.1 vs T2 (iv) 0.3 ± 0.1, T1 (im) 0.5 ± 0.2 vs T2 (im) 0.3 ± 0.1/h) and the mean times of elimination (T1 (iv) 1.3 ± 0.8 vs T2 (iv) 2.3 ± 0.6 and T1 (im) 1.6 ± 0.7 vs T2 (im) 2.6 ± 0.9 h.
DISCUSSION
High concentrations in serum and milk of ceftazidime (10 mg/kg body weight) administered iv and im to LTG and NLTG were found 12 h after the administration of the antibiotic. These concentrations exceeded the MIC for sensitive organisms13,21. The high concentrations of ceftazidime in milk agree with those obtained in lactating cows16. The pharmacokinetic profile in NLTG was described by means of an open 2-compartment model, as previously described in humans7,19, calves21, sheep15, non-lactating cows16 and rabbits1. In LTG as well as in lactating cows an open 3-compartment model was used16. Following the AIC, it was possible to determine that the tricompartmental model is the most appropriate to adjust the data of the concentration time in serum of ceftazidime administered by the iv route to LTG. It differs in the slopes (phases) required to obtain an adequate description of the curve in results coming from NLTG, where the model of choice was bicompartmental. This difference within the same animal species could be the result of the different state of production of the mammary glands of the animals included in this study.
A single-compartment model was used in order to describe the pharmacokinetic behaviour of ceftazidime administered im.
In the description of the concentration time of ceftazidime administered via the iv route to NLTG and LTG, provided by using models of 2 and 3 compartments, a rapid distribution phase was observed (t1/2λ1 = 0.2 ± 0.3 and 0.1 ± 0.03 h, respectively) followed by a slower distribution phase (t1/2λ2 = 1.6 ± 0.6 h) in the tricompartmental model used in LTG. A much slower phase of distribution to deeper tissues was observed in lactating cows (28.1 ± 19.2 h) than the phase found in goats in this study16. This could be attributed to the distinctive physiological and productive conditions of the animal species in this study. In studies in rabbits1, sheep15 and humans20 the mean times of rapid distribution were higher (0.258 ± 0.054, 0.22 ± 0.09 and 0.62 ± 0.01 h, respectively) than those observed in this study.
The half-life in serum of ceftazidime administered iv to NLTG (2.3 ± 0.6) was higher than those found in cows (1.4 ± 0.3 h)16, dogs (0.82 h) and rats (0.23 h)10, humans (1.8 ± 0.2 h)25, and sheep (1.6 ± 0.2 h)15, and similar to those obtained in rabbits (2.22 ± 0.351 h)1 and calves (2.31 h)21. The half-life in LTG (1.6 ± 0.3 h) was higher than that obtained in lactating cows (1.1 ± 0.2 h)16. The faster elimination rate constant after iv administration of ceftazidime to LTG (0.5 ± 0.1/h) compared with that obtained in NLTG (0.3 ± 0.1/h) with higher mean elimination times, agrees with a higher clearance in LTG (103.4 ± 35.8 ml/kg) vs NLTG (68.1 ± 35.8 m/kg) (not statistically demonstrated).
The volume of distribution at steady state was not much different in quantity between LTG (200.5 ± 79.4) and NLTG (238.1 ± 60.3 ml/kg) and also compared with the one obtained in dogs (218 ± 7ml/kg)10, although it was lower than those observed in cows (lactating = 489.8 ± 136.9 and non-lactating = 390.2 ± 212.9 ml/kg)16, non-lactating sheep (356.1 ± 208.0 ml/kg)15 and non-lactating calves (294.0 ml/kg)21. These volumes of distribution at steady state were among the values observed for drugs with little distribution into the extravascular tissues6. When comparing LTG with NLTG, the volume of distribution is not different because the drug passes to the mammary compartment, i.e. a a deep compartment. However, not all the drug that arrives in the mammary glands is eliminated through this route; it can start circulating again, giving rise to a clearance in lactating animals that is not statistically different from the non-lactating animals' clearance results.
Although the AUC for the iv route appears different in LTG (103.4 ± 39.0 µg/ml ) and NLTG (124.2 ± 34.9 µg/ml ) , this was not supported statistically, and could be due to the passage of the antibiotic into milk.
When ceftazidime was administered im to LTG and NLTG it was well absorbed and showed a high bioavailability (113.0 ± 17.8 and 96.0 ± 18.0 %), fast constants of absorption (4.6 ± 3.4 and 3.4 ± 0.7/h) from the site of administration and high Cmax (67.5 ± 14.0 and 31.9 ± 12.9 µg/ml ). The mean times of elimination of ceftazidime administered im to LTG (1.6 ± 0.7 h) and NLTG (2.6 ± 0.9 h) were similar to those obtained for the iv route in the same animals. Similarities were found in the mean times of elimination of ceftazidime administered iv (2.3 ± 0.39 h) and im (2.1 ± 0.17 h) to calves21, suggesting that the absorption does not interfere with the terminal half-life.
Ceftazidime is a weak organic acid and its liposoluble form (non-ionised) that allows it to pass from plasma to milk and vice versa depends on the pKa (dissociation constant of the drug), the pH of the environment and the drug's ability to bind to serum or plasma proteins. Although ceftazidime is eliminated mainly by the kidney and to a much lesser extent by extrarenal mechanisms20, marked penetration of the antibiotic into milk was observed (penetration (iv) 18.3 ± 13.5 and (im) 14.3 ± 10.6 %). Those values were lower than those observed in bovines (penetration (iv) 47.7 ± 38.2 and (im) 51.1 ± 39.0 %)19. This lower penetration can be attributed to the low milk production of goats during Trial 1. In another study, when cephalothin was administered to goats, a correlation was found between the areas under the curve, the maximum concentrations and the time to reach them in milk, and the volume of milk produced by the animals18.
When ceftazidime was administered via the iv and im route (10 mg/kg body weight) to LTG and NLTG, the interdose intervals necessary to obtain a T > MIC value between 40 and 60 % for microorganisms with MIC = 4 µg/ml were 8 to 12 hours. However, in order to confirm the doses and intervals, it is necessary to have specific information about clinical treatments in goats.
Ninety-six hours after administration of ceftazidime iv and im, residues of the drug determined by a standard method used by the dairy industry were not found in milk.
CONCLUSION
In conclusion, the high concentration time values and the pharmacokinetic profile of ceftazidime observed in serum and milk makes it a promising agent for the empirical treatment of infections in lactating and non-lactating goats.
ACKNOWLEDGEMENTS
This study was supported by a grant provided by the Commission of Scientific Research of the Province of Buenos Aires, Argentina. RR is member of scientific board of the Scientific Research Commission of the Province of Buenos Aires, CICPBA, and FJR is member of scientific board of CONICET.
REFERENCES
1. Abd El-Aty A M, Goudah A, Abo El-Sooud K 2001 Pharmacokinetics, intramuscular bioavailability and tissue residue profile of ceftazidime in rabbit model. Deutsche Tierärztliche Wochenschrift 108:168-171 [ Links ]
2. Baggot J D 1977 Principles of drug disposition in domestic animals: the basis of veterinary clinical pharmacology. W B Saunders, Philadelphia [ Links ]
3. Blanco J D, Jorgensen J H, Castaneda Y S, Crawford S A 1983 Ceftazidime levels in human breast milk. Antimicrobial Agents and Chemotherapy 23:79-480 [ Links ]
4. Commission on Life Sciences, National Research Council 1996 Guide for the care and use of laboratory animals. The National Academies Press, Washington, D.C. [ Links ]
5. Gibaldi M, Perrier D 1982 Pharmacokinetics. In Dekker M (ed.), Multicompartment models (2nd edn). Dekker, New York: 47-109 [ Links ]
6. Goodman M and GilmanAC2003 Las Bases Farmacologicas de la Terapeutica (9th edn). McGraw-Hill Interamericana. D.F. Mexico: 1207-1236. [ Links ]
7. Harding S M, Munro, A J, Thornton J E, Ayrton, J, Hogg M I 1981 The comparative pharmacokinetics of ceftazidime and cefotaxime in healthy volunteers. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 263-272 [ Links ]
8. Herbst D 1982 Identification and determination of four β-lactam antibiotics in milk. Journal of Food Protection 45:450-451 [ Links ]
9. Lam Y W F, Duroux M H, Gambertoglio J G, Barriere S L, Guglielmo B J 1988 Effect of protein binding on serum bactericidal activities of ceftazidime and cefoperazone in healthy volunteers. Antimicrobial Agents and Chemotherapy 32:298-302 [ Links ]
10. Matsui H, Komiya M, Ikeda C H, Tachibana A 1984 Comparative pharmacokinetics of YM-13115, ceftriaxone, and ceftazidime in rats, dogs, and rhesus monkeys. Antimicrobial Agents and Chemotherapy 26:204-207 [ Links ]
11. Monfrinotti A, Ambros L, Prados A P, Kreil V, Rebuelto M 2010 Pharmacokinetics of ceftazidime after intravenous, intramuscular and subcutaneous administration to dogs. Journal of Veterinary Pharmacology and Therapeutics 33:204-207 [ Links ]
12. O'Callaghan C H, Acred P, Harper P B, Ryan D M, Kirby S M, Harding S M 1980 GR 20263, a new broad-spectrum cephalosporin with anti-pseudomonal activity. Antimicrobial Agents and Chemotherapy 17:876-883 [ Links ]
13. Oonaka K, Furuhata K, Hara M, Fukuyama M 2010 Powder infant formula milk contaminated with Enterobacter sakazakii. Japanese Journal of Infectious Diseases 63:103-107 [ Links ]
14. Ritschel W A 1986 Handbook of basic pharmacokinetics. In RitschelWA(ed.) Pharmacokinetics of single dose administration (2nd edn). Drug Intelligence Publications, Hamilton: 282-301 [ Links ]
15. Rule R, Rubio M, Perelli M C 1991 Pharmacokinetics of ceftazidime in sheep and its penetration into tissue and peritoneal fluids. Research in Veterinary Science 51:233-238 [ Links ]
16. Rule R, Quiroga G H, Rubio M, Buschiazzo H O, Buschiazzo P M 1996 The pharmacokinetics of ceftazidime in lactating and non-lactating cows. Veterinary Research Communications 20:543-550 [ Links ]
17. Rule R, Quiroga G, Buschiazzo H, Lacchini R, Buschiazzo P 1998 Rate of decline of cefotaxime and ceftazidime in milk following intramammary administration to healthy and mastitic dairy cows. Veterinary Record 43:310-311 [ Links ]
18 Rule R, Cordiviola C, Vita M, Lacchini R 2004 Correlations between milk production and kinetic variables in milk cephalothin administered to lactating goats. Veterinarni Medicina 49:370-372 [ Links ]
19. Ryan D M, Mason U, Harding S M 1981 The penetration of ceftazidime into extra-vascular fluid. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 283-288 [ Links ]
20. Saito A 1983 Studies on absorption, distribution, metabolism and excretion of ceftazidime in Japan. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 225-262 [ Links ]
21. Soback S, Ziv G 1989 Pharmacokinetics of ceftazidime given alone and combination with probenecid to unweaned calves. American Journal of Veterinary Research 50:1566-1569 [ Links ]
22. Thornsberry C 1985 Review of in vitro activity of third-generation cephalosporins and other newer beta-lactam antibiotics against clinically important bacteria. American Journal of Medicine 79(2A): 14-20 [ Links ]
23. Toutain P L 2002 Pharmacokinetic/pharmacodynamic integration in drug development and dosage-regimen optimization for veterinary medicine. AAPSPharmSciTech 4:160-188 [ Links ]
24. Turnidge J D 1998 The pharmacodynamics of beta-lactams. Clinical Infectious Diseases 27:10-22 [ Links ]
25. Wise E R, Armstrong G C, Brown R M, Andrews J M 1981 The pharmacokinetics and tissue penetration of ceftazidime and cefamandole in healthy volunteers. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 277-282 [ Links ]
26. Yamaoka K, Nakagawa T, Uno T 1978 Application of Akaike's information criterion (AIC) in the evaluation of linear pharmacokinetic equations. Journal of Pharmacokinetics and Biopharmaceutics 6:165-175 [ Links ]
Received:September 2010
Accepted:26 October 2011
* Author for correspondence: Calle 63 Nro. 216 (1900), La Plata. Argentina. E-mail: robertorule@yahoo.com.ar