Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Journal of the South African Veterinary Association
On-line version ISSN 2224-9435
Print version ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.82 n.4 Pretoria Dec. 2011
ARTICLE ARTIKEL
A study of some infectious causes of reproductive disorders in cattle owned by resource-poor farmers in Gauteng Province, South Africa
S M NjiroI,*; A G KidanemariamI; A M TsotetsiI; T C KatsandeII; M MnisiI; B A LubisiI; A D PottsI; F BaloyiII; G MoyoII; J MpofuII; A KalakeII; R WilliamsI
IOnderstepoort Veterinary Institute, PO Box X04, Onderstepoort, 0110 South Africa
IIGauteng Department of Agriculture and Rural Development
ABSTRACT
Two hundred and thirty-nine cattle from Gauteng Province in South Africa were tested for various pathogens causing reproductive diseases including bovine viral diarrhoea/mucosal disease (BVD/MD) virus, infectious bovine rhinotracheitis/infectious pustular vulvovaginitis (IBR/IPV) virus, Neospora caninum and Brucella abortus using various tests. For BVD/MD virus, 49.37 % tested positive, 74.47 % for IBR/IPV virus, 8.96 % for Neospora caninum and 3.8 % for Brucella abortus. The result for Brucella abortus is higher than the national average, possibly due to the small sample size. A high seroprevalence of antibodies to both BVD/MD virus and IBR/IPV virus was evident. These 2 viruses should be considered, in addition to Brucella abortus, when trying to establish causes of abortion in cattle. The clinical significance of Neospora caninum as a cause of abortion in Gauteng needs further investigation. One hundred and forty-three bulls were tested for Campylobacter fetus and Trichomonas fetus, and a low prevalence of 1.4 % and 2.1 % respectively was found in this study. The clinical implications of these findings are discussed.
Keywords: abortion, Brucella abortus, BVD/MD virus, Campylobacter fetus, cattle, IBR/IPV virus, Neospora caninum, resource-poor farmers, Trichomonas fetus.
INTRODUCTION
Infertility and abortions cause major losses in animal production. While brucellosis is a well-known infectious cause of abortions, other less well known pathogens may also cause abortions or infertility. An abortion represents a loss of expected additional milk and meat, wastes breeding time and results in additional costs due to the special diet and care required for pregnant animals. Animals culled to control abortion-causing infections need to be replaced. Abortions can therefore cost the producer and the State a great deal of money. For instance in Argentina, such losses have been estimated to cost approximately US$165 million every year3. The early and accurate detection of the specific pathogen involved in infections that result in abortion and infertility is essential so that efficacious interventions can be implemented. Infections with Campylobacter fetus var. venerealis and Trichomonas fetus occur in both the commercial and small-scale emerging farming sectors in South Africa. These pathogens cause endometritis, pyometra, abortion, infertility and considerable economic losses, particularly in endemic regions15. Trichomonosis causes infertility, delayed return to oestrus after mating, early embryonic death and sometimes abortion. It may directly cause death of the embryo or may do so via endometritis and marked leucocytic diapedesis into the endometrium25. Consequently, early detection and screening of the herd for these infections is a prerequisite for successful and economically viable cattle farming.
This study sought to establish the prevalence of a number of abortion-and infertility-causing pathogens in cattle belonging to emerging, resource-poor farmers in Gauteng Province. Among the pathogens investigated through serological tests were Brucella abortus, bovine viral diarrhoea/mucosal disease (BVD/MD) virus, infectious bovine rhinotracheitis/ infectious pustular vulvovaginitis (IBR/ IPV) virus and Neospora caninum. Neospora caninum is a protozoan parasite of animals and a major cause of abortions in cattle7. Brucella abortus is among the earliest known causes of abortions in cattle, having been discovered in 1897. As a zoonosis, the pathogen has attracted much attention in terms of control and where vaccination is properly implemented, prevalence has dropped dramatically, to the extent that in some circles brucellosis is said to be of historical significance only12. The BVD/MD virus infects the developing foetus and is capable of causing abortions in pregnant animals. It is 1 of the major causes of abortions in cattle and has a worldwide distribution15. The IBR/IPV virus occurs in cattle herds throughout the world and can cause separate respiratory and genital syndromes, keratoconjunctivitis, meningoencephalitis in calves and abortions in cows. Seroprevalence varies from herd to herd. In Turkey it was found to be between 20 % and 74 %1. In this study the infectious causes of reproductive disease in the study herds are reported and conclusions and recommendations to control and prevent these diseases are provided.
MATERIALS AND METHODS
Sample size
Cattle herds belonging to emerging farmers (those with non-commercial livestock operations as identified by animal health technicians) were sampled as described below. Sample size was determined using the epidemiological formula described by Thrusfield24.
Blood sampling for serology
Blood samples were taken from cattle at 21 localities throughout Gauteng Province (Fig. 1). Samples were taken in vacutainer tubes without anticoagulant from 11 to 25 animals per herd. A total of 239 samples were taken (Table 1). Blood was taken directly from the coccygeal vein and left to settle and clot in a cooler box with ice packs. Serum was decanted into fresh, labelled containers in the laboratory. The serum samples were frozen at -20 ºC until needed for testing.
Sheath washes
Sheath wash samples were collected from bulls from various areas of Gauteng (Fig. 1). One hundred and forty-three bulls from 19 cattle farming areas in Pretoria, Germiston and Randfontein were sampled by preputial washings as per standard protocol and tested for Trichomonas fetus and Campylobacter fetus using culture and PCR techniques. Samples were collected from bulls ranging in age from 2 to 11 years 2 or 3 times from each bull at weekly intervals. A total of 333 sheath wash samples were collected between 30 November 2009 and 10 June 2010. Briefly, smooth rubber pipettes were introduced into the full length of the preputial cavity and the preputial orifice held firmlly closed with 1 hand around the pipette to prevent PBS from escaping. Approximately 20-30 m of PBS was introduced into the preputial cavity using a funnel attached to the open end of the pipette. The preputial cavity was closed and agitated vigorously for 2 to 3 minutes to free any organisms present in the crypts of the mucous membrane of the penis and prepuce. The washings were then transferred to universal containers and kept in a cool box with ice packs until transported to the laboratory, where they were received within 6 to 8 hours of collection.
Culture and polymerase chain reaction (PCR) testing for Campylobacter fetus and Trichomonas fetus. On arrival at the laboratory, the sheath wash samples were centrifuged at 1200 g for 10 minutes. The supernatant was aspirated with a syringe and filtered through 0.65 µm Millipore filters. Skirrow's agar was inoculated with a drop of filtered and unfiltered sample. The sediment was directly examined by light microscopy on a wet mount slide prepared from the sample to detect Trichomonas fetus.
A portion of the sediment was inoculated into Steve's culture medium and incubated at 37 ºC in an ordinary incubator for up to 72 hours. Amphotericin B (antifungal) at a concentration of 1.5 % and 100 000 units of penicillin (antibacterial) were added to the media to control mould and bacterial growth.
For Campylobacter, plates were incubated at 37 ºC under microaerophilic conditions with 5-10 % oxygen, 5-10 % carbon dioxide and 5-9 % hydrogen for optimal growth. Conditions of culture and incubation were systematically verified by using control strains of C. fetus subsp. fetus and C. fetus subsp. venerealis. Following incubation, all plates were examined for the presence of small (1-3 mm diameter), smooth and shiny colonies having a slightly grey to pink appearance16. Suspect colonies were Gram-stained and if slender, curved Gram-negative rods were observed, the colonies were re-streaked onto Columbia blood agar plates to purify and bulk up growth for further testing. Preliminary tests used to establish whether the suspect isolates belonged to the genus Campylobacter included catalase and oxidase production, growth at 25 ºC, anaerobic growth at 37 ºC, and no hydrogen sulphide production on Triple Sugar Iron (TSI) agar.
In addition to culture, DNA was extracted from samples using the Silica-Guanidium thiocyanate method. The PCR test was performed using primers specific for a unique sequence in the Campylobacter fetus and/or Trichomonas fetus DNA with the crude DNA extract as template9,18. The following species-specific primer sequences were used in this study. For Campylobacter fetus the forward primer set was 5'-GTTAGGGAAGAACAATGA CGG-3' and the reverse primer was 5'-TTATCTCTAAGA GATTAGTTGG-3'. For Trichomonas fetus the forward primer set was 5'-CGGGTC TTCCTATATGAG ACAGAACC-3' and the reverse primer was 5'-CTGCCGTTG GATCAGTTT CGTTAA-3'. Cycling conditions for targeted genes were denaturation at 95 ºC for 15 s, followed by 35 cycles at 94 ºC for 30 s, 60 ºC annealing for 30 s and extension at 72 ºC for 30 s.
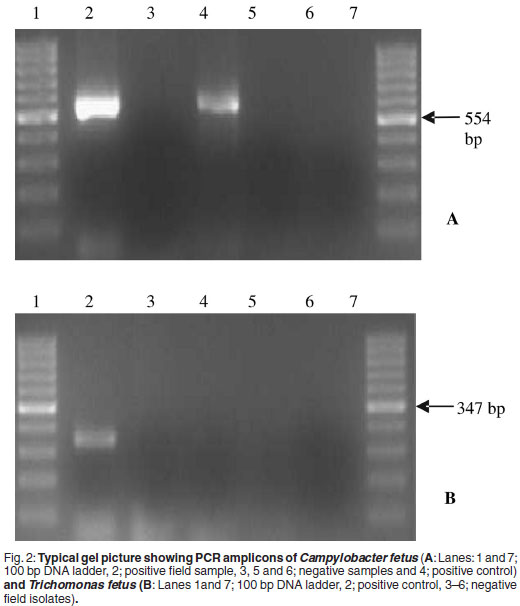
The PCR product was analysed by electrophoresis in a 1.5 % gel containing ethidium bromide and visualised under UV light for the presence of either a 554 bp product for Campylobacter fetus or a 347 bp product for Trichomonas foetus (Fig. 2).
Serological testing
Each serum sample was tested for antibodies against each of the pathogens as follows:
• Brucella abortus: samples were screened with the Rose Bengal Test, agglutination test, and those testing positive were confirmed with the complement fixation test.
• Neospora caninum: Neospora caninum antibody (Herdcheck®) ELISA test kits were purchased from IDEXX and the test carried out on each sample according to the kit manufacturer's instructions.
• The BVD/MD virus: BVD/MD virus antibody (Herdcheck®) ELISA test kits were purchased from IDEXX and the test carried out on each sample according to the kit manufacturer's instructions.
• The IBR/IPV virus: IBR/IPV virus antibody (Herdcheck®) ELISA test kits were purchased from IDEXX and the test carried out on each sample according to the kit manufacturer's instructions.
RESULTS
Nine out of 237 samples (3.8 %), representing 4 out of 15 herds, tested positive for Brucella abortus on the complement fixation test. Thirteen out of 145 samples (8.96 %) tested positive for Neospora caninum on the antibody (Herdcheck®) ELISA test. One hundred and eighteen out of 239 samples (49.37 %) tested positive for the BVD/MD virus on the antibody (Herdcheck®) ELISA test and 178 out of 239 samples (74.47 %) tested positive for the IBR/IPV virus on the antibody (Herdcheck®) ELISA test (Table 1).
All the bulls that were sampled in the study except 3 (140/143) tested negative for Campylobacter fetus and Trichomonas fetus on the 2 or 3 samples collected from each bull (Table 2). Three bulls (2.1 %) tested positive for Trichomonas fetus on culture and PCR and of these 3, 2 (1.4 %) also tested positive for Campylobacter fetus on culture and PCR. These bulls were from Kaalfontein farm in Magaliesburg area (Table 2, Fig. 1). It is important to note that the 3 positive bulls all belonged to the same farmer. A total of 17 Bulls was tested in that area giving a herd prevalence of 17.6 %.
DISCUSSION
Brucellosis
In South Africa there is a legislative requirement that all heifers between 4 and 8 months old must be vaccinated with the live, attenuated S19 vaccine, which has been in force since 1968, or the RB51 vaccine for older cows. This has significantly reduced the national prevalence of brucellosis, from 10.5 % in 1976 to 6 % 3 years later. By 1984/5 the figure was further reduced to 1.9 % and by 1988/89 it was down to 1.4 %10. In the work reported here, only 9 samples out of the 237 tested were positive for brucellosis on the complement fixation test. Currently national eradication schemes in force in South Africa are directed against bovine brucellosis and bovine tuberculosis. For brucellosis, for example, the 3.8 % prevalence among emerging farmers established in the present study is more than twice the national average, which was reported to be 1.4 % in 1988/8911. This high prevalence is of major concern as much effort is being made to eradicate the disease. The discrepancy may also be attributable to a much smaller sample size in the present study. Steps should, however, be taken to ensure that all emerging farmers in Gauteng comply with the requirement to have every heifer vaccinated for brucellosis by the time they are 8 months of age. Older cows should be vaccinated with the RB51 vaccine.
BVD/MD virus
Various serological surveys have placed the prevalence of the BVD/MD virus in infected herds at between 49 % and 85 %. In a study of cattle in 2 herds in Brazil, BVD/MD virus herd prevalence of 49 % (34 out of 69 animals sampled tested positive) in 1 herd and 62 % (108 out of 174 animals sampled tested positive) in the other were found14. In Iran, 141 serum samples taken from 20 herds of cattle were tested for antibodies against BVD/MD and a prevalence of 72.25 % was recorded9.In Chile, serum samples were taken from 948 cows chosen from 40 herds and tested for antibodies against the BVD/MD virus. A seroprevalence of 73.8 % was observed20. In the present serological survey of 239 samples taken from 24 herds, a seroprevalence of 49.4 % was found. This falls within the limits of what other investigators have found.
IBR/IPV virus
This virus occurs widely in cattle. According to some authors, the IBR/IPV virus is considered the most commonly diagnosed viral cause of abortion in cattle12 Various serological surveys have demonstrated the prevalence of the IBR/IPV virus in infected herds at between 19.5 % and 86 %. The national prevalence in the United Kingdom has been estimated at 70 %2. Cattle in 2 herds in Brazil were tested for antibodies against the IBR/IPV virus, among other abortion causing pathogens. Herd prevalences of 39 % (22 out of 54 animals sampled tested positive) in 1 herd and 86 % (150 out of 174 animals sampled tested positive) in the other were found14. In a study of 313 animals from 5 herds in Turkey, 61 (19.5 %) tested positive for antibodies against the IBR/IPV virus using the virus neutralisation test22. This was a much lower prevalence compared with those reported in earlier studies in Turkey. The authors attributed this drop in prevalence to the effectiveness of control measures taken to control the virus in Turkey. In the present study at 75.9 %, the prevalence of the IBR/IPV virus was within the limits found elsewhere.
The clinical significance of the high prevalence rate of BVD/MD (49.4 %) and IBR/IPV (75.9 %) reported in this study is, however, not clear and should be investigated further through virus isolations from aborted fetuses or demonstration of active infection.
Neospora caninum
Although this protozoan parasite is widely recognised as a major cause of abortions in cattle, results of various surveys reveal that its prevalence varies greatly from place to place. In a survey of 2 farms in Algeria, serum samples were taken from 33 out of 87 fcattle in 1 farm and 31 out of 150 in a 2nd farm. These were tested for antibodies against Neospora caninum using an ELISA kit from IDEXX. An average prevalence rate of 32.8 % was obtained6. By contrast, a survey carried out in Virginia, USA, in 1997-1998 suggested that neosporosis may not be a significant problem12. In the present study, a prevalence rate of 8.96 % was found, leading to the conclusion that neosporosis in South Africa cannot be ignored among emerging farmers.
Of the animals tested serologically, 41.37 % were positive for both BVD/MD virus and IBR/IPV virus, 4.82 % were positive for these 2 as well as either Neospora caninum or Brucella abortus. Such scenarios might point to the possibility of the immunosuppressive properties of the BVD/MD virus21 making it easier for other pathogens to also infect the same animal.
Campylobacter fetus and Trichomonas fetus
Campylobacter fetus and Trichomonas fetus were isolated in only 1 of the cattle herds sampled, in which both Campylobacter fetus and Trichomonas fetus were cultured and detected by PCR from 2 bulls and Trichomonas fetus was detected by PCR from a 3rd bull. The overall prevalence of 1.4 % and 2.1 % of campylobacteriosis and trichomonosis, respectively, found in this study should therefore be interpreted with caution as it does not necessarily reflect the situation in the entire province. A prevalence of 28.7 % of Campylobacter fetus in sheath wash samples in communal grazing areas in South Africa has been previously reported19. The incidence of campylobacteriosis in sub-Saharan Africa varies. In Malawi, the reported incidence was 11.5 % in 294 zebu and 11.1 % of 54 exotic bulls tested serologically13. When the vaginal mucus agglutination test was performed on the cows, it showed that 53.8 % of the herds and 13.4 % of the samples were infected. Using the same test in Zimbabwe, 33 % of the cows sampled were found to be positive21.An incidence rate of 6.1 % among 194 cows sampled on farms was reported in West Bengal, India5.
The prevalence of Campylobacter fetus and Trichomonas fetus observed in this study was lower than what is generally reported. Despite this low isolation rate of campylobacters and trichomonads, irregular returns to service and infertility were frequent in some of the study herds. The factors associated with poor isolation and detection of Campylobacter fetus and Trichomonas fetus have been well documented. These include contamination with urine, semen, faeces and/or blood which are known to interfere with the sensitivity of the diagnostic tests26. Efforts were, however, made to improve the isolation and detection of the organisms by collecting samples from each bull 2-3 times at 1 week intervals as per standard sampling protocol for Campylobacter fetus and Trichomonas fetus17. The tests used are the OIE recommended standard tests.
In conclusion, the results of this study demonstrate the potential importance of BVD/MD, IBR/IPV, Neospora caninum and Brucella abortus as causes of reproductive failure in cattle in Gauteng Province. The clinical significance of these infectious diseases still needs to be fully investigated through isolations from aborted fetuses or demonstration of active infection. Government disease control practices have in the past only focused on control of Brucella abortus among the infectious causes of reproductive diseases in South Africa. Based on the high seroprevalence of other reproductive diseases in this study, other infectious causes of reproductive failures also need attention. Campylobacter fetus and Tritrichomonas fetus were not identified as major problems in emerging cattle farming areas in Gauteng province. Use of improved and validated PCR tests will, however, enhance the detection of these causative agents. A further study that involves more provinces and cattle in commercial farming areas is recommended.
ACKNOWLEDGEMENTS
We are indebted to David Letsoalo, Daniel Chipana and Johannes Kekana for their ungrudging support in sample collection and driving to the study sites throughout the duration of the study and to the Gauteng Province Animal Health Technicians for directing us to the relevant farms and also helping with sample collection. This project was funded by the Gauteng Province Department of Agriculture and Rural Development.
REFERENCES
1. Albayrak H, Yazici Z, Okur-Gumusova S 2007 Seroprevalence to bovine Herpesvirus Type 1 in sheep in Turkey. Veterinarski Arhiv 77:257-263 [ Links ]
2. Banks M 1999 Living with IBR. Holstein Journal 1:84. [ Links ]
3. Campero C M, Moore D P, Oseon A C, Cipolla A L. Odriozola E 2003 Aetiology of bovine abortion in Argentina. Veterinary Research Commununications 27:359-369 [ Links ]
4. Catalyst May 2005 http://www.conmark.org/files/Catalyst Newsletter May05.pdf [ Links ]
5. De B N, Chatterjee A, Bidyanta J, Chakroborty M, Deb S K, Mondal P, Sen G P 1982 Note on the problems of breeding cows with special reference to common coital infections. Indian Journal of Animal Sciences 52:700-702 [ Links ]
6. Dechicha A, Gharbi S, Kebbal S, Chatagnon G, Tainturier D, Ozrout R, Guetarni D 2010 Serological survey of etiological agents associated with abortion in two Algerian dairy cattle breeding farms. Journal of Veterinary Medicine and Animal Health 2:001-005 [ Links ]
7. Dubey J P, Schares G, Ortega-Mora L M 2007 Epidemiology and control of neosporosis and Neospora caninum. Clinical Microbiology Reviews 20:323-367 [ Links ]
8. Duffy J H, Clark B L, Monsborough M J 1975 The influence of age on the susceptibility of bulls to Campylobacter fetus subsp venerealis. Australian Veterinary Journal 51:294-297 [ Links ]
9. Felleisen R S J, Lambelet N, Bachmann P, Nicolet J, Muller N, Gottstein B 1998 Detection of Trichomonas fetus by PCR and DNA enzyme immunoassay based on rRNA gene unit sequences. Journal of Clinical Microbiology 36:513-519 [ Links ]
10. Garoussi M T, Haghparast A, Hajenejad M R 2009 Prevalence of bovine viral diarrhoea virus antibodies among the industrial dairy cattle herds in suburb of Mashad-Iran. Tropical Animal Health and Production 41:663-667 [ Links ]
11. Godfroid J, Bosman P P, Herr S, Bishop G C 2004 Bovine brucellosis. In Coetzer J A W, Tustin R C (eds) Infectious diseases of livestock (2nd edn) Vol. 3. Oxford University Press, Cape Town: 1510-1527 [ Links ]
12. Hovingh E 2009 Abortions in dairy cattle - 1 Common causes of abortions. Virginia Polytechnic Institute and State University, Blacksburg [ Links ]
13. Klastrup N O, Halliwell R W 1977. Infectious causes of infertility/abortion in cattle in Malawi. Nordisk Veterinaermedicin 29:325-330 [ Links ]
14. Mineo T W P, Alenius S, Naslund K, Montassier H J, Bjorkman C 2006 Distribution of antibodies against Neospora caninum, BVDV and BHV-1 among cows in Brazillian dairy herds with reproductive disorders. Revista Brasileira de Parasitologia Veterinária 15:188-192 [ Links ]
15. Njiro S M, Nkosi C M 2009 Detection of the bovine viral diarrhoea/mucosal disease (BVD/MD) virus in tissues from aborted ruminant foetuses using immunohistochemistry. Journal of the South African Veterinary Association 80:229-232 [ Links ]
16. Nuru S, Dennis S M 1976 Abortion and reproductive performance of cattle in northern Nigeria: a questionnaire survey. Tropical Animal Health and Production 8:213-219 [ Links ]
17. OIE 2009 Bovine genital campylobacteriosis. Manual of diagnostic tests and vaccines for terrestrial animals (mammals, birds and bees) (6th edn). Office Internationaledes Épizootes, Paris: 661-670 [ Links ]
18. Oyarzabal O A, Wesley I V, Harmon K M, Schroeder-Tucker L, Barbaree J M, Lauerman L H, Backert S, Conner D E 1997 Specific identification of Campylobacter fetus by PCR targeting variable regions of the 16S rDNA. Veterinary Microbiology 58:61-71 [ Links ]
19. Pefanis S M, Herr S, Venter C G, Kruger L P, Queiroga C C, Amaral L 1988 Trichomoniasis and campylobacteriosis in bulls in the Republic of Transkei. Journal of the South African Veterinary Association 59:139-140 [ Links ]
20. Reinhardt G, Riedemann S, Ernst S, Aguilar M, Enriquez R, Gallardo J 1990. Seroprevalence of bovine viral diarrhea/ mucosal disease in southern Chile. Preventive Veterinary Medicine 10:73-78 [ Links ]
21. Slauson D O, Cooper B J 1982 Mechanisms of disease; a textbook of comparative general pathology. Williams & Wilkins, Los Angeles [ Links ]
22. Tan M T, Yildirim W, Erol N, Gungor A B 2006 The seroprevalence of bovine herpes virus type 1 (BHV-1) and bovine leukemia virus (BLV) in selected dairy cattle herds in Aydin Province, Turkey. Turkish Journal of Veterinary and Animal Sciences 30:353-357 [ Links ]
23. Terblanche J 1979 Bovine vibriosis. Rhodesia Agricultural Journal 76:43-45 [ Links ]
24. Thrusfield, M. 1995 Veterinary epidemiology (2nd edn). Blackwell Science, Oxford [ Links ]
25. Vandeplassche M 1982 Reproductive efficiency in cattle: a guideline for projects in developing countries. FAO Animal Production and Health Paper No. 25. Food and Agriculture Organization of the United Nations, Rome [ Links ]
26. Wilson I G 1997 Inhibition and facilitation of nucleic acid amplification. Applied and Environmental Microbiology 63:3741- 3751 [ Links ]
Received: January 2011
Accepted: October 2011
* Author for correspondence. E-mail: njiros@arc.agric.za














