Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
Journal of the South African Veterinary Association
versión On-line ISSN 2224-9435
versión impresa ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.82 no.3 Pretoria ene. 2011
ARTICLE ARTIKEL
Pathology and immunohistochemistry of papillomavirus-associated cutaneous lesions in Cape mountain zebra, giraffe, sable antelope and African buffalo in South Africa
J H WilliamsI,*; E van DykII; P J NelI,III; E LaneIV; E Van WilpeV; R G BengisVI; L-M de Klerk-LoristVI; J van HeerdenVII
IDepartment of Paraclinical Sciences, Faculty of Veterinary Science, University of Pretoria, Private Bag X04, Onderstepoort, 0110 South Africa
IIDepartment of Companion Animal Clinical Studies, Faculty of Veterinary Science, University of Pretoria, Private Bag X04, Onderstepoort, 0110 South Africa
IIIFree State Department of Economic Development, Tourism and Environmental Affairs, Private Bag X20801, Bloemfontein, 9300 South Africa
IVResearch and Scientific Services, National Zoological Gardens, PO Box 754, Pretoria, 0001 South Africa
VElectron Microscopy Unit, Department of Anatomy and Physiology, Faculty of Veterinary Science, University of Pretoria, Private Bag X04, Onderstepoort, 0110 South Africa
VINational Department of Agriculture, PO Box 12, Skukuza, Kruger National Park, 1350 South Africa
VIIKimberley Veterinary Clinic, 16 Dalham Road, Kimberley, 8301 South Africa
ABSTRACT
Skin lesions associated with papillomaviruses have been reported in many animal species and man. Bovine papillomavirus (BVP) affects mainly the epidermis, but also the dermis in several species including bovine, the best-known example being equine sarcoid, which is associated with BVP types 1 and 2. This publication describes and illustrates the macroscopic and histological appearance of BPV-associated papillomatous, fibropapillomatous or sarcoid-like lesions in Cape mountain zebra (Equus zebra zebra) from the Gariep Dam Nature Reserve, 2 giraffes (Giraffa camelopardalis) from the Kruger National Park, and a sable antelope (Hippotragus niger) from the Kimberley area of South Africa. An African buffalo (Syncerus caffer) cow from Kruger National Park also had papillomatous lesions but molecular characterisation of lesional virus was not done. Immunohistochemical staining using polyclonal rabbit antiserum to chemically disrupted BPV-1, which cross-reacts with the L1 capsid of most known papillomaviruses, was positive in cells of the stratum granulosum of lesions in Giraffe 1, the sable and the buffalo and negative in those of the zebra and Giraffe 2. Fibropapillomatous and sarcoid-like lesions from an adult bovine were used as positive control for the immunohistochemistry and are described and the immunohistochemistry illustrated for comparison. Macroscopically, both adult female giraffe had severely thickened multifocal to coalescing nodular and occasionally ulcerated lesions of the head, neck and trunk with local poorly-circumscribed invasion into the subcutis. Necropsy performed on the 2nd giraffe revealed neither internal metastases nor serious underlying disease. Giraffe 1 had scattered, and Giraffe 2 numerous, large, anaplastic, at times indistinctly multinucleated dermal fibroblasts with bizarre nuclei within the sarcoid-like lesions, which were BPV-1 positive in Giraffe 1 and BPV-1 and -2 positive in Giraffe 2 by RT-PCR. The sable antelope presented with a solitary large lesion just proximal to the right hind hoof, which recurred after excision, and was BPV-1 positive by RT-PCR. Other wart-like growths were present elsewhere on the body. The Cape mountain zebra either succumbed from their massive lesions or were euthanased or removed from the herd because of them. The lesions were BPV-1 and/or -2 positive by RT-PCR. The buffalo lesions were wart-like papillomatous projections in the inguinal and udder region. Stratum granulosum cells that stained immunohistochemically positive in the various species appeared koilocyte-like, as described in human papillomaviral lesions.
Key words: African buffalo, bovine, Cape mountain zebra, fibropapilloma, giraffe, immunohistochemistry, koilocyte, papillomavirus, pathology, sable antelope, sarcoid-like.
INTRODUCTION
Papillomaviruses are widespread, possess oncogenic properties, and are generally species-specific, affecting many different species, in which they usually induce benign papillomas or fibropapillomas of cutaneous and mucous epithelia, especially of thin or traumatised regions of skin3,8 . Bovine papillomavirus (BPV) induces exophytic papillomas or 'warts'and fibropapillomas in cattle3 and has been associated with malignant transformation to carcinoma of the urinary bladder and upper alimentary tract in cattle experimentally fed bracken fern (Pteridium aquilinum)28. Some papilloma-viruses can also infect other species, resulting in a different pathological outcome from that in the normal host, e.g. cottontail rabbit papillomavirus causes papillomas in the natural host which rarely progress to carcinomas, but often induces skin cancer in domestic rabbits, and BPV can induce fibroblastic tumours in C3H/eB mice and malignant fibroblastic tumours in hamsters2. It is recorded that a domestic short-haired cat developed a multinodular exophytic papillomatous lesion on the dorsum of its nose after prior actinic damage, the viral DNA of which sequenced with 98 % similarity to human papillomavirus type 913.
Six different papillomaviruses occur in cattle, namely types 1, 2 and 5 which cause cutaneous fibropapillomas, types 3 and 6 which cause skin papillomas, and type 4 which is associated with papillomas of the alimentary tract3. BPV types 1 and/or 2 or closely homologous genetic variants especially of BPV type 1 have also been causatively associated with papillomatous, fibropapillomatous to sarcoidlike lesions in various other species. These include BPV-1 in a captive Burchell's zebra (Equus burchelli boehmi)11, both BPV types 1 and 2 in 2 inbred populations of Cape mountain zebra 12,14,15,27, BPV in horses2,8, donkeys18, domestic shorthaired cats20, water buffalo22 and llamas and alpacas21. BPV 2 was revealed in the stratum granulosum of papillomatous lesions on the muzzle and distal limbs of Patagonian Merino sheep. It stained immunohistochemically with rabbit anti-bovine papillomavirus primary antibody25. BPV 2 has been found in papillomas of a European bison medial to the medial canthus of the left eye10. Impala and giraffe in Kenya were reported in 1978 to have viral skin papillomas but BPV was neither ruled in nor out6.
In 172 hyperplastic and neoplastic epithelial lesions from 8 different mammalian and 1 avian species, immunohistochemically positive papillomavirus structural antigens were only found in well-differentiated cells of the stratum granulosum, stratum spinosum and stratum corneum23. In human cervical intraepithelial neoplasia, human papillomavirusinfected cells are characteristically found in the upper layer of the epithelium; they have enlarged irregular nuclei with clear cytoplasm and are called 'koilocytes'24.
Equine sarcoids are locally aggressive fibroblastic benign tumours of skin17. They have been clinically classified as fibroblastic tumours with a variable epidermal component, which present in 6 types, with some subtypes, that correlate closely with the histological appearance8. The types include superficial or 'occult' type which presents as local alopecia, scaling and skin thickening; verrucous or 'warty'type; nodular types; fleshy and fibrovascular fibroblastic sarcoids, often resembling granulation tissue; and a malignant type that is locally aggressive and extends widely into adjacent skin and subcutis. The majority of sarcoids in horses have mixed components of 2 or more types.
Each species in which BPV is associated with cutaneous lesions appears to have its own distinct sarcoid-specific variants when BPV DNA is sequenced2,20,21, the exception being the recently reported water buffalo with multiple cutaneous and perivulvar fibropapillomas, where the long control region DNA sequence was identical to that of BPV-1, and in which viral particles were also produced22.
This publication describes and illustrates the macroscopic and histological appearance of BPV-associated papillomatous, fibropapillomatous or sarcoid-like lesions in an outbreak in inbred Cape mountain zebra (Equus zebra zebra) from the Gariep Dam Nature Reserve, 2 giraffe (Giraffa camelopardalis) and an African buffalo (Syncerus caffer) from the Kruger National Park, and a sable antelope (Hippotragus niger) from the Kimberley area of South Africa. There have been anecdotal reports of similar lesions in various game species, especially giraffe, in both South Africa and neighbouring regions. To date no investigative pathological studies have been reported.
MATERIALS AND METHODS
The history, clinical appearance and presentation, epidemiology and molecular diagnostic techniques used in the zebra included in this paper have been described in detail elsewhere14,15,19,27.
Lesions from all cases described in this paper were submitted in 10 % neutral buffered formalin, digitally photographed where whole lesion/s were present, routinely trimmed, embedded in wax blocks, sectioned at 5 microns, and stained with haematoxylin and eosin (HE). They were examined by light microscopy, described and digitally photographed. Immunohistochemistry was performed using polyclonal rabbit anti-papillomavirus antibody, the immunogen being chemically disrupted BPV type 1 (DakoCytomation N-Series Primary Antibody, DakoCytomation Inc, Carpinteria, USA), using the avidin-biotin technique 4. This antibody reacts with the L1 capsid of most known papillomaviruses. Simultaneously with each staining session, a known naturally-occurring bovine fibropapilloma, previously shown by electron microscopy to have papilloma-viral particles in the epidermal stratum granulosum cells that also stained positively with the BPV antibody, served as a positive control.
Prior to preservation in formalin, fresh tissues from the zebra27, giraffe and sable26 were prepared for transmission electron microscopy according to standard procedure as reported and described elsewhere26,27 and tissues from these cases/ animals were also analysed by reverse transcriptase PCR (RT-PCR) as described in the same reports. The buffalo tissue was similarly prepared; unfixed tissue was examined ultrastructurally and for-malin-fixed tissue immunohistochemically and results are reported here. Buffalo samples were not subjected to RT-PCR testing due to financial constraints.
RESULTS
The results obtained by the different diagnostic techniques applied are summarised in Table 1. The macroscopic and microscopic findings are described in detail below.
Bovine positive control
The bovine fibropapilloma was an exophytic cylindrical lesion approximately 2 cm in diameter and 3 cm in vertical height with a central core of fibroblastic tissue, an irregular outer covering of markedly hyperplastic and hyperkeratotic epidermis, and with the stratum granulosum containing scattered koilocytes, i.e. virus-infected cells with clear cytoplasm (Fig. 1). Dermal papillae with branching and connecting rete pegs extended into and across the fibroblastic core. Dermal fibroblastic nuclei were mostly small, slender, wavy to straight, basophilic and these cells were producing collagen which stained similarly to normal collagen with HE. No mitoses were found in 10 high-power fields (hpfs). From the same bovine, a sarcoid-like expansile dermal lesion was also submitted. This lesion had mostly thick, rounded or irregular, branching dermal papillae extending from the hyperplastic, hyperkeratotic epithelium into the underlying pleomorphic fibroblastic tissue with very few slender rete pegs as normally seen in equine sarcoids. Eight mitoses were counted per 10 hpfs in the fibroblastic part of this lesion and cells in this region were mostly pleomorphic with vesicular nuclei and producing intermediately-eosinophilicstaining collagen.

Cape mountain zebra
In 1995, an outbreak of equine sarcoid occurred in an inbred18 population of Cape mountain zebra in the Gariep Dam Nature Reserve13,14. Most affected animals had 1 lesion and no animals had more than 4 lesions; the lesions were predominantly fibroblastic (57 %), but verrucous (16 %), nodular (10 %), or combinations thereof (Fig. 4) occurred. Male zebra had mostly inguinal lesions and females were affected especially on the head and neck (Figs 2 and 3). Tumours averaged an annual increase in lesion size of up to 260 %, and movement of affected animals was impeded. Serum leakage to the exterior due to extensive epidermal ulceration of the lesion surfaces resulted in hypoproteinaemia, weakness and death in several cases. The incidence increased to 17.6 % at 1 stage during the outbreak13,14. Twenty-three biopsies from sarcoid lesions of 19 affected animals were received and examined including 4 biopsies from 1 animal and 2 from another. The dermal sarcoid component was white and uniformly firm.
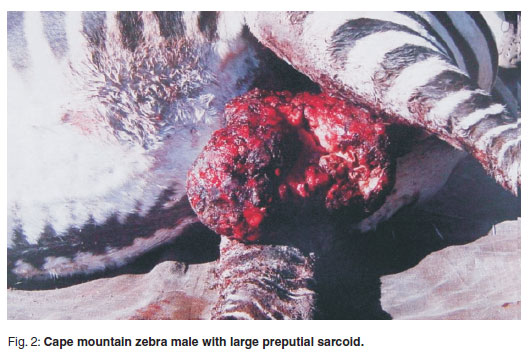


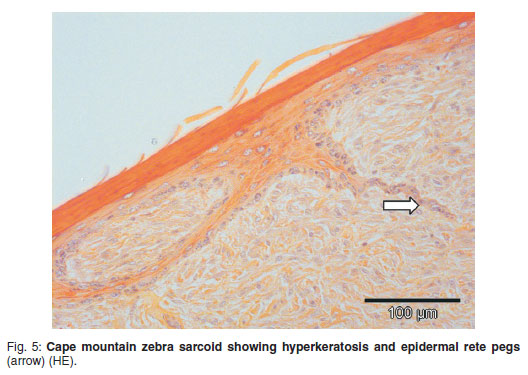
On microscopic examination, the masses extended from the dermal-epidermal interface into deep dermis and subcutis, with scattered small blood vessels containing very little blood (Fig. 5). All dermal adnexal structures were obliterated except for occasional remnant hair follicles. Where epidermis was still intact, it was either thin or hyperplastic with dyskeratosis, with marked superficial orthokeratotic hyperkeratosis. Intermittent thin or branched rete pegs formed tendrils into the underlying sarcoid. 'Picketing'as seen in many equine sarcoids was not a feature in the zebra lesions3,5. The dermis was expanded from the epidermal interface by pleomorphic fibroblasts with intercellular mature and immature collagen in varying amounts, the cells forming streams, interlacing bundles, occasional whorls or being haphazardly arranged. The more superficial cells often had larger more vesicular nuclei, which occasionally appeared anaplastic, as well as more mitotic figures and deeper regions were populated with smaller spindle cells with smaller more basophilic oval or wavy to elongated nuclei.
Scattered lymphocytes, plasma cells and occasional eosinophils and mast cells were found throughout the dermal component. Ten of the biopsy specimens were superficially ulcerated with superficial serocellular crusting, a predominantly neutrophilic exudate, contamination by extraneous material and bacteria and a purulent inflammatory reaction zone with underlying granulation tissue, often with eosinophils and neutrophilic vascular leucocytosis and exocytosis. Eleven of 23 biopsy samples had no mitoses per 10 hpf, 2 had 1 mitosis/10 hpf, 3 had 2/10 hpf. 3 had 4/10 hpf, 1 had 8 mitoses/10 hpf and there was 1 with 17 mitoses counted in 10 hpf. Immunohistochemistry and transmission electron microscopy26 failed to show papillomavirus in either epidermal or dermal regions.
Giraffe 1
An adult female giraffe with numerous, almost confluent, wartlike lesions of the neck and back and multifocal verrucous to nodular lesions of the trunk and limbs26 was shot near the Shingwedzi rest camp in Kruger National Park in May 2007.
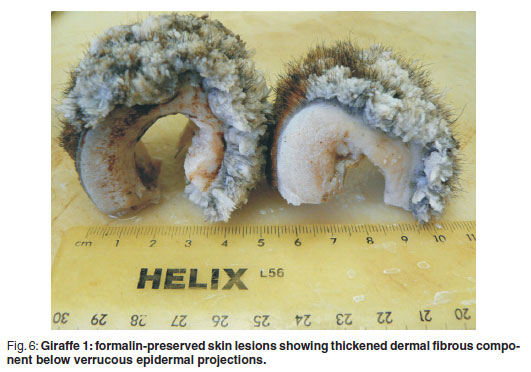
The formalin-preserved skin samples had markedly thickened epidermis and dermis and varying diameter white circular clusters of exophytic frond-like thin papillary outgrowths within and between each of which protruded slightly longer hairs. The epidermal-dermal and hypodermal interfaces were generally indistinct, white and widened up to approximately 1 cm. The dermis was tough to section (Fig. 6).
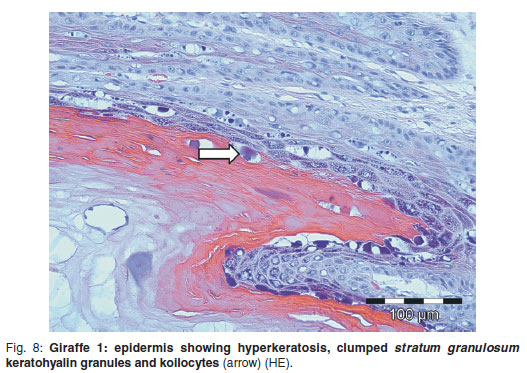
The epidermal and dermal lesions microscopically resembled a combination of papilloma and equine sarcoid respectively without rete pegs, although their structure was very different from the bovine control. The epidermis was markedly hyperplastic, acanthotic and thrown up into regular pointed peaks and troughs (Fig. 7), with the stratum granulosum having numerous koilocytes with small pycnotic nuclei and variably-sized basophilic to amphophilic keratohyalin granules within the clear cytoplasm (Fig. 8). The stratum corneum was markedly widened by orthokeratotic hyperkeratosis which sloughed in parallel lamellae with numerous small loose keratin scales most superficially. There were no rete pegs.

The superficial dermis in all areas of lesion was replaced by more densely cellular active fibroblastic cells with small vesicular wavy nuclei and pale eosinophilic, immature collagen, replacing adnexal structures except for scattered hair follicles proximal to the sweat glands. In some sections this tissue infiltrated the hypodermis and subcutis, entrapping mature, darkly eosinophilic and poorly cellular normal collagen bundles and replacing or compressing sweat glands. Scattered cells throughout were large, with pleomorphic, palely-vesicular, bizarre nuclei and occasional intra-nuclear cytoplasmic invaginations or bi-or vague multi-nucleation. Distinct mitotic figures were not found. In the adnexal zone, hair follicles contained hair shafts.
Immunohistochemical staining for papillomavirus was positive in the stratum granulosum in scattered small pycnotic nuclei of koilocyte-like cells (Fig. 9).

Giraffe 2
A 2nd female giraffe aged 15 years was euthanased a year after Giraffe 1, in the vicinity of the Skukuza rest camp of the Kruger National Park. The lesions were found predominantly on the head, neck and shoulders, and individually they were more nodular, thicker and generally much larger than those of Giraffe 126.
The fresh skin specimens had irregular, variably hyperkeratotic but narrow (1-2 mm) black-pigmented epidermis with sparse short protruding hairs (Fig. 10). The dermis below was up to 3-4 cm thick, whitish to pale pink, firm, rubbery, homogenous tissue forming distinct or indistinct, variably-sized, circular nodules, some of which were superficially ulcerated.
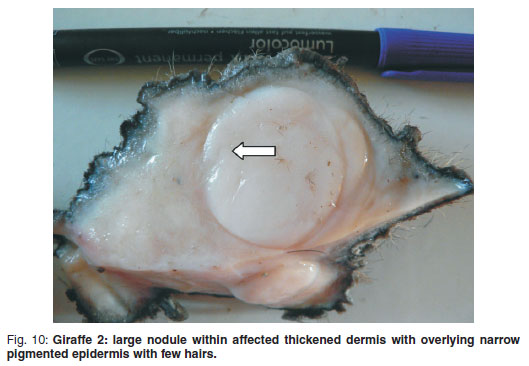
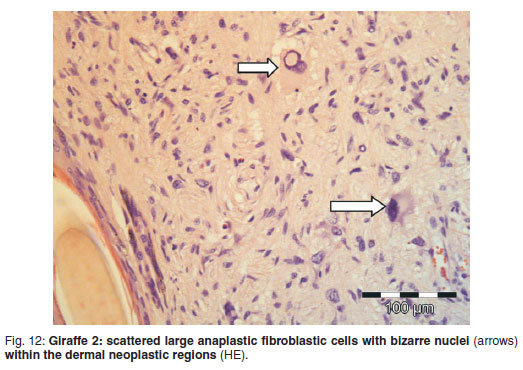
Microscopically, the occasional areas of skin with no associated dermal neoplasia showed only mild hyperkeratosis over a regularly-ridged epidermis with normal adnexal structures. There was an abrupt transition from normal skin to lesions with marked epidermal orthokeratotic hyperkeratosis, containing multifocal occasional fungal spores and hyphae within the expanded stratum corneum, often adjacent to hairs, but not extending distally into hair follicles. Neither stratum granulosum koilocytes nor rete pegs were present (Fig. 11). The subepidermal, dermal, hypodermal and subcutaneous regions, even in some places below normal epidermis, were similar to those described in Giraffe 1. The transformed fibroblasts had mostly vesicular spindle or oval nuclei. The pale eosinophilic immature collagen matrix formed whorls, palisades and storeyform patterns. In all areas of tumour, large, pleomorphic fibroblasts in some places numbered up to 12 per hpf (Fig. 12). These cells usually had several nucleoli of varying shapes. Occasional larger nuclei were granular, resembling early mitotic figures; otherwise recognisable mitotic figures were not found.

Immunohistochemical staining failed to reveal papillomavirus in these lesions.
Histopathology of mildly autolysed internal organs submitted in 10 % formalin after necropsy showed follicular and paracortical reactive lymphoid hyperplasia with sinus histiocytosis and medullary plasmacytosis of the prescapular lymph node and mild splenic follicular lymphoid hyperplasia with germinal centres and occasional post-reactive hyalinosis. Other observations included mild global proliferative glomerulonephritis, mild microvesicular hepatic degeneration, stress-induced adrenocortical hyperplasia, moderate pancreatic granule depletion, mild multifocal myocardial perivascular fibrosis, lymphoplasmacytic and eosinophilic colitis with multifocal mucous metaplasia, multifocal perivascular lymphocytic interstitial pneumonia, focal encapsulated lymphoplasmacytic pneumonia with central mineralised tissue debris, mild chronic pyloric eosinophilic enteritis with hepatic capsular adhesion, mild agonal alveolar emphysema, and visceral congestion. Special stains revealed no acid-fast bacteria within lung or lymph node sections or in the area of focal pneumonia.
Sable antelope
A single sable antelope cow out of a herd of 30 translocated from their natural habitat to a game farm in the Kimberley area was lame due to a growth 6 cm in diameter on the lateral aspect of the distal region of the 2nd phalanx, proximal to the right hind hoof. The mass recurred after excision and other well-defined masses were found on the right shoulder and lip. Three specimens of formalin-preserved tissue were submitted for histopathological examination.
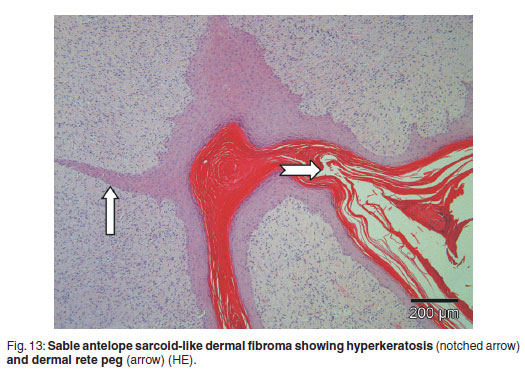
The non-ulcerated tissue with angular, thick superficial papillary projections had narrow attachments to the underlying dermis comprising immature moderately cellular fibroblastic tissue. This tissue was covered by thin, intermittently pigmented epidermis with moderate abrupt orthokeratotic hyperkeratosis proximally and thin, often connecting or branching dermal rete pegs projecting into the underlying dermal fibrous tissue (Fig. 13). The pleomorphic, small vesicular dermal fibroblasts produced pale eosinophilic immature collagen, sometimes forming whorls or interlacing bundles. Papillary fibrous tissue contained scattered blood-filled small vessels, but the underlying dermis was poorly vascular. Few koilocytes were found in the stratum granulosum. The most superficial keratin laminae in places had remnant nuclei and aggregations of serum with necrotic cellular debris, suggesting prior inflammatory response.
The ulcerated biopsied tissue comprised the same cellular fibrous tissue and immature collagen, but with overlying perpendicularly vascularised granulation tissue. A broad band of superficial inflammatory cells, predominantly lymphocytes and plasma cells with scattered neutrophils and degenerated leucocytes, mingled with overlying necrotic fibrous tissue. Epidermis and adnexal structures were absent. Mitotic figures in the ulcerated region numbered up to 7/10 hpfs, but none were identified in the non-ulcerated regions.
Immunohistochemical staining for BPV showed few occasional small shrunken nuclei staining positively in non-vacuolated cells of the stratum granulosum of the epidermis. The lesion resembled a verrucous equine sarcoid.
African buffalo
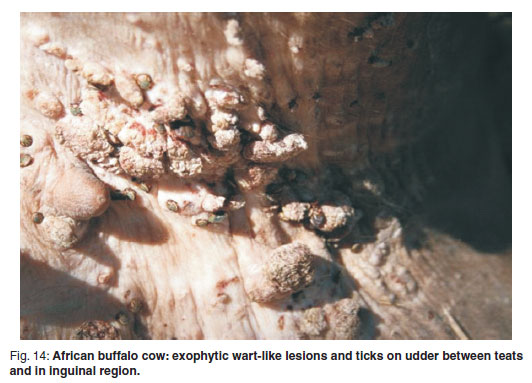
A 2-year old buffalo heifer within a herd at Ntlokweni in the southwestern part of the Kruger National Park was seen during routine procedures, after chemical immobilisation, to have exophytic papillomatous lesions, measuring from 1 to 5 cm in diameter, clustered on the udder between the teats (Fig. 14). Isolated lesions were also present in the groin and on the inner thigh. The geographical position of the herd was 25º39'12"S and 31º31'12"E. A sample of lesion was placed unfixed in a sterile tube for electron microscopy and a duplicate placed in 10 % formalin for histopathology.
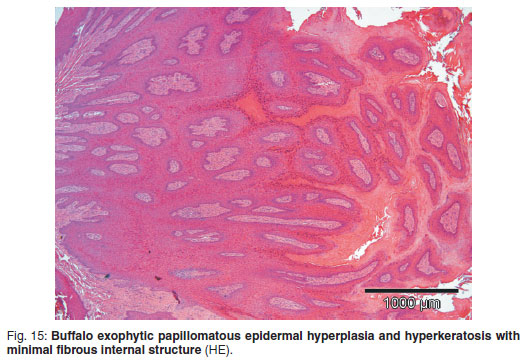
The formalin-fixed specimen was wart-like with multiple thin papillary projections, measured approximately 2.5 cm diameter × 2 cm in depth and was curled towards the dermis. Microscopically this lesion resembled a classical papilloma, comprising a locally-extensive area of vigorous exophytic papillary epidermal hyperplasia with marked lamellar orthokeratotic hyperkeratosis which flaked off superficially (Fig. 15). Very few vacuolated koilocyte-like cells were found in the stratum granulosum. Multiple epidermal projections were supported internally with scant interlinking dermal fibrous connective tissue containing scattered lymphocytes, plasma cells and some neutrophils, especially at the dermal--epidermal interface. Directly below the mass, adnexal structures were scarce, comprising mainly occasional hair follicles.
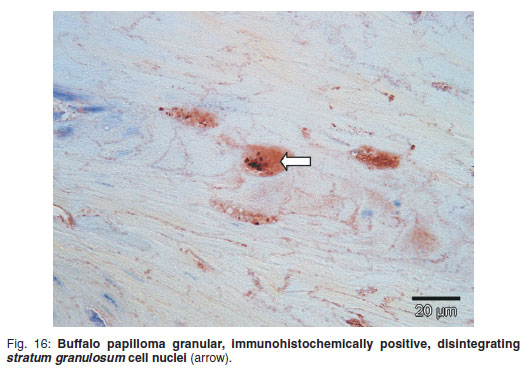
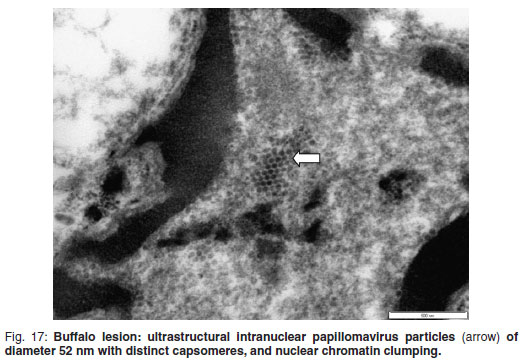
Immunohistochemistry showed granular nuclear staining within vague vacuolated degenerated cells which were structurally very indistinct (Fig. 16). The unfixed buffalo lesional material, examined ultrastructurally with negative staining, contained intranuclear papillomavirus particles measuring 52 nm in diameter and with distinct capsomeres (Fig. 17), and chromatin clumping was present.
DISCUSSION
The papillomaviruses normally infect epithelial cells of animals and humans, resulting in proliferative, typically benign, self-limiting and spontaneously regressing lesions called warts, papillomas or condylomas2. Some papillomaviruses are linked to malignancy, especially cervical carcinoma, linked to human PV types 16 and 18, in women2. Equine penile squamous cell carcinomas and occasional non-squamous cell carcinomas have recently been associated in a series in North America with equine papilloma-virus type 2 DNA sequences7. Some types of papillomaviruses also infect fibroblasts, inducing fibro-epithelial tumours; BPV types 1 and 2 are amongst these and are known to cause benign fibropapillomas in cattle3.
During acute virus infection, replication of the genome is strictly linked to the state of differentiation of the infected cells; in papillomatosis the virus initially infects the basal keratinocytes. The early region genes are expressed in the undifferentiated basal and suprabasal layers. Viral DNA replicates in the differentiating spinous and granular layers and expression of the late structural proteins is limited to the terminally differentiated cells of the squamous layer, where the new virus particles are encapsidated and released into the environment as the cells die2. Initiation of malignant transformation of fibroblasts is linked to the deregulated expression of the early virus genes, resulting in uncontrolled proliferation and loss of differentiation of infected cells. The major transforming protein E5 is responsible for various complex biochemical cellular pathways2.
The histological lesions from both giraffes in the current study showed scattered large pleomorphic anaplastic fibroblasts with bizarre nuclei which at times were indistinctly multinucleated or folded. Giraffe 2 had a denser population of these cells and also had neither epidermal virus, nor koilocytes, in comparison with Giraffe 1. Both giraffes, however, still registered BPV DNA on RT-PCR testing 26. It would appear that this is a local histological development of malignancy and possibly the end stage of viral cell transformation due to BPV in this species. Giraffe 2 had internal histological changes reminiscent of aging, stress and most likely bouts of parasitic and other infections. There were no metastases of the skin tumours to regional lymph nodes nor to internal organs despite the increased evidence of histological malignancy, and the regional lymph node showed histological reactivity as expected in an immunologically competent individual.
Most equine sarcoids contain detectable BPV types 1 and/or 2 viral DNA16, and are also known to express the BPV types 1 and 2 major transforming oncoprotein E5, but do not produce infectious virus particles, hence being considered a nonproductive infection where viral DNA exists episomally1,9. Only the early genes are transcribed in order to maintain a copy number and to control cell growth. Thus virus capsids are not formed, possibly due to expression of capsid proteins needing the cellular environment only found within the well-differentiated keratinocytes of the host species. Equine sarcoids with BPV DNA have been detected widely and there is little evidence for expression of the BPV structural proteins or for virus capsid formation2. BPV has not been isolated from clinical cases nor has BPV DNA been found in equine papillomas, equine non-sarcoid tumours or equines without sarcoids, but it has been found in some cases of dermatitis2. Experimental extracts of sarcoid from horses do not induce warts in cattle2.
Intra-type sequence variation occurs within papillomavirus types, influencing the cellular location and function of the oncoproteins, consequently affecting the pathogenesis and transforming ability of the virus2. Each species appears to have its own distinct variant of BPV. The sequence of E5 protein also changes and may contribute to the differences in pathogenesis of disease in each species2. BPV E5 transforming oncoprotein was found in all equine sarcoids in 1 study2. Viral gene expression, especially of E5, may contribute to virus persistence and disease pathogenesis by down-regulating MHC class 1 expression2. Transformation of fibroblasts in non-host species is by BPV genomes maintained episomally2.
In most cases papillomavirus lesions regress following activation of the host immune response; however, several immune evasion methods have been described. Equine sarcoids are non-regressing lesions, suggesting that such evasion mechanisms are evoked in this species2. If MHC class 1 expression is down-regulated, cytotoxic T lymphocytes may not be able to detect infected cells, and hence may be a factor in BPV persistence. Sarcoids are also recurrent lesions. Aberrant perinuclear localisation of p53 has been demonstrated in 44 % of equine sarcoids, suggesting that mutational independent inactivation of p53 occurs commonly in sarcoids2.
The modes of infection, epidemiology, susceptibility of various game animal species and individuals, as well as reasons for production of the types of lesions produced in the animals of this paper are, at this stage, speculative, but the lesions and their course in the cases described here resembled to quite a large degree that of equine sarcoid, in its various forms, as caused by BPV types 1 and 2. It is generally experienced that sarcoid appears to be predisposed at sites of trauma or prior skin damage, thin skin, and where insects cause irritation to skin, and this was probably the case in the zebras and most likely from insect activity or inter-animal trauma.
There is a known breed predisposition for sarcoids in horses2,8 with thinner-skinned breeds like Arabians being most prone, and Lipizzaners being most resistant8. The Cape mountain zebra in Gariep Dam Nature Reserve were known to be inbred19 and this possibly led to susceptibility to the BPV types 1 and 2 DNA that were found to be associated with the lesions. Horses and cattle on surrounding farms at the time of the outbreak showed no papillomas or sarcoid-like lesions14,15 suggesting that BPV might be ubiquitous in the environment, possibly residing in insects or other vectors, or on fomites as yet undiscovered. Over the years of the outbreak, removing affected individuals either physically or by euthanasia seemed to decrease the subsequent incidence15. It appeared at the time that the initial nodular lesions progressed to become fibroblastic in type when they outgrew the overlying epidermis, which eventually ulcerated (P J Nel pers. obs.), and that fibroblastic lesions could be a natural progression of nodular lesions. Another isolated and inbred population of Cape mountain zebra in the Bontebok Nature Reserve experienced a similar outbreak of sarcoid-like lesions around 200212. The lesion prevalence in this population was 53 %. A concurrent study on the incidence of sarcoid in genetically diverse Burchell's zebra in the Kruger National Park was 1.9 %12. Virus particles were not found in the zebra lesions by either immunohistochemistry or electron microscopy, a similar finding to that in equine sarcoid.
Only Giraffe 126, the sable26 and buffalo had detectable virus particles, mostly in the stratum granulosum nuclei of the epidermis with transmission electron microscopy and also immunohistochemically. It may be speculated that the more serious and advanced lesions in Giraffe 2 were a natural late progression from the lesion stage of Giraffe 1, and for unknown reason/s viral particles were no longer being formed in the epidermis, but the continuing episomal DNA effect of E5 protein was still active in dermal fibroblasts. Such lesion stage information gleaned by histology, immunohistochemistry and/or electron microscopy might be relevant when assessing whether an affected animal could be infectious to others of its own kind or to other species in the vicinity.
In game species in large areas of wilderness, individuals with sarcoid-like lesions appear to occur individually and sporadically, as found with Giraffes 1 and 2, as mentioned in anecdotal reports, and as reported in Burchell's zebra12. The lesions may be debilitating and even life-threatening, as seen in the Cape mountain zebra, or predispose to secondary bacterial or other parasitic infections if ulcerated. Lesions in areas of potential contact by animals of the same species, such as a buffalo calf suckling from teats adjacent to papillomatous lesions, or by predator ingestion of affected animals, could conceivably lead to direct infection if contact occurred.
CONCLUSION
Histological, immunohistochemical, ultrastructural and molecular investigation techniques are warranted in more individuals of various game species with papillomatous, fibropapillomatous or sarcoid-like cutaneous lesions. Such information would build a broader database of this at times seriously debilitating and occasionally life-threatening condition and help to elucidate epidemiological mechanisms of infection and therefore possible intervention, prevention or treatment measures. Increasing numbers of game species are being captive-bred, artificially fed, and/or kept on relatively small game farms, leading to inbreeding, habitat pressure, an increasing wildlife - livestock interface, and increasing population pressure within their own species and between species. Together with changing vector dynamics associated with global warming, these could be ideal circumstances for infectious agents such as BPV to increase in importance.
ACKNOWLEDGEMENTS
The South African Veterinary Foundation is sincerely thanked for their monetary contribution towards the costs of the molecular diagnostic work done on these animal specimens by Dr van Dyk. The kind assistance of the late Ryno Watermeyer is sincerely appreciated and remembered at the microscope with camera in the Department of Tropical Diseases at the Veterinary Faculty at Onderstepoort. The staff of the histopathology laboratory of the Section of Pathology at Onderstepoort are also gratefully acknowledged for their care in producing the histological sections and specifically Mrs Marie Smit and Mrs Rephima Phaswane for the immunohistochemical staining. Professor A. Kotze of the Genetics Department of the University of the Free State, Bloemfontein, South Africa, is acknowledged for allowing funds from the United Fish and Wildlife Service - Wildlife Without Borders-Africa programme to process the necropsy histology of Giraffe 2.
REFERENCES
1. Amtmann E, Muller H, Sauer G 1980 Equine connective tissue tumours contain unintegrated bovine papillom virus DNA. Journal of Virology 35: 962-964 [ Links ]
2. Chambers G, Ellsmore V A, O'Brien P M, Reid S W J, Love S, Campo M S, Nasir L 2003 Review article: Association of bovine papillomavirus with the equine sarcoid. Journal of General Virology 84: 1055-1062 [ Links ]
3. Ginn P E, MansellEKL, Rakich P M 2007 Skin and appendages. In Maxie G M (ed.) Jubb, Kennedy, and Palmer's pathology of domestic animals (5th edn), Vol. 1 Elsevier Saunders, London: 749-767 [ Links ]
4. Haines D M, Chelack B J 1991. Technical considerations for developing enzyme immunohistochemical staining procedures on formalin-fixed paraffin-embedded tissues for diagnostic pathology. Journal of Veterinary Diagnostic Investigation 3: 101-112 [ Links ]
5. Jackson C 1936 The incidence and pathology of tumours of domesticated animals in South Africa. Onderstepoort Journal of Veterinary Science and Animal Industry 6: 378-385 [ Links ]
6. Karstad L, Kaminjolo J S 1978 Skin papillomas in an impala (Aepyceros melampus) and a giraffe (Giraffa camelopardalis). Journal of Wildlife Diseases 14: 309-313 [ Links ]
7. Knight C G, Munday J S, Peters J, Dunowska M 2011 Equine penile squamous cell carcinomas are associated with the presence of equine papillomavirus type 2 DNA sequences. Veterinary Pathology Online DOI: 10.1177/0300985810396516 [ Links ]
8. Knottenbelt D C 2005 A suggested clinical classification for the equine sarcoid. Clinical Techniques in Equine Practice 4: 278-295 [ Links ]
9. Lancaster W D 1981 Apparent lack of integration of bovine papillomavirus DNA in virus-induced equine and bovine tumour cells and virus-transformed mouse cells. Virology 108: 251-255 [ Links ]
10. Literák I, Tomito Y, Ogawa T, Shirasawa H, Šmid B, Novotný L, Adamec M 2006 Papillomatosis in a European bison. Journal of Wildlife Diseases 42: 149-153 [ Links ]
11. Löhr C V, Juan-Sallés C, Rosas-Rosas A, Parás García A, Garner M M, Teifke J P 2005 Sarcoids in captive zebras (Equus burchellii): association with bovine papillomavirus type I infection. Journal of Zoo and Wildlife Medicine 36: 74-81 [ Links ]
12. Marais H J, Nel P J, Bertschinger H J, Schoeman J P, Zimmerman D 2007 Prevalence and body distribution of sarcoids in South African Cape mountain zebra (Equus zebra zebra). Journal of the South African Veterinary Association 78: 145-148 [ Links ]
13. Munday J S, Hanlon E M, Howe L, Squires R A, French A F 2007 Feline cutaneous papilloma associated with human papillomavirus type 9. Veterinary Pathology 44: 924-927 [ Links ]
14. Nel P J 2007 An outbreak of equine sarcoid in a population of Cape mountain zebra (Equus zebra zebra) - a retrospective study. MSc dissertation, University of Pretoria [ Links ]
15. Nel P J, Bertschinger H, Williams J, Thompson P 2006 Descriptive study of an outbreak of equine sarcoid in a population of Cape mountain zebra (Equus zebra zebra)in the Gariep Nature Reserve. Journal of the South African Veterinary Association 77: 184- 190 [ Links ]
16. Otten N, Von Tscharner C, Lazary S, Antczak D F, Gerber H 1993 DNA of bovine papillomavirus type 1 and 2 in equine sarcoids: PCR detection and direct sequencing. Archives of Virology 132: 121-131 [ Links ]
17. Ragland W H, Keown G H, Spencer G R 1970 Equine sarcoid. Equine Veterinary Journal 2: 2-11 [ Links ]
18. Reid S W J, Smith K T, Jarret W F H 1994 Detection, cloning and characterisation of papillomaviral DNA present in sarcoid tumours of Equus asinus. Veterinary Record 135: 430-432 [ Links ]
19. Sasidharan S P 2004 Comparative genetics of selected southern African mountain zebra (Equus zebra zebra and Equus zebra hartmannae) populations. MSc dissertation, University of Pretoria. [ Links ]
20. Schulman F Y, Krafft A E, Janczewski J 2001 Feline cutaneous fibropapillomas: clinicopathologic findings and association with papillomavirus infection Veterinary Pathology 38: 291-296 [ Links ]
21. Schulman F Y, Krafft A E, Janczewski T, Reupert R, Jackson K, Garner M M 2003 Camelid mucocutaneous fibropapillomas: clinicopathologic findings and association with papillomavirus Veterinary Pathology 40: 103-107 [ Links ]
22. Silvestre O, Borzacchiello G, Nava D, Iovane G, Russo V, Vecchio D, D'Ausilio F, Gault E A, Campo M S, Paciello O 2009 Bovine papillomavirus type 1 DNA and E5 oncoprotein expression in water buffalo fibropapillomas. Veterinary Pathology 46: 636-641 [ Links ]
23. Sironi G, Caniatti M, Scanziani E 1990 Immunohistochemical detection of papillomavirus structural antigens in animal hyperplastic and neoplastic epithelial lesions. Journal of Veterinary Medicine A 37: 760-770 [ Links ]
24. Stevens A, Lowe J S, Young B (eds) 2002 Wheater's Basic histopathology: a colour atlas and text (4th edn). Elsevier Sciences Limited, Edinburgh: 200-256 [ Links ]
25. Uzal F A, Latorraca A, Ghoddusi M, Horn M, Adamson M, Kelly W R, Schenkel 2000 An apparent outbreak of cutaneous papillomatosis in Merino sheep in Patagonia, Argentina. Veterinary Research Communications 24: 197-202 [ Links ]
26. Van Dyk E, Bosman A-M, Van Wilpe E, Williams J H, Bengis R G, van Heerden J, Venter E H 2011 Detection and characterisation of papillomavirus in skin lesions of giraffe and sable antelope in South Africa. Journal of the South African Veterinary Association 82: 80-85 [ Links ]
27. Van Dyk E, Oosthuizen M C, Bosman A-M, Nel P J, Zimmerman D, Venter E H 2009 Detection of bovine papillomavirus DNA in sarcoid affected and healthy free-roaming zebra (Equus zebra) populations in South Africa. Journal of Virological Methods 158: 141-151 [ Links ]
28. Wosiacki S R, Barreiro A B, Alfieri A F, Alfieri A A 2005 Semi-nested PCR for detection and typing of bovine papillomavirus type 2 in urinary bladder and whole blood from cattle with enzootic haematuria. Journal of Virological Methods 43: 5428-5434 [ Links ]
Received: February 2011.
Accepted: May 2011.
* Author for correspondence. E-mail: june.williams@up.ac.za














