Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Journal of the South African Veterinary Association
On-line version ISSN 2224-9435
Print version ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.82 n.3 Pretoria Jan. 2011
CLINICAL COMMUNICATION KLINIESE MEDEDELING
Antebrachial chondrodysplasia in New Zealand white rabbits (Oryctolagus cuniculus)
T PulkerI,*; A CarstensII; J WilliamsIII
IUniversity of Pretoria Biomedical Research Centre, Private Bag X04, Onderstepoort, 0110 South Africa
IIDepartment of Companion Animal Clinical Studies, Faculty of Veterinary Science, University of Pretoria, Private Bag X04, Onderstepoort, 0110 South Africa
IIIDepartment of Paraclinical Sciences, Faculty of Veterinary Science, Private Bag X04, Onderstepoort, 0110 South Africa
ABSTRACT
Rabbits obtained from a South African rabbit breeder exhibited deformities of the distal forelimbs. The clinical, radiological and histological presentation of mid-antebrachial valgus formation (a.k.a distal foreleg curvature) in these rabbits was consistent with an autosomal recessive trait associated with a chondrodystrophic lesion of the distal ulna epiphysis 1st described in the 1960s. The impact this might have on South African farming enterprises and laboratory facilities has not been established, but the heritability and welfare implications of this condition make it a concern. Mildly affected animals can adapt to the deformity with some housing adjustments, but severely affected animals may require humane euthanasia.
Keywords: antebrachium, chondrodysplasia, hereditary conditions, Oryctolagus cuniculus, rabbit, valgus.
INTRODUCTION
The use of rabbits in South Africa is confined mainly to farming enterprises and laboratory animal centres. Low-cost protein sources are constantly being sought to guarantee food security and rabbit meat is 1 of the avenues being explored. Diseases in a rabbitry can have profound effects on meat quality and economic viability. Pathological lesions observed during meat inspections in northern Italy abattoir rabbits mainly affected the integument, digestive and urinary systems6. Non-traumatic developmental musculoskeletal disorders are not commonly detected in abattoir rabbits during the slaughter process6. This may in part be due to the fact that many of the dysplastic conditions in rabbits are lethal mutations, associated with low survival rates. Production losses can be expected due to the decrease in progeny reaching slaughter age8. However, 1 form of chondrodysplasia in rabbits, known as distal foreleg curvature, is not associated with a change in growth rate or survival3,4, but these animals should be removed from the breeding stock to prevent dissemination of this mutation.
In a laboratory setting, the presence of 'silent' conditions can have varied or even unknown effects on research results. Although non-traumatic developmental musculoskeletal disorders in rabbits are not often diagnosed, these disorders have the potential to compromise the animal's well-being. Once recognised, various measures can be implemented to counteract this negative impact such as non-slippery flooring, improved access to food, water and shelter and regular monitoring.
CASE HISTORY
Nine of 18 approximately 3-month-old female New Zealand white rabbits, obtained from a local rabbit breeder and housed at the University of Pretoria Biological Research Centre (UPBRC), had varying degrees of lateral deviation of the forelimbs from the mid ulna-radius diaphyses distally, with the manus directed laterally away from the body. Three of the 9 animals were unilaterally affected on visual inspection. Physical examination of these animals revealed no clinical abnormalities apart from the deviated forelimbs associated with difficulty in adducting these limbs.
Radiographic findings of the forelimbs of 2 of these animals were suggestive of some form of thoracic limb chondrodysplasia. Additional radiographs were recommended, including an unaffected rabbit to serve as a control.
Four of the bilaterally affected rabbits (including the initial 2 animals sent for radiographs) and 1 unaffected rabbit were radiographed at the Onderstepoort Veterinary Academic Hospital, Department of Diagnostic Imaging, after intramuscular sedation with 35 mg/kg ketamine (Anaket-V, Bayer) and 5 mg/kg xylazine (Chanazine 2 %, Bayer). Both craniocaudal and mediolateral views of the antebrachiae, elbows and manus were taken. Craniocaudal views of the tibia, and distal and mediolateral views of the femur and distal views of both hind limbs were taken of the 2 affected animals and the unaffected animal.
Owing to severe locomotor deficits 8 months later 1 of the bilaterally affected rabbits was euthanased on humane grounds with 200 mg/kg sodium pentobarbitone intravenously (Euthapent, Kyron Laboratories) and necropsied.
RESULTS
Radiographic findings of the 4 affected animals were similar. Mild cranial (143- 160º) and moderate medial (131-148º) bowing of the mid-radial and, to a similar extent, mid-ulnar diaphyses was noted in all affected animals with no indication of metabolic bone disease (Fig. 1A,B). These deformities were not evident on the unaffected animal's radiographs (Fig. 2). The changes were restricted to the radius and ulna, as no radiographic changes indicative of chondrodysplasia were discovered on hind-limb and humeral views. Mild to moderate elbow degenerative joint disease was observed in all 5 radiographed animals. As similar changes were noted in the unaffected animal, this was considered an incidental finding. Bilateral stifle degenerative joint disease was evident in all 3 rabbits that had hind-limb radiographs taken. Two of these animals had bowing of the antebrachium and the 3rd had a normal antebrachium.
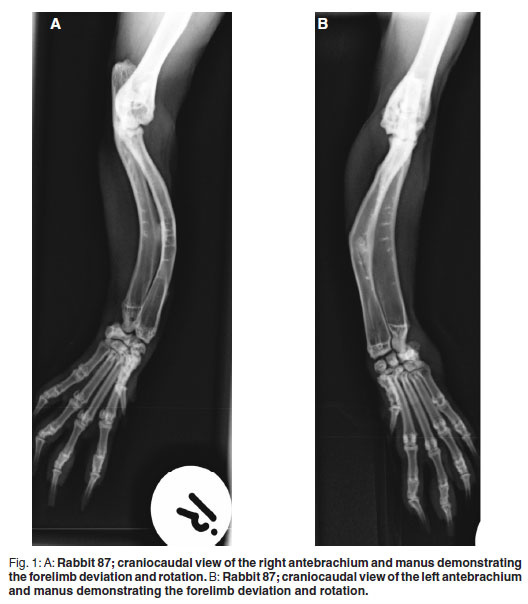

The only abnormality found macroscopically during necropsy examination of 1 of the affected rabbits was the bilaterally symmetrical bowing and shortening of the radii/ulnae. These were fixed in formalin in toto after which they were formalin in toto after which they were placed in 8 % formic acid, which was replaced every 4-5 days, to decalcify the bones. Only 0.5 cm of the distal and proximal radii/ulnae could be sectioned after 2 weeks of decalcification, suggesting normal mineralisation of these bones. Histologically these sections showed healthy bone and periosteum with medullary cavities containing healthy bone marrow.
At the time of diagnosis, the mean weight of the affected rabbits was 4.3 ± 0.33 kg while the unaffected animals had a mean weight of 4.69 ± 0.43 kg. Upon implementation of a restricted diet, for weight control reasons, 3 months later the mean weight for the affected animals was 4.88 ± 0.4kg and 5.31 ± 0.39 kg for the unaffected animals. Over the rest of the year, the mean weights for the animals fluctuated between 4.6-4.8 kg (affected) and 4.7-4.9 kg (unaffected).
DISCUSSION
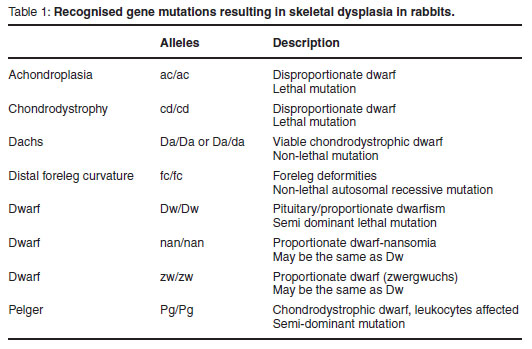
Skeletal dysplasias are a group of defects associated with short limbs, abnormal bone shape and/or increased bone fragility, which primarily affect bone formation or remodelling7. Despite the formulation of an extensive classification system for human skeletal dysplasias, a complete system for the corresponding animal conditions has not been reported. Skeletal dysplasias are a complex and diverse group of abnormalities. A brief overview of the recognised conditions in rabbits is summarised in Table 11.
Animal skeletal dysplasias are often propagated by inbreeding or excessive use of an animal carrying the relevant gene. It is, however, important to exclude non-genetic causes of dysplasia before condemning a stud animal. For the rabbits in question it was not possible to rule out infectious agents, toxin exposure or mineral deficiencies that could result in metabolic bone disease during gestation and early development, as the animals were only obtained in early adulthood. These causes were considered unlikely, however, owing to a lack of further clinical abnormalities and the localised nature of the lesion.
A relatively common clinical manifestation in rabbits, referred to as 'splay leg', presents as the inability to adduct any or all of the limbs. Splay leg can be used to describe the physical posture of mutants with syringomyelia, hypoplasia pelvis, femoral luxation, hereditary distal foreleg curvature or achondroplasia restricted to shoulder and hip joints1. It has also been used to describe young animals raised on slippery surfaces that present with the same inability to adduct the limbs5,8. Usually the latter cases can be corrected by placing the animal on non-slippery floors1,2,5,8. Two of the affected rabbits were housed together on plastic mesh flooring and straw. Although this non-slippery flooring improved mobility, the adduction problem was not resolved.
Radiographic findings suggested chondrodysplasia. This is a form of generalised skeletal dysplasia defined as a defect in or disordered development of cartilage7. The clinical, radiological and histological findings of these 4 affected rabbits correspond with lesions described in Beveren, Dutch, Belgian and French Silver rabbits in the 1960s3,4. The cause of the deformity in these breeds was found to be a chondrodystrophic lesion in the distal ulna epiphysis. The lesion appeared from 2 weeks of age and progressed until approximately 2 months, from which time regression occurred rapidly with complete radiographic and histological resolution of the lesion by approximately 4 months of age. The only indication of the condition after this was the permanent deformity of the distal forelegs. Animals displaying this deformity had no other clinical abnormalities; body growth and physical condition remained unchanged. This correlates with the findings that the 4 affected animals had no other clinical abnormalities and were in good physical condition. The radiographs were taken when the rabbits were over 4 months of age once the lesion had healed and only the permanent deformity could be visualised. The rabbit sent for necropsy was over a year of age at the time of necropsy, hence the lack of histological deformities in the radius and ulna. Although no significant difference in weights between the affected and unaffected animals could be detected and the growth of the animals was similar, the affected animals were slightly lighter than the unaffected animals, indicating potentially lower meat production from these animals.
This distal ulna epiphyseal lesion has been described in a comprehensive overview.3,4 It was found to be an interruption of normal endochondral ossification, resulting in persistent chondrocytes that continued to multiply. Without degradation of the cartilaginous matrix, an increasingly broad cartilaginous zone developed, delaying calcification. This irregular cartilage plate during the progressive stage of bone growth gave rise to the bow shape of the ulna shaft. As a firm anatomical connection exists between the radius and ulna, the curvature of the radius was secondary to the ulna deformity. Similarly, the positional changes of the paw and carpus were secondary to the ulna lesion3. No other skeletal changes or signs of dwarfism were described. Various breeding tests indicated an autosomal recessive mode of inheritance. Serum and bone calcium, phosphorus and haematological examinations were performed as well as a comprehensive study of the lesion progression both radiologically and histologically3.
As this is to the authors' knowledge the 1st report of chondrodysplasia in New Zealand white rabbits in South Africa, the importance and prevalence of this condition are not known. Even though this condition is unlikely to have a major effect on meat production, there are welfare implications that need to be considered in both farming enterprises and laboratory facilities. Heritability is a concern in specified pathogen-free breeding establishments in South Africa owing to the diminishing genetic pool and difficulty in importing new genetic material. Mildly affected animals are generally able to compensate for the deformities with a few housing adjustments and regular welfare monitoring. Severely affected animals may require humane euthanasia.
ACKNOWLEDGEMENTS
The authors thank Mrs I Janse van Rensburg, Ms M Rheeder, Mr H Kgaga, the veterinary technologists and support staff at the UPBRC who were involved in the daily care and management of these animals, and the staff at the Onderstepoort Veterinary Academic Hospital involved in the radiography of these animals.
REFERENCES
1. Lindsey J R, Fox R R 1974 Inherited diseases and variations. In Weisbroth S H, Flatt R E, Kraus A L (eds) Biology of the laboratory rabbit. Academic Press, New York: 377-401 [ Links ]
2. Meredith A, Flecknell P 2006 BSAVA manual of rabbit medicine and surgery (2nd edn). British Small Animal Veterinary Association, Gloucester [ Links ]
3. Pearce L 1960 Hereditary distal foreleg curvature in the rabbit I. Manifestations and course of the bowing deformity: genetic studies. Journal of Experimental Medicine 3: 801-821 [ Links ]
4. Pearce L 1960 Hereditary distal foreleg curvature in the rabbit II. Genetic and pathological aspects. Journal of Experimental Medicine 3: 823-830 [ Links ]
5. Quesenberry K E, Carpenter J W 2004 Ferrets, rabbits and rodents clinical medicine and surgery (2nd edn). Saunders, Philadelphia [ Links ]
6. Rampin F, Piccirillo A, Schiavon E, Poppi L, Grilli G 2008 Detection of pathological lesions in slaughtered rabbits. Italian Journal of Animal Science 7: 105-111 [ Links ]
7. Thompson K 2007 Bones and joints. In Grant Maxie M, JubbKVF (eds) Jubb, Kennedy and Palmer's pathology of domestic animals (5th edn) Vol. 1. Elsevier Saunders, New York: 24-47 [ Links ]
8. Walden N B 1990 The TG Hungerford Vade mecum series for domestic animals. Series C number 13, Rabbits: a compendium. University of Sydney Post Graduate Foundation in Veterinary Science, Sydney [ Links ]
Received: November 2010.
Accepted: June 2011.
* Author for correspondence. E-mail: tamsyn.pulker@up.ac.za














