Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
Journal of the South African Veterinary Association
versión On-line ISSN 2224-9435
versión impresa ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.82 no.3 Pretoria ene. 2011
ARTICLE ARTIKEL
A pilot study of common health problems in smallholder pigs in Angónia and Boane districts, Mozambique
C MatosI; C SitoeI; S AfonsoII; J BanzeI; J BaptistaII; G DiasII; F RodriguesIII; A AtanásioI; A NhamussoI; M-L PenrithI,IV,V,*; A L Willingham IIIVI
IDirecção de Sciencias Animais, Instituto de Investigação Agrícola de Moçambique
IIFaculdade de Veterinária, Universidade Eduardo Mondlane, Maputo, Moçambique
IIIDirecção Nacional de Pecuária, Ministério de Agricultura, Maputo, Mozambique
IVDepartment of Veterinary Tropical Diseases, Faculty of Veterinary Science, University of Pretoria, Private Bag X04, Onderstepoort, 0110 South Africa
VTAD Scientific, Pretoria, South Africa
VIW HO/FAO Collaborating Center for Neglected and Other Parasitic Zoonoses, Section for Parasitology, Health and Development, Department of Veterinary Disease Biology, Faculty of Life Sciences, University of Copenhagen, Dyrlaegevej 100, 1870 Frederiksberg C, Denmark
ABSTRACT
A pilot survey was conducted in 2 districts in Mozambique to determine the most important health problems facing smallholder pig producers. While African swine fever is the most serious disease that affects pigs at all levels of production in Mozambique, it is likely that productivity is reduced by the presence of mange and gastrointestinal parasites, while in traditional systems the conditions are favourable for the development of porcine cysticercosis caused by the pork tapeworm Taenia solium, which poses a health risk to communities. Results of the pilot survey confirmed that, with the exception of African swine fever, ecto- and endoparasites are probably the most important health risks for producers. Porcine cysticercosis is more prevalent among pigs in traditional, free-ranging systems, while mange becomes a serious factor when pigs are permanently confined.
Keywords: African swine fever, Mozambique, parasites, pig, porcine cysticercosis, smallholders.
INTRODUCTION
A pilot project whose objective was to propose ways to improve health and production in smallholder pigs in Mozambique in order to achieve better control of African swine fever (ASF) and porcine cysticercosis and thus improve the income of producers was conducted in 2 districts in Mozambique. Since control of both ASF and cysticercosis requires that pigs should be permanently confined, existing systems were studied in participation with the producers, the problems posed by those systems identified, and the constraints that would be imposed by permanent confinement of pigs considered.
The researchers were confronted by most of the problems inherent in on-farm studies, particularly those involving short-cycle species, and especially by the loss to ASF of pigs that were included in the initial sampling. It was originally planned that the study would include an evaluation of the effect of anti-parasitic treatment during the course of the project, but to carry this out scientifically proved impossible due to off-take of pigs and also the impracticality of using some of the pigs as untreated controls, since this would have been unfair to the owners.
The purpose of this article is to present the results of the pilot study, to compare them with published data available on smallholder pigs elsewhere, and to evaluate the main threats to smallholder pig health based on the findings of the study.
MATERIALS AND METHODS
Localities
The 2 districts chosen were Angónia District in Tete Province and Boane District in Maputo Province (Fig. 1). Angónia District is characterised by pigs farmed in a traditional, free-ranging system in which pigs spend as much time as possible unconfined in order to feed themselves by grazing and scavenging, being confined at most during the season when crops are on the land, and/or at night to protect them from predators. The Boane District is close to the capital city, Maputo, and, while some pigs are still traditionally farmed, many of the producers have adopted a more commercial approach to farming, with an eye on the market offered by the proximity of the city, and pigs are mostly confined. Angónia District is situated in an area where ASF is endemic1,9, while Boane is situated in the area south of the Save River that was traditionally free of ASF but that has since the first incursion of the disease in 1994 been subject to sporadic outbreaks7.
Sample size
The sample size was calculated using the number of pigs in the district based on official figures from the livestock department and the known or estimated prevalence of ASF and porcine cysticercosis in each district. Pigs over 6 months of age were targeted during the initial survey, and pregnant sows and sows with unweaned litters were excluded to avoid stress and loss of piglets. After adjustment of the number of pigs calculated to exclude piglets under 6 months of age and sows as specified above, 49 pigs from Boane were selected and 91 from Angónia. The pigs were identified with numbered ear tags and although few identified pigs were still available to be sampled at the end of the project, samples were taken from piglets from the same herds. Selection of participating farmers (herds) was performed in consultation with the district extension officers. All of the participating farmers volunteered to take part in the project.
Collection of samples
Faecal samples were collected from the rectum into plastic bags and transferred to the laboratory on ice.
Skin scrapings were performed to obtain mange mites; other ectoparasites were collected by forceps.
Blood samples were collected by venipuncture using the jugular, brachial, or ear vein into blood tubes without coagulant and the serum was decanted and used for antibody detection.
Sample processing
Eggs of helminths and oocysts of coccidia were obtained by flotation using a saturated salt solution. The samples were evaluated quantitatively using the McMaster method as described by Thienpont et al.13, and the worm eggs were identified according to the same reference.
Material from skin scrapings was mounted on a glass slide in lactophenol, covered with a cover slip and examined microscopically for mange mites.
Ectoparasites were identified according to Soulsby10 to genus or species level.
Antibodies to ASF were detected using the Indirect ELISA based on the method used at the Onderstepoort Veterinary Institute, South Africa9.
Antibodies to cysts of T. solium were detected by an antibody ELISA using antigen obtained from purified cyst fluid prepared in house according to the method of Ito et al.5
Antiparasitic treatment
Infested pigs were treated at 3-monthly intervals (Boane) or 4-monthly intervals (Angónia) by the application of a dip of which the active ingredient was Amitraz against ectoparasites and injection of ivermectin at the recommended dose for pigs of 1 m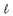 /33 kg against gastrointestinal parasites and immature mange mites. Various brands of these products were used during the project, as no single commercial product was consistently available; for this reason the trade names are omitted.
/33 kg against gastrointestinal parasites and immature mange mites. Various brands of these products were used during the project, as no single commercial product was consistently available; for this reason the trade names are omitted.
Advice to farmers
Participating farmers were offered advice on ways to improve the hygiene of the area in which the pigs were kept and were provided with information during workshops and in the form of posters and pamphlets about parasites, common diseases of pigs including ASF, and good husbandry.
RESULTS
Pig lice (Haematopinus suis) were the ectoparasites found in the highest proportion in both districts, with 44.9 % of pigs in Boane and 59.3 % of pigs in Angónia being infested. Counts were not made, but large numbers of lice were often present, with many eggs visible on the hairs.
Sarcoptic mange was present only in pigs in Boane District, where 25 % of pigs were infected. A small number of pigs in Angónia had fleas (Echidnophaga sp.) (6.6 %) and/or ixodid ticks (11 %). One tick species was identified as a member of the genus Rhipicephalus.
Mixed infections occurred in about 10-12 % of the animals: 5 pigs in Boane (mange + lice) and in 11 pigs in Angónia (lice + ticks on 2 pigs, lice + fleas on 3 pigs, lice + fleas + ticks on 3 pigs).
Gastrointestinal parasites identified by eggs included Ascaris suum, Hyostrongylus rubidus, Oesophagostomum sp., Strongyloides ransomi and Trichuris suis. All were present at both sites, as well as oocysts of coccidia (Table 1).
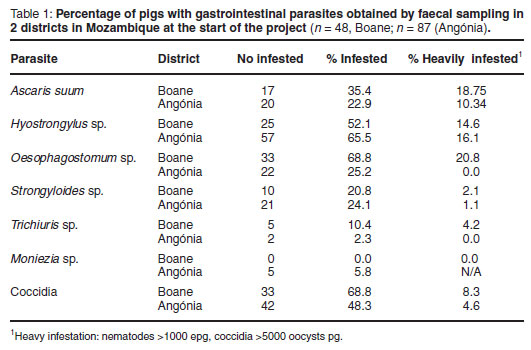
At the end of the project, 37 piglets in Boane from the same herds that had been sampled initially and treated during the project were evaluated for ectoparasites and sampled for gastrointestinal parasites and ASF antibodies. For various reasons, mainly logistic, only 14 pigs were sampled in Angónia, and the results are therefore not presented, but little difference in the proportion of internal parasites was observed. In Boane, the percentage of pigs with lice had dropped to 25 %, but mange had increased to 42.5 %. The percentage of pigs infested was lower for all the nematodes (A. suum 24.3 %, H. rubidus 27 %, Oesophagostomum sp. 37.8 %, Strongyloides sp. 2.7 %, T. suis 2.7 %), and of coccidia slightly lower (56.8 %). The percentage of pigs with heavy infestations was also lower (A. suum 13.5 %, H. rubidus 0%, Oesophagostomum sp. 2.7 %, Strongyloides sp. 0 %, T. suis 0 %, coccidia 2.7 %).
Antibodies to T. solium cysts were found in 1 pig in Boane district (2.1 %, n = 48) and 15 in Angónia district (17.9 %, n = 84).
Initially, none of the pigs from Boane district (n =48) and 3 pigs from Angónia district (3.3 %, n =91) had antibodies to ASF. Of the 14 pigs sampled at the end of the project in Angónia district, 2 (14.3 %) had antibodies, while all of the 37 pigs from Boane were negative.
DISCUSSION
At the start of the project, it was assumed that ASF and porcine cysticercosis were the most serious health problems likely to affect smallholder pigs in Mozambique. Both of these diseases are best controlled by keeping pigs permanently confined6-8, but confining pigs not only places the responsibility of feeding the pigs on the owner, it also poses hygienic problems that, among other things, favour infestation with ecto-and endoparasites. The 2 districts were chosen with a view to comparing the health status of pigs in a completely traditional free-range system with pigs in a semi-urban situation where they are kept confined most of the time. Additionally, it was intended to compare endo-and ectoparasite burdens between treated and untreated pigs at the beginning and the end of the project, but this proved impossible because for ethical reasons it was necessary to treat all the pigs and also because of loss of pigs, mainly due to the ASF outbreaks in Angónia and 1 study site in Boane. However, sampling of piglets from the treated herds at the end of the project suggested that treatment may have been effective in reducing gastrointestinal parasite burdens in Boane, and a general improvement in pig health in that district was reported by extension officers and participating farmers, some of whom had instituted their own treatment programmes.
Outbreaks of ASF occurred in both districts during the life of the project. In Angónia, where the disease is endemic, outbreaks occur at frequent intervals9. Previous serological surveys showed that about 40 % of pigs sampled had antibodies to ASF9, indicating that ASF is endemic in the area, as it is in the adjacent Mchinje district of Malawi3,4. The percentage of positive pigs was much lower in the present survey. This is likely due to a biased age selection, with the probability that most of the pigs sampled were between 6 and 8 months of age, too old for maternal antibodies and possibly born after the last outbreak. An outbreak was reported to have occurred in July/August, about a month after the samples were taken. Reporting of outbreaks in Angónia is sporadic at best, but the local farmers regard the disease as seasonal, suggesting that it occurs at approximately annual intervals, probably because the farming system, which determines when the pigs are confined, is seasonal.
An outbreak of ASF also occurred in Boane from August to October 2004, after an absence of the disease from the provinces south of the Save River since 20017. The effect on affected farmers was devastating, but emergency control measures immediately instituted by the provincial livestock services and supported by the project enabled farmers whose pigs were not yet infected to protect their herds. Subsequent investigation of the virus that included molecular characterisation indicated that it was probably introduced from Cabo Delgado province. This event proved that confining pigs will not protect them from ASF unless additional measures are taken to ensure that they do not come into contact with infected pork or other material contaminated with the virus.
The prevalence of cysticercosis determined for Angónia (17.9 %) corresponds almost exactly with the prevalence found for the Mbuli district in Tanzania by lingual examination of pigs, where the farming system is virtually identical6. That study showed that villages where latrines were used and/or pigs were kept confined had a much lower prevalence of cysticercosis6. In the present study, a questionnaire survey conducted among farmers showed that the percentage of households with latrines was 78.1 % in Boane and 97.5 % in Angónia (G Dias, unpubl. data, 2004/5), but it was generally admitted that latrines are not always used, especially by the children. In Angónia the pigs certainly have access to human faeces over long periods when they are not confined at all, while in Boane the farmers who participated in the study rarely release their pigs intentionally, so that access is greatly reduced. It is also possible that pollution of the area with human faeces is lower in a periurban than a rural situation.
Apart from these 2 conditions, there was no evidence of diseases of any importance that affected the pigs, apart from ecto-and endoparasites. Pig lice were the most prevalent ectoparasites in both districts. No attempt was made to quantify the infestation, but some of the pigs were heavily infested. The most, perhaps only, serious ectoparasite found is the mange mite, Sarcoptes scabiei var. suis. Although only 25 % of the pigs sampled at Boane were infested, some of the infestations were heavy and clearly had an effect on the pigs. While the treatment applied during the project may have contributed to the lower prevalence of pig lice found at the end of the project, it apparently had no effect on the mange. This is not surprising, as it is well known that mange in pigs is not easy to eradicate and requires a targeted programme to break the life cycle of the mites in order to be really effective14. Mange was not found in any of the pigs in Angónia. A small number of pigs in Angónia had fleas, ticks and Moniezia sp., the last 2 probably picked up on pastures shared with cattle and goats.
The following helminth parasites have been reported to infect pigs in Mozambique: Fasciola gigantica, Echinococcus granulosus, Taenia hydatigena, T. solium, A. suum, Ascarops strongylina, Choerostrongylus pudendotectus, Metastrongylus elongatus, Oesophagostomum dentatum, Physocephalus sexalatus, Seteria (Artinema) congolensis and Stephanurus dentatus2. From the parasites listed above, many of which are not gastrointestinal, eggs of only 2 species were found in the present study, namely A. suum and Oesophagostomum sp. However, eggs of other helmiths not listed in the earlier publication2 were found, which suggests the need for new surveys.
Worm burdens were similar in the 2 districts, with the exception of Oesophagostomum sp., which had a much higher prevalence in Boane. The prevalence of H. rubidus and Strongyloides sp. was slightly higher in Angónia, and that of A. suum was higher in Boane. The percentage of pigs with high egg counts was relatively low in both districts, but in particular in Angónia. The pattern agrees with that found for pigs in traditional systems in Cameroon12 and Burkina Faso11. The prevalence of coccidia was higher in Boane, which is not surprising, considering that many of the pigs were kept under wet, unhygienic conditions highly favourable for coccidia. No anticoccidial treatment was applied, but some of the participating farmers succeeded in improving their hygiene on the advice of the researchers, which it is hoped contributed to the lower proportion of infection found in Boane at the end of the project.
There is little doubt that, apart from ASF, which is devastating when it occurs because of the high mortality, nutrition is the most serious problem for the farmers, particularly when their pigs are confined. However, it is believed that by reducing the parasite burden of the pigs, farmers could achieve better productivity, since the pigs would be able to make better use of the food that is available. Improving hygiene of the pigs' environment can contribute significantly to reducing parasite burdens. However, once pigs are permanently or semi-permanently confined, it will be important to develop cost-effective strategies to treat mange, preferably by using a product that is effective against both mange mites and pig lice.
ACKNOWLEDGEMENTS
We would like to express our gratitude to DANIDA for financing the project through the Mozambique-Denmark Cooperation Agreement, and to the Executive Secretary of the Conselho Técnico de Investigação Agrária, Dr Alsácia Atanásio and her staff and the Interim Research Coordinating Committee for their support in all administrative matters. We thank Dr Rosa Costa for her unfailing support in her capacity as Director of the lead institute in Mozambique, as well as the (then) Director of the Veterinary Faculty, Dr Luis Neves, and the (then) Director of Livestock Services, Dr Fernando Songane, for allowing their researchers to participate in the project. Our grateful thanks to the District Directors of Agriculture for Boane and Angónia, the chief livestock officers for Tete and Maputo Provinces, Drs Carlos Santana Afonso and Américo de Conceição, Drs Filipe Vilela and Felicidade Macome of the Estação Zootécnica e Agronómica, Angónia, for their support, and in particular the extension officers (Eugénia dos Santos, Ricardo Jamisse and José Muianga) and all the pig farmers who participated in the project.
REFERENCES
1. Bastos A D S, Penrith M-L, Macome F, Pinto F, Thomson G R 2004 Co-circulation of two genetically distinct viruses in an outbreak of African swine fever in Mozambique: no evidence for individual infection. Veterinary Microbiology 103: 169-182 [ Links ]
2. Cruz e Silva J A 1971. Contribuição para o estudo dos helmintes parasitas dos vertebrados de Moçambique. Memórias da Junta de Investigação do Ultramar No. 61: 1-497 [ Links ]
3. Haresnape J M, LunguSAM, Mamu F D 1987 An updated survey of African swine fever in Malawi. Epidemiology and Infection 99: 723-732 [ Links ]
4. Haresnape J M, Mamu F D 1986 The distribution of ticks of the Ornithodoros moubata complex (Ixodoidea: Argasidae) in Malawi and its relation to African swine fever epizootiology. Journal of Hygiene, Cambridge 96: 535-544 [ Links ]
5. Ito A, Plancarte A, Ma L, Kong Y, Flisser A, Cho S Y, Liu Y H, Kamhawi S, Lightowlers M W, Schantz P M 1998. Novel antigens for neurocysticercosis: simple method for preparation and evaluation for serodiagnosis. American Journal of Tropical Medicine and Hygiene 59: 291-294 [ Links ]
6. Ngowi H A, Kassuku A A, MaedaGEM, Boa M E, Carabin H, Willingham,ALIII 2004 Risk factors for the prevalence of porcine cysticercosis in Mbulu District, Tanzania. Veterinary Parasitology 120: 275-283 [ Links ]
7. Penrith M-L, Lopes Pereira C, Lopes da SilvaMMR, Quembo C, Nhamusso A, Banze J 2007 African swine fever in Mozambique: review, risk factors and considerations for control. Onderstepoort Journal of Veterinary Research 74: 149-160 [ Links ]
8. Penrith M-L, Thomson G R, Bastos A D S 2004 African swine fever. In Coetzer J A W, Tustin R C (eds) Infectious diseases of livestock (2nd edn) Vol. 2. Oxford University Press South Africa, Cape Town: 1087-1119 [ Links ]
9. Penrith M-L, Thomson G R, BastosADS, Phiri O C, Lubisi B A, Botha B, Esterhuysen J, Du Plessis E C, Macome F, Pinto F 2004 An investigation into natural resistance to African swine fever in domestic pigs from an endemic area in southern Africa. Revue scientifique et technique, Office International des épizooties, 23: 665-677 [ Links ]
10. Soulsby E J L 1982 Helminths, arthropods and protozoa of domesticated animals. Baillière Tindall, London [ Links ]
11. Tamboura H H, Banga-Mboko H, Maes D, Youssao I, Traore A, Bayala B, Dembele M A 2006 Prevalence of common gastrointestinal nematode parasites in scavenging pigs of different ages and sexes in Eastern Centre province, Burkina Faso. Onderstepoort Journal of Veterinary Research 73: 53-60 [ Links ]
12. Tchoumboue J, Tong, J C, Awah-Ndukum J 2000 Prevalence of gastro-intestinal parasites of pigs in the Western Highlands of Cameroon. Bulletin of Animal Health and Production in Africa 48: 183-186 [ Links ]
13. Thienpont D, Rochette F, VanparijsOFJ 1979 Diagnosing helminthiasis through coprological examination. Janssen Research Foundation, Beerse [ Links ]
14. Urquhart G M, Amour J, Duncan J L, Dunn A M, Jennings F W 1996 Veterinary parasitology (2nd edn). Blackwell Science Limited, Oxford [ Links ]
Received: May 2011.
Accepted: September 2011.
* Author for correspondence. E-mail: marylouise@vodamail.co.za














