Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Journal of the South African Veterinary Association
On-line version ISSN 2224-9435
Print version ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.82 n.3 Pretoria Jan. 2011
ARTICLE ARTIKEL
The pathology of acute Nolletia gariepina poisoning of cattle
E C Du PlessisI,II,*; L ProzeskyII; C J BothaII
IVETPATH, A Division of Idexx Laboratories, PO Box 12731, Onderstepoort, 0110 South Africa
IIDepartment of Paraclinical Sciences, Faculty of Veterinary Science, University of Pretoria, Private Bag X04, Onderstepoort, 0110 South Africa
ABSTRACT
Toxicity in cattle by the shrub Nolletia gariepina was induced experimentally by intraruminal administration of 3 g/kg dried, milled plant material as a single dose. The animals had to be starved for 24 hours before dosing, as dosing on a full rumen did not induce any signs of toxicity during 5 days of observation and clinical pathology monitoring. Clinical signs were not specific and varied according to the duration (acute versus subacute) of the toxicological process. Clinical pathological parameters indicated renal and to a lesser extent hepatic damage, with raised serum concentrations of urea, creatinine, aspartate aminotransferase (AST) and gamma glutamyl transferase (GGT). Increased urinary sodium and potassium concentration and GGT activity, as well as proteinuria, were evident. Histological and electron microscopic examinations revealed acute renal tubular epithelial cell degeneration and necrosis, especially of the proximal convoluted tubules. Mild hepatocellular degeneration was also noticeable.
Keywords: cattle, clinical pathology, nephrotoxicosis, Nolletia gariepina, pathology, South Africa.
INTRODUCTION
An outbreak of a nephrotoxic syndrome occurred in cattle on 2 farms in the Kalahari sandveld in the Vanzylsrus-Kuruman area of South Africa during the year 2000. Extensive on-site examination and laboratory investigation of the grazing, feed supplements and water on the farms could not incriminate any known nephrotoxin as the cause of the syndrome. Finally a shrub, identified as Nolletia gariepina, which grew extensively on the farms and was heavily grazed during the period in question, was shown to be responsible1,6. This paper describes the pathology and clinical pathology of experimental N. gariepina poisoning in cattle.
MATERIALS AND METHODS
Plant material
Nolletia gariepina plants collected in June 2000 from 1 of the farms involved in the outbreak were transported to the Onderstepoort Veterinary Institute (OVI), where they were dried, milled and stored at 5 ºC until required5. A sample was submitted to the National Botanical Institute, Pretoria for confirmation of identity and a voucher specimen has been lodged there.
Experimental animals
The experimental design was approved by the ethical committee of the OVI.
Four 8-month-old Nguni heifers were used in the experiment. None of them had been exposed previously to N. gariepina and they were all born and raised on the OVI research farm. All animals were identified and individually penned outside on concrete floors at the Toxicology Large Animal Unit of the OVI. The cattle were weighed before commencement of the trial. They were all fitted with rumen canulae under local anaesthesia on the 1st day of the experiment (D 0).
Experimental procedures
A dose of 3 g/kg of dried, milled plant material mixed with 1 ℓ of tap water was administered via the rumen canula to 2 heifers, Cases 1 and 2, on D 9, without prior starvation. The other 2 heifers, Cases 3 and 4 were used as negative controls and received 1 ℓ of tap water only.
Nine days after the 1st dosing (D 18), Case 2 was dosed for a 2nd time with 3 g/kg of plant material after being starved for 24 h. She was euthanased on D 19 (the 1st day after the 2nd dosing). On D 19 a 2nd dose of 3 g/kg of plant material was administered intraruminally to Case 1 after the animal had been starved for 24 h. The animal was euthanased on D 21 of the trial (the 2nd day after the 2nd dosing). Case 3 was dosed with tap water on D 18 and euthanased on D 19 of the trial, on the same day as Case 2. Case 4 was dosed with tap water on D 19 and euthanased on D 21 concomitantly with Case 1.
Clinical signs
The animals were monitored daily for habitus, rectal temperature, pulse, respiration, rumen movements, urination and feed and water intake. Each heifer 's concentrate feed, in the form of pellets (OVI formulated ration) and Eragrostis hay, fed as ad libitum roughage, were weighed and recorded to determine the individual intake per day.
Clinical pathology
Blood was collected from the jugular vein and urine was collected at natural voiding from all 4 heifers on 2 occasions, D 5 and D 7, before the 1st dosing. After the 1st dosing, serum and urine were collected on D 11 and D 13 from all 4 heifers and after the 2nd dosing; the same samples (serum and urine) were collected from Cases 2 and 3 on D 19, on the day of euthanasia. Serum and urine were also collected from Cases 1 and 4 on D 20, 1 day after they were dosed, and serum was collected on D 21. Urine could not be collected on D 21 because Case 1 was anuric.
All serum and urine samples were submitted to the Clinical Pathology Laboratory of the Veterinary Academic Hospital, Onderstepoort. Measurements were determined using the Technicon methods for the RA-1000 Analyzer. Urinalysis and enzyme activity determinations were performed within 1 day after collection. Analysis of the urine with the aid of a dipstick (Uricheck, Rapimed Diagnostics) was performed within 1 h of collection.
The following clinical pathology parameters were determined on the blood and urine samples:
Serum samples
• Serum urea nitrogen (SUN) and creatinine (SC) concentrations, to determine whether renal excretion was affected.
• Sodium and potassium concentrations for use in the tests for fractional clearance of sodium and potassium.
• Serum inorganic phosphate to detect proximal tubular dysfunction
• Gamma glutamyl transferase (GGT) and aspartate aminotransferase (AST) activities were recorded at 25 ºC to screen for possible liver damage.
• Calcium concentration for detection of abnormalities in tubular reabsorption.
Urine samples
• Conventional urine strip test (dipstick), for rapid determination of urinary pH and to screen for the presence of leukocytes, nitrites, protein, glucose, ketones, urobilinogen, bilirubin and blood.
• Macroscopic urine appraisal for colour and clarity.
• Urine sediment examination for casts and cells.
• Urine GGT concentration for detection of renal proximal tubular damage. The analysis was performed immediately after collection because of the poor storage potential of GGT.
• Urine protein for use in determination of the urine protein to creatinine ratio (UP/C) for assessment of glomerular permeability.
• Sodium, potassium and creatinine concentrations to determine fractional clearance of electrolytes and UP/C.
Urinary protein loss
Urine protein to creatinine ratio was used as an alternative to 24-hour collection of urine for determination of protein loss11. The method depends on the principle that creatinine excretion remains fairly constant between 24-hour periods and that the concentrations of both urinary creatinine and urinary protein depend on the urine volume. Protein loss/24 hours (mg/kg/24 h) can be determined from the UP/C ratio4.
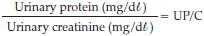
Fractional clearance of electrolytes
Fractional clearance of sodium and potassium were determined in the experimental and control cases from single measurements of the electrolyte and creatinine concentrations in simultaneous urine and blood samples. The ratio quantifies the clearance of the electrolyte in relation to the clearance of endogenous creatinine. The procedure does not require a timed volumetric collection and corrects for variations in water intake7. The urinary creatinine to serum creatinine ratio is used to correct for fluid reabsorption10. The formula used gives the clearance (FC) of an electrolyte (E) as a ratio of creatinine (CR) clearance5,7,10,16. Serum and urinary concentrations of the electrolyte in question are measured in mEq/ℓ , while serum and urinary creatinine concentrations are measured in mg/dℓ . Fractional clearance of the electrolyte is then expressed as a percentage of creatinine clearance5.
| FCE = (urinary E/serum E) | |
| × (serum CR/urinary CR) × 100 | |
Macroscopic pathology
All the experimental animals were euthanased by intravenous injection of an overdose of sodium pentobarbitone (Eutha-Nase, Bayer AH), and necropsies were performed immediately.
Microscopic pathology
Tissue specimens (approximately 10 mm3) from several organs were collected and fixed by immersion in 10 % neutral buffered formalin. Samples included brain, lung (cranioventral and caudodorsal lobes), heart (left and right ventricles, interventricular septum, apex, papillary muscle), thyroid gland, liver, gallbladder, spleen, adrenal gland, kidney (several sections from both kidneys), urinary bladder, mesenteric lymph node, pancreas, alimentary tract (reticulum, rumen, omasum, abomasum, duodenum, jejunum, ileum, caecum, colon), skeletal muscle (semimembranous/semitendinous muscles and diaphragm) and bone marrow. Tissues from all the cases were embedded in paraffin wax, and sections were cut at 5 µm and stained routinely with haematoxylin and eosin (HE).
Electron microscopic examination
Renal and hepatic tissue specimens were collected from all cases within minutes of death, cut into 1 mm3 blocks and fixed in labelled glass vials containing 2.5 % glutaraldehyde in 4 % 0.1 M Millonig's buffer. Samples were routinely processed for electron microscopy, examined using a transmission electron microscope and photographed.
RESULTS
Clinical signs
The negative controls remained healthy throughout the study period, with a healthy appetite and normal water intake.
Neither Cases 1 nor 2 developed clinical signs after the 1st dosing on D 9, which was performed without prior starvation. All clinical parameters remained essentially normal for 9 days.
Case 1 developed clinical signs within 16 h of a 2nd dose administered after 24 h of starvation. Abdominal pain was evident from the tucked-up appearance of the abdomen, tremors of the abdominal muscles and hunched back. The animal was depressed (2+ habitus) and tended to lie down (recumbent). Pale, almost colourless urine was voided. Within 24 h the clinical picture worsened, and the animal could barely rise and exhibited limb and neck weakness. The animal was anorexic and chewed and licked constantly.
Case 2 was recumbent and in shock within 24 h of dosing. Her eyes were severely sunken from dehydration and the extremities were cold. The neck was twisted back with the head resting on the flank. The animal had no interest in the surroundings (stupor) and its feed remained untouched.
Clinical chemistry
The results of the clinical chemistry are summarised in Table 1. After the 1st dosing on D 9, without prior starving of animals, all clinical pathology parameters in all 4 heifers remained constant and within the normal ranges for cattle on 2 occasions (D 11 and 13). Notable abnormalities were only detected on the 1st day after the 2nd dosing (D 19 and 20, respectively), when the heifers were starved prior to the dosing (Table 1).
Urine macroscopic appraisal and dipstick analyses
The urine colour and clarity varied between dark clear yellow to pale clear yellow. In both the control and test animals the pH values were quite variable throughout the trial. With the urinary dipstick no abnormalities could be detected in the 2 control animals throughout the trial period. Both experimental animals had mild to moderate proteinuria on D 19 and 20, respectively, after dosing, approximately equivalent to 100 (Case 1) and 500 (Case 2) mg protein/d . Mild to moderate haematuria was also present, which may have contributed to the proteinuria. Case 1 had a low level of glucose.
Macroscopic pathology
Case 1
The kidneys were light brown in colour and bulged slightly on cut surface. Hepatic lesions included a distended gallbladder and on cut surface the parenchyma had a light brown colour with a slightly orange tint. Gross enlargement of the liver was not apparent. Both the rumen and intestinal tract contained little ingesta, indicating anorexia, and the rumen pH tested low at 4. No urine was present in the urinary bladder.
Case 2
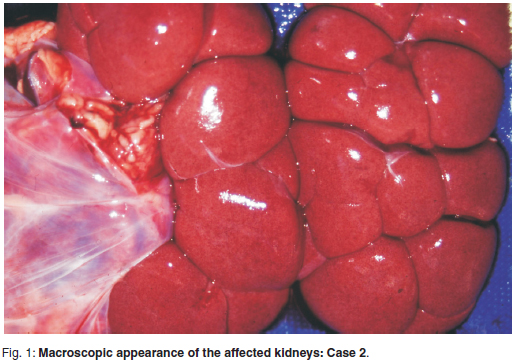
The animal had severely sunken eyes and the skin was inelastic, indicating dehydration of about 3 %. Moderate haemorrhages was visible around both kidneys that were pale brown in colour (Fig. 1) and moderately larger and softer than those of the control animals. Urine in a distended bladder had a normal colour and appearance. A large amount of dark green bile filled the gall bladder. The rumen contained scanty, pasty contents, which had an aromatic smell from the presence of N. gariepina, and the omasum was filled with dry material (stasis and dehydration) whereas the small intestine was empty.
Cases 3 and 4
No lesions were evident in either of the animals and the gastrointestinal tract was filled with normal ingesta.
Microscopic pathology
Kidney
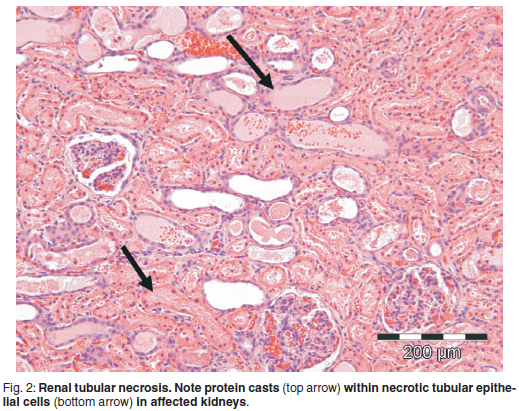
In both experimentally poisoned cattle severe nephrosis was the most striking lesion present. No lesions were visible in the glomeruli. The epithelium of the proximal convoluted tubules (PCT) in the cortex and straight tubules in the cortico-medullary junction was most severely affected. Necrotic epithelial cells showed dehiscence from the basement membrane with the presence of large numbers of protein casts in the tubular lumens (Fig. 2). Single neutrophils were evident in the interstitial tissue between the PCT and mild neutrophilia was evident in the blood vessels, indicating a mild inflammatory reaction in response to the renal tubular necrosis. A few tubules exhibited moderately basophilic renal tubular epithelial cells with large, vesicular nuclei indicative of early regeneration. In the medulla most of the collecting tubules contained eosinophilic hyaline casts, with nuclear remnants from the necrotic cells.
No lesions were present in kidney samples from the control animals.
Liver
Case 1 displayed mild hepatocellular swelling, with accumulation of multiple small vacuoles in the cytoplasm indicating degeneration (sublethal) injury. A few neutrophils were present in the sinusoids. In Case 2 more severe hepatic involvement was evident. Numerous scattered hepatocytes throughout the parenchyma displayed increased eosinophilia of the cytoplasm with the cells rounding off and nuclear karyorrhexis or karyopyknosis. The sinusoids were inapparent because of swelling of the hepatocytes. Here also, a few leukocytes had infiltrated into the sinusoids and central veins. No lesions were visible in the portal tracts and bile ducts.
Spleen
Case 1 revealed haemosiderin-laden macrophages in the red pulp, in association with mild neutrophilia. No lesions were evident in the white pulp follicles.
In Case 2 multifocal areas of karyorrhexis and karyolysis of lymphocytes in the follicular centres were evident. Moderate accumulation of haemosiderin-laden macrophages was evident multifocally in the red pulp sinusoids, which appeared to be relatively lymphocyte-poor. In both the negative control cases the spleen was unremarkable.
Brain
In Cases 1 and 2 moderate dilation of the perivascular spaces was evident diffusely, indicating a moderate amount of brain oedema. No lesions were present in the brain tissue of the negative control cases.
None of the other organs examined revealed any significant lesions.
Electron microscopic examination
Kidney
As expected no lesions were noted in the 2 control animals.
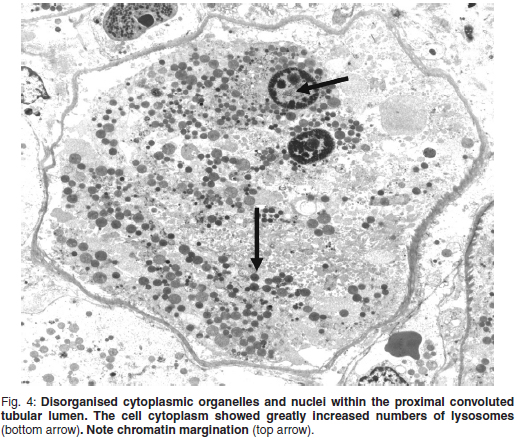
Examination of the glomeruli of the 2 affected animals did not reveal any abnormalities. Severe lesions were, however, evident in the PCT. Most epithelial cells lining the tubules showed loss of the cell membrane, thus dispersing the internal cytoplasmic organelles and nuclei as a disorganised and loose mass within the tubular lumen, which was demarcated by the intact basement membrane (Fig. 3).

The cell cytoplasm showed greatly increased numbers of lysosomes compared with the normal cells in the 2 control animals (Fig. 4). These lysosomes probably represent autolysosomes, as single membranes bound them, and some may have represented residual bodies with undigested electron-dense lipid residues. The normal basal distribution of the mitochondria, with parallel orientation to the cell axis, was lost. Their long, rod-like appearance had changed to a rounded form. Numerous mitochondria contained intramitochondrial inclusions. These were irregular, of medium density and had woolly, filamentous borders, which gave them a flocculant and woolly appearance. Some of them were very electron-dense and the woolly nature was hard to discern. The inclusions are known as flocculant or woolly densities, as well as 'dense matrical deposits' and 'amorphous matrical deposits' and represent an early manifestation of irreversible cell injury8. The distal convoluted tubules lay directly adjacent to the PCT in some fields and were thus easily compared. The epithelial cells were intact and all cytoplasmic organelles and the nuclei appeared minimally affected.
Liver
The hepatic structures in the controls all appeared normal.
Electron microscopic changes in the hepatocytes of the animals that were poisoned were considerably milder than those in the PCT epithelium. Small numbers of ribosomes were distributed throughout the cytoplasm, which indicated a degree of degranulation of the endoplasmic reticulum. Variably sized, single-membrane-bound vacuoles were visible in the cytoplasm of a few cells (Fig. 5). Some of them contained mitochondria and other sequestered material that could not be identified because of the advanced stage of breakdown and degradation, confirming that the vacuoles were autolysosomes.

DISCUSSION
Experimental dosing of cattle with N. gariepina reproduced the nephrotoxic syndrome that was observed in the field cases. The clinical, post-mortem and histological features were similar to those noted in the field cases and confirmed acute renal failure resulting from ingestion of the shruband corroborate the findings of a previous study that described the pathophysiology and pathology in sheep following dosing with of N. gariepina1,6,13. Feed deprivation was confirmed to be a predisposing factor for poisoning to develop. This phenomenon had been noted in the field outbreak, where poisoning only occurred after the animals were penned for 36-48 h and then released into camps where N. gariepina grew abundantly6. Although the plant is highly toxic, poisoning in cattle was not reported on the farms before these outbreaks, nor afterwards, even though the plants remained within the camps grazed by the cattle13.
During the laboratory trial, the 1st dosing on D 9, without prior starvation, did not result in any notable clinical or clinical pathological signs, but dosing after 24 hours' starvation resulted in acute nephrosis. Adequate filling of the rumen thus appears to be an important factor in preventing poisoning. Dilution of the poisonous substance by ruminal contents may play a role, but this hypothesis will have to be examined further.
Both experimental cases were euthanased because of the rapid deterioration in their condition. The toxicity developed very acutely and had a short course. Clinical signs were non-specific and probably related to shock associated with the kidney damage. Various clinical pathological parameters confirmed renal damage associated with a renal tubular lesion, mainly affecting the PCT. Mild or moderate proteinuria was detected and urine protein loss, calculated from the UP/C, increased markedly. Increased urinary protein loss may develop as a result of glomerular leakage or lack of proximal tubular reabsorption or both11,13. In the absence of histological and electron microscopic glomerular lesions, the proteinuria was assumed to result from a loss of protein in the urine as a result of tubular lesions. Proteinuria arising from tubular lesions results from the normal passage of low molecular weight proteins (globulins) through the glomerular filter with defective tubular reabsorption.
Low levels of glucose were present in the urine of 1 case. This can occur in normoglycaemic animals as a result of decreased tubular reabsorption of glucose by diseased renal tubules, as nearly all filtered glucose is reabsorbed within the 1st 20 % of the PCT5.
The presence of large numbers of renal tubular epithelial cells and moderate numbers of granular casts in the urine sediment confirmed severe tubular damage as was noted both light-and electron microscopically.
Increased SUN and SC indicated a reduced glomerular filtration rate (GFR) as a result of accumulation of the glomerular filtrate within the injured and necrotic tubules. This confirmed that the majority of nephrons were non-contributory to the final urine9,10. Both SUN and SC are estimates of GFR. Of the 2, SC is considered more accurate because of the method of renal excretion, although it is not proportionally related to creatinine clearance and GFR because of extrarenal losses of creatinine11. Extrarenal factors involved with urea metabolism, such as rumen urea metabolism and acid-base status, may have been involved in the modest increase of SUN in the experimental animals before they were dosed. Glomerular filtration rate must be reduced to 25 % of normal or less before SUN increases above the normal range.
Increases in fractional clearance of sodium and potassium also indicated tubular damage. Single sample fractional clearance determinations are used to evaluate the role of the renal tubules in the maintenance of homeostasis. Decreased GFR resulting from renal failure leads to an increase in the fractional clearance of some substances in the urine to maintain clinically normal plasma concentrations of these substances for as long as possible. This would explain why the serum electrolyte concentrations, notably sodium and potassium, remained quite stable in the azotaemic experimental animals. Tubular adaptation occurs in the remaining functional nephrons, with increased tubular secretion of both sodium and potassium into the urine. Fractional clearance of sodium (FCNa) can be used to distinguish between acute renal failure (ARF) caused by prerenal lesions and that caused by acute tubular necrosis (ATN). If FCNa stays below 1 %, the cause of ARF is prerenal, while FCNa above1%is seen with ATN10,15. This is because tubular damage leads to reduced sodium reabsorption in the PCT. In prerenal ARF the tubules are intact and reabsorb all filtered sodium. As both sodium and potassium are mainly reabsorbed in the PCT, these results also confirmed damage primarily to the PCT.
Gamma glutamyl transferase excretion in the urine originates from the kidneys2,3,12, and the enzymuria that developed in the test cases indicates damage to the PCT13,14,15. In sheep, mercuric chloride poisoning induces a severe increase in urinary GGT and the analysis can be used in the evaluation of renal damage1.
The histological and electron microscopic lesions confirmed that the PCT is the main site of action of the toxin present in N. gariepina, and that the rest of the nephron, including the glomerulus, is largely unaffected. These examinations also confirmed an intact basement membrane in the affected PCTs, consistent with toxic rather than hypoxic damage3,12. Renal tubular epithelial cell regeneration appeared sooner than has been described previously, with tubular epithelial cell proliferation already evident within 24 h of the toxic insult. Regenerating cells had a basophilic appearance, often encountered in induced nephropathies9.
Histological and clinical pathological investigations also showed mild to moderate, reversible injury to the hepatic parenchyma, which confirmed the observations seen in the field outbreaks. Increased serum AST concentration was indicative of hepatocellular damage11 and although GGT activity is present in many tissues, notable elevation in activity in the serum is primarily observed in liver disease13 and indicates hepatobiliary disease associated with cholistasis11. Mild hepatocellular swelling and degeneration (sublethal injury), present in both cases, are reversible lesions9. They may have developed as a result of primary toxic damage to the hepatocytes, or secondary to hypoxia or metabolic conditions that developed during renal failure.
The electron microscopic changes in the liver sections revealed only a few pathological and non-specific changes, most of which were reversible and mild in nature. Degranulation of the rough endoplasmic reticulum is an early and mild lesion and has been described in hepatocytes exposed to toxic substances such as carbon tetrachloride8. There is evidence that it leads to impaired protein synthesis. An increased number of autolysosomes also indicates sublethal intracellular injury8.
ACKNOWLEDGEMENTS
We wish to express our sincere thanks and appreciation to the following people: The staff of the Toxicology stables at the Onderstepoort Veterinary Institute (OVI) for their help with the experimental animals, especially Ms L Labuschagne and DrJPJ Joubert; the laboratory staff of the Pathology Section at the OVI for technical assistance with post-mortem examinations, as well as processing, sectioning and staining of the histopathological sections; the Electron Microscopy Unit, Faculty of Veterinary Science, for processing of electron microscopy samples; Ms E Myburgh from the Clinical Pathology Laboratory, Faculty of Veterinary Science for chemical analysis of blood and urine samples; Professor F Reyers for advice on the clinical chemistry aspects of the experiment; Professor J Lawrence for editorial assistance; Elma Vorster for invaluable secretarial assistance and Peter Mokonoto for his assistance with the scanner.
REFERENCES
1. Acamovic T, Steward C S, Pennycott T W (eds) 2004 Poisonous plants and related toxins. CABI, Wallingford [ Links ]
2. Braun J P, Benard P, Burgat V, Rico A G 1983 Gamma glutamyl transferase in domestic animals. Veterinary Research Communications 6: 77-90 [ Links ]
3. Confer A W, Panciera R J 2001 The urinary system. In McGavin M D, Carlton, W W, Zachary J F (eds) Thomson's special veterinary pathology (3rd edn). Mosby, St Louis: 235-277 [ Links ]
4. Dibartola S P, Chew D J, Jacobs J 1980 Quantitative urine analysis including 24hour protein excretion in the dog. Journal of the American Animal Hospital Association 16: 537-546 [ Links ]
5. Duncan J R, Prasse K W, Mahaffey E A 1994 Veterinary laboratory medicine: clinical pathology (3rd edn). Iowa State University Press, Ames [ Links ]
6. Du Plessis E C 2004 Pathological investigation of the nephrotoxic effects of the shrub Nolletia gariepina (DC) Mattf. in cattle. MMedVet thesis, University of Pretoria [ Links ]
7. Fleming S A, Hunt E L, Riviere J E, Anderson K L 1991 Renal clearance and fractional excretion of electrolytes over four 6-hour periods in cattle. American Journal of Veterinary Research 52: 5-8 [ Links ]
8. Ghadially F N 1988 Ultrastructural pathology of the cell and matrix (3rd edn). Buttterworths, London [ Links ]
9. Gopinath C, Prentice D E, Lewis D J 1987 Atlas of experimental toxicological pathology. MTP Press, Norwell [ Links ]
10. Jamison R L, Myers B D, Neild G 1997 Acute renal failure. In Jamison R L, Wilkinson R (eds) Nephrology. Chapman & Hall Medical, London: 765-783 [ Links ]
11. Kaneko J J, Harvey J W, Bruss M L 1997 Clinical biochemistry of domestic animals. Academic Press, San Diego [ Links ]
12. Maxie M G 1993 The urinary system, In Jubb K V F, Kennedy P C, Palmer N (eds), Pathology of domestic animals (4th edn) Vol. 2. Academic Press, Philadelphia 425-522 [ Links ]
13. Meintjies R A, Botha C J, Prozesky L 2005 Toxicity, pathophysiology and pathology in sheep following dosing of the nephrotoxic plant Nolletia gariepina (DC) Mattf. Onderstepoort Journal of Veterinary Research 72: 39-53 [ Links ]
14. RaoDST, Joshi H C, Kumar M 1988 Leucine amino peptidase and gamma glutamyl transpeptidase activity in bracken-ferninduced haematuria in calves and rats. Indian Journal of Animal Sciences, 58: 544-547 [ Links ]
15. Stonard M D 1990 Assessment of renal function and damage in animal species. A review of the current approach of the academic, governmental and industrial institutions represented by the Animal Clinical Chemistry Association. Journal of Applied Toxicology 10: 267-274 [ Links ]
16. Whiting P H, Brown P A 1996 The relationship between enzymuria and kidney enzyme activities in experimental gentamycin nephrotoxicity. Renal Failure 18: 899-909 [ Links ]
Received: March 2011.
Accepted: August 2011.
* Author for correspondence. E-mail: liza@idexxsa.co.za














