Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
Journal of the South African Veterinary Association
versão On-line ISSN 2224-9435
versão impressa ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.82 no.2 Pretoria Jan. 2011
ARTICLE ARTIKEL
Piroplasm parasites of white rhinoceroses (Ceratotherium simum) in the Kruger National Park, and their relation to anaemia
D GovenderI,II; M C OosthuisenII; B L PenzhornI,*
IScientific Services, South African National Parks, Private Bag X402, Skukuza, Mpumalanga, 1350 South Africa
IIDepartment of Veterinary Tropical Diseases, Faculty of Veterinary Science, University of Pretoria, Private Bag X04, Onderstepoort, 0110 South Africa
ABSTRACT
As part of a larger survey to map the geographical distribution of Babesia and Theileria parasites in the southern African rhinoceros population, white rhinoceroses were sampled during routine immobilisations in the Kruger National Park. Polymerase chain reaction (PCR) and reverse line blot (RLB) hybridisation assays were used to screen for the presence of piroplasms and complete blood counts were used to assess associated changes in clinical parameters. Of the 195 rhinoceroses sampled, 71 (36.4 %) tested positive for the presence of Theileria bicornis, with no significant change in the haematological parameters measured, while 18 (9.2 %) tested positive for Theileria equi. None of the rhinoceroses sampled tested positive for Babesia bicornis, a parasite associated with mortalities in black rhinoceroses.
Keywords: anaemia, Babesia bicornis, Theileria bicornis, Theileria equi, white rhinoceros.
INTRODUCTION
There have been many records of both Babesia and Theileria spp in wild animals22,27. In most cases these were incidental findings, but piroplasmosis has been implicated in losses amongst wildlife, generally related to stress22. The clinical spectrum of piroplasm infection ranges from asymptomatic infection to death. Parasite replication often produces only mild anaemia with few associated symptoms, but it may also cause severe anaemia with numerous clinical manifestations, including high fever, hypotension, pulmonary oedema, disseminated intravascular coagulation, haemoglobinuria, and multiple organ failure8.
There is a paucity of information on the occurrence of piroplasms in African rhinoceroses in general, but particularly in white rhinoceroses (Ceratotherium simum), where a single published paper could be traced1. This paper reported the results of an ad hoc survey of 106 white rhinoceroses in Zululand (northern KwaZulu-Natal, South Africa) during 1967 to 1969, where basic microscopy revealed a 32.1 % prevalence of a Theileria-like piroplasm and a 1.9 % prevalence of a large Babesia sp1. The latter is of concern as the recently described Babesia bicornis has been associated with fatalities in black rhinoceroses (Diceros bicornis) in East and South Africa2,19. The recently described Theileria bicornis, on the other hand, appears to be apathogenic in black rhinoceroses19.
The southern white rhinoceros (C. s. simum) population in South Africa increased from near extinction in 1900 to more than 11 320 animals at the beginning of the 21st century4,23,24, with over 8000 individuals occurring in the Kruger National Park (KNP) alone. Efforts to restock private reserves within their former range and to strengthen and augment present populations have resulted in a rise in rhinoceros immobilisations and translocations. This may pose a risk, however, as little is known about susceptibility of rhinoceroses to various infections and the role that translocations may play in the epidemiology of disease5. Stress associated with capture, confinement in bomas, extreme environmental conditions, pregnancy and poor nutrition can all increase the possibility of opportunistic infections and recrudescence of latent infections11,15,16,17.
Changes to the haematological picture in babesiosis can be quite varied, with anaemia, leukopenia or leukocytosis and/or thrombocytopenia all being recorded29. The haemolytic anaemia associated with piroplasm infection is thought to be induced by trophozoite intra-erythrocyte binary fission, secondary immune-mediated haemolytic anaemia, oxidative damage to erythrocytes3,18,21 and the presence of a haemolytic factor in serum, which has been described in Babesia gibsoni infection20.
Routine immobilisation of white rhinoceroses in KNP for management purposes offered the opportunity to determine the prevalence of Babesia and Theileria spp. in their blood, using the reverse line blot (RLB) hybridisation assay, and to evaluate whether these piroplasms, when present, are pathogenic to their host (as manifested by development of anaemia).
MATERIALS AND METHODS
Study area
The study was conducted on animals selected in the southern Morula Section of the KNP, South Africa (24º0'41"S 31º29'7"E) bordered by the Sabie and Crocodile Rivers. This area represents the highest white rhinoceros density in KNP (Fig. 1.)

Sample collection
White rhinoceroses (n = 195) were selected randomly during routine immobilisation between January 2003 and September 2006. These included both males and females of all age classes, a stratified sample representing the age and sex class ratios found in the KNP at the time of the project. Blood was collected from the auricular vein, using 6 m Vacutainer® (Franklin Lakes, USA) tubes with EDTA anticoagulant, once the animal was immobilised and in lateral recumbency. Blood tubes were then labelled with the animal-specific details as well as date and GPS location. Specimens were placed in a cooler box and transported to the laboratory.
Vacutainer® (Franklin Lakes, USA) tubes with EDTA anticoagulant, once the animal was immobilised and in lateral recumbency. Blood tubes were then labelled with the animal-specific details as well as date and GPS location. Specimens were placed in a cooler box and transported to the laboratory.
Sample processing
At the laboratory, the blood tubes were placed on a roller for c. 5 min or until the contents were properly mixed. Five µ of the sample was run on an automated blood counter machine (ABC Vet) to attain complete blood counts. The remainder of the blood sample was pipetted into 3.7 m
of the sample was run on an automated blood counter machine (ABC Vet) to attain complete blood counts. The remainder of the blood sample was pipetted into 3.7 m NUNC tubes and frozen as EDTA-buffered whole blood in a -10 ºC freezer. Samples were then batched and taken to the Molecular Biology Laboratory, Department of Veterinary Tropical Diseases, Faculty of Veterinary Science, University of Pretoria.
NUNC tubes and frozen as EDTA-buffered whole blood in a -10 ºC freezer. Samples were then batched and taken to the Molecular Biology Laboratory, Department of Veterinary Tropical Diseases, Faculty of Veterinary Science, University of Pretoria.
DNA extraction
DNA was extracted from 200 µ of EDTA-buffered whole blood using the QIAamp® DNA Extraction Kit (Qiagen, Southern Cross Biotechnology, South Africa) following the manufacturer 's instructions.
of EDTA-buffered whole blood using the QIAamp® DNA Extraction Kit (Qiagen, Southern Cross Biotechnology, South Africa) following the manufacturer 's instructions.
PCR
One set of primers was used to amplify a 460-to 520-bp fragment of the 18S SSU rRNA spanning the V4 hypervariable region7,14. The forward primer, RLB-F [5'-GAC ACA GGG AGG TAG TGA CAA G-3'] and the biotin-labelled reverse primer, RLB-R [5'-Biotin-CTA AGA ATT TCA CCT CTA ACA GT-3'], hybridised with regions conserved for Theileria and Babesia spp. Methods described were similar to those followed by Gubbels et al.7, with minor changes. Reaction conditions in a 25 µ volume were as follows: 12.5 µ
volume were as follows: 12.5 µ of Platinum Quantitative PCR Supermix-UDG (Invitrogen), 20 pmol (0.25 µ
of Platinum Quantitative PCR Supermix-UDG (Invitrogen), 20 pmol (0.25 µ ) of both the forward and reverse primers (Inqaba Biotec, South Africa), 2.5 µ
) of both the forward and reverse primers (Inqaba Biotec, South Africa), 2.5 µ of the extracted DNA template and 9.5 µ
of the extracted DNA template and 9.5 µ of molecular grade water. A touch-down PCR programme was followed, starting with 3 min at 37 ºC; 10 min at 94 ºC; and 10 cycles of 94 ºC for 20 s, 67 ºC for 30 s, 72ºC for 30 s with decreasing of the annealing temperature after every 2nd cycle by 2 ºC 5 times. These cycles continued until the annealing temperature reached 57ºC. Finally, 40 cycles of 94 ºC for 20 s; 57 ºC for 30 s and 72 ºC for 30 s were performed. The PCR was completed with a final extension cycle of 7 min at 72 ºC, and left at 4 ºC.
of molecular grade water. A touch-down PCR programme was followed, starting with 3 min at 37 ºC; 10 min at 94 ºC; and 10 cycles of 94 ºC for 20 s, 67 ºC for 30 s, 72ºC for 30 s with decreasing of the annealing temperature after every 2nd cycle by 2 ºC 5 times. These cycles continued until the annealing temperature reached 57ºC. Finally, 40 cycles of 94 ºC for 20 s; 57 ºC for 30 s and 72 ºC for 30 s were performed. The PCR was completed with a final extension cycle of 7 min at 72 ºC, and left at 4 ºC.
RLB hybridisation
Two Theileria/Babesia-specific membranes were used to allow for the maximum number of samples to be run concurrently. Membrane 1 was prepared according to the method described by Gubbels et al.7,25. Membrane 2 was a ready prepared Isogen Life Science membrane (Maarssen, the Netherlands). Both membranes utilised the same probe sequences (Table 1) and probe concentrations. The RLB hybridisation method was adapted from Gubbels et al.7, the only difference being that 10 µ of PCR product was used instead of 40 µ
of PCR product was used instead of 40 µ .
.

Data analysis
The Statistical Analysis System (SAS®) was used to analyse the data. The chi-square test and the Mann-Whitney non-parametric test were used for the statistical comparison between haemoprotozoal prevalence, age and sex class, and season, as well as for the haematology results. The significance level was set at P < 0.05.
RESULTS
Frequencies of infection
Using the RLB hybridisation assay, none of the 195 samples tested positive for the presence of B. bicornis, while a prevalence of 36.4 % was found for T. bicornis (Table 2). There were no significant differences between the various age/sex classes when the 5 groups were compared (Table 2); when compared with the rest of the population, however, the subadult females showed a significantly higher prevalence (χ2 = 5.68; P = 0.01).

There was an unexpected 9.2 % prevalence of Theileria equi in the study population. There appeared to be a significant correlation (χ2 = 19.26; P < 0.0001) between membrane used and detection of these parasites, with the Isogen Life Science membrane detecting higher rates of infection compared with the 'in-house' prepared membrane. All animals positive for T. equi were also positive for T. bicornis; the signal for T. equi, although strong, was significantly weaker than the T. bicornis signal or the T. equi-positive control.
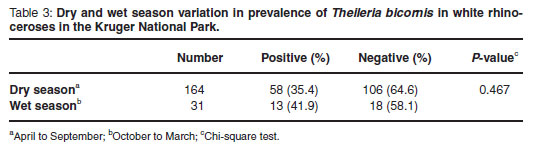
Seasonal differences in prevalence
Monthly and seasonal occurrences of T. bicornis were compared (Fig. 2 and Table 3). Although the prevalence of infection was higher during the wet season, this did not differ significantly from the prevalence during the dry season.

Blood profiles
Haematology values are shown in Table 4. There was no significant change to the blood profile with T. bicornis infection, i.e. no significant change in haematocrit, red blood corpuscles (RBCs), mean corpuscular volume (MCV), mean corpuscular haemoglobin (MCH), mean corpuscular haemoglobin concentration (MCHC) or platelets (PLTs) (Table 4). There was a slight elevation in white blood corpuscles count (WBC) (leucocytosis) in infected animals, but the difference was not significant (P = 0.068; Mann-Whitney non-parametric test).
DISCUSSION
The prevalence of T. bicornis in the KNP white rhinoceros population is similar to that reported in Zululand (northern part of KwaZulu-Natal)1. The RLB hybridisation assay was not able to detect B. bicornis in any of the samples. These could be either true negative results or due to a very low B. bicornis parasitaemia which was below the detection limit of the test. Blood smears of a debilitated black rhinoceros in KNP showed erythrocytes parasitised by large Babesias (R Bengis, Skukuza State Veterinary Services, pers. comm., 2008). The 2 rhinoceros species in the KNP are hosts to the same tick species10, suggesting that B. bicornis could be expected to occur in both rhinoceros species.
Given the density of white rhinoceroses in the KNP, a concerted effort was made to select only healthy, nutritionally unstressed animals for translocation. This may have biased our results and account for the absence of B. bicornis in the study population.
The complete blood-count parameters that were measured did not show significant changes, although there was an indication of an increase in white blood cells in infected rhinoceroses. Anaemia, although sometimes seen as a classical early detection sign for babesiosis, was not detected in animals infected with T. bicornis.
The subadult female proportion of the population showed a significantly higher rate of infection with T. bicornis compared with the rest of the population. Subadult females are most likely the segment of the population undergoing the most dramatic changes: they are becoming reproductively active, participate in courtship and mating and are often early pregnant when captured. This may result in high stress levels and a hormonal milieu that may induce immune suppression, making these animals more susceptible to haemoparasites6,26,28.
A 9.2 % prevalence of T. equi was seen in the study population. Only animals positive for T. bicornis were positive for T. equi and the RLB signal, although strong, was significantly weaker than the T. bicornis signal or the T. equi-positive control. This could suggest possible cross reaction of the T. equi probe with the T. bicornis amplicons. When investigating the T. equi RLB probe region within the V4 hypervariable area, it was shown that T. equi probe differed by 8 base nucleotides which, under normal circumstances, should be sufficient to prevent hybridisation of the probe and the PCR amplicons. If the temperature of the hybridisation was not optimal, however, some crossreaction may have been possible. It should also be kept in mind that it has previously been shown with 18S rRNA gene sequencing and phylogenetic analysis that T. bicornis clustered closely with T. equi9,19. Sequencing and cloning of the parasite 18S rRNA gene of the samples in question will have to be done to clarify the matter.
The apparent lack of seasonal variation in T. bicornis infection, although it might be expected due to the seasonality of their tick vectors, might be explained by the fact that these parasites are known to cause chronic asymptomatic infection that can persist for months or years12. Studies of Babesia suggest that the parasite-induced erythrocyte membrane antigen variability and cytoadherence result in long-term asymptomatic infection of the vertebrate host, increasing the probability of transmission to arthropod vectors and to new hosts, thereby assuring pathogen survival13.
CONCLUSION
Although none of the rhinoceroses sampled tested positive for B. bicornis, a parasite associated with mortalities in black rhinoceroses, this study confirmed a relatively high prevalence of T. bicornis in the white rhinoceros population in the KNP. It also revealed that T. bicornis did not seem to be detrimental to the infected animals. This could represent an endemically stable situation, where coevolution of parasite and host has been pushed to the degree where long-term asymptomatic infection ensures parasite survival and persistence in the environment.
ACKNOWLEDGEMENTS
We thank the National Research Foundation (though grant GUN 65022 to B L Penzhorn) and the Wildlife Group of the South African Veterinary Association for funding this research; SANParks for laboratory support and access to biological samples; Miss Jenny Joubert and Mr Khosi Maseko for sample collection and storage; Mrs Rina Owen for assisting with the statistical analysis; Dr Raksha Bhoora and Dr Kgomotso Sibeko for assisting with the analysis of samples. This project (V045/07) was approved by the Research Committee of the Faculty of Veterinary Science and the Animal Use and Care Committee of the University of Pretoria.
REFERENCES
1. Bigalke R D, Keep M E, Keep P J, Schoeman J H 1970 A large Babesia sp. and a Theilerialike piroplasm of the square-lipped rhinoceros. Journal of the South African Veterinary Medical Association 41: 292-294 [ Links ]
2. Brocklesby D W 1967 A Babesia species of the black rhinoceros. Veterinary Record 80: 484 [ Links ]
3. de Gopegui R R, Peñalba B, Goicoa A, Espada Y, Fidalgo L E, Espino L 2007 Clinico-pathological findings and coagulation disorders in 45 cases of canine babesiosis in Spain. Veterinary Journal 174: 129-132 [ Links ]
4. Emslie R 2004 Rhino population sizes and trends. Pachyderm 37: 107-110 [ Links ]
5. Fischer-Tenhagen C, Hamblin C, Quandt S, Frölich K 2000 Serosurvey for selected infectious disease agents in free-ranging white and black rhinoceros in Africa. Journal of Wildlife Diseases 36: 316-323 [ Links ]
6. Glaser R, Kiecolt-Glaser J K 2005 Stressinduced immune dysfunction: implications for health. Nature Reviews, Immunology 5: 243-251 [ Links ]
7. Gubbels J M, de Vos A P, van der Weide, Viseras M J, Schouls L M, de Vries E, Jongejan F 1999 Simultaneous detection of bovine Theileria and Babesia species by reverse line blot hybridization. Journal of Clinical Microbiology 37:1782-1789 [ Links ]
8. Hunfeld K P, Hildebrandt A, Gray J S 2008 Babesiosis: recent insights into an ancient disease. International Journal for Parasitology 38: 1219-1237 [ Links ]
9. Katzer F, McKellar S, Kirvar E, Shiels B 1998 Phylogenetic analysis of Theileria and Babesia equi in relation to the establishment of parasite populations within novel host species and the development of diagnostic tests. Molecular and Biochemical Parasitology 95: 33-44 [ Links ]
10. Knapp S E, Krecek R C, Horak I G, Penzhorn B L 1997 Helminths and arthropods of black and white rhinoceroses in southern Africa. Journal of Wildlife Diseases 33: 492-502 [ Links ]
11. Kock N D, Jongejan F, Kock M D, Kock R A, Morkel P 1992 Serological evidence for Cowdria ruminantium infection in freeranging black (Diceros bicornis) and white (Ceratotherium simum) rhinoceroses in Zimbabwe. Journal of Zoo and Wildlife Medicine 23: 409-413 [ Links ]
12. Krause P J 2000 Babesiosis. Seminars in Pediatric Infectious Diseases 11: 182-188 [ Links ]
13. Krause P J, Daily J, Telford S R, Vannier E, Lantos P, Spielman A 2007 Shared features in the pathobiology of babesiosis and malaria. Trends in Parasitology 23: 605-610 [ Links ]
14. Matjila P T, Penzhorn B L, Bekker C P, Nijhof A M, Jongejan F 2004 Confirmation of occurrence of Babesia canis vogeli in domestic dogs in South Africa. Veterinary Parasitology 122: 119-125 [ Links ]
15. McCulloch B, Achard P L 1969 Mortalities associated with capture, translocation, trade and exhibition of black rhinoceroses. International Zoo Yearbook 9: 184-195 [ Links ]
16. Mihok S, Kock R, Masake R 1995 Health implications of translocations of endangered species in Africa: trypanosomiasis in rhinoceros. Proceedings of the Joint Conference of the AAZV/WDA/AAWA: 372-373 [ Links ]
17. Miller R E, Boever W J 1982 Fatal hemolytic anemia in the black rhinoceros: case report and a survey. Journal of the American Veterinary Medicine Association 181: 1228-1231 [ Links ]
18. Murase T, Ueda T, Yamato O, Tajima M, Maede Y 1996 Oxidative damage and enhanced erythrophagocytosis in canine erythrocytes infected with Babesia gibsoni. Journal of Veterinary Medical Science 58: 259-261 [ Links ]
19. Nijhof A M, Penzhorn B L, Lynen G, Mollel J O, Morkel P, Bekker C P, Jongejan F 2003 Babesia bicornis sp. n. and Theileria bicornis sp. n.: tick-borne parasites associated with mortality in the black rhinoceros (Diceros bicornis). Journal of Clinical Microbiology 41: 2249-2254 [ Links ]
20. Onishi T, Ueda K, Horie M, Kajikawa T, Ohishi I 1990 Serum hemolytic activity in dogs infected with Babesia gibsoni. Journal of Parasitology 76: 564-567 [ Links ]
21. Orinda G O, Commins M A, Waltisbuhl D J, Goodger B V, Wright I G 1994 A study of autoantibodies to phosphatidyl-serine in Babesia bovis and Babesia bigemina infections in cattle. Veterinary Immunology and lmmunopathology 40: 275-281 [ Links ]
22. Penzhorn B L 2006 Babesiosis of wild carnivores and ungulates. Veterinary Parasitology 138: 11-21 [ Links ]
23. Pienaar D J 1994 Kruger's diversity enriched. Custos May: 22-25 [ Links ]
24. Pienaar U de V 1970 The recolonisation history of the square-lipped (white) rhinoceros Ceratotherium simum simum (Burchell) in the Kruger National Park (October 1961 - November 1969). Koedoe 13: 157-169 [ Links ]
25. Schnittger L, Yin H, Qi B, Gubbels M J, Beyer D, Niemann S, Jongejan F, Ahmed J S 2004 Simultaneous detection and differentiation of Theileria and Babesia parasites infecting small ruminants by reverse line blotting. Parasitology Research 92: 189-196 [ Links ]
26. Serafeim A, Gordon J 2001 The immune system gets nervous. Current Opinion in Pharmacology 1: 398-403 [ Links ]
27. Uilenberg G 2006 Babesia - A historical overview. Veterinary Parasitology 138: 3-10 [ Links ]
28. Zhang Y, Zhang Y, Miao J, Hanley G, Stuart C, Sun X, Chen T, Yin D 2008 Chronic restraint stress promotes immune suppression through Toll-like receptor 4-mediated phosphoinositide 3-kinase signaling. Journal of Neuroimmunology 204: 13-19 [ Links ]
29. Zobba R, Ardu M, Niccolini S, Chessa B, Manna L, Cocco R, Pinna Parpaglia M L 2008 Clinical and laboratory findings in equine piroplasmosis. Journal of Equine Veterinary Science 28: 301-308 [ Links ]
Received: January 2011.
Accepted: March 2011.
* Author for correspondence. E-mail: banie.penzhorn@up.ac.za














