Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Journal of the South African Veterinary Association
On-line version ISSN 2224-9435
Print version ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.82 n.2 Pretoria Jan. 2011
SHORT COMMUNICATION KORT MEDEDELING
Variability of yellow tulp (Moraea pallida Bak.) toxicity
L D Snyman*; R A Schultz*; H van den Berg*
Section of Toxicology, ARC-Onderstepoort Veterinary Institute, Private Bag X05, Onderstepoort, 0110 South Africa
ABSTRACT
Yellow tulp (Moraea pallida Bak.), collected predominantly during the flowering stage from a number of sites in South Africa, showed large variation in digoxin equivalent values, indicating variability in yellow tulp toxicity. Very low values were recorded for tulp collected from certain sites in the Northern Cape.
Keywords: digoxin equivalent, Moraea pallida, toxicity, yellow tulp.
Poisoning with cardiac glycoside-containing plants, of which yellow tulp (Moraea pallida Bak.) (Fig. 1) is the most important member, is collectively the most important plant-associated poisoning of livestock in southern Africa2. The cardiovascular, gastrointestinal, nervous, and respiratory systems are affected1,2, with posterior paresis as the most usual and outstanding nervous sign5. Yellow tulp is widely distributed and occurs under a variety of climatic conditions, topographical situations and soil types in most provinces of South Africa as well as in Botswana1 and Namibia (Fig. 2). In the present study, fluorescent polarisation immuno-assay3 analyses of yellow tulp, collected predominantly during the flowering stage from a number of sites in the country, indicate large variation in toxicity The toxin concentrations, expressed as digoxin equivalents, are indicated in Table 1.

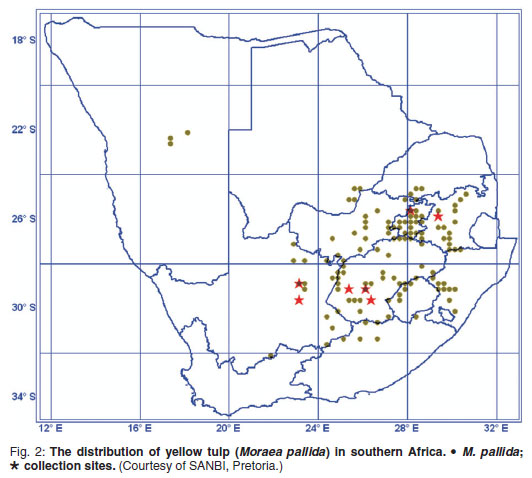
The concentrations of digoxin equivalents for yellow tulp, recorded at the Onderstepoort Veterinary Institute from an artificially established, irrigated, grass and tulp plot and from a site at Middelburg, Mpumalanga, were high while those collected from the majority of sites in the Griekwastad district were very low. This might explain the apparent absence of yellow tulp poisoning in the Northern Cape2. The reason for the large variation in toxicity among the different collections is unknown. Identification of the factors responsible for the variation might be useful in predicting yellow tulp poisoning under various conditions.
The low toxicity of yellow tulp collected from the Griekwastad district was exploited for easy preparation of an identification (ID) factor, used to avert cattle to yellow tulp6. Conditioned aversion to some toxic plants has been successfully induced in livestock by the oral administration of an ID factor together with the aversion factor for that particular plant - in the case of yellow tulp this aversion factor is the toxic principle, epoxyscillirosidin1,4,5. The extremely low toxicity of the yellow tulp collected at Site 4 was exploited by preparing ID factors from this - instead of the local tulp for use in areas such as the Highveld where M. pallida is very toxic. The simplified method for extracting the ID factor6 allows for some contamination with the aversion factor (epoxyscillirosidin); which is potentially dangerous, as the ID factor is administered in conjunction with the toxic aversion factor (epoxyscillirosidin) during the process of inducing aversion. Using relatively less toxic tulp for preparing the ID factor could therefore significantly reduce the risk of inadvertent poisoning during the aversion process.
ACKNOWLEDGEMENTS
The South African National Biodiversity Institute is thanked for the use of data from the National Herbarium, Pretoria Computerised Information System (PRECIS). Funding was provided by the Gauteng Department of Agriculture Conservation and Environment through the Directorate: Technology Development and Support and the North West Province (Department of Agriculture, Conservation, Environment and Tourism).
REFERENCES
1. Kellerman T S, Coetzer J A W, Naudé T W, Botha C J 2005 Plant poisonings and mycotoxicoses of livestock in southern Africa (2nd edn). Oxford University Press, Cape Town [ Links ]
2. Kellerman T S, Naudé T W, Fourie N 1996 The distribution, diagnoses and estimated economic impact of pant poisonings and mycotoxicoses in South Arica. Onderstepoort Journal of Veterinary Research 63: 65-90 [ Links ]
3. Schultz R A, Kellerman T S, van den Berg H 2005 The role of fluorescence polarisation immuno-assay in the diagnosis of plantinduced cardiac glycoside poisoning of livestock in South Africa. Onderstepoort Journal of Veterinary Research 72: 189-201 [ Links ]
4. Snyman L D, Kellerman T S, Schultz R A, Joubert J P J, Basson K M, Labuschagne L 2004 Conditioned feed aversion as a means of preventing intake of yellow tulp (Homeria pallida) by livestock. In Acamovic T, Steward C S, Pennycott T W (eds) Poisonous plants and related toxins. CABI Publishing, Wallingford, Oxon, UK: 531-539 [ Links ]
5. Snyman L D, Schultz R A, Joubert J P J, Botha C J, Labuschagne L 2009 Evaluation of activated charcoal as treatment for yellow tulp poisoning in cattle. Journal of the South African Veterinary Association 80: 274-275 [ Links ]
6 Snyman L D, Schultz R A, Labuschagne L 2011 Amended method of averting cattle to yellow tulp (Moraea pallida). In Riet-Correa F, Pfister J, Schild A L, Wierenga T (eds) Poisoning by plants, mycotoxins and related toxins. CAB International, Wallingford, Oxon, UK. Proceedings of the 8th International Symposium on Poisonous Plants, João Pessoa, Paraíba, Brazil, 4-8 May 2009 (in press) [ Links ]
Received: February 2011.
Accepted: April 2011.
* Authors for correspondence. E-mail: snyman@oranet.co.za / schultza@arc.agric.za / vandenbergh@arc.agric.za














