Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Journal of the South African Veterinary Association
On-line version ISSN 2224-9435
Print version ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.82 n.2 Pretoria Jan. 2011
CLINICAL COMMUNICATION KLINIESE MEDEDELING
Nicotian glauca poisoning in ostriches (Struthio camelus)
C J BothaI,*; P A SteenkampII; A OlivierIII; L C BekkerI
IDepartment of Paraclinical Sciences, Faculty of Veterinary Science, University of Pretoria, Private Bag X04, Onderstepoort, 0110 South Africa
IICouncil for Scientific and Industrial Research, Biosciences, Private Bag X2, Modderfontein, 1645 South Africa
IIIOstrich Research Laboratory, PO Box 241, Oudtshoorn, 6620 South Africa
ABSTRACT
Putative Nicotiana glauca (wild tobacco) poisoning was diagnosed in a flock of ostriches near Oudtshoorn, South Africa. Post mortem examinations (n = 7) were performed on ostriches (Struthio camelus) that had died. Suspicious leaf remnants (weighing 80-770 g), packed in a layer on top of other plant material, were carefully separated from the proventricular content and submitted for chemical determination of anabasine, the major toxic principle contained by this plant. A standard solid phase extraction method was used followed by an optimised liquid chromatography-mass spectrometry procedure. Anabasine was detected in the leaf remnants (114-177 µg/g dry weight) removed from the proventriculus of the ostriches that succumbed as well as in control N. glauca leaves (193 µg/g dry weight). The analytical methods used in this study revealed the presence of anabasine in the suspicious leaf remnants, indicating that the birds had been exposed to N. glauca and had died of this poisoning.
Keywords: anabasine, liquid chromatography-mass spectrometry, Nicotiana glauca, ostriches, poisoning, tree tobacco, wild tobacco.
INTRODUCTION
Nicotiana glauca (wild tobacco, tree tobacco), a member of the Solanaceae family, is a slender shrub or a small erect tree growing up to 5 m high with long, lax branches8 (Fig. 1). This plant is a native of Argentina that has become widespread in South Africa, occurring mostly on disturbed soil such as old lands, trampled areas and road verges8. It is also a common weed along inland rivers and is occasionally even found as an ornamental in gardens3,8.

Although N. glauca is a very poisonous plant, it is seldom incriminated in poisoning of livestock in South Africa and is therefore considered to be mainly of academic interest3. Nevertheless, if sufficient amounts of this plant are ingested by stock, poisoning and even death can result3,5. Mortalities have been reported in ostriches (Struthio camelus)2,7. Since 3 reports at the turn of the previous century7 there has been a dearth of scientific literature on outbreaks in ostriches. In 1903, Hutcheon2 reported that farmers implicated wild tobacco as a cause of death in their birds. He described the following symptoms: 'staggering gait, spasmodic contractions of the voluntary muscles followed by stupor, the birds sit down and throw their heads about, finally, the neck is doubled right back and the head laid on the body; death being due to coma'.
Nicotiana glauca contains the piperidinepyridine alkaloid, anabasine, which is closely related to nicotine, derived from N. tabacum1. Anabasine acts as an agonist at peripheral nicotinic receptors. The nicotinic receptors of the autonomic ganglia and the neuromuscular junctions are initially stimulated, but this is invariably followed by prolonged blocking. Strong stimulation of the central nervous system also occurs resulting in tremors followed by convulsions. Death is ascribed to respiratory paralysis3,4.
CASE HISTORY
During October 2007 a flock of ostriches, 9-10 months old and weighing 70-80 kg, on a farm near Oudtshoorn, in the Little Karoo of South Africa, was moved to a lucerne pasture during the morning. Two dead birds were noticed 2-3 h later. On closer inspection a further 7 birds were either affected or dead. The affected birds exhibited ataxia and torticollis (neck bent downwards) and succumbed within 2-3 h. The rest of the flock was removed from the pasture and no further mortalities occurred. On post mortem examination no gross lesions were noticed, except excessive mucus in the oral cavity and at the choanae. The proventriculus contained broadly ovate leaves, which appeared to be wild tobacco leaves, packed in a layer on top of other plant material. In 7 birds the suspicious leaf remnants were carefully separated from the rest of the proventricular content, weighed and submitted for chemical analysis to determine the presence of anabasine. The detection of this alkaloid would confirm exposure of the birds to N. glauca and explain the mortalities encountered.
Chemical analysis
Extraction of proventricular samples and plant material
The proventricular leaf samples, and 1 plant sample (N. glauca leaves) collected in the Douw Steyn Poisonous Plant Garden, Faculty of Veterinary Science at Onderstepoort and included as a positive control, were dried separately at 40 ºC to a constant weight in an oven (Labotec, Midrand, South Africa). The dried samples were finely milled using a grinder equipped witha1mm sieve (MF10 Basic, Ika-Werke, Staufen, Germany), followed by addition of 25 mℓ 0.02 M sulphuric acid (Merck, Darmstadt, Germany) to 3 g milled material. The extractions were performed at 40 ºC for 30 min in a shaking water bath (Labotec, Midrand, South Africa). The plant material was filtered and re-extracted with 25 mℓ 0.02 M sulphuric acid. The aqueous extracts were combined and the pH of each sample adjusted to approximately 9.5 with 25 % ammonia solution (Merck, Darmstadt, Germany)6.
Solid phase extraction (SPE )
The aqueous extract of each sample was passed through a SPE cartridge (Oasis HLB, Waters, Milford, USA). The absorbed compounds were eluted from the solid phase material with 1.5 mℓ methanol (Merck, Darmstadt, Germany). The eluates were dried under a stream of nitrogen at 40 ºC and stored at -20 ºC until reconstitution with 1ℓ m ultra-pure water (HPLC grade (20 MΩ) obtained from a Millipore Elix 5/Advantage 10 water-conditioning system, Millipore, USA) for liquid chromatograhy-mass spectrometry (LC-MS) analysis.
Quantitative LC-MS analysis
Aliquotes of standard solutions of anabasine ((±)-Anabasine (90 %), Sigma-Aldrich, St Louis, USA) and the crude extracts were analysed using a Waters Thermabeam system comprising a 2695 Solvent Delivery System, a 2996 photodiode array (PDA) detector, column heater and Thermabeam (TMD) electron ionisation mass spectrometry (EIMS) detector (Waters, Milford, USA). Chromatographic separation was achieved with a 250 × 2 mm 110 Å, 5 µm, Gemini C18 column (Phenomenex, Torrance, USA) maintained at 40 ºC. The starting eluent consisted of ultra-pure water (containing 10 mM formic acid; 98 %, Sigma-Aldrich, St Louis, USA) and acetonitrile (SpS (Super Purity Solvent), Romil Ltd, Cambridge, UK) (95:05) at 0.2 mℓ /min. A gradient chromatographic method was used starting at 95 % Milli-Q water (containing 0.1 % (v/v) formic acid) and 5 % acetonitrile at a flow rate of 0.2 mℓ /min. These conditions were kept stable for 1 min followed by a linear gradient to 10 % water and 90 % acetonitrile at 40 min. This eluent composition was kept constant for 5 min followed by a rapid curve (gradient curve 2) to the initial conditions. The runtime was 60 min.
The PDA detector was placed 1st in line and full scan spectra were collected between 200 and 600 nm at a sample rate of 1 spectrum/s and a resolution of 1.2 nm. The TMD detector was placed after the PDA detector and operated in positive scan mode (50-650 amu) with a gain of 10; collecting 1 spectrum/s. The nebuliser temperature was set at 90 ºC, the expansion region temperature at 80 ºC and the source temperature at 225 ºC. The total volume of post-column eluent was sent to the TMD detector and helium was used as nebulisation gas at 30ℓ/h. The TMD detector was tuned every day prior to starting an analysis run and caffeine was injected as test compound to ensure functionality of the total system.
Single-ion monitoring experiments were completed by selecting the base peak ion in the EI spectrum of anabasine (84 Da) and collecting 1 spectrum/s with a gain setting of 10. The interface and nebuliser settings were as described above. Injection volumes ranged between 1 and 10 µℓ depending on the concentration of the sample, but quantitative work was performed utilising 10 µℓ injection volumes.
RESULTS
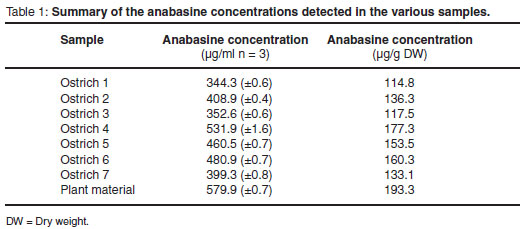
The analyses indicated that all the material tested contained anabasine. The results are reflected in Table 1 and Figs 2-4. They are discussed in more detail below.
DISCUSSION
The published method6 followed to extract anabasine from leaf remnants (weighing 80-770 g) collected from the 7 ostriches as well as N. glauca plant material was successful; however, the analytical procedure had to be optimised for the system utilised. The Gemini C18 column displayed adequate retention for anabasine without compromising peak shape and sensitivity and was used for all analytical work. Owing to the instrumental configuration used, where all the column effluent is sent to the mass detector, the presence of unrelated compounds can lead to a high background noise and a resulting loss of chromatographic resolution and spectral interferences. It was established that by dissolving the samples in water rather than methanol the background noise could be significantly reduced without affecting the response of anabasine.
An array of anabasine standards were used to evaluate the calibration curves produced by using the PDA detector at a fixed wavelength (260 nm) and the mass detector at a single mass ion (84 Da). The extracted control N. glauca plant material was analysed on the LC-MS system using the optimised analytical conditions. The UV result is depicted in Fig. 2 and the MS analysis in Fig. 3. Inserted into these figures are the UV and EI spectra of anabasine, respectively.
The EI spectrum of the compound suspected to be anabasine was submitted to the NIST 2005 mass spectral library (Waters, Milford, USA) and was positively confirmed as anabasine. The match results were 905 in a forward match, 906 in a reversed match with a 81.1 % probability.
The optimised analytical procedure was also utilised for the analysis of the leaf remnants removed from the proventriculus of the 7 ostriches. Anabasine was detected in all the samples (Fig. 4). The analytical results for the proventricular leaf samples indicated the presence of anabasine in large quantities with an average concentration of 141.8 µg/g dry weight. The quantitative analytical results are summarised in Table 1 as derived from the data obtained from the PDA detector.
The utilisation of the NIST 2005 mass spectral library also proved very successful as the search criteria obtained positively identified anabasine as the toxic compound present in the plant material and the proventricular leaf samples of the 7 ostriches.
The analytical results confirmed the ingestion of N. glauca by the ostriches, which, most probably, resulted in the clinical signs and deaths. The acute mortalities following ingestion of N. glauca demonstrate the susceptibility of this ratite species. As far as could be ascertained this is the 1st scientific report of wild tobacco poisoning in ostriches for over a century, which, in addition, is supported by modern analytical procedures. Although gas chromatographymass spectrometry (GC-MS) has previously been used for the definitive diagnosis of N. glauca toxicosis in cattle5, this report describes an optimised LC-MS method to confirm this rare veterinary intoxication.
REFERENCES
1. Cheeke P R 1998 Natural toxicants in feeds, forages and poisonous plants (2nd edn). Interstate Publishers, Danville, Illinois [ Links ]
2. Hutcheon D 1903 The poisoning of stock. Agricultural Journal of the Cape of Good Hope 23: 390-399 [ Links ]
3. Kellerman T S, Coetzer J A W, Naudé T W, Botha C J 2005 Plant poisonings and mycotoxicoses of livestock in southern Africa (2nd edn). Oxford University Press South Africa, Cape Town [ Links ]
4. Manoguerra A S, Freeman D 1982/1983 Acute poisoning from the ingestion of Nicotiana glauca. Journal of Toxicology: Clinical Toxicology 19: 861-864 [ Links ]
5. Plumlee K H, Holstege D M, Blanchard P C, Fiser K M, Galey F D 1993 Nicotiana glauca toxicosis of cattle. Journal of Veterinary Diagnostic Investigation 5: 498-499 [ Links ]
6. Steenkamp P A, van Heerden F R, van Wyk B-E 2002 Accidental fatal poisoning by Nicotiana glauca: identification of anabasine by high performance liquid chromatography/photodiode array/mass spectrometry. Forensic Science International 127: 208-217 [ Links ]
7. Steyn D G 1934 The toxicology of plants in South Africa. Central News Agency, South Africa [ Links ]
6. Vahrmeijer J 1981 Poisonous plants of southern Africa that cause stock losses. Tafelberg Publishers, Cape Town [ Links ]
Received: January 2011.
Accepted: April 2011.
* Author for correspondence. E-mail: christo.botha@up.ac.za














