Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Journal of the South African Veterinary Association
On-line version ISSN 2224-9435
Print version ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.82 n.2 Pretoria Jan. 2011
ARTICLE ARTIKEL
In vitro antimicrobial susceptibility of Staphylococcus aureus strains from dairy herds in KwaZulu-Natal
T Schmidt
Allerton Provincial Veterinary Laboratory, Private Bag X2, Cascades, 3202 South Africa. E-mail: tracy.schmidt@kzndae.gov.za
ABSTRACT
Staphylococcus aureus is 1 of the most important causes of bovine mastitis and is responsible for significant economic losses to the dairy industry worldwide. One of the principal approaches used in treating intramammary infections is the administration of antimicrobials. Due to the propensity of S. aureus to develop resistance, antimicrobial susceptibility monitoring is necessary to ensure that treatment regimens are effective. As part of this investigation, 90 S. aureus strains isolated from mastitis cases submitted to Allerton Provincial Veterinary Laboratory during 2008 and 2009 were evaluated for their susceptibility to a panel of 10 antimicrobials. Only 8 of the 90 S. aureus isolates tested (8.9 %) were found to be susceptible to all of the antimicrobials evaluated. A very high level of resistance to the beta-lactam antibiotics was noted: 47.8 % of the isolates were resistant to penicillin and 65.6 % were resistant to ampicillin. Minimal resistance to oxacillin, cephalothin and trimethoprim-sulfamethoxazole (1.1 %) was found. Seventeen (18.9 %) of the isolates tested were found to be resistant to 3 or more antimicrobials. The need for vigilant monitoring of bacterial resistance trends in the dairy industry is warranted as the potential public health implications are significant.
Keywords: antimicrobial susceptibility, bovine mastitis, Staphylococcus aureus.
INTRODUCTION
Staphylococcus aureus is a formidable pathogen and nowhere is this more evident than in the dairy industry, where considerable losses are incurred annually due to intramammary infections caused by this bacterium11.
The success of S. aureus as a pathogen is due to the variety of strategies the bacterium has evolved which enable it to evade the immune system and counter therapeutic assaults. Once S. aureus has breached the physical barriers of the teat canal, the host's local immune response is challenged by the production of an impressive array of virulence factors that confer protection on the bacteria, enabling them to become established within the udder microenvironment. Enzymes such as hyaluronidase, staphylokinase and proteinases assist tissue invasion, while antiphagocytic factors such as the extracellular polysaccharide capsule compromise the process of phagocytosis, a crucial component of the host's cellular immune response13.
In the event that the invading bacteria are phagocytosed, they are in fact able to survive and even replicate within the phagocyte. The intracellular existence confers protection on the bacteria from the onslaught of the immune response as well as the effects of antimicrobials. With the eventual death of the phagocytic cell the bacteria are released where they are able to induce further damage through the production of multiple haemolysins and other tissue toxins13. In severe cases, S. aureus is able to induce fibrosis and the formation of microabscesses that further aid the bacteria by limiting the penetration of antibiotics into the site of infection17.
Mastitis caused by S. aureus ranges in severity from subclinical to a peracute, gangrenous form. Subclinical mastitis is the most common and likely outcome of a S. aureus infection and it is also the most problematic, as it usually proceeds undetected, constantly eroding profit margins2. The economic losses incurred due to S. aureus infections are difficult to quantify and include, but are not limited, to the following: decreased milk production, reduced milk quality, veterinary and treatment costs, premature culling of cows and consequent loss of genetic potential7.
The successful implementation of a mastitis control programme is therefore imperative. This requires the prompt identification of S. aureus-infected animals before the bacterium has the opportunity to infect other animals in the herd. Management approaches to handling infected animals usually involve segregation, culling or treatment2,16, with the therapeutic approach often being the favoured recourse.
The ability of S. aureus to develop or acquire biochemical strategies which confer resistance to different antimicrobials is an additional tactic in this pathogen's impressive arsenal. One of the diagnostic tools available to practitioners to assist with the selection of an appropriate treatment is the in vitro testing of isolates against a representative panel of antimicrobial drugs. The susceptibility pattern of the implicated strain enables decisive action to be taken by the veterinarian in terms of treatment10,14.This avoids the needless application of ineffective antimicrobials and prevents unnecessary costs from being incurred.
The purpose of this investigation was to evaluate the in vitro susceptibility of S. aureus isolates to different antimicrobial classes that are used in the dairy industry and to use these data to establish the incidence of resistance amongst these pathogens in the KwaZulu-Natal commercial dairy sector.
MATERIALS AND METHODS
Samples
Allerton Provincial Veterinary Laboratory has a large Mastitis Control and Milk Hygiene Section which, for many years, has provided a diagnostic service to the extensive dairy industry in KwaZulu-Natal. Composite and individual quarter milk samples are submitted routinely or on an ad hoc basis by clients as part of their mastitis management plan. The S. aureus strains evaluated during this investigation were isolated during the course of routine diagnostic work carried out at Allerton between January 2008 and December 2009. A total of 90 S. aureus isolates from 60 different commercial dairy herds were evaluated for their susceptibility to different classes of antimicrobials.
Bacteriological culture and isolation
Milk samples were cultured onto Columbia blood agar (Oxoid) supplemented with 5 % sheep blood. Inoculated plates were incubated at 35-37 ºC for approximately 36 h before being examined. All staphylococcal colonies showing yellow pigmentation were tested for coagulase production using diluted rabbit plasma (Bio-Rad) according to the method described by Quinn et al.12. Isolates which tested positive for coagulase production using the overnight tube coagulation test were identified as S. aureus. In the event that multiple S. aureus isolates were cultured and identified in a batch of samples from the same herd, visual inspection of colonies was used to select representative isolates for antimicrobial susceptibility testing.
Antimicrobial susceptibility testing
Antimicrobial susceptibility testing was carried out in accordance with the guidelines published by the Clinical and Laboratory Standards Institute (formerly the National Committee for Clinical Laboratory Standards)3. Briefly, a suspension of each test isolate was prepared in 0.9 % physiological saline to a turbidity equivalent to a 0.5 McFarland standard. Each suspension was streaked onto Mueller Hinton Agar (Oxoid) following which antimicrobial discs (Oxoid) were positioned onto the plates. The panel of antimicrobials tested was selected in such a way as to ensure that each of the classes of antimicrobials available as mastitis remedies was represented. Selection was also to a certain extent restricted by the availability of published interpretive data. The panel of antimicrobials tested is summarised in Table 1.
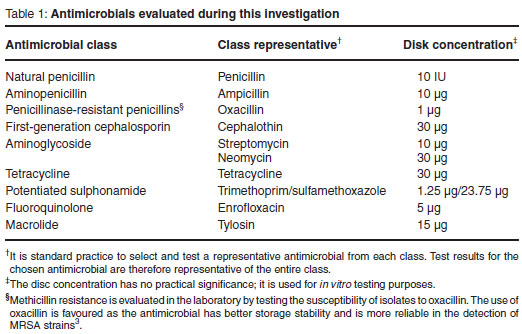
Inoculated plates were incubated at 35-37 ºC for 24 h (±2 h). The zones of inhibition were measured to the nearest millimetre and compared with published CLSI interpretative data. The results were reported as susceptible, intermediate or resistant. Test performance was monitored by using a S. aureus reference strain, ATCC 25923.
RESULTS
The susceptibility data of the 90 S. aureus isolates evaluated during the assessment period are summarised in Table 2, while the percentage of isolates showing resistance to the different antimicrobials tested is graphically depicted in Fig. 1.
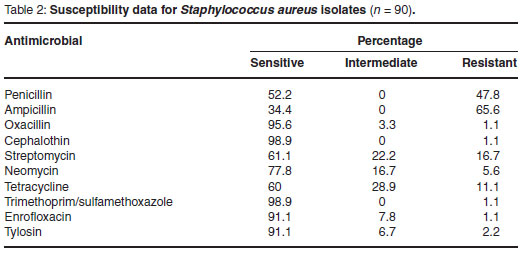

Only 8 of the 90 S. aureus isolates tested (8.9 %) were found to be susceptible to all of the antimicrobials evaluated. Overall the greatest degree of resistance was observed to the beta-lactam antibiotics penicillin (47.8 %) and ampicillin (65.6 %). Minimal resistance (1.1 %) to oxacillin, cephalothin and trimethoprim/sulfamethoxazole was observed.
Seventeen (18.9 %) of the isolates tested were found to be resistant to 3 or more antimicrobials. The susceptibility patterns of these multi-drug-resistant isolates are summarised in Table 3.
DISCUSSION
A high percentage of the isolates tested were resistant to the beta-lactam antibiotics penicillin and ampicillin. The figures obtained are in agreement with those obtained in similar studies carried out elsewhere. A study in China reported that 77.3 % of the S. aureus mastitis isolates tested were resistant to penicillin/ampicillin, while studies in Denmark, Brazil and Argentina reported figures of 75 %, 55.1 % and 40 %, respectively1,4,9,14. Although the percentage resistance to this class of drugs has varied between countries, all studies indicated that the greatest resistance observed amongst S. aureus strains was to the beta-lactams. The extensive resistance to this class of antimicrobials is not altogether surprising considering the fact that the penicillins are 1 of the oldest groups of antimicrobials and have been available for many years. They are among the most common antimicrobials used in the dairy industry; 97 % of the intramammary preparations available in South Africa are penicillins or penicillin-dihydrostreptomycin combinations. The remaining 3 % of intramammary preparations available are tetracyclines and cephalosporins5,8.
As part of a recent National Surveillance and Monitoring Programme coordinated by the Faculty of Veterinary Science of the University of Pretoria, a number of S. aureus bovine mastitis isolates from different provinces were screened for antimicrobial resistance15. Each isolate was tested against a panel of antimicrobials and the minimum inhibitory concentration (MIC) of each antimicrobial determined. Comparatively much lower levels of resistance to the penicillins were recorded in the National Surveillance and Monitoring Programme study than were obtained in this study. The National Surveillance Programme reported that approximately 10 % of the S. aureus isolates tested were resistant to ampicillin whereas the figures obtained in this study were much higher (65.6 %). This may be attributed in part to the different test methodologies used but may also represent the different target populations tested. The levels of resistance to the aminoglycosides were similar in the 2 studies. The national programme reported that 14.4 % and 6.3 % of S. aureus isolates tested were resistant to gentamicin and neomycin respectively, whereas in this investigation 17 % of the isolates tested were resistant to streptomycin and 5.6 % were resistant to neomycin. Lower levels of resistance to enrofloxacin were recorded in this study, 1.1 % compared with 10.6 % recorded in the National Survey. Unfortunately, because the composition of the antimicrobial panel used in the National Surveillance Programme was designed for the purposes of general surveillance and not specifically for the evaluation of mastitis pathogens, oxacillin and a firstgeneration cephalosporin were not incorporated in the test panel.
The most common strategy used by S. aureus to circumvent the action of the penicillins is by the production of the enzyme beta-lactamase, which hydrolyses the beta-lactam ring, rendering the entire compound inactive6. The use of methicillin, a beta-lactamase-resistant penicillin, initially overcame the problem experienced with beta-lactamase-producing bacteria. Unfortunately certain groups of bacteria, including the staphylococci, have evolved new strategies that led to the emergence of methicillin-resistant strains. This has had the greatest impact in human medicine, where methicillin resistant S. aureus (MRSA) has emerged as a major nosocomial pathogen. Until recently the problem was limited to hospitals, but the MRSA strains have started to spread in the human community at large. The presence of MRSA strains has been reported in animals but accounts of isolations from dairy cattle have been rare6. One of the isolates evaluated during this investigation was found to be resistant to oxacillin, and by virtue of group representation, methicillin resistant. Two further isolates were found to be moderately susceptible to oxacillin. The presence of MRSA strains is hereby confirmed in KZN dairy herds but to date this particular resistance pattern appears to have a limited distribution. Owing to the public health significance of MRSA, ongoing monitoring for methicillin-resistant strains in the dairy industry is warranted6.
Seventeen of the S. aureus isolates tested were found to be multi-drug resistant, i.e. resistant to 3 or more of the antimicrobials tested. One of these isolates was in fact found to be resistant to 5 of the antimicrobials, namely penicillin, ampicillin, oxacillin, tetracycline and the potentiated sulphonamide (trimethoprim/sulfamethoxazole). The isolate was found to be sensitive to cephalothin but according to CLSI guidelines methicillin-resistant staphylococci should be reported as resistant to all beta-lactams, including cephalosporins, despite any apparent in vitro susceptibility3. The occurrence of multi-drugresistant strains is a cause of great concern as these strains may be readily transmitted to other dairy cows or even workers in the dairy. Treatment options are limited and culling of animals becomes necessary to remove these strains from circulation.
The disc diffusion method is 1 of the standardised protocols recommended by the CLSI for the in vitro antimicrobial susceptibility testing of bacterial isolates. The method has found application in many diagnostic laboratories due to ease of use, flexibility and economic feasibility12. It needs to be borne in mind that all in vitro tests were initially developed and used in the human diagnostic field. Therefore, most of the interpretive data available for use are derived from studies carried out in human medicine. To date very little research has been carried out using veterinary isolates and evaluating the clinical efficacy of antimicrobials against these isolates in different animal hosts10. Diagnostic results should therefore be used as a guideline only. A further consequence of the lack of experimental data in animal hosts is that there are limited interpretative data available that permit the synergistic effect of different drug combinations to be assessed in vitro.
Ultimately the in vivo success of antimicrobial therapy is reliant upon multiple factors. In addition to the virulence of the causative agent, the physicochemical characteristics of the drug, the prevailing udder microenvironment and the in vivo interaction of all components significantly influence the success of treatment13. Where S. aureus is implicated, the successful treatment of infected animals is significantly compromised by the strategies of a cunningly resourceful pathogen.
CONCLUSION
Staphylococcus aureus has gained notoriety in the dairy industry due to its success as a pathogen and its consequent impact on animal health and profit margins. One of the principal approaches adopted in combatting S. aureus is the administration of antimicrobials. Few accounts exist that document the susceptibility of S. aureus strains implicated in bovine mastitis to the different classes of antimicrobials used in the local dairy industry. Although treatment guidelines will ultimately be based on the susceptibility pattern of the specific S. aureus strain implicated in a particular herd, the data obtained from this study may provide practitioners with an insight into existing resistance patterns and assist when immediate treatment action needs to be taken. The data obtained from this investigation indicate that resistance to the beta-lactam antibiotics, penicillin and ampicillin, is common. Resistance to the other classes of antimicrobials tested varies between 1.1 % and 16.7 %. The detection of strains exhibiting intermediate susceptibility and, in 1 case, resistance to methicillin is a cause for concern, as is the occurrence of multi-resistant strains which are present within some KwaZulu-Natal dairy herds.
The data obtained in this study support the need for continued rigorous monitoring of drug resistance patterns. This will ensure that appropriate treatment regimens are implemented that will reduce the indiscriminate use of antimicrobials that has often been implicated in the emergence of resistant bacterial strains.
ACKNOWLEDGEMENTS
The author wishes to acknowledge the technical staff in the Mastitis Control Section at Allerton PVL (Y. Rajpal, M. Khanyi and S. Mantanga) and Drs Chisi and Perrett for reviewing this manuscript.
REFERENCES
1. Aarestrup F M, Jensen N E 1998 Development of penicillin resistance among Staphylococcus aureus isolated from bovine mastitis in Denmark and other countries. Microbial Drug Resistance 4: 247-256 [ Links ]
2. Barkema H W, Schukken Y H, Zadoks R N 2006 The role of cow, pathogen, and treatment regimen in the therapeutic success of bovine Staphylococcus aureus mastitis. Journal of Dairy Science 89: 1877-1895 [ Links ]
3. CLSI 2008 Performance standards for antimicrobial disk and dilution susceptibility tests for bacteria isolated from animals; Approved standard M31-A3 (3rd edn). Clinical and Laboratory Standards Institute, Wayne, NJ [ Links ]
4. De Oliveira A P, Watts J L, Salmon S A, Aarestrup F M 2000 Antimicrobial susceptibility of Staphylococcus aureus isolated from bovine mastitis in Europe and the United States. Journal of Dairy Science 83: 855-862 [ Links ]
5. Eager H A 2008 A survey of antimicrobial usage in animals in South Africa with specific reference to food animals. MVSc thesis, University of Pretoria, Pretoria [ Links ]
6. Giguére S, Prescott J F, Baggot J D, Walker R D, Dowling R M 2006 Antimicrobial therapy (4th edn). Blackwell Publishing, Cornwall [ Links ]
7. Haveri M 2008 Staphylococcus aureus in bovine intramammary infection: molecular, clinical and epidemiological characteristics. University of Helsinki, Helsinki, Finland [ Links ]
8. Swan G.E. (managing editor) 2004. Speciality index. MIMS IVS. MIMS, a division of Johnnic Publishing Limited, Johannesburg 42(4): 23-35 [ Links ]
9. Li J-P, Zhou H-J, Yuan L, He T, Hu S-H 2009 Prevalence, genetic diversity, and antimicrobial susceptibility profiles of Staphylococcus aureus isolated from bovine mastitis in Zhejiang Province, China. Journal of Zhejiang University 10: 753-760 [ Links ]
10. Moroni P, Pisoni G, Antonini M, Villa R, Boettcher P, Carli S 2006 Antimicrobial drug susceptibility of Staphylococcus aureus from subclinical bovine mastitis in Italy. Journal of Dairy Science 89: 2973-2976 [ Links ]
11. Petrovski K R, Trajcev M, Buneski G 2006 A review of the factors affecting the costs of bovine mastitis. Journal of the South African Veterinary Association 77: 52-60 [ Links ]
12. Quinn P J, Carter M E, Markey B, Carter G R 1994 Clinical Veterinary Microbiology. Mosby, Madrid, Spain [ Links ]
13. Quinn P J, Markey B K, Carter M E, Donnelly W J, Leonard W C 2002 Veterinary microbiology and microbial disease. Blackwell Publishing, City, Cornwall, UK [ Links ]
14. Rabello R F, SouzaCRVM, Duarte R S, LopesRMM,Teixeira L M, CastroACD 2005 Characterization of Staphylococcus aureus isolates recovered from bovine mastitis in Rio de Janeiro, Brazil. Journal of Dairy Science 88: 3211-3219 [ Links ]
15. SANVAD 2007 South African National Veterinary Surveillance and Monitoring Programme for Resistance to Antimicrobial Drugs. Department of Veterinary Tropical Diseases, Faculty of Veterinary Science, University of Pretoria, Pretoria [ Links ]
16. Zadoks R N, van Leeuwen W B, Kreft D, Fox L K, Barkema H W, Schukken Y H, van Belkum A 2002 Comparison of Staphylococcus aureus isolates from bovine and human skin, milking-equipment, and bovine milk by phage typing, pulsed-field gel electrophoresis, and binary typing. Journal of Clinical Microbiology 40: 3894-3902 [ Links ]
17. Ziv G, Storper M 1985 Intramuscular treatment of subclinical staphylococcal mastitis in lactating cows with penicillin G, methicillin and their esters. Journal of Veterinary Pharmacology and Therapeutics 8: 276-283 [ Links ]
Received: October 2010.
Accepted: April 2011.














