Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Journal of the South African Veterinary Association
On-line version ISSN 2224-9435
Print version ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.81 n.3 Pretoria Sep. 2010
CLINICAL COMMUNICATION KLINIESE MEDEDELING
Surgical uterine drainage and lavage as treatment for canine pyometra
K G M De Cramer
Rant en Dal Animal Hospital, PO Box 704, Rant en Dal, Mogale City, South Africa. E-mail: kdcramer@mweb.co.za
ABSTRACT
Pyometra is a common post-oestral syndrome in bitches. Classical treatment consists of either ovariohystorectomy or medical intervention. Surgical uterine drainage and lavage via direct trans-cervical catheterisation using a 5 % povidone-iodine in saline solution was performed successfully in 8 bitches with pyometra. All bitches conceived and whelped without complications subsequent to this treatment. It is concluded that this method offers an effective alternative treatment for canine pyometra with shorter recovery times as well as good clinical recovery and pregnancy rates in bitches destined for further breeding.
Keywords: dog, pyometra, uterine lavage.
INTRODUCTION
By definition pyometra is a pus-filled uterus. Pyometra is typically a post-oestral syndrome in adult bitches associated with a variety of clinical and pathological manifestations of genital and multisystemic disease2-5,7,11,20,27. Pyometra is most commonly associated with polydipsia, polyuria, abdominal distension, vomition, poor appetite, weight loss and anaemia20. A purulent vaginal discharge may, however, be the only symptom in some cases of pyometra when the cervix is open11.It appears that the degree of patency of the cervix dictates the severity of the clinical signs to a large extent11. Canine pyometra is a life-threatening disease28,34. It is postulated that intrauterine bacteria, which ascend from the vagina during pro-oestrus and oestrus, induce the disease during metoestrus by acting on the progesterone-primed endometrium directly via toxin production, or indirectly by the release of inflammatory mediators33. There is a clear association between cystic endometrial hyperplasia (CEH) and pyometra11,20,27. CEH is a progressive degenerative process and is generally accepted to be the initiating lesion and contributes to failure to shed normally transient uterine infections by compromising the uterine environment and/or local defence mechanisms38. Parity and age are significant risk factors in the development of pyometra20,32.
Escherischia coli, beta-haemolytic streptococci, Pasteurella multocida, coagulasepositive Staphylococcus spp. and Klebsiella spp. are amongst the organisms mostly isolated from pyometra cases, either in pure culture or mixed infections6,9,10,25.As pyometra may have fatal complications, clients opting to retain their bitch's reproductive capacity should be warned of potential risks of medical or surgical treatments. Pyometra cases show marked haematological and biochemical changes. These changes include elevated total white blood cell counts (WCC), marked left shift, toxic degeneration of neutrophils, elevated serum ALP levels and detection of plasma endotoxin in some cases16. Clinical improvement of pyometra cases has been associated with reduction in plasma progesterone concentrations, increased vaginal discharges, reduction in diameter of the uterus as determined by ultrasonography and normalisation of haematological and biochemical profiles3,5,13,15,18,29. Treatment of canine pyometra with prostaglandins (PG) is successful with clinical recovery rates approaching 80 % and post-treatment pregnancy rates attained varying from 40%to80%19,21,29,31,37 . Prostaglandin F2α (PGF2α) facilitates smooth muscle contraction in the uterine wall leading to expulsion of uterine contents in cases of pyometra21. It is this expulsion of uterine contents that is suspected to be responsible in part for recovery39. The duration of prostaglandin administration is thus dictated by the uterine dimension, time it takes for vaginal discharges to subside and ultimately by return of the WCC to normal and the patient to clinical normality. To compare the success rate of treatment protocols for pyometra clinical recovery rates, pregnancy rates, litter size, recurrence rates and duration of treatment should be considered. In some cases, the duration of treatment is of concern. In a retrospective study, some dogs were treated with PG for up to 26 days and in another for over 3 weeks before cure was achieved19,26. There are cases of canine pyometra refractory to PGF2α therapy6 and cases that recur at subsequent cycles13,15,29. This accentuates the importance of immediate breeding after treatment.
Combination therapy using prostaglandins with either dopamine agonists or progesterone receptor antagonists have been used to successfully treat spontaneously occurring cases of pyometra13. In one study using cabergoline and cloprostenol clinical recovery was attained within 14 days in over 90 % of cases, with a pregnancy rate of 64 %13. Combination therapy is probably the preferred choice in closed cervix pyometra as aglepristone was reported to open a closed cervix in approximately 48 h following the 1st administration15. When combined with cloprostenol the clinical recovery rate was 84 % but the treatment frequently lasted up to 28 days15. Intra-uterine antibiotics and antiseptics including povidoneiodine have been used successfully in treating uterine infections in cows14,23,24. Intra-uterine infusions of saline, EDTA-Tris, chlorhexidine gluconate and antibiotics have similarly been successfully used in mares12,22,40 .
The uterine drainage and lavage as illustrated in this study was performed in an attempt to shorten the duration of treatment of pyometra in the bitch as well as achieve both high clinical recovery and pregnancy rates post-treatment.
MATERIALS AND METHODS
Case selection
The study was performed in a commercial breeding establishment and in a private practice. Any bitch that developed pyometra was automatically included in the trial irrespective of age or breed until 8 bitches were collected. Informed owner consent was obtained in all cases. The case selection of the individual bitches was based on their exhibiting some or all of the typical signs of pyometra characterised by depression, anorexia, polydipsia, polyuria, pyrexia and purulent vaginal discharge. All showed an enlarged fluidfilled uterus on ultrasonography and typical leucogram changes. Two of the 8 bitches, the Kanaan and the Greyhound, showed severe signs of depression, dehydration, severe tachycardia, toxic mucous membranes and anaemia. A vaginal discharge was noted in 5 of the bitches. Prior to antimicrobial therapy, a deep vaginal guarded swab was collected and sent for bacteriological culture and antibiotic sensitivity assessments (Table 1).
Reproductive history of the bitches presented
Marmalade. Beagle, 5 years multigravid bitch had 3 previous litters. In her last 2 cycles she was not bred. Pyometra was diagnosed ±6 weeks post-oestrus.
Ansie. German shepherd, 4 years maiden bitch that cycled normally. She developed pyometra 8 weeks post-oestrus.
Callie. German shepherd, 5 years old, maiden bitch. Developed pyometra at approximately 7 weeks post-oestrus.
Quali. German shepherd bitch, age unknown. Previous reproductive history unknown. Developed pyometra ±4 weeks post-oestrus.
Vasta. German shepherd, multigravid bitch, aged 6 years. Developed pyometra 10 weeks post-oestrus.
Kanaan bitch. Kanaan cross bitch, 6 years. Reproductive history unknown. Pyometra was diagnosed but it was not recorded when she was last in oestrus.
Siberian husky, 4 years, multigravid bitch, developed pyometra 5 weeks postoestrus.
Greyhound, 10 years, multigravid but had not conceived in the last 2 years. Pyometra was diagnosed 3 weeks postoestrus.
Pre-surgical therapy
Two cases presented in severe shock and were 1st stabilised by using shock therapy consisting of extensive fluid therapy and high doses of prednisolone sodium succinate (Solu-delta Cortef, Upjohn) at 10 mg/kg IV. Corticosteroid therapy has been used in varied doses for sepsis and related syndromes for more than 50 years, but its use remains controversial1. As soon as the diagnosis of pyometra was made, all the bitches were treated with 20 mg/kg clavulanate potentiated amoxycillin (Synulox, Beecham Pharmaceuticals) twice daily and gentamycin (Genta 20, Phenix) at 4 mg/kg (BID) until the result of the antibiogram indicated the use of other antibiotics. Flunixin meglumin (Finadyne, Centaur Laboratories) was also given at 2 mg/kg (SID) in conjunction with ringers lactate infusion at rates above maintenance. The surgical flushing commenced within 48 hours of the diagnosis.
Surgical method
The flushing medium selected was a 50:50 mixture of 'Betadine' (povidoneiodine 100 mg/mℓ, Adcock-Ingram) and normal saline to constitute a 5 % betadine-saline mixture. The 8 bitches were premedicated with atropin sulphate 0.5 %, (Centaur laboratories) and acetylpromazine maleate (Aceprom 2 %, Centaur laboratories). Induction of anaesthesia was achieved by using sodium thiopentone (Intraval, Maybaker) and maintenance was achieved by halothane gas inhalation. The perineal area was washed using water and antiseptic soap. The bitch was prepared surgically as for a laparotomy.
In addition to a surgeon and a surgical assistant, the procedure required a 3rd assistant to manipulate the irrigation pipette and flushing medium (the 'nonsterile assistant', see Fig. 1). A standard abdominal midline incision was made to allow exteriorisation of the uterine body (see Fig. 2) and localisation of both cervix and vagina. The non-sterile assistant introduced a sterile proctoscope into the vagina as far cranially as possible before removal of the introducer from the proctoscope. A sterile bovine artificial insemination pipette with syringe attachment was introduced via the lumen of the proctoscope and advanced cranially until the surgeon was able to locate its tip through the vaginal wall. The non-sterile assistant then removed the proctoscope from the vagina. The surgeon located the tip of the pipette with 1 hand and simultaneously stabilised the cervix with the other hand. The non-sterile assistant carefully advanced the pipette cranially until the pipette was in close proximity to the cervix. The surgeon then manipulated the cervical opening onto the tip of the pipette and slid the cervix over the pipette. The tip of the pipette was palpable in the uterine body and visible when pushed against the uterine wall (see Fig. 3). The non-sterile assistant then attached a 50 mℓ syringe to the end of the pipette. Depending on the viscosity of the exudate, the assistant could apply suction and aspirate content from the uterus. In most cases the viscosity of the exudates prevented this action. The surgical assistant was required to occlude the utero-tubal junctions of both horns using gentle digital pressure while the non-sterile assistant infused the flushing medium into the uterus. Considerable care was taken to ensure that no pus was forced into the abdomen via the fallopian tubes. Considerable care was also taken by the assistant handling the pipette in the vagina not to penetrate the uterine wall.
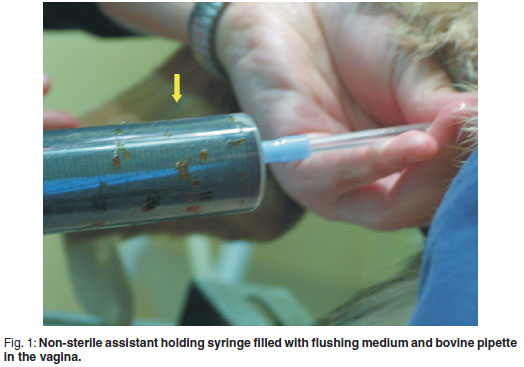


The maximum volume infused into the uterus at any one time depended on the size of the uterus and varied between 20 and 50 mℓ. With the surgical assistant occluding the utero-tubal junctions, the surgeon massaged the uterine wall of the uterus, mixing the exudate and flushing medium. The non-sterile assistant thereafter aspirated the contents of the uterus back into the syringe prior to emptying the syringe, re-filling and injecting more flushing medium into the uterus. This was repeated until that part of the uterus was evacuated. This procedure was repeated progressing towards the tip of the horn and then repeated in the other horn.
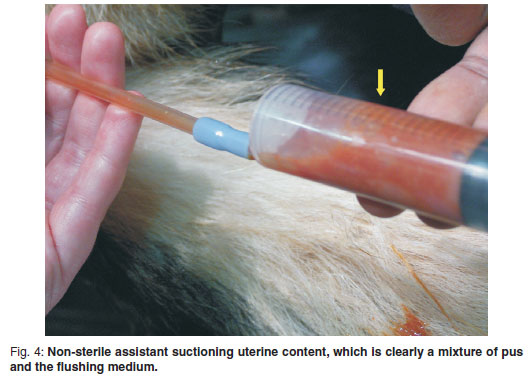
The aspirated exudate initially appeared purulent and viscous. It changed from a thick viscous consistency to a watery consistency (see Fig. 4) and resembled the colour of the introduced flushing medium as the procedure progressed. A volume range of 300-500 ml was used to flush the entire uterus. As the viscosity of the uterine content decreased, this content was observed to escape from around the catheter into the vagina. As the procedure progressed the uterine dimensions were observed to decrease (see Fig. 5). At the end of the procedure the remaining uterine content was milked caudally to escape from the uterus by a combination of aspiration via the pipette lumen or manually along the outer diameter of the pipette into the vagina. The pipette could then be removed from the cervix and an irrigation of the vagina was performed using the flushing medium to remove any pus that may have collected in the vagina. The surgical wound was closed using a standard method. No difficulty was encountered in passing the pipette through the cervix or guiding it into the uterine horns in any of the bitches including the 3 bitches that did not show vaginal discharges and were considered to be closed cervix pyometra cases. The entire procedure was uneventful in all the bitches.

Post-surgical case management
Fluid thearapy at maintenance rates was administered for the 1st day following surgery. Gentamycin and Finadyne were given for a maximum of 5 days, whereas Synulox was given till the vaginal discharge subsided. The results of the culture and sensitivity tests revealed that either the Synulox and/or gentamycin were effective against 6 of 8 samples tested. The results of the sensitivity tests did not warrant changing the antimicrobial therapy in at least these 6 bitches. The appetite, habitus, vital signs and volume and nature of any vaginal discharge were recorded daily for 14 days post-surgery. The colour of the vaginal discharges varied from light straw-coloured to dark brown and appeared to be muco-purulent in nature. The volumes were scant and subsided in all but 1 bitch by day 4. The Greyhound bitch showed a vaginal discharge for 12 days. All the bitches ate normally within 2 days of surgery. A blood sample was obtained via cephalic venipuncture for haematology at 14 days post-surgery. By 14 days after the flushing, the WCC had returned to normal in all bitches.
RESULTS
The subsequent oestrus cycle
At the 1st signs of pro-oestrus, the bitches were presented for routine oestrous monitoring. The bitches were all treated empirically with 20 mg/kg Synulox from the time of 1st mating until 5 days into the subsequent dioestrus. The bitches were then monitored for vaginal discharges daily until pregnancy diagnosis by ultrasound at Day 27 of dioestrus. The inter-oestrus interval appeared to be unaffected by the surgical intervention.The bitches all showed a normal oestrous cycle and were mated at least twice each, every alternate day from the 1st day of standing heat until dioestrus. All the bitches conceived and whelped normally. The litter size was considered normal in all but 1 of the bitches (Table 1).
DISCUSSION
No previous reports were found on the use of intra-uterine antiseptics in bitches. The purpose of this study was to evaluate whether surgical uterine flushing and lavage of the diseased (pyometra) uterus could be used as a safe alternative treatment option and also possibly shorten the duration of treatment as well as achieve high clinical recovery and pregnancy rates. The results obtained in this study show that the duration of treatment was 12 days or shorter which compares favourably with other studies and the 100 % recovery and pregnancy rates achieved exceed any of those in previous studies5,6,13,15,18,19,29,31,37. These results need to be scrutinised and attempts made at possible explanations of its success. One possible explanation is the extent to which the septic exudate was efficiently removed from the uterine lumen by direct manual manipulation and a significant reduction in diameter of the uterus achieved during the flushing procedure. It is speculated that this was achieved by both the physical effect of flushing as well as manual manipulation and 'milking' of the uterus itself. The success of the current method described may in part be explained by the efficient expulsion of uterine contents as is the case when PG is used39.
In the choice of antiseptic, its microbicidal properties, safety, efficacy in the presence of purulent debris and the strength of dilution at which it exhibits these properties are important considerations. Although povidone-iodine was found to be temporarily cytotoxic after 30 min contact with murine fibroblasts, an unexpected revitalisation was observed 24 hours later30. In one study it was demonstrated that povidone-iodine can be used safely in spinal surgeries, and does not influence wound healing, bone union and clinical outcome at a dilution of 0.35 %8. The microbicidal action spectrum of povidone-iodine is broad even after short onset times36. Unlike local antibiotics and some other antiseptic substances, no resistance has been reported with povidone-iodine36. With protein load (0.2 % albumin), which mimics the presence of pus, the optimum in microbicidal effectiveness shifts to concentrations >10%of povidone-iodine35. The direct antiseptic effect of the flushing medium itself may thus have contributed to its success. The fact that a residual, undetermined amount of flushing medium must have remained in the uterus might have had an osmotic effect and/or irritant post-flushing or lasting antiseptic effect. Likewise it may be assumed that an undetermined but probably small amount of pus may have remained in the uterus post-flushing. It was very clear that the mixing of the flushing medium in the uterus led to a significant reduction in the viscosity of the exudate. This reduction in viscosity eased the flushing procedure as it progressed but must also have aided in improved drainage post-surgery. It is the author's opinion that the removal of the exudates from the uterus, as evidenced by reduction in uterine diameter, must have been one of the main contributing factors to successful and shorter course of treatment achieved.
It has been suggested that placement of intra-uterine catheters cause mechanical drainage and reduction in uterine diameter as monitored by sequential hysterography17,25. Mechanical stimulation of the uterine wall was supported by a case which was subjected to unilateral drainage of the uterus for a few days25. Since drainage took place along and not through the plastic catheters, it is difficult to understand how one catheter could exert its selective effect by facilitating escape of exudate from the drained horn. A more plausible explanation for the asymmetrical involution of the uterus could be that the catheter stimulated contraction of the uterine horn either directly or indirectly. It may be speculated in the current study that the flushing medium induces increased motility in the same fashion. The direct irritant properties might also have had a stimulating and rejuvenating effect on the endometrial epithelium. The flushing of the uteri may also have had the effect of releasing endogenous prostaglandins which in turn could have induced luteolysis, uterine motility and expulsion of septic debris, but this was not confirmed in this study.
The beneficial effects of drugs such as PF2α, prolactin inhibitors and antigestagens in the treatment of pyometra may be due to their effect on reducing the action of progesterone on the uterus. These drugs may, through mechanisms such as opening the cervix and increasing uterine contractility, be beneficial adjuncts to uterine lavage. It was interesting to note that in the 3 cases considered to have a closed cervix pyometra (based on absence of vaginal discharges), the cervix was easily passed using direct manual manipulation. These cases had vaginal discharges post-flushing, indicating that the manual intervention and possibly the reduction in viscosity both opened or stretched the cervical opening and allowed remaining contents to escape from the cervix which was apparently closed prior to the intervention.
Considering the fact that bacterial contamination appears to occur during oestrus and early dioestrus, it was logical to institute antimicrobial therapy during the vulnerable phase of post-flushing oestrous cycles. Whether this preventive treatment truly played a role in controlling the recurrence of pyometra, was not conclusively determined, as no controls were available to test this hypothesis. It is not clear from the literature whether this practice is of proven value or not.
In this study, the possibility of the flushing procedure and/or media inducing any uterine pathology was not established. The fact that all the bitches whelped normal-sized litters for their breed in the 1st cycle following flushing indicates that uterine pathology was either insignificant or absent. Information is lacking as to whether the extent and degree of CEH may be in any way reversed or altered by any medication and/or surgical intervention as demonstrated by histopathology before and after such interventions. In future studies it might therefore be of value to both biopsy the uterine wall and monitor progesterone profiles before and after the flushing as this may reveal whether flushing alters the extent and degree of CEH in canines suffering from pyometra or induces pathology. This study only evaluated the conception rates of treated bitches at the 1st cycle following treatment. More long-term studies are required to determine long-term fertility in these bitches. Further studies may also highlight whether uterine drainage and lavage might be efficacious in cases refractory to PG treatment.
The direct visual examination of the uterus by laparotomy has the added advantage of closer examination of the uterine wall for signs and severity of cystic endometrial hyperplasia (CEH) and other uterine/ovarian pathology which may have a bearing on the longterm reproductive performance which is either difficult or impossible to evaluate using ultrasonography.
In conclusion, this study showed that surgical uterine drainage and lavage offers an effective alternative treatment for canine pyometra with shorter recovery times as well as good clinical recovery and pregnancy rates in bitches destined for further breeding. These results justify further studies with more cases to establish whether this method is statistically advantageous over current methods. It would also be of interest to establish whether this method could be used in conjunction with recognised methods in order to optimise a treatment regimen for canine pyometra.
ACKNOWLEDGEMENTS
Sr Z van Niekerk is thanked for assistance in surgical procedures. Srs Mathee, Viviers and Cronje are thanked for conscientious monitoring and data collection during the project. Profs J O Nöthling and M L Schulman are thanked for their guidance.
REFERENCES
1. Annane D, Bellissant E, Bollaert P E, Briegel J, Confalonieri M, De Gaudio R, Keh D, Kupfer Y, Oppert M, Meduri G U 2009 Corticosteroids in the treatment of severe sepsis and septic shock in adults: a systematic review. Journal of the American Medical Association 301: 2362-2375 [ Links ]
2. Austad R, Blom A K, Borresen B 1979 Pyometra in the dog. III. - A pathophysiological investigation. III. Plasma progesterone levels and ovarian morphology. Nordisk Veterinaer Medicin 31: 258-262 [ Links ]
3. Bartoskova A, Vitasek R, Leva L, Faldyna M 2007 Hysterectomy leads to fast improvement of haematological and immunological parameters in bitches with pyometra. Journal of Small Animal Practice 48: 564-568 [ Links ]
4. Bosschere H D, Ducatelle R, Tshamala M 2002 Is mechanically induced cystic endo metrial hyperplasia (CEH) a suitable model for study of spontaneously occurring CEH in the uterus of the bitch? Reproduction in Domestic Animals 37: 152-157 [ Links ]
5. Breitkopf M, Hoffmann B, Bostedt H 1997 Treatment of pyometra (cystic endometrial hyperplasia) in bitches with an antiprogestin. Journal of Reproduction and Fertility. Supplement 51: 327-331 [ Links ]
6. Brown J M 1985 Use of prostaglandin F2a in treatment of uterine diseases in the bitch. Modern Veterinary Practice 66: 381-382 [ Links ]
7. Chaffaux S, Thibier M 1978 Peripheral plasma concentrations of progesterone in the bitch with pyometra. Annales de Recherches Vétérinaires 9: 587-592 [ Links ]
8. Chang FY, Chang MC, Wang ST, Yu WK, Liu C L, Chen T H 2006 Can povidoneiodine solution be used safely in a spinal surgery? European Spine Journal 15: 1005- 1014 [ Links ]
9. Coster R d, D'ieteren G, Josse M, Jacovljevic S, Ectors F, Derivaux J 1979 Clinical, histological, bacteriological and hormonal aspects of chronic metritis in the bitch. Annales de Médecine Vétérinaire 123: 233-247 [ Links ]
10. Dhaliwal G K, Wray C, Noakes D E 1998 Uterine bacterial flora and uterine lesions in bitches with cystic endometrial hyperplasia (pyometra). Veterinary Record 143: 659-661 [ Links ]
11. Dow C 1958 The cystic hyperplasia - pyometra complex in the bitch. Veterinary Record 49: 1102-1111 [ Links ]
12. Dyk E van, Immelman A, Heerden J S 1982 The use of amikacin in the treatment of endometritis caused by Pseudomonas aeruginosa in the mare. Journal of the South African Veterinary Association 53: 124-126 [ Links ]
13. England G C W, Freeman S L, Russo M 2007 Treatment of spontaneous pyometra in 22 bitches with a combination of cabergoline and cloprostenol. Veterinary Record 160: 293-296 [ Links ]
14. Feldmann M, Tenhagen genannt Emming S, Hoedemaker M 2005 Treatment of chronic bovine endometritis and factors for treatment success. Deutsche tierärztliche Wochenschrift 112: 10-16 [ Links ]
15. Fieni F 2006 Clinical evaluation of the use of aglepristone, with or without cloprostenol, to treat cystic endometrial hyperplasiapyometra complex in bitches. Theriogenology 66: 1550-1556 [ Links ]
16. Fransson B, Lagerstedt A S, Hellmen E, Jonsson P 1997 Bacteriological findings, blood chemistry profile and plasma endotoxin levels in bitches with pyometra or other uterine diseases. Journal of Veterinary Medicine (A)44: 417-426 [ Links ]
17. Funkquist B, Lagerstedt A S, Linde C, Obel N 1983 Intra-uterine drainage for treatment of pyometra in the bitch. Zentralblatt für Veterinarmedizin (A)30: 72-80 [ Links ]
18. Gábor G, Siver L, Szenci O 1999 Intravaginal prostaglandin F2a for the treatment of metritis and pyometra in the bitch. Acta Veterinaria Hungarica 47: 103-108 [ Links ]
19. Gilbert R O, Nothling J O, Oettle E E 1989 A retrospective study of 40 cases of canine pyometra-metritis treated with prostaglandin F-2 alpha and broad-spectrum antibacterial drugs. Journal of Reproduction and Fertility Suppl. 39: 225-229 [ Links ]
20. Hardy R M, Osborne C A 1977 Canine pyometra - a polysystemic disorder. Current Veterinary Therapy 1229-1234 [ Links ]
21. Jackson P G G 1979 Treatment of canine pyometra with dinoprost. Veterinary Record 105: 131 [ Links ]
22. Jackson P S, Allen W R, Ricketts S W, Hall R 1979 The irritancy of chlorhexidine gluconate in the genital tract of the mare. Veterinary Record 105: 122-124 [ Links ]
23. Kaneene J B, Coe P H, Smith J H, Rapnicki P, Smith C L, Gerloff B, Morrow D A 1986 Drug residues in milk after intrauterine injection of oxytetracycline, lincomycinspectinomycin, and povidone-iodine in cows with metritis. American Journal of Veterinary Research 47: 1363-1365 [ Links ]
24. Koujan A, Eissa H M, Hussein M A, Ayoub M M, Afiefy M M 1996 Therapeutic efficacy of povidone-iodine (Betadine) and dichloroxylenol (Septocid) in Holstein cows affected with endometritis and/or cervicitis. Acta Veterinaria Hungarica 44: 111-119 [ Links ]
25. Lagerstedt A S, Obel N, Stavenborn M 1987 Uterine drainage in the bitch for treatment of pyometra refractory to prostaglandin F2a. Journal of Small Animal Practice 28: 215-222 [ Links ]
26. Lange K, Gunzel-Apel A R, Hoppen H O, Mischke R, Nolte I 1997 Effects of low doses of prostaglandin F2 alpha during the early luteal phase before and after implantation in beagle bitches. Journal of Reproduction and Fertility Suppl. 51: 251-257 [ Links ]
27. Low D G 1954 Pyometra in the bitch. Veterinary Medicine, Small Animal Clinician 49: 527-530 [ Links ]
28. Marretta S M, Matthiesen D T, Nichols R 1989 Pyometra and its complications. Problems in Veterinary Medicine 1: 50-62 [ Links ]
29. Meyers-Wallen V N, Goldschmidt M H, Flickinger G L 1986 Prostaglandin F2a treatment of canine pyometra. Journal of the American Veterinary Medical Association 189: 1557-1561 [ Links ]
30. Muller G, Kramer A 2006 Comparative study of in vitro cytotoxicity of povidoneiodine in solution, in ointment or in a liposomal formulation (Repithel) and selected antiseptics. Dermatology 212, Suppl. 1: 91-93 [ Links ]
31. Nelson R W, Feldman E C, Stabenfeldt G H 1982 Treatment of canine pyometra and endometritis with prostaglandin F2 alpha. Journal of the American Veterinary Medical Association 181: 899-903 [ Links ]
32. Niskanen M, Thrusfield M V 1998 Associations between age, parity, hormonal therapy and breed, and pyometra in Finnish dogs. Veterinary Record 143: 493-498 [ Links ]
33. Noakes D E, Dhaliwal G K, England G C 2001 Cystic endometrial hyperplasia/pyometra in dogs: a review of the causes and pathogenesis. J. Reprod. Fertil. Suppl 57: 395-406 [ Links ]
34. Pretzer S D 2008 Clinical presentation of canine pyometra and mucometra: a review. Theriogenology 70: 359-363 [ Links ]
35. Reimer K, Schreier H, Erdos G, Konig B, Konig W, Fleischer W 1998 Molecular effects of a microbicidal substance on relevant microorganisms: electron microscopic and biochemical studies on povidone-iodine. Zentralblatt für Hygiene und Umweltmedizin 200: 423-434 [ Links ]
36. Reimer K, Wichelhaus T A, Schañfer V, Rudolph P, Kramer A, Wutzler P, Ganzer D, Fleischer W 2002 Antimicrobial effectiveness of povidone-iodine and consequences for new application areas. Dermatology 204: 114-120 [ Links ]
37. Sokolowski J H 1980 Prostaglandin F2 alpha-THAM for medical treatment of endometritis, metritis, and pyometritis in the bitch. Journal of the American Animal Hospital Association 16: 119-122 [ Links ]
38. Verstegen J, Dhaliwal G, Verstegen-Onclin K 2008 Mucometra, cystic endometrial hyperplasia, and pyometra in the bitch: advances in treatment and assessment of future reproductive success. Theriogenology 70: 364-374 [ Links ]
39. Wheaton L G, Barbee D D 1993 Comparison of two dosages of prostaglandin F2a on canine uterine motility. Theriogenology 40: 111-120 [ Links ]
40. Youngquist R S, Blanchard T L, Lapin D, Klein W 1984 The effects of EDTA-Tris infusion on the equine endometrium. Theriogenology 22: 593-599 [ Links ]
Received: March 2010.
Accepted: July 2010














