Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
Journal of the South African Veterinary Association
versão On-line ISSN 2224-9435
versão impressa ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.81 no.2 Pretoria Jan. 2010
ARTICLE ARTIKEL
Changes in Mithun (Bos frontalis) spermatozoa during epididymal passage
H K BhattacharyyaI,*; K M BujarbaruahII; B K GoswamiIII; R K BiswasIV; I KhanV
ITeaching Veterinary Clinical Complex, Faculty of Veterinary Sciences and Animal Husbandry, Shuhama-Alusteng, Srinagar - 190 006, Jammu and Kashmir, India
IIAnimal Sciences, Indian Council of Agricultural Research (ICAR), New Delhi - 110 001, India
IIILakhimpur College of Veterinary Science, Assam Agricultural University, Azad, North Lakhimpur, Assam, India
IVDepartment of Gynecology, Obstetrics and A.I., College of Veterinary Science, Khanapara, Guwahati - 781022, Assam, India
VFaculty of Fisheries, Shuhama-Alusteng, Srinagar, Jammu and Kashmir, India
ABSTRACT
Genital organs of 10 healthy, adult Mithun bulls (6-8 years old) that were slaughtered at the dwellings of tribal people for meat were collected. Immediately after collection, spermatozoa from 3 different regions of the epididymis, i.e. the head, body and tail, were obtained to study morphological changes of the spermatozoa during passage through these regions. The prevalence of proximal cytoplasmic droplets significantly decreased from the head to the tail of the epididymis. Conversely, the percentage of distal cytoplasmic droplets increased significantly from the head to the tail region. The incidence of tailless heads rose significantly from head to body and then reduced significantly in the tail region. The percentage of total head abnormalities did, however, not change markedly, but total mid-piece and tail abnormalities differed significantly between the three epididymal regions.
Keywords: epididymal passage, male reproduction, Mithun, sperm morphology.
INTRODUCTION
The Mithun (Bos frontalis) is a semi-wild bovid13. The World Conservation Monitoring Cell lists this 'ceremonial ox' of northeastern mountainous regions of India as endangered. It plays an important role in the socioeconomic life of the tribal communities but is yet to be scientifically studied in detail4. The Mithun is usually kept as a free-ranging animal in forests. Tribal people slaughter the Mithun for its meat, which they consider to be precious.
The number of Mithuns has declined greatly in recent times, creating the need to identify fertile males to use as sires in order to conserve the species. In the bull, the most striking morphological change in the spermatozoa during their passage through epididymis is the migration of the cytoplasmic droplet from the proximal to the distal end of the mid-piece9,12. Other changes have also been described3,8. Knowing the morphological changes in spermatozoa during epididymal passage in the Mithun will help define semen quality in the species. Thus the present study examines morphological changes as the spermatozoa pass through the epididymis.
MATERIALS AND METHODS
The present study was conducted in the National Research Centre on Mithun (NRCM), Indian Council of Agricultural Research (ICAR), Nagaland, India. Genital organs of 10 healthy, adult Mithun bulls aged 6-8 years were collected from the dwellings of tribal people when they sacrificed them during marriage ceremonies. The animals were found to be in good health and no physical abnormalities were noticed pre-or post-slaughter. Different parts of the external and internal genitalia were also free of gross pathological changes.
The study was conducted from January to mid June. Maximum and minimum temperatures varied between 21.0 and 9.4 ºC in January and between 30.0 and 25.71 ºC in June. Monthly rainfall was 9.4-289.5 mm and relative humidity 72.5-83.8%. Immediately after collection the genital organs were transported to the laboratory in normal saline solution at 37 ºC. The epididymis was dissected from the testis and epididymal fluid was obtained by incision with a scalpel followed by gentle pressure on the central part of each of three regions of the epididymis, namely the head, body and tail. The epididymal fluid containing spermatozoa from these regions was suspended separately in buffered formal saline. The suspended fluid was further suitably diluted in buffered formal saline to facilitate the study of sperm characteristics, namely proximal cyotoplasmic droplets, distal cyotoplasmic droplets, tailless heads and mid-piece and tail abnormalities, using a phase contrast microscope. Sperm head abnormalities were studied under oil immersion (×100) using a light microscope. The epididymal fluid suspension was used to prepare the thin smears stained with Carbol-fuchsin-eosin stain17,11. Hundreds of spermatozoa per region of each epididymis were examined. The Mann-Whitney test was used to test for differences between the right and left parts of the epididymis, while the Kruskal-Wallis test was used to test for differences between the head, body and tail of the epididymis.
RESULTS
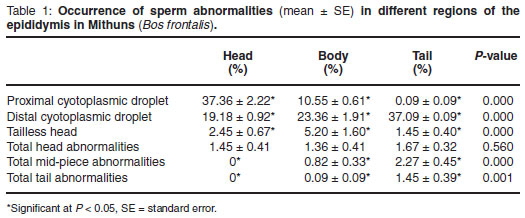
As the spermatozoa pass through the epididymis there is a successive and significant reduction in the prevalence of proximal cyotoplasmic droplets between the head, body and tail, and droplets were virtually absent in the tail (Table 1). On the other hand, the prevalence of distal cyotoplasmic droplets increased significantly in succession in the 3 epididymal regions. The occurrence of tailless heads rose significantly from head to body and then decreased significantly in the tail region (Table 1). Percentage of total sperm head abnormalities did not differ significantly between the 3 regions. Abnormalities of the mid-piece and tail were absent in the head and increased significantly from there to the body and further to the tail. No significant difference was found between the right and left parts of the epididymis.
DISCUSSION
The increase in the prevalence of distal cytoplasmic droplets from the head to the tail regions of the epididymis corresponds to the migration process of the cytoplasmic droplet from a proximal to distal position during epididymal transit. The significant decrease in proximal cytoplasmic droplet and significant increase in distal cyotoplasmic droplet percentages in the spermatozoa from the head to the tail regions of the epididymis was also reported by earlier workers in bulls1, rams2, boars16 and goats15. The finding in the current study that the prevalence of tailless spermatozoa is higher in the body of the epididymis than in the head or tail is similar to that of Rao14, who showed a higher prevalence in the distal part of the head compared to the tail. Since the neck region of spermatozoa is vulnerable and fragile, the sperm head can become detached from the mid-piece with the initiation of motility, which takes place at the base of the sperm head5. Spermatozoa acquire motility between the head and tail of the epididymis and hence the scope for detachment of the head of the spermatozoa could be greatest in the lower head or body, which might explain the significantly higher incidence of tailless heads in the body region. The significant reduction in the incidence of tailless heads in the tail could be attributed to the dissolution and liquefaction3 or phagocytosis within the tail epididymis. In agreement with the current study showing no effect of epididymal passage on the prevalence of sperm head abnormalities, Igboeli10 observed that the prevalence of sperm head abnormalities is also similar in the head and tail of the epididymis of domestic bulls. The present finding in respect of tail abnormalities is in general agreement with observations of earlier workers2,6 that most of the tail abnormalities arise after the spermatozoa leave the testis. Cury and Vinha7 also reported that the incidence of tail abnormalities increased with the passage of spermatozoa through the epididymis. The increase in the percentage of mid-piece and abnormalities in the tail compared to the body could be ascribed to the exposure of the spermatozoa to varying biochemical microenvironments during their passage through the epididymis that might influence the development of sperm tail abnormalities. The present study revealed that the changes in sperm characteristics during epididymal passage in Mithun bulls are similar to those previously recorded in bulls but with some variation.
ACKNOWLEDGEMENT
The authors are grateful to the Director, NRCM, ICAR, Jharnapani, Nagaland, for providing facilities.
REFERENCES
1. Amann R P, Almquist J O 1962 Reproductive capacity of dairy bulls. VII. Morphology of epididymal sperm. Journal of Dairy Science 45: 1516-1526 [ Links ]
2. Amann R P, Hay S R, Hammerstedt R H 1982 Yield, characteristics, motility and cAMP content of sperm isolated from seven regions of ram epididymis. Biology of Reproduction 27: 723-733 [ Links ]
3. Amann R P, Almquist J O 1962 Reproductive capacity of dairy bulls. VI. Effect of unilateral vasectomy and ejaculation frequency on sperm reserves; aspects of epididymal physiology. Journal of Reproduction and Fertility 3: 260-268 [ Links ]
4. Bhattacharyya H K, Rafiqul Islam, Bujarbaruah K M 2005 Mithun: a unique large ruminant of northeastern India. Livestock International 9: 22-23 [ Links ]
5. Bhosrekar M R 1990 Semen production and artificial insemination (2nd edn). Privately published, Alahabad, India [ Links ]
6. Blom E 1950 Interpretation of spermatic cytology in bulls. Journal of Fertility and Sterility 1: 223-238 [ Links ]
7. Cury L T, Vinha N A 1982 Changes in the morphology of goat spermatozoa during their passage through the epididymis. Arquivos da Escola de Veterinaria da Universidade Federal de Minas Gerais 34: 267-271 (cf. Animal Breeding Abstract 51: 310) [ Links ]
8. Gustafson B 1966 Luminal contents of the bovine epididymis under conditions of reduced spermatogenesis, luminal blockage and certain sperm abnormalities. Acta Veterinaria Scandinavica 7: Suppl. 17 [ Links ]
9. Hancock J I 1955 The disintegration of bull spermatozoa. Veterinary Record 67: 825-826 [ Links ]
10. Igboeli G 1968 Bull and rabbit testes and epididymal spermatozoa at normal and body temperatures studied with the aid of ligatures, PhD thesis, Cornell University, Ithaca, New York [ Links ]
11. Lagerlof N 1934. Morphologische Untersuchungen uber Veranderungen in spermatobild und in den Hoden Bei Bullen mit Verminderter order aufgehobener. Fertilitat Acta Pathology Microbiology Scandinavian Suppl: 1-19 [ Links ]
12. Nicander I 1958 Studies on the regional histology and cytochemistry of the ductus epididymis in stallions, rams and bulls. Acta Morphology Nederland Scandinavia 1: 337-362 [ Links ]
13. Pathak N N, Barua K K 1980 The Mithun of Nagaland and Arunachal. Science Report 17: 483 [ Links ]
14. Prasad C S, Reddy J M 2000 Morphological changes of goat spermatozoa during epididymal transit. Indian Journal of Animal Reproduction 21: 27-29 [ Links ]
15. Rao A R 1971 Changes in the morphology of sperm during their passage through the genital tract in bulls with normal and impaired spermatogenesis. PhD thesis, Royal Veterinary College, Stockholm, Sweden [ Links ]
16. Wekerle L, Szollosi E, Sarlos P, Schnaider L 1984 Morphological examination of sperm cells obtained from the testes and epididymis of boars. Magyar Allatorvosok Lapja 40: 663-666 (cf. Animal Breeding Abstract 54: 53-56) [ Links ]
17. Williams W W 1920 Technique of collecting semen for laboratory examination with a review of several diseased bulls. Cornell Veterinarian 10: 87-94 [ Links ]
Received: September 2008.
Accepted: June 2009.
* Author for correspondence. E-mail: drhiranyabh@yahoo.co.in














