Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
South African Journal of Chemistry
versão On-line ISSN 1996-840X
versão impressa ISSN 0379-4350
S.Afr.j.chem. (Online) vol.72 Durban 2019
http://dx.doi.org/10.17159/0379-4350/2019/v72a21
RESEARCH ARTICLE
Synthesis of Indeno[1,2-b]benzofurans using TPAB as Highly Efficient and Recoverable Catalyst
Ahmad Poursattar Marjani*; Jabbar Khalafy*; Somayeh Akbarzadeh
Department of Organic Chemistry, Faculty of Chemistry, Urmia University, Urmia, Iran
ABSTRACT
Herein we report the synthesis of some novel indeno[1,2-b]benzofuran derivatives by reaction of ninhydrin with cyclic 1,3-dicarbonyl compounds including cyclopentane-1,3-dione, cyclohexane-1,3-dione, dimedone, 4,4-dimethylcyclohexane-1,3-dione, meldrum's acid, barbituric acid, thiobarbituric acid, 1,3-dimethylbarbituric acid and indane-1,3-dione. We also used tetra-propylammonium bromide (TPAB) as an organocatalyst.
Keywords: Cyclic 1,3-dicarbonyl compounds, ninhydrin, indeno[1,2-b]benzofurans, TPAB.
1. Introduction
The synthesis of indeno[1,2-i>]benzofurans through the reaction of ninhydrin with 1,3-diketones, aryl aldehydes and malononitrile has been carried out under different reaction conditions. Numerous types of reagents, like AcOH,1 I2/MeOH,2 BF3-OEt2,3 EtOH/US4are known for their catalyzing effect on this reaction. In spite of acceptable results obtained from the above-mentioned methods, their usage is limited by some disadvantages such as high cost, low yields of product, high reaction temperatures and the use of some less than desirable solvents.
The indeno[1,2-i>]benzofurans are important heterocyclic compounds due to their wide range of pharmaceutical and biological activities, such as their anti-microbial, anti-oxidant, analgesic and anti-inflammatory properties.1,5
Tetrapropylammonium bromide (TPAB) is a readily available, and inexpensive chemical and one of the most commonly utilized phase-transfer catalysts (PTC) with numerous applications in diverse catalytic processes, including for instance in the synthesis of ZSM-type zeolites.6
Following our previous research work involving the synthesis of novel heterocyclic compounds,7 we report here the production of some new indeno[1,2-i>]benzofuran derivatives in excellent yield and relatively short reaction times, through the reaction of ninhydrin with cyclic 1,3-dicarbonyl compounds, using TPAB as a recyclable catalyst.
2. Experimental
The chemicals used in the current research were purchased from Acros and Merck companies. FTIR spectra were recorded with a Thermo Nicolet (Nexus 670) instrument. 1H and 13C NMR spectra were recorded on a Bruker spectrometer at 300 and 75 MHz in CDCl3 or DMSO-d6 as solvents and using TMS as the internal standard. Mass spectra were recorded on a Varian Matt 311 spectrometer. Melting points were determined with an Electrothermal 9200 apparatus. Elemental analyses were performed using a Leco Analyzer 932.
General Procedure for the Synthesis of Products 3a-i
A mixture of ninhydrin (1, 1 mmol) and cyclic 1,3-dicarbonyl compounds 2a-i (1 mmol) was heated in the presence of TPAB (20 mol%) in dichloromethane (2 mL) under reflux for an appropriate time (Scheme 1). The reaction progress was investigated by TLC using CH2Cl2/hexane/methanol, 15:15:1. After completion of the reaction, the solvent was removed under reduced pressure and the obtained precipitate was filtered and washed with H2O/EtOH (1:2), resulting to the desired products 3a-i in good yields of 87-98 %.
Recovery of TPAB
In the separation of products 3a-i, after extraction of filtrate with CHClj, the aqueous layer was separated and evaporation of water gave TPAB, which may be used for reusability process.
4a,9a-Dihydroxy-2,3,4«,9«-tetrahydrocyclopenta[b]indeno[2,1-rf]furan-1,9-dione (3a): This compound was obtained from cyclopentane-1,3-dione, time reaction 26 min, white powder, yield 87 %; m.p. 159-161 °C; FT-IR vmax: 3494, 2926, 2563, 1749, 1714,1604,1545,1370,1291,1142,954,848, 768, 717,640,581,476, 426 cm-1; áH: 7.98 (4H, bs, Ar), 3.61 (2H, bs, 2xOh), 2.38 (4H, bs, 2xCH2); áC: 194.7, 191.5, 158.6, 143.2, 142.1, 141.3, 130.8, 127.8, 126.6,119.3,115.7, 72.2,36.1,29.3; m/z: 259 [M+1]+ (95), 258 [M]+ (100), 242 (46), 230 (95), 173 (39), 134 (28), 105 (62), 104 (57), 77 (42), 76 (38). Found: C, 65.12; H, 3.90 %. Calc. for C14H10O5 (258); C, 65.29; H, 3.80 %.
4b,9b-Dihydroxy-6,7,8,9b-tetrahydro-9if-indeno[1,2-b]benzo furan-9,10(4MZ)-dione (3b): This compound was obtained from cyclohexane-1,3-dione, time reaction 20 min, yellow needles, yield 93 %; m.p. 165-168 °C [lit.,1174 °C].
4b,9b-Dihydroxy-7,7-dimethyl-6,7,8,9b-tetrahydro-9if-indeno [1,2-b]benzofuran-9,10(4MZ)-dione (3c): This compound was obtained from dimedone, time reaction 25 min, white needles, yield 98 %; m.p. 198-200 °C [lit.,4 210 °C].
4b,9b-Dihydroxy-6,6-dimemyl-6,7,8,9b-tetrahydro-9if-indeno[ 1,2-b]benzofuran-9,10(4Wi)-dione (3d): This compound was obtained from 4,4-dimethylcyclohexane-1,3-dione, time reaction 25 min, white powder, yield 97 %; m.p. 152-154 °C; FT-IR vmax: 3389, 3253, 3096, 2941, 2880, 2679, 2495, 2360, 1711, 1660, 1609,1469,1392,1330,1294,1263,1178,1158,1066,1006,949,920, 881,852,778,668,577,520,499,448 cm-1; áH: 7.90-7.88 (2H, m, Ar), 7.74-7.65 (2H, m, Ar), 5.51 (2H, bs, 2xOH), 2.26 (2H, bs, CH2), 1.27 (2H, bs, CH2), 1.03 (6H, s, 2xCH3) ppm; áC: 196.1, 196.0, 163.5, 149.4,148.0,132.6,121.7,117.9,116.2,115.2,109.7, 72.7,45.5,34.7, 23.4, 22.4 ppm; m/z: 301 [M+1]+ (19), 300 [M]+ (85), 243 (18), 226 (22), 202 (46), 167 (45), 146 (19), 134 (23), 104 (42), 83 (100), 55 (15). Found: C, 67.99; H, 5.37 %. Calc. for C16H16O5 (300); C, 67.82; H, 5.47 %.
4b,9b-Dihydroxy-2,2-dimethyl-4b,9b-dihydro-4íí,5iï-indeno [2',1':4,5]furo[2,3-d][1,3]dioxine-4,5-dione (3e): This compound was obtained from Meldrum's acid, time reaction 26 min, white powder, yield 87 %; m.p. 110-113 °C; FT-IR vmax: 3434,3254,2974, 1727, 1605, 1463, 1389, 1342, 1223, 1097, 976, 764, 654 cm-1; δH: 7.28-7.15 (4H, m, Ar), 5.33 (2H, bs, 2xOH), 1.44 (6H, s, 2xCH3) ppm; δC: 202.2, 192.7, 167.4, 162.3, 144.5, 140.5, 134.3, 131.7, 129.4, 122.0, 113.7, 86.0, 80.1, 24.6 ppm; m/z: 304 [M]+ (4), 290 (15), 202 (10), 167 (25), 149 (68), 125 (19), 114 (100), 97 (39), 86 (40), 71 (51), 57 (67). Found: C, 59.22; H, 3.98 %. Calc. for C15H12O7 (304); C, 59.40; H, 3.76 %.
4b,9b-Dihydroxy-4b,9b-dihydro-2ii-indeno[2',1':4,5]furo [2,3-d] pyrimidine-2,4,5(1ff,3.ff)-trione (3f): This compound was obtained from barbituric acid, time reaction 30 min, yellow solid, yield 91 %; m.p. 165-167 °C; FT-IR vmax: 3607, 3456, 3175, 2970,1697,1583,1471, 1370,1247,1182,1045,1009, 970, 852, 772, 730, 639, 580, 525, 424 cm-1; δH: 9.43 (2H, bs, 2xNH), 7.91-7.85 (4H, m, Ar), 3.37 (2H, bs, 2xOH) ppm; áC: 204.9, 203.0, 202.9, 165.6,152.2,137.0,134.7,132.6,124.3,122.0,119.3,87.3, 79.6 ppm; m/z: 288 [M]+ (13), 271 (4), 229 (17), 211 (4), 186 (4), 143 (20), 128 (14), 114 (100), 104 (45), 86 (38), 76 (38), 50 (8). Found: C, 54.18; H, 2.80; N, 9.72 %. Calc. for C13H8N2O6 (288); C, 54.00; H, 2.92; N, 9.61 %.
4b,9b-Dihydroxy-2-thioxo-2,3,4b,9b-tetrahydro-4if-indeno [2',1':4,5]furo[2,3-d]pyrimidine-4,5(1ff)-dione (3g): This compound was obtained from thiobarbituric acid, time reaction 30 min, white needle, yield 92 %; m.p. 190-192 °C; FT-IR vmax: 3585, 3515, 3070, 2969, 2879, 1712, 1590, 1528, 1433, 1378, 1289, 1248, 1194, 1014, 971, 889, 848, 771, 724, 626, 533 cm-1; δH: 10.89 (2H, s, 2xNH), 7.92-7.87 (4H, m, Ar), 3.65 (2H, bs, 2xOH) ppm; δC: 202.1,174.5,163.8,147.3,137.3,135.0,129.0,126.9,124.4,122.1, 116.4,97.1,85.4 ppm; m/z: 304 [M] + (5), 290 (6), 186 (3), 160 (3), 144 (34), 132 (28), 114 (100), 104 (78), 86 (35), 72 (33), 76 (75), 59 (8). Found: C, 51.32; H, 2.65; N, 9.21 %, Calc. for C13H8N2O5S (304); C, 51.43 %, H, 2.52, N, 9.35 %.
4b,9b-Dihydroxy-1,3-dimethyl-4b,9b-dihydro-2íí-indeno[2',1': 4,5]furo[2,3-rf]pyrimidine-2,4,5(1ff,3ií)-trione (3h): This compound was obtained from 1,3-dimethylbarbituric acid, time reaction 30 min, yellow solid, yield 89 %; m.p. 128-130 °C; FT-IR vmax: 3366,2959, 2859,1669,1455,1382,1263,1157,1102, 990, 924, 884,747,623,493 cm-1; δH: 8.07-8.05 (2H, m, Ar), 7.93-7.90 (2H, m, Ar), 4.99 (1H, s, OH), 4.43 (1H, s, OH), 3.30 (6H, s, 2xCH3) ppm; δC: 198.5,197.4,197.3,150.2,138.0,135.7,132.1,131.6,124.6,122.3, 111.6,94.1,83.2,26.5,25.2 ppm; m/z: 316 [M]+ (5), 300 (5), 186 (6), 156 (41), 132 (38), 104 (100), 76 (91), 50 (8). Found: C, 56.97; H, 3.82; N, 8.86 %. Calc. for C15H12N2O6 (316); C, 56.88; H,3.96; N,8.71 %.
4b,10b-Dihydroxy-4b,10b-dihydrodiindeno[1,2-b:2',1'-rf]furan-10,11-dione (3i): This compound was obtained from indane-1,3-dione, time reaction 27 min, white powder, yield 88 %; m.p. 179-181 °C; FT-IR vmax: 3418, 3081, 2906, 1707, 1587, 1464, 1344, 1265,1155,1083,940,887, 797, 755,603,443 cm-1; δH: 7.95 (4H, m, Ar), 7.70-7.51 (4H, m, Ar), 4.99 (2H, bs, 2xOH) ppm; δC: 199.8, 197.2, 169.2, 167.8, 147.4, 141.2, 140.5, 138.6, 128.4, 128.1, 122.0, 120.8,113.7,113.0, 107.0, 105.5, 90.0, 72.6 ppm; m/z: 306 [M]+ (3), 305 (8), 290 (30), 276 (16), 263 (15), 233 (16), 205 (10), 189 (18), 176 (25), 145 (14), 104 (100), 90 (9), 76 (58). Found: C, 70.59; H, 3.29 %. Calc. for C18H10O5 (306); C, 70.81; H, 3.39 %.
3. Results and Discussion
The synthesis of the heterocyclic compounds started with arylglyoxals as core and used TPAB as an efficient organocatalyst with acceptable results.8 As part of the project to develop catalytic applications of TPAB in synthesis of various heterocyclic compounds, it was found that the reaction of ninhydrin (1) with cyclic 1,3-dicarbonyl compounds 2a-i using TPAB (20 mol%) in dichloromethane under reflux conditions, afforded indeno [1,2-b]benzofuran derivatives 3a-i in excellent yield (Scheme 1).
In a preliminary study, the reaction was conducted without any catalyst, as a result, no product was obtained even after 24 h under reflux conditions (Table 1, entry 1). To determine the optimized reaction conditions for the synthesis of indeno [1,2-b] benzofurans, the reaction of ninhydrin (1,1 mmol) with dimedone (2c, 1 mmol) was chosen as a model reaction to produce compound 3c, and its behaviour was examined in the presence of several potential catalysts such as TPAB for phase transfer, p-TSA as acid source, L-cysteine and L-proline as zwiterions and sodium alginate as a salt, with different molar ratios and various solvents at room temperature ranging to reflux. To find out the appropriate solvent, for the reaction, model reaction was repeated in various solvents such as H2O, acetone, CH2Cl2 and EtOH. Reaction in water or dichloro-methane in the absence of any catalyst at room temperature or under reflux condition gave no product (Table 1, entry 1). After evaluating a number of different conditions (see Table), the optimum yield (98 %) was obtained when TPAB was used as catalyst, CH2Cl2 as solvent, 25 min of reaction time and under reflux conditions (Table 1, entry 21). To peruse the amount of catalyst in our trial reaction, the procedure was optimized using different molar concentrations of TPAB. To this end, an excellent yield of adduct 3c was obtained using 20 mol%. On the contrary, lower quantities of the catalyst gave moderate yields of the product at longer reaction times (results not shown). Furthermore, using larger amounts of TPAB was not effective in terms of yields and resulted in large amounts of the starting materials remaining unconverted. The respective results of the investigations are displayed in Table 1.
Based on our survey and optimization of the reaction conditions, the generality and the scope of the organocatalyst in the synthesis process of 3a-i were assessed by reaction of ninhydrin (1) with different cyclic 1,3-dicarbonyl compounds 2a-i (Scheme 1). As depicted in Scheme 1, the desired compounds were obtained in excellent yields and short reaction times from all of the reactions. Formation of the indeno[1,2-i>]benzofurans system were confirmed by studying FT-IR, Ή-NMR, 13C-NMR, mass spectral data and microanalysis, beside comparison of the obtained spectral data of known compounds with those reported in the literature.
In terms of spectroscopic characterization, the 1H NMR spectrum of product 3d showed a characteristic broad singlet at δ = 5.51 ppm attributable to the OH groups, which was exchanged by D2O. Aromatic protons appeared as multiplet at δ = ~7.65-7.90 ppm, methylene groups gave broad singlets at δ = 2.26 and 1.27 ppm, and methyl groups displayed a singlet at δ = 1.03 ppm. The 13C NMR spectrum showed 16 peaks for all the different carbons as expected. Moreover, the FT-IR (KBr) spectrum of 3d, showed the characteristic absorptions bands at 1711 and 1660 cm-1 due to the vibrations of different carbonyl groups, and its mass spectrum showed the molecular ion at m/z 300 and a peak at m/z 83 with 100 % abundance as base peak.
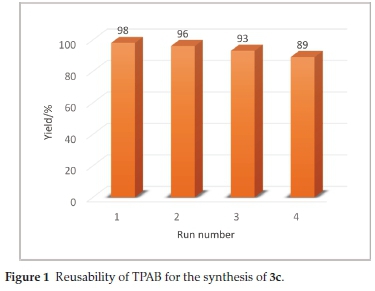
The recyclability of TPAB was also investigated for the synthesis of 3c. In this respect, the catalyst was recovered and reused more than four times for obtaining 4i>,9fc-dihydroxy-8,8-dimethyl-4b,8,9,9fc-tetrahydro-7H-indeno[1,2-i>]benzofuran-6,10-dione (3c). No significant loss of catalytic efficiency was observed (Fig. 1).
4. Conclusions
In conclusion, a highly efficient and easy procedure was reported for the synthesis of a new series of indeno[2,1-i>]benzo-furan derivatives 3a-i in excellent yields, by reaction of ninhydrin and cyclic 1,3-dicarbonyl compounds using 20 mol% TPAB as an organocatalyst in CH2Cl2 as solvent. The reasonable reaction times, simple work-up, high yields and recoverability of the catalyst are the main merits of this procedure. The final obtained products are suitable for the synthesis of a series of polycyclic heterocycles that may show potential antimicrobial and inhibitory properties.1
Acknowledgements
The authors are grateful to Urmia University for its support of this work.
Supplementary Material
Supplementary information is provided in the online supplement.
ORCID iDs
A. Poursattar Marjani: orcid.org/0000-0002-5899-4285
J.Khalafy: orcid.org/0000-0003-1202-2664
References
1 S.H. Mehdi, R. Hashim, R.M. Ghalib, M.F.C.G. da Silva, O. Sulaiman, S.Z. Rahman, V. Murugaiyah and M.M. Marimuthu, Synthesis, characterization, antimicrobial and enzymatic activity of 4b,9b-dihy-droxy-7,8-dihydro-4bH-indeno[1,2-b]benzofuran-9,10(6H,9bH)-dione, J. Mol. Struct., 2001,1006, 318-323. [ Links ]
2 a) J.M. Kim, K.Y. Lee and J.N. Kim, Oxidative aromatization of 2-acylcyclohexane-1,3-dione derivatives using Iodine in methanol, Bull. Korean Chem. Soc.,2003,24,1057-1058. [ Links ]b) J.E. Na,J. M. Kim, S. Lee and J.N. Kim, Synthesis of benzo[b]indeno[2,1-d]furanone skeleton from ninhydrin and cyclohexane-1,3-dione derivatives, Bull. Korean Chem. Soc., 2003,24,1725-1726. [ Links ] c) J.E. Na, S. GowriSankar, S. Lee and J.N. Kim, Selective methylation of the ninhydrin-phenol adducts with I2 in MeOH, Bull. Korean Chem. Soc., 2004, 25, 569-572. [ Links ]
3 T. Chanda and M.S. Singh, Developments toward the synthesis and application of 3-hydroxyindanones, Org. Biomol. Chem., 2016, 14, 8895-8910. [ Links ]
4 Y. Saini, R. Khajuria, L.K. Rana, G. Hundal, VK. Gupta, R. Kant and K.K. Kapoor, Unprecedented reaction of ninhydrin with ethyl cyano-acetate and diethyl malonate on ultrasonic irradiation, Tetrahedron., 2016, 72, 257-263. [ Links ]
5 a) K.R. Prabhakar, V.P. Veerapuri, P. Bansal, K.P. Vipan, K.M. Reddy, A. Barik, B.K.D. Reddy, P. Reddanna, K.I. Priyadarsini and M.K. Unnikrishnan, Identification and evaluation of antioxidant, analgesic/anti-inflammatory activity of the most active ninhydrin phenol adducts synthesized, Bioorg. Med. Chem. 2006, 14, 7113-7120. [ Links ] b) S. Mahendran, S. Badami, S. Ravi, B.S. Thippeswamy and V. P. Veerapur, Antioxidant, analgesic and anti-inflammatory properties of new ninhydrin adduct of embelin, Pharm. Chem. J., 2011, 45, 547-551. [ Links ]
6 a) R.J. Argauer and G.R. Landolt, Acid-free nitration of benzene and toluene in zeolite NaZSM-5, U.S. Patent. 3 702 886 1972. [ Links ] b) C. Bolm, A.S. Magnus and J.P. Hildebrand, Catalytic synthesis of aldehydes and ketones under mild conditions using TEMPO/Oxone, Org. Lett., 2000, 2, 1173-1175. [ Links ]
7 a) J. Khalafy, M. Ezzati, P. Madadi, A. Poursattar Marjani and H. Yaghoobnejad Asl, Synthesis of some 5-[2-aryl-2-oxoethyl]-1,3-dimethylpyrimidine-2,4,6-trione derivatives by a one-pot, three-component reaction, S. Afr. J. Chem., 2017, 70, 132-136. [ Links ] b) M. Ezzati, J. Khalafy, A. Poursattar Marjani and R.H. Prager, The catalyst-free syntheses of pyrazolo[3,4-b]quinolin-5-one and pyrazolo[4',3':5,6] pyrido[2,3-d]pyrimidin-5,7-dione derivatives by one-pot, three-component reactions, Tetrahedron, 2017, 73, 6587-6596. [ Links ] c) A. Poursattar Marjani, J. Khalafy, F. Majidi Arlan and E. Eyni, A simple protocol for the green synthesis of a new series of pyrimido[4,5-b] [1,6]naphthyridines in the presence of silver nanoparticles (AgNPs), ARKIVOC, 2019, v, 1-9. [ Links ]
8 a) M. Ezzati, J. Khalafy, A. Poursattar Marjani and R.H. Prager, An efficient one-pot, four-component synthesis of pyrazolo[3,4-b]pyridines catalyzed by tetrapropylammonium bromide (TPAB) in water, Aust. J. Chem., 2018, 71,435-41. [ Links ] b) A. Poursattar Marjani, J. Khalafy and A. Farajollahi, Synthesis of ethyl 2-amino-4-benzoyl-5-oxo-5,6-dihydro-4H-pyrano[3,2-c]quinoline-3-carboxylates by a one-pot, three-component reaction in the presence of TPAB, J. Heterocycl. Chem., 2019, 56, 268-274. [ Links ] c) A. Poursattar Marjani, J. Khalafy and S. Akbarzadeh, Synthesis of pyrazolopyridine and pyrazoloquinoline derivatives by one-pot, three-component reactions of arylglyoxals, 3-methyl-1-aryl-1H-pyrazol-5-amines and cyclic 1,3-dicarbonyl compounds in the presence of tetrapropylammonium bromide, Green Process Synth., 2019, 8, 533-541. [ Links ] d) N. Etivand, J. Khalafy and A. Poursattar Marjani, Facile, one-pot, four-component synthesis of a new series of imidazo[1,2-a]pyridines in presence of TPAB in EtOH under reflux conditions, Res. Chem. Intermediat., 2019, 45, 3379-3394. [ Links ]
Received 5 February 2019
Revised 3 July 2019
Accepted 21 July 2019
* To whom correspondence should be addressed. E-mail: a.poursattar@gmail.com / a.poursattar@urmia.ac.ir
Supplementary Data
The supplementary data is available in pdf: [Supplementary data]














