Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
South African Journal of Chemistry
versão On-line ISSN 1996-840X
versão impressa ISSN 0379-4350
S.Afr.j.chem. (Online) vol.71 Durban 2018
http://dx.doi.org/10.17159/0379-4350/2018/v71a22
RESEARCH ARTICLE
Monitoring of So2, nox and Nh3 emission from burning of solid wastes at awotan and lapite dumpsites, Ibadan, Nigeria
Ayodele Rotimi IpeaiyedaI; Bamidele Ayodele FalusiII
IDepartment of Chemistry, University of Ibadan, Ibadan, Nigeria
IIDepartment of Chemistry, Federal College of Education (Special), Oyo, Nigeria
ABSTRACT
Emission of gaseous pollutants from open dumpsites has been identified as a major source of air pollution in urban cities such as Ibadan, Nigeria. Air quality assessments at Awotan and Lapite dumpsites where burning of solid wastes occurs without adequate control were presented in this study. The open burning of solid wastes pose environmental challenges associated with the emission of SO2,NOx and NH3 whose adverse impact on humans in the vicinity of dumpsites is inevitable. The concentrations obtained for SO2,NOx and NH3 were in the range of 260-379 ug m-3; 354-470 ug m-3 and 590-907ug m-3, respectively, at Awotan dumpsite. The corresponding concentrations at Lapite dumpsite were 342-428 jug m-3; 521-741 jug m-3 and 1085-1374 jug m-3, respectively. Levels of SO2 and NOx exceeded the European Union and Nigeria national standard air quality limits. There is a need to develop better operating practices in terms of period and method of combustion of solid wastes at designated dumpsites in Ibadan.
Keywords: Solid wastes, sulphur dioxide, oxide of nitrogen, ammonia, open dumpsites
1. Introduction
Urbanization and the quest to improve living standard in most cities of developing countries have resulted in an increase in quantity of municipal solid waste (MSW) generation. In most developing countries open burning of wastes is still performed in an uncontrolled manner, thereby making waste generation an issue of environmental and health concern. Rapid combustion of wastes occurs in most landfills resulting in direct emission of odours, particulate matter, greenhouse gases and other gaseous pollutants to atmosphere. It is apparent that unconventional burning activities in either dumpsites or landfills change the composition of the atmosphere leading to, for instance, increased acid deposition, stratospheric ozone loss and potentially climate change.1,2 Gaseous pollutants such as oxides of nitrogen (NOx), sulphur dioxide (SO2) and carbon monoxide (CO) are implicated in these global environmental effects and their control is an issue of current public concern.3,4 However, control strategies must be based on an understanding of the concentrations of gaseous pollutants from pollution sources such as landfills.
In Nigeria, as in many developing countries in the world, numerous studies examining concentrations of NOx and SO2 from vehicular and industrial emissions have been reported.5-8 Pollution resulting from the emission of atmospheric gaseous pollutants at various municipal solid waste dumpsites have received little or no attention. The economy in Nigeria has led to an influx of people migrating from rural areas to Ibadan city in search for a better quality of life. Ibadan is the third largest city in Nigeria, with a population of over 3 million and a total area of about 3080 km2.9 Migration to this city is increasing the annual rate of solid waste generated. The city now generates more than 996 102 tons of solid waste annually.10 Much of the wastes is collected and managed by the state Solid Waste Management Agency (SWMA). Lapite and Awotan dumpsites are designated for the disposal of solid wastes. The combustion of wastes in an uncontrolled manner at the dumpsites is subjecting the neighbourhood to health risks from obnoxious smell, smoke and gaseous emissions including greenhouse gases (GHGs). The substantial emission of SO2, NOx and NH3 to the atmosphere is inevitable and this inordinate emission has the potential to adversely impact the people residing in the vicinity of the dumpsites. Despite the observed annual rate of increment in waste generation, there seems to be lack of information regarding the levels of gaseous air pollutants emanating from these dumpsites. Hence, there is a need to account for the contribution of this pollution source to atmospheric gaseous pollutants. This information can guide future national gaseous emission control programmes. Therefore, the purpose of this study was to determine the concentrations of sulphur (IV) oxide (SO2), nitrogen oxides (NOx), and ammonia (NH3) from Awotan and Lapite dumpsites in Ibadan. A regression model was intended to be generated for subsequent air pollutant emission estimation from the solid wastes generated in the city.
2. Experimental Methods
2.1. Description of Study Area, Sampling and Characterization of Solid Wastes
The sampling of solid wastes and collection of air samples were carried out at Lapite and Awotan dumpsites located in Ibadan (Fig. 1) between July and October 2016. Permission was sought from SWMA to conduct sampling on these two frequently used dumpsites in Ibadan. Lapite was noticed to be very active in receiving the highest proportion of waste generated in Ibadan, while Awotan received comparatively less. The two dumpsites were chosen for this study because of their proximity to residential areas. Lapite dumpsite (latitude 7°34'08"N and longitude 3°54'39"E) occupies about 20 hectares, while Awotan dumpsite (latitude 7°27'48"N and longitude 3°50'56"E) occupies about 25 hectares. Solid waste samples were collected from the dumpsites for the purpose of characterization. A quadrant (60 x 60 cm2) was purposively placed randomly upon the waste heaps at north, south, west, and east locations of the dumpsites. All forms of solid wastes that were directly under the area covered by the quadrant were collected into a polythene bag. The process was repeated randomly three times at each location. Solid wastes from each location were packed into different polythene bags and 5.0 kg of solid waste was weighed out of each polythene bag into another empty polythene bag and mixed thoroughly. Thereafter, 75.0 kg of the mixed solid waste sample was accurately weighed to obtain a representative sample for each location and various components were segregated. The processes of collection and segregation of solid wastes were repeated at every sampling period. A total number of 80 (1 sample x 80 days) mixed solid waste samples were segregated for each dumpsite.
2.2. Sampling and Determination of Gaseous Air Pollutants
During the sampling periods, air was absorbed into various reagents for SO2,NOx and NH3 determinations using air sampling trains.11-13 Each sampling train that was made of a wooden box (55 cm x 33 cm x 25 cm) with four impingers. Air was aspirated into the impingers by a vacuum pump. The impingers were connected to the vacuum pump through capillary tubes fitted with polypropylene funnels and glass orifices for the passage of air at the specific flow rate of the connected glass orifices. The modified West and Gaeke procedure was used for the determination of SO2 in air samples.14-16 The determination of NOx based on the Griess-Saltzman colorimetric azo-dye forming reagent was done following the intersociety committee method of analysis.17,18 The colorimetric method following nesslerization procedure was used for the determination of NH3.19,20 At each sampling site, the sampling trains were mounted on stands about 2 m high, at a suitable open space in the north, south, west and east locations of the dumpsite. This constituted four sampling locations in each dumpsite. The sampling trains were configured with critical orifices whose flow rates were calibrated for SO2 (549 mL min-1), NOx (1012 mL min-1) and NH3(860 mL min-1). The first impinger was filled with 15 mL of 0.1 M potassium tetrachloromercurate (II) for absorbing SO2. Sulphur dioxide (SO2) from air reacted with potassium tetrachloromercurate (II) to form dichloro-sulphitomercurate complex, which was made to react with pararosaniline and formaldehyde solutions to form an intensely purple coloured pararosaniline methysulphonic acid.14-16 The second impingers contained chromic acid which converted the nitrogen monoxide present in the air to nitrogen dioxide. The third impinger contained 15 mL of a mixture of sulphanilic acid (5.0g) dissolved in glacial acetic acid (140 mL) and 20 mL of 0.001 g mL-1 of N-(1-naphthyl)-ethylenediamine dihydro-chloride (NINE) for absorbing NOx. The diazotization reaction of NINE in glacial acetic solution with NOx contained in air formed a pinkish coloured azo-dye solution.17,18 The fourth impinger was filled with 15 mL of 0.5 M sulphuric acid solution for absorbing NH3 in the air. This solution was subsequently treated with 2.0 mL of Nessler reagent to give a brown coloured solution.19,20 Each sampling was operated for 1 h. In order to determine the degree of variation during a daytime cycle of activities within the dumpsites, air was sampled at four locations in each dumpsite for seven defined 1-h periods of the day. The sampling periods were 8-9 am, 9.15-10.15 am, 10.30-11.30 am, 1-2 pm, 2.15-3.15 pm, 3.30^.30 pm and 4.45-5.15 pm for all locations. The sampling was done five times a week for four months. A total number of 2240 air samples (4 locations x 7 hours x 80 days) were collected for each dumpsite. Calibration curves were prepared from standard sodium sulphite, sodium nitrite and ammonium chloride solutions. Reagents used were of analytical reagent (AR) grade. Reagent blanks were analyzed for each daily sampling and analysis. The absorbance values of the solutions after colour development were measured using Spectrumlab 752s UV/Vis spectrophotometer. The measurements of absor-bance were carried out for all the absorbing solutions from the four locations (north, south, west and east) in each dumpsite.
The reading was carried out in duplicate and average data were obtained for each set of absorbance measurements. Duncan's multiple range test was used to investigate ifmonthlyconcentra-tions of SO2, NOx and NH3 were significantly different.
2.3. Air Pollutant Emission Estimation
An emission factor approach was used to predict SO2,NOx and NH3 emission from the dumpsites. The emission factor for the estimation of air pollutants level is a function of solid waste received by the dumpsite. This approach was adopted for air pollutants estimation based on average yearly solid waste generated at Awotan and Lapite dumpsites using Equation 1. API is the air pollutant emitted in tons yr-1, EFP is the emission factor of pollutant in kg mg-1 and AYSWG is the average yearly solid waste generated in tons per annum. The emission factors of NOx, SO2, and NH3from municipal solid waste were 3.0 kg mg-1,4.4 x 10-1 kg mg-1, and 7.30 kg mg-1, respectively, as established by the Environmental Protection Agency.21
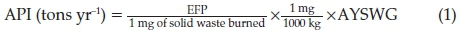
The information on the amount of solid waste moved to each dumpsite monthly for 2012 to 2015 was obtained from the state Solid Waste Management Board (SWMB). The total solid waste generated per capita at each dumpsite was calculated using the data obtained from SWMB. The API values for NOx, SO2 and NH3 emitted for the years 2012 to 2015 were subsequently estimated for Awotan and Lapite dumpsites.
Many mathematical air quality models have previously been developed to assess spatial variation in air pollution. Current approaches for such assessment include the use of interpolation methods and land-use regression models.22 A curvilinear regression was employed to fit a curve using the estimated API values for each pollutant emitted from each dumpsite and years for which API values were estimated as input data. The SPSS computer package was used to fit first a straight line, then a quadratic curve followed by a cubic curve to the data. The cubic equations were adequate to provide a good fit to the data. A general regression model can be expressed as Equation 2.

where Y is the dependent variable (estimated API for air pollutant); x is considered an independent variable (period in terms of years for which API was estimated); a is the constant of regression and b, c, d are the coefficients of regression. The constant and the coefficients are obtained using least-square method, which minimizes the error associated with the regression.23
3. Results and Discussion
3.1. Characterization of Municipal Solid Waste
Waste generation in Nigeria was reported to be about 25 million tons annually, at a rate of 0.44-0.68 kg capita-1 day-1.24,25 According to SWMA in Ibadan, solid wastes of 503 309.68 metric tons per annum (Table 1) was generated in 2016 with an estimated population of 2 550 593 going by 2006 population census.26 With this population, per capita rate of municipal solid wastes generation in Ibadan is about 0.54 kg capita-1 day-1. This rate of solid wastes generation was much higher than 0.46 kg capita-1 day-1 reported in the literature for Ibadan city.27 The solid waste generation rates for the last three years as shown in Table 1 reveals a 23 % increase in the amount of solid waste generated in 2016 compared with the previous year.
The respective components of solid wastes from Awotan and Lapite dumpsites were primarily composed of biodegradable materials (2.1 ± 1.4,3.1 ± 0.4 %), glass (31.8 ± 2.4,34.6 ± 1.6 %), metal (15.3 ± 1.3,12.3 ± 1.1 %), textile and plastics (32.6 ± 1.3, 31.3 ± 1.0 %),leather(3.9 ± 0.5,3.5 ± 0.6 %) andpaper(14.3 ± 1.4, 15.2 ± 0.9 %). The decreasing order of the waste components was textile and plastic>glass>metals>paper>leather>biode-gradable (Fig. 2). The sources of solid wastes such as glass and metals placed in open dumpsites are mainly a function of location, which can be of residential, commercial or industrial origin.28 In this study, high quantities of non-combustible solids were observed compared with other components because the wastes deposited at the dumpsites were mainly from the residential areas. Metals, paper and leather were of small proportion owing to the fact that scavengers were actively busy on the dumpsites picking up the wastes for recycling. The collected items from the dumpsites are sold in bulk to various companies in Nigeria.
3.2. Emission Estimation of SO2, NOx and NH3 from Solid Waste Dumpsites
Lapite and Awotan dumpsites received 30.5 % and 12.6 % of the wastes generated per annum in the year 2016 (Table 1). Overall monthly average estimates of API were 67.5, 460.5 and 1151.2 tons annum-1 for emission of SO2,NOX and NH3, respectively, from Lapite dumpsite while corresponding emission of 27.9, 190.1 and 475.2 tons annum-1 from Awotan dumpsite were estimated for year 2015 (Fig. 3). Thus, higher levels of the three gaseous air pollutants were estimated to be emitted at Lapite dumpsite than Awotan dumpsite. Similar high levels of these air pollutants were observed in a study conducted in Ogbomoso, Nigeria.29 The curvilinear regression expressions shown in Fig. 3 indicates how estimates of SO2,NOXand NH3emitted (API) vary with the period in terms of years for which solid wastes were generated at both dumpsites. One way to forecast the likely amount of air pollutants emitted is to plot the estimated API against the periods and to fit the regression curve. The regression model seems to perform best for the amount of air pollutants emitted predominantly from point sources.30 The estimated API and the periods were the input data to the computer regression programme, which generated the cubic equations shown in Fig. 3 for SO2,NOX and NH3. These cubic expressions were found to adequately provide a good fit to the values of API with R2 values of 1.000 for all the expressions. Thus, the expression can simply be relevant to estimate the levels of SO2,NOX and NH3 to be emitted in subsequent years for both dumpsites. The levels and distribution of these air pollutants can be considerably influenced by meteorological parameters such as wind direction, wind speed, relative humidity and rainfall.31 However, these parameters were not considered in this present study. Therefore, consideration and detailed evaluation of the meteorological parameters of air at the dumpsites is recommended for further study to improve the predictions of the air pollutant concentrations.
3.3. Concentrations of SO2, NOx and NH3 at Dumpsites
The levels of SO2, NOx and NH3 observed on monthly basis at Awotan dumpsites are shown in Table 2, while their variations on hourly basis are shown in Fig. 4. The respective SO2, NOx and NH3 concentrations of 324 ± 28, 401 ± 45 and 741 ± 89 ug m-3 were observed at Awotan dumpsite on a monthly basis. These levels were lower than the corresponding levels of 388 ± 32, 651 ± 78 and 1290 ± 110ag m-3 at Lapite dumpsite (Table 2). This resulted in an increase in concentrations of about 20.0 %, 62.0 % and 74.0 % above the concentrations obtained at Awotan dumpsite. The increasing order of concentrations obtained at the two dumpsites is SO2<NO2<NH3. The monthly concentrations of SO2, NOx and NH3 were significantly different with the exception of NH3 for the first two months (Table 2). The burning period of wastes at the two dumpsites is inconsistent. The inconsistency in the burning period could be responsible for the variation in the levels of pollutants emanating from the dumpsites.
There are variations in the levels of SO2, NOx and NH3 with respect to the daily sampling period. The lowest levels of SO2, NOx and NH3 were obtained between 8.00 and 9.00 am at both dumpsites. The levels gradually increased until 2.15-3.15 pm at which the highest levels were obtained. The exception in the increasing trend was noted for ammonia level at Lapite dumpsite for which the highest level was obtained in the morning between 9.15 and 10.15 am. Beyond 3.15 pm, the levels of SO2, NOx and NH3 gradually decreased (Fig. 4).
The levels of SO2,NOx and NH3 obtained in this study were compared with specific air quality guidelines for Nigeria and some other countries (Table 3). The levels of SO2 obtained at Awotan (324 ag m-3) and Lapite dumpsites (388 ag m-3) were above the national allowable limit and other international air quality guidelines. The emission of NOx resulting from combustion is mainly in the form of NO, which is generated to the level of available oxygen at temperatures above 1000 oC. Since NO is only slightly soluble in water, a 30 min trial of its inhalation is free of side effects except to infants and precarious patients.32 However, NO is oxidized usually within an hour to NO2 by radicals from the photo reaction of volatile organic compounds possibly at the dumpsite.33 For environmental purposes, using concentration of NO2 as surrogate for the concentration of NOx seems to suffice for the assessment of NOx quality in air.34 On this basis, the levels of NOx observed in this study were above the NO2 level recommended by UK and US air quality guidelines (Table 3).
Table 4 shows the levels of SO2,NOx and NH3 as reported in the literature for some notable dumpsites in Nigeria cities and other countries. Dumpsites in other cities such as Effurun, Asaba, Yenogoa and Benin have higher SO2 levels compared to 324 and 388 ug m-3 obtained in this study for Awotan and Lapite dumpsites, respectively. In the case of NOx concentrations, higher levels were found at Lapite dumpsite than other dumpsites mentioned in Table 4. Ammonia levels of 1290 ug m-3 for Lapite dumpsite were higher than the levels in other dumpsites, with the exception of Effurun dumpsite where 1357.2 ag m-3 was reported (Table 4).
4. Conclusion
This study revealed that there was enhanced air pollution of the dumpsites' environment with SO2,NOx and NH3 The amount of wastes deposited at Lapite dumpsite was more than what Awotan dumpsite receives annually. Hence, air quality assessment at Lapite dumpsite indicated high levels of these air pollutants than at Awotan dumpsite. The current air quality at both dumpsites indicated that SO2,NOx and NH3 levels were above the national and international air quality guidelines.
Information on air quality with respect to SO2,NOx and NH3 levels has now been established in this study for designated Awotan and Lapite dumpsites in Ibadan. Thus, the assessment of these air pollutants at these dumpsites has provided additional baseline air quality data. In addition, the information becomes a useful tool to determine necessary control techniques or good approaches to be applied by the waste management board for air pollutant emission management at dumpsites. The effective techniques among many may include modification of combustion process and enforcement of waste characterization by the waste management board. The emission of SO2,NOx and NH3 into the atmosphere at Awotan and Lapite dumpsites has potential to constitute a major public health hazard, especially to the dumpsite workers that are regularly exposed to the emissions and to people residing in the vicinity of dumpsites. Regular monitoring of the levels of these air pollutants into the atmosphere at the dumpsites is therefore suggested.
References
1 D.J. Wuebbles, A. Jain, J. Edmonds, D. Harvey and K. Hayhoe, Global change state of the science. Environ. Pollut., 1999,100(1-3), 57-86. [ Links ]
2 R.T. Watson and D.L. Albritton, Climate Change 2001: Synthesis Report, Third Asessment of the Intergovernmental Panel on Climate Change, Cambridge University Press, 2001. [ Links ]
3 P.M. Lemieux, C.C. Lutes and D.A. Santoianni, Emissions of organic air toxics from open burning: a comprehensive review, Prog. Energy Combust. Sc., 2004, 30(1), 1-32. [ Links ]
4 J. Sonibare and L. Jimoda, Criteria air pollutants from some anthropogenic combustion processes in Lagos, Nigeria, Energy Sources, Part A, 2009, 31(11), 923-935. [ Links ]
5 A.A. Susu, K.E. Abhulimen and A.B. Adereti, Modeling of air pollution systems with chemical reactions: application to gas flares in Nigeria, Int. J. Comput. Methods Eng. Sci. Mech, 2005, 6(3), 201-213. [ Links ]
6 A. Abdulkareem, J. Odigure and S. Abenege, Predictive model for pollutant dispersion from gas flaring: a case study of oil producing area of Nigeria, Energy Sources, Part A, 2009, 31(12), 1004-1015. [ Links ]
7 A.O. Amakiri, A. Monsi, S.C. Teme, P.N. Ede, O.J. Owen and E.M. Ngodigha, Air quality and micro-meterological monitoring of gaseous pollutants/flame emissions from burning crude petroleum in poultry house, Toxicol. Environ. Chem, 2009, 91(2), 225-232. [ Links ]
8 O. Oguntoke, O.K. Otusanya and H.J. Annegarn, Emission of pollutants from wood waste incineration at sawmills in Abeokuta metropolis, Nigeria, Int. J. Environ. Stud, 2013, 70(6), 964-975. [ Links ]
9 J. Falade, Public acquisition of land for landscaping and open space management, J. Nigeria Institute ofTown Planners, 1998, 11, 1-13. [ Links ]
10 O.S. Amuda, S.A. Adebisi, L.A. Jimoda and A.O. Alade, Challenges and possible panacea to the municipal solid wastes management in Nigeria, J. Sustainable Development Stud., 2014, 6(1), 64-70. [ Links ]
11 J. Jacobs and S. Hochheiser, Continous sampling and ultramicrodetermination of nitrogen dioxide in air, Anal. Chem., 1958, 30(3), 426-428. [ Links ]
12 A. Mishra and R. Shrivastava, Study of seasonal concentration of SPM, RSPM, SO2 and NOx in the ambient air of Rewa city. Asian J. Sci. Technol., 2017, 8(7), 5084-5087. [ Links ]
13 C. Häni, J. Sintermann, T. Kupper, M. Jocher and A. Heftel, Ammonia emission after slurry application to grassland in Switzerland, Atmos. Environ., 2016,125, 92-99. [ Links ]
14 P.W. West and G. Gaeke, Fixation of sulfur dioxide as disulfito-mercurate (II) and subsequent colorimetric estimation, Anal. Chem., 1956, 28(12), 1816-1819. [ Links ]
15 P. Sharma, P. Sharma, S. Jain and P. Kumar, An integrated statistical approach for evaluating the exceedence of criteria pollutants in the ambient air of megacity Delhi, Atmos. Environ., 2013, 70, 7-17. [ Links ]
16 P. Onianwa, S.O. Fakayode and B. Agboola, Daytime atmospheric sulphur dioxide concentrations in Ibadan city, Nigeria, Bull. Chem. Soc. Ethiop., 2001, 15(1), 71-78. [ Links ]
17 J.P. Lodge Jr, Methods ofAir Sampling and Analysis, CRC Press, Boca Raton, 1988. [ Links ]
18 D. Schwela, GAP Forum Air Pollution Monitoring Manual, in Global Atmospheric Pollution Forum, 2011. [ Links ]
19 J.W. O'Dell. EPA Method 350.1 Determination of ammonia nitrogen by semi-automated colorimetry, Environmental Monitoring Systems Laboratory Office of Research and Development, U.S. EPA, Cincinnati, Ohio, 1993, pp. 1-15. [ Links ]
20 R.F. Rathbone and R.K. Majors, Techniques for measuring ammonia in fly ash, mortar, and concrete, Powerplant Chemistry, 2003, 5, 757-763. [ Links ]
21 W.M. Joyner, Compilation of Air-pollutant Emission Factors. Volume 1. Stationary-point and Area Sources, Supplement A. No. PB-87-150959/ XAB; AP-42-SUPPL-A, Environmental Protection Agency, Research Triangle Park, NC (USA). Office of Air Quality Planning and Standards, 1986. [ Links ]
22 G. Hoek, R. Beeleb, K. de Hoogh, D. Vienneau, J. Gulliver, P. Fischer and D. Briggs, A review of land-use regression models to assess spatial variation of outdoor air pollution, Atmos. Environ., 2008, 42(33), 7561-7578. [ Links ]
23 V. Vigneron, M. Ponthieu, G. Barina, J.-M. Audic, C. Duquennoi, L. Mazeas, N. Bernet and T. Bouchez, Nitrate and nitrite injection during municipal solid waste anaerobic biodegradation, Waste Manage., 2007, 27(6), 778-791 [ Links ]
24 T.C. Ogwueleka, Municipal solid waste characteristics and management in Nigeria, Iran J. Environ. Health Sci. Eng., 2010, 6(3), 173-180. [ Links ]
25 K.A. Ayuba, L.A. Manef, A.H. Sabrina and S.W.N. Azmin, Current status of municipal solid waste management practise in FCT Abuja, Res. J. Environ. Earth Sci., 2013, 5(6), 295-304. [ Links ]
26 H.E. Jenkins, R.B Aylward, A. Gasasira, C.A. Donnelly, M. Mwanza, J. Corander, S. Garnier, C. Chauvin, E. Abanda, M.A. Pate, F. Adu, M. Bada and N.C. Grassly, Implications of a circulating vaccine-derived poliovirus in Nigeria, N. Eng. J. Med., 2010, 362(25), 2360-2369. [ Links ]
27 I.K. Adewumi, M.O. Ogendengbe, J.A. Adepetu and Y.L. Fabiyi, Planning organic fertilizer industries for municipal solid wastes management, J. Appl. Sci. Res., 2005,1(3), 285-291. [ Links ]
28 C. Chiemchaisri, W. Chiemchaisri, S. Kumar and P.N. Wicrama-rachchi, Reduction of methane emission from landfill through micro-bial activities in cover soil: a brief review, Crit. Rev. Env. Sci. Technol., 2012. 42(4), 412-434. [ Links ]
29 L.A. Jimoda, J.A. Adeniran, J.A. Sonibare and A.A. Ayandiran, Investigation of NO2/NO, SO2, CO and volatile organic compounds emission from solid waste in Ogbomoso, Civil Environ. Res., 2013, 3, 145-152. [ Links ]
30 G.C. Pratt, C.Y. Wu, D. Bock, J.L. Adgate, G. Ramachandran, T.H. Stock, N. Morandi and K. Sexton, Comparing air dispersion model predictions with measured concentrations of VOCs in urban communities, Environ. Sci. Technol., 2004, 38(7), 1949-1959. [ Links ]
31 B. Kim, J. Lee, J. Jang, D. Han and K.-H. Kim, Prediction on the seasonal behaviour of hydrogen sulfide using a neural network model, Scientific World J., 2011,11, 992-1004. [ Links ]
32 M. Beghetti, K. Morris, P. Cox, D. Bohn and I. Adatia, Inhaled nitric oxide differentiates pulmonary vasospasm from vascular obstruction after surgery for congenital heart disease, Intensive Care Med., 1999, 25, 1126-1130. [ Links ]
33 M. Placet, C.O. Mann, R.O, Gilbert and M.J, Niefer, Emissions of ozone precursors from stationary sources: a critical review, Atmos. Environ., 2000, 34(12), 2183-2204. [ Links ]
34 L. Cox and R. Blaszczak, Nitrogen Oxides (NOx) Why and How they are Controlled, DIANE Publishing Co., 1999, pp. 1-57 [ Links ]
35 G.R.E.E. Ana, M.K. Shridhar and J.F. Olawuyi, Air pollution in a chemical fertilizer complex in Nigeria: the impact on the health of the workers, J. Environ. Health Res., 2005, 4(2), 57-62. [ Links ]
36 A.J. Cohen, H.R. Anderson, B. Ostra, K.D, Pandey, M. Krzyzanowski, N. Künzli, K. Gutschmidt, A. Pope, I. Romieu, J.M. Samet and K. Smith, The global burden of disease due to outdoor air pollution, J. Toxicol. Environ. Health, Part A, 2005, 68(13-14), 1301-1307. [ Links ]
37 J. Baldasano, E. Valera and P. Jimenez, Air quality data from large cities, Sci. Total Environ., 2003, 307(1), 141-165. [ Links ]
38 J. Bare, TRACI 2.0: the tool for the reduction and assessment of chemical and other environmental impacts 2.0, Clean Technol. Environ. Policy, 2011. 13(5), 687-696. [ Links ]
39 K. Wallingford, NIOSH (National Institute for Occupational Safety and Health) indoor air quality in office buildings, National Inst. for Occupational Safety and Health, Cincinnati, OH (USA), 1987, p. 1. [ Links ]
40 A. Rim-Rukeh, An assessment of the contribution of municipal solid waste dump sites fire to atmospheric pollution, Open J. Air Pollution, 2014, 3(3), 53-60. [ Links ]
41 World Health Organization, Air Quality Guidelines: Global Update 2005: Particulate Matter, Ozone, Nitrogen Dioxide, and Sulfur Dioxide, World Health Organization Europe, Copenhagen, Denmark, 2006, p. 484. [ Links ]
42 B. Ghauri, A. Lodhi and M. Mansha, Development of baseline (air quality) data in Pakistan, Environ. Monit. Assess., 2007, 127(1), 237-252. [ Links ]
43 EPA, Inventory of US Greenhouse Gas Emissions and Sinks: 1990-2009, U.S. Environmental Protection Agency 2012, Washington, DC, 2011. [ Links ]
Received 12 February 2017
Revised 29 October 2018
Accepted 31 October 2018
* To whom correspondence should be addressed. E-mail: ayosade2003@yahoo.com. ORCID iD: A.R. Ipeaiyeda: orcid.org/0000-0001-9674-1513














