Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Chemistry
On-line version ISSN 1996-840X
Print version ISSN 0379-4350
S.Afr.j.chem. (Online) vol.69 Durban 2016
http://dx.doi.org/10.17159/0379-4350/2016/v69a25
RESEARCH ARTICLE
Identification of chelidonic acid as the predominant ligand involved in ni uptake in the hyperaccumulator berkheya coddii
Chrisanne NaickerI; Andrew KindnessII; Letitia PillayII, *
ISchool of Chemistry & Physics, University of KwaZulu-Natal, Private Bag X54001, Durban, 4000, South Africa
IIMolecular Sciences Institute, School of Chemistry, University of the Witwatersrand, Private Bag 3, Johannesburg, 2050, South Africa
ABSTRACT
Nickel uptake in Berkheya coddii, a Ni hyperaccumulator (>3 % Ni m/m), was studied using plants from serpentine outcrops in Barberton, Mpumalanga Province, South Africa. Size exclusion chromatography (SEC), high resolution mass spectrometry (HR-MS) and high performance liquid chromatography (HPLC) were used to identify and quantify organic and amino acids typically associated with hyperaccumulation. Calculated molar ratios were used to identify potential Ni-ligand associations with amino and organic acids. The former showed no single acid present at levels sufficient to complex adequately with the high levels of Ni in B. coddii. Only elevated proline concentrations in younger B. coddii leaves were recorded; however, proline may be produced as a stress response to elevated metal concentrations. A combination of chromatography and spectroscopy led to the identification of chelidonic acid as the ligand playing a significant role in Ni uptake in B. coddii. Chelidonic acid and Ni in leaves were quantified in a 3:1 molar ratio. However, during spiking experiments where soluble Ni was added to soil, the chelidonic acid and Ni concentrations increased to a 6:1 molar ratio. The other amino and organic acids present in the plant showed no response to an increase in soluble Ni, thus indicating chelidonic acid has a role in the B. coddii hyperaccumulation process. This finding is the first to link chelidonic acid to hyperaccumulation and will have significant impact on the potential of B.coddii for phyto-remediation.
Keywords: Berkheya coddii Roessler, hyperaccumulator, Ni uptake, organic acids, amino acids, chelidonic acid.
1. Introduction
Plants have the ability to regulate appropriate concentration levels of essential metals, e.g. Ni (<500 mg kg-1), Fe, Mn, Cu and Zn (10-1000 mg kg-1), as well as non-essential metals and metalloids, e.g. Hg, Cd, Pb and As (<100 mg kg-1).1 Essential metals play an important role in enzymatic processes critical to plant sustainability; however, high concentrations may have adverse effects ultimately leading to plant death.2 Plants can exhibit one of three responses when exposed to high levels of metals in soil, i.e. exclusion (metal is not transported to above ground tissue), unrestricted uptake (metal taken up until plant dies) and hyperaccumulation (high levels of the metal stored within plant tissue).3 Hyperaccumulating plants are able to tolerate and accumulate highconcentrations ofparticularmetals without experiencing toxic effects.4 The process of hyperaccu-mulation is deemed an extreme physiological response in heavy metal tolerance.5 Ni is the most common element involved in hyperaccumulation and there are approximately 450 reported Ni hyperaccumulators.6 A Ni hyperaccumulator is defined as having a 'concentration of greater than 1000 mg Ni kg-1 dry leaf tissue'.4 This threshold was set at 100-1000 times higher than the normal Ni concentration in plants grown on non-metalliferous soils and 100 times greater than the expected levels from non-accumulating plants on serpentine soils.7
Plants contain a number of potential ligands, e.g. organic acids, amino acids, proteins and peptides which are required for physiological processes. These ligands also play a role in mechanisms of metal uptake in both hyperaccumulator and non-hyperaccumulator plants. Most metals are considered to be bound to low molecular mass compounds or proteins.8 Several organic acids and amino acids have been reported to bind to the accumulated metal in hyperaccumulating plants.9 Ni has been associated with complex formation with the following organic acids: citric acid,9-10 malic acid10c, 11 and a malate/citrate combination.12Alyssum species analyzed using XAS to investigate the coordination of Ni within the plant species, were reported to contain Ni(His)2 complexes.13 However, upon analyses using XANES and EXAFS, Montargès-Pelletier et al. found that it was not histidine responsible for complexing to Ni, but rather malate ligands.12 The hyperaccumulators Alyssum lesbiacum, Pisum sativum and Brassica juncea have a directly proportional relationship between histidine and Ni concentration.13-14
1.1. Berkheya coddii
Berkheya coddii has been extensively studied as a potential candidate for phytoremediation and phytomining (metals extracted from soil and used for commercial processes.15 However, the mechanism which allows B. coddii to accumulate Ni is not well understood. Manipulating the mechanism of Ni uptake, translocation and storage may enhance phytoextraction potential of the plant.16 Several studies involving the storage of Ni within the plant were carried out to gain insight into possible mechanisms. Dimethylglyoxime (DMG) staining has been used to identify Ni deposits within the plant.16 This technique is inadequate in terms of quantifying Ni in the plant and can thus be used only for the sole purpose of qualitative identification.17Using micro-particle induced X-ray emission (PIXE) analysis of the plant showed that most Ni is stored in tissues that have no physiological role within plant processes; however, some Ni was present in areas of the plant that are involved in transport and detoxification.18 Previous work has also indicated that Ni is mainly stored in the outer cuticles within the leaf and has a high presence in the xylem (the main route of transportation).19Nuclear microprobe studies indicated Ni to be concentrated in the margins and mesophyll of the leaf and in the midrib epidermis of young leaves.20 Laser ablation coupled with DMG staining and inductively coupled plasma-mass spectrometry (ICP-MS) on the roots gave an insight into the plant's uptake mechanism. Ni was concentrated in the stele (central part of stem) as opposed to the cortex of the roots, which indicated the presence of a selective or active uptake mechanism.21 Contradicting results were reported from studies using magnetic resonance imaging (MRI) on the roots, with results indicating a passive uptake mechanism and a specific pattern of Ni concentration at the plane of the roots.22 Robinson el al.23 reported that the higher the content of Ni in the soil, the lower uptake of macro and micronutrients in the plant. Studies involving the effects of Ni uptake on insects were also investigated. The amount of Ni present in insects that feed on B. coddii could have an impact on metal movement into other ecosystems, thus affecting food webs.24 An early study into the uptake mechanism of B. coddii reported malic acid to be responsible for Ni uptake in the plant.25 However, later analysis calculated a Ni:malic acid molar ratio of (1:0.013), which indicated that malic acid could not be involved in the uptake mechanism.26B. coddii was also studied as a control plant to investigate the coordination of Ni to nicotianamine.7 The study reported nicotianamine as the ligand potentially responsible for binding to Ni in B. coddii but did not present conclusive quantification of molar ratios to confirm this.
Understanding how Ni is accumulated within B. coddii would be very useful for the plants role in phytoremediation. The objectives of this study were to identify and quantify the predominant ligand responsible for complexing Ni in the leaves of B. coddii.
2. Materials and Methods
2.1. Experimental Site and Facilities
Sampling was carried out in Barberton, Mpumalanga Province, South Africa. B. coddii plants were collected from the sites in January (summer). Samples of ultramafic soil were also collected, along with the plants. Plants were transplanted into large pots along with the ultramafic soil collected. B. coddii plants were watered once a day with tap water and the leaves sprayed using a plant mister. The plants grew well in a warm environment. All acids and chemicals used were purchased from Sigma-Aldrich unless stated otherwise.
2.2. Amino Acid Analysis in B. coddii
Three plants grown from seed were harvested at 16 weeks. Plants were separated into roots, stems, young leaves, middle leaves and bottom leaves (oldest). Sections of the plant were thoroughly washed with tap water to remove any soil and dirt residue. Extraction by grinding in an agate mortar and pestle with millipore water followed an adapted procedure.27 The ground sample was filtered and added to 1 mL of a 50 % acetonitrile, 0.1 % formic acid solution. The solution was sonicated for an hour, then centrifuged and diluted (10-fold) throughout. A volume of 10 of the solution was added to the Waters AccQ Tag Kit constituents, and placed in a heating block at a temperature of 55 °C for 10 min. Heated samples (three replicates) were then analyzed using LC-MS.28 Mass spectral analysis (HR-MS) was carried out on a Waters Synapt G2 connected to a Waters UPLC. The conditions were as follows; column: Waters BEH C18, 2.1 x 100 mm, mobile phase of 70 % water/30 % methanol, a flow rate of 0.25 mL min-1, ES negative mode, with a capillary voltage of 3 kV and a cone voltage of 15 V. Nickel from the extract was analyzed using a Perkin Elmer, Optical Emission Spectrophotometer (OES), Optima 5300DV in axial mode at wavelength of 231.06 nm.
2.3. Organic Acid Analysis
A mass of 10 g of Sephadex G-10 was used as the stationary phase. An aqueous, 4 M solution of a Tris(hydroxymethyl) methylamine solution was made using Millipore water in a 100 mL volumetric flask. The pH was adjusted to 4 using concentrated HCl. The column was then used to run the plant water extracts and collect nine 5 mL fractions. An aliquot of 2 mL from the collected fraction was diluted to 10 mL and analyzed for Ni by ICP-OES (conditions listed above) and for organic acids by HPLC. HPLC analysis was carried out using a Shimadzu - Prominence LC-30 AD (UV detector SPD-M20A), with a flow rate of 1.00 mL min-1, a detection wavelength of 220 nm and a Phenomenex C18 column. The mobile phase consisted of 0.05 M KH2PO4 (pH adjusted to 2.7 with conc. H3PO4). The fractions were analyzed against a range of organic acid standards to determine which acids were detected in B.coddii plant-water extracts. In order to ensure reproducibility of retention times, fractions and organic acid standards were run a total of 5 times.
2.4. Ni Pre-treatment in B. coddii
Three B. coddii plants were grown from seedlings to 8 weeks of age. The plants were then re-potted (200 mm pots) for spiking studies. A100 mM NiCl2 solution was used to spike plants. Every second day, 20 mL of the solution was added to the plant for a total of 4 weeks. Cumulatively, a total of 280 mL (28 mmol) of the solution was added to each plant. Leaves (3-5) from each plant were randomly collected every 7 days, combined, and extracted using the method previously described. Control plants were also present with no addition of Ni solution. Analysis to quantify the Ni and chelidonic acid were carried out in triplicate by ICP-OES and HPLC, respectively.
3. Results and Discussion
3.1. Amino Acids in B. coddii
A total of 12 free amino acids were detected and quantified in various plant sections, i.e. roots, stems, bottom leaves (oldest), middle leaves, top leaves (youngest) and seeds of B. coddii (no Ni pre-treatment, i.e. no extra addition of Ni, 16 weeks old). This amino acid study was performed to ascertain any possible role that amino acids may have on Ni uptake in B. coddii.
Nickel concentrations in leaves of B. coddii increase as the leaf grows and ages (Table 1). The larger the leaf surface area, the higher the Ni accumulation. Amino acid concentrations in the plant correlate with findings in other Ni hyperaccumulators where they play no role in coordination.7 All amino acids except glutamine were present in higher concentrations in younger leaves compared to older leaves. With the exception of proline, amino acid concentrations were higher in the seeds compared to leaves, stems and roots. Concentrations in roots and stems were lower than other plant sections. High proline concentrations were apparent in young and middle leaf sections. Proline typically accumulates in plants that undergo stress.29 These stresses can be brought about by drought, high levels of salt, excessive light, oxidative and biotic stresses and heavy metal stresses.30 In addition, the function of proline in the younger plants is to protect enzyme activities and protein integrity.31 Younger B. coddii plants experience more stress to metal accumulation than older plants due to the sudden response to accumulation of metals.
For proline to be involved in Ni uptake and storage it needs to bind in at least a 1:1 molar ratio. Molar ratios between Ni and proline did not yield any ratios of significance, particularly for the young leaf (0.006535 Pro:1 Ni) and middle leaf (0.0001468 Pro:1 Ni). This indicates that proline is not responsible for binding to Ni in B. coddii, but involved in physiological processes due to the stressful environment. Proline distribution suggests that as B. coddii grows, physiological processes within the leaves adapt to Ni accumulation. This suggests that a larger concentration of the Ni binding ligand could be present in older leaves thus relieving plant stresses and requiring lower concentrations of proline within the leaves.32
Other amino acid concentrations do not appear to be significant. Concentrations of histidine in B. coddii correlate with typical levels in plants of <400 mg kg-1.33 Histidine levels present in the plant indicate that the acid functions as a plant growth and aid development.34 A study carried out by Nasibi et al.35 reported that arginine played a role in Ni accumulation in plants by producing nitric oxide donors that assist with reducing Ni toxicity. The concentrations of arginine measured suggest that it is involved in combating Ni toxicity since concentrations are higher in the leaves. Asparagine has been linked to glutamine and arginine as the major nitrogen transporters in plants.36Asparagine and glutamine are known to be present in high concentrations in seeds of plants and this is reflected in the concentrations present in B. coddii seeds.37
Cysteine concentration compared to other amino acids in its cycle is low. Cysteine is generally associated with high sulphur content and sulphur ligands, which have been reported not to be present in most Ni hyperaccumulators.3 Amino acid analysis did not yield any substantial concentrations of nicotianamine, the ligand previously reported to be responsible for Ni coordination in B. coddii.7Nicotianamine is an amino acid (originating from its precursor methionine) in the oxaloacetate cycle that occurs in plants. Methionine concentrations in various sections of B. coddii are <100 mg kg-1. If nicotianamine was being produced for Ni uptake, concentrations of methionine should be high enough to reach a molar ratio of 1:1. The calculated molar ratio 1.54 x 10-6:1 (methionine to Ni), indicates that this is not the case.
Berkheya coddii seeds contain higher Ni and amino acid concentrations (apart from proline in young leaves) than the stems and roots; this may be due to the plant requiring Ni and sufficient amino acid concentrations for its physiological processes during germination. The high amino acid levels in the seeds are present to combat any toxic effects Ni will have on the plant during germination. However, these results contradict those of Groeber et al.38who found no requirement for Ni during the germination of seeds of B. coddii, although concentrations of Ni in seeds (cotyledons mainly) were 0.4-0.5 %. Roots and stems have lower Ni concentrations because they have no apparent role in the storage process.
Molar ratios between amino acids and Ni were calculated, with none being of any significance to indicate a role in Ni binding. In order to fully understand the relationship between amino acids and Ni uptake, further work should be carried out (over an extended period).
3.2. Organic Acids in B. coddii
Organic acids in B. coddii (root, stem and leaves) were analyzed using RP-HPLC to identify potential ligands responsible for Ni uptake and to ascertain their distribution within the plant. Twenty-five organic acids that play a role in plant processes were investigated with those detected (Table 2).
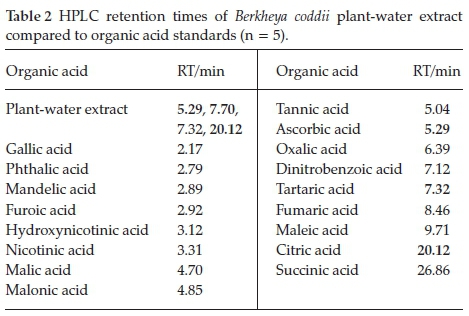
A previous study into organic acids present in the leaves of B. coddii indicated an unidentifiable peak present in the sample.32In this study, investigation showed that this previously unidentified peak (at 7.70 min) was unmatched with any of the listed organic acids. The other three peaks were identified to be citric, tartaric and ascorbic acid and were subsequently quantified. Quantification yielded molar ratios <<<1:10-8 indicating that the identified organic acids (citric, tartaric and ascorbic acid) do not play a major role in Ni accumulation. The peak at 7.70 min did not match the retention times of other organic acids tested.
3.3. Extraction and Isolation of Unknown Ni-ligand
A plant-water extract was passed through a size exclusion column. Nine fractions of 5 mL were collected. Each fraction was analyzed for Ni by ICP-OES and organic acids using RP-HPLC. Fractions 3 and 4 had the highest concentrations of both Ni and the unidentified peak from the HPLC chromatogram (7.70 min).
The UV spectra (Fig. 1) obtained for the unidentified peak in both fractions match the spectrum reported by Pillay.26 These findings suggest a relationship between Ni and the unknown peak in B. coddii. Previous findings show that Ni is extracted most abundantly in water and indicates the ligand is polar in nature.26
3.4. Identification of Chelidonic Acid
High resolution-mass spectrometry (HR-MS) carried out on the unidentified peak of the plant-water extract indicated that the ligand had a molar mass of 184.10 g mol-1 and a molecular formula C7H4O6. MS-MS was then performed after HR-MS to elucidate the unknown compound based on smaller fragmentation patterns. No metal isotopes were detected in either of the spectra indicating that the complex dissociated upon interaction with the LC column. The preliminary structure of chelidonic acid was confirmed using RP-HPLC. A comparison of the plant-water extract to a standard of chelidonic acid was carried out. The standard chromatogram and UV spectrum matched those of the plant water extract. Lambda max values obtained for the chelidonic acid correlate with the values obtained in the plant extract viz. 196, 224 and 273 nm. Additionally, a sample of synthesized chelidonic acid was analyzed by HR-MS and confirmed that the compound in the extract was chelidonic acid (Fig. 2). Based on retention time, matching UV spectra (Fig. 1) and HR-MS, the unknown organic acid peak in the plant-water extract was confirmed as chelidonic acid.
3.5. Quantification of Chelidonic Acid in B. coddii
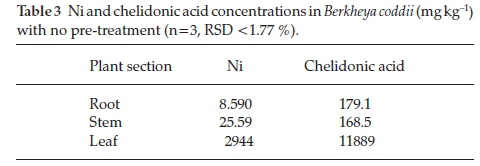
Chelidonic acid and Ni concentrations in the root, stems and leaves of B. coddii were quantified. Plant sections were exhaustively extracted and analyzed by HPLC and ICP-OES. The first extraction yielded 99.97 % chelidonic acid and 98.56 % Ni.
Ni has been reported to be stored within the leaves ofB. coddii.23The concentration of chelidonic acid in leaves of B. coddii is approximately 10-fold higher compared to other plant sections (Table 3) indicating that the primary location of the acid is within the leaves. Reported chelidonic acid concentrations in leaves, stems and root of non-hyperaccumulators are <200 mg kg-1indicating that it plays a role in the hyperaccumulation process.39
3.6. Effect of Ni Pre-treatment on Chelidonic Acid and Amino Acid Concentrations in B. coddii
Pre-treatment of B. coddii plants with Ni was carried out to examine the relationship between Ni and chelidonic acid. B. coddii was spiked with a 100 mM Ni solution over the period of a month to investigate the relationship between Ni and chelidonic acid (Fig. 3).
The control plant (grown in ultramafic soil) showed a gradual increase in Ni concentrations over the 28-day period at an average rate of approximately 3 mg kg-1 per day. Plants that were exposed to soluble Ni showed a distinct difference in uptake and over the 28 days, the average intake was approximately 34 mg kg-1 per day (Fig. 3a). The plants response to addition of soluble Ni is clearly to increase the Ni uptake. Chelidonic acid, measured in conjunction with the Ni was also quantified (RP-HPLC). Results over a period of 28 days show a sharp increase which resembles an exponential trend in chelidonic acid concentration when treated with Ni (n =3, %RSD = 0.093) compared to a gradual increase in the control (n = 3, %RSD = 0.015) where Ni also gradually increased (Fig. 3b).
Molar ratios indicate whether a ligand is able to complex to a metal by deducing if there are sufficient reactants to allow complexation to occur. A ratio equal to or higher than the ratio required for a ligand and metal to complex indicates that complexation is able to occur. Ligands responsible for complexation in B. coddii are produced in excess.31 Molar ratios calculated between chelidonic acid and Ni show significant changes to the ratio over the 28-day period when the plant was exposed to soluble Ni (Table 4).
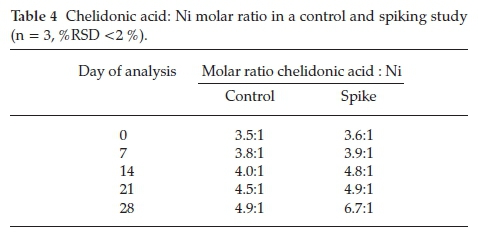
Typically, a chelidonic acid Ni-complex forms in a 2:1 molar ratio.40 In this study, as the volume of Ni addition increases, the chelidonic acid production increases over 3-fold for the pre-treated plants compared to the control, and on the last day of the experiment the ratio is 6:1 (pre-treated plant). The increase suggests that chelidonic acid plays a role in Ni uptake in B. coddii. There are significant differences in the molar ratios between the control and spiked plants. The control shows a gradual increase over the month of the study while spiking increases the ratio. These data indicate that B. coddii responded to an increase in soluble Ni by producing excess concentrations of chelidonic acid. Amino acid concentrations on day 0 and day 28 of the treatment were analyzed to ascertain the effects of soluble Ni (spikes) on the response of amino acids (Table 5).

These preliminary results showed that there are no significant changes in amino acid concentrations after 28 days, thus indicating no amino acid response to the additional soluble Ni. These minor changes to amino acid concentrations were similarly reported in a study carried out on another Ni hyperaccumulator Stackhousia tryonii by Bhatia et al.41A pre-treatment study over a longer period (6 months) may be able to deduce more comprehensively the type of roles amino acids play in Ni uptake. The lack ofamino acid response in comparison to the chelidonic acid response suggests that chelidonic acid plays a major role in Ni uptake in B. coddii.
4. Conclusion
This study reports the first association of chelidonic acid to Ni hyperaccumulator plants. It effectively shows that previously reported ligands associated with uptake in B.coddii are unlikely. Neither malic acid nor nicotianamine (previously reported as ligands responsible for Ni uptake) are present in sufficient amounts to adequately complex Ni. Amino acid analyses suggest that despite the significant role they play in plant growth, their influence on Ni hyperaccumulation in B. coddii is not significant.
The ligand predominantly responsible for Ni binding was identified as chelidonic acid. This was confirmed by HR-MS and HPLC. Further, chelidonic acid appears to respond to increased Ni concentrations in the plant while no other amino or organic acids tested had significant responses. The chelidonic acid:Ni molar ratios fall within a range that can be expected for Ni complexation. This finding represents a significant impact on the potential use of B. coddii in phytoremediation.
A number of additional experiments (e.g. solubilizing Ni or adding chelidonic acid to soil), may allow for enhanced Ni uptake at contaminated sites. More detailed analysis of amino acid response to the addition of soluble metals should be carried out to establish comprehensively their roles within this plant. Confirmation of chelidonic acid as having a major role that binds to Ni should also be confirmed by EXAFS. Additionally, other plants known to contain significant amounts of chelidonic acid, e.g. Chelidonium majus could be studied to ascertain their Ni accumulation capability.
Acknowledgements
The authors acknowledge the Central Analytical Facility, Stellenbosch University, for amino acid analysis. Laboratory work was carried out at facilities at UKZN.
References
1 C. Anderson, A. Deram, D. Petit, R. Brooks, R. Stewart and R. Simcock, in Trace Elements in Soils: Bioavailability, Flux and Transfer, (I. Iskandar, M. Kirkham, eds.), vol. 4, Lewis Publishers, Boca Raton, Florida, USA, 2001, pp. 63-76. [ Links ]
2 W. Wei, Y. Wang, Z.-G. Wei, H.-Y. Zhao, H.-X. Li and F. Hu, Roles of organic acids and nitrate in the long-distance transport of cobalt in xylem saps of Alyssum murale and Trifolium subterraneum, Biol. Trace Elem. Res., 2009,131, 165-176. [ Links ]
3 D.L. Callahan, A.J. Baker, S.D. Kolev and A.G. Wedd, Metal ion ligands in hyperaccumulating plants, J. Biol. Inorg. Chem., 2006, 11, 2-12. [ Links ]
4 R. Brooks, J. Lee, R.D. Reeves and T. Jaffré, Detection of nickeliferous rocks by analysis of herbarium specimens of indicator plants, J. Geochem. Explor, 1977, 7, 49-57. [ Links ]
5 A.J. Baker, Accumulators and excluders - strategies in the response of plants to heavy metals, J. Plant Nutr., 1981, 3, 643-654. [ Links ]
6 A. van der Ent, A.M. Baker, R. Reeves, A.J. Pollard and H. Schat, Hyperaccumulators of metal and metalloid trace elements: facts and fiction, Plant Soil, 2013, 362, 319-334. [ Links ]
7 D. Callahan, The Coordination of Nickel in Hyperaccumulating Plants, Ph.D. thesis, University of Melbourne, Melbourne, 2007. [ Links ]
8 D.E. Salt, R.C. Prince, A.J. Baker, I. Raskin, I.J. and Pickering, Zinc ligands in the metal hyperaccumulator Thlaspi caerulescens as determined using X-ray absorption spectroscopy, Environ. Sci. Technol., 1999, 33, 713-717. [ Links ]
9 D. Schaumlöffel, L. Ouerdane, B. Bouyssiere and R. Lobinski, Speciation analysis of nickel in the latex of a hyperaccumulating tree Sebertia acuminata by HPLC and CZE with ICP MS and electrospray MS-MS detection, J. Anal. Atom. Spectrom, 2003,18, 120-127. [ Links ]
10 a) J. Lee, R.D. Reeves, R.R. Brooks and T. Jaffré, Isolation and identification of a citrato-complex of nickel from nickel-accumulating plants, Phytochemistry, 1977,16,1503-1505; [ Links ] b) J. Lee, R.D. Reeves, R.R. Brooks and T. Jaffré, The relation between nickel and citric acid in some nickel-accumulating plants, Phytochemistry, 1978, 17, 1033-1035; [ Links ] c) F.A. Homer, R.D. Reeves, R.R. Brooks and A.J. Baker, Characterization of the nickel-rich extract from the nickel hyperaccumulator Dichapetalum gelonioides, Phytochemistry, 1991, 30, 2141-2145; [ Links ] d) S. Sagner, R. Kneer, G. Wanner, J.-P. Cosson, B. Deus-Neumann and M.H. Zenk, Hyperaccumulation, complexation and distribution of nickel in Sebertia acuminata, Phytochemistry, 1998, 47, 339-347; [ Links ] e) S. Bidwell, University of Melbourne (Melbourne), 2000. [ Links ]
11 a) R. Brooks, S. Shaw and A. Asensi Marfil, The chemical form and physiological function of nickel in some Iberian Alyssum species, Physiol. Plantarum, 1981, 51,167-170; [ Links ] b) R. Gabbrielli, C. Mattioni and O. Vergnano, Accumulation mechanisms and heavy metal tolerance of a nickel hyperaccumulator, J. Plant Nut.r, 1991, 14, 1067-1080. [ Links ]
12 E. Montargès-Pelletier, V. Chardot, G. Echevarria, L. J. Michot, A. Bauer and J.-L. Morel, Identification of nickel chelators in three hyperaccumulatingplants: an X-ray spectroscopic study, Phytochem., 2008, 69, 1695-1709. [ Links ]
13 U. Krämer, J.D. Cotter-Howells, J.M. Charnock, A.J. Baker and J.A.C. Smith, Free histidine as a metal chelator in plants that accumulate nickel, Nature, 1996, 635-638. [ Links ]
14 a) Y. Kameda, E. Ito, A. Takeshita, R. Nakazawa and H. Takenaga, Detoxicational effect of the exogenous histidine on the growth of pea (Pisum sativum L.) treated with nickel, J. Agri. Sci. Tokyo, 2002, 47, 45-8; [ Links ] b) L. Kerkeb and U. Krämer, The Role of free histidine in xylem loading of nickel in Alyssum lesbiacum and Brassica juncea, Plant Physiol., 2003,131, 716-724. [ Links ]
15 a) B. Robinson, R. Brooks, A. Howes, J. Kirkman and P. Gregg, The potential of the high-biomass nickel hyperaccumulator Berkheya coddii for phytoremediation and phytomining, J. Geochem. Explor., 1997, 60, 115-126; [ Links ] b) R.R. Brooks, Plants that Hyperaccumulate Heavy Metals: their Role in Phytoremediation, Microbiology, Archaeology, Mineral Exploration and Phytomining, CAB International, 1998; [ Links ] c) R.R. Brooks, M.F. Chambers, L.J. Nicks and B.H. Robinson, Phytomining Trends Plant Sci., 1998, 3, 359-362; [ Links ] d) D.E. Salt, R. Smith, I. Raskin, Phyto-remediation, Annual Rev. Plant Biol., 1998, 49, 643-668; [ Links ] e) R. Brooks, B. Robinson and A. Howes, A. Chiarucci, An evaluation of Berkheya coddii Roessler and Alyssum bertolonii Desv. for phytoremediation and phytomining of nickel, S. Afr. J. Sci., 2001, 97, p. 558-560. [ Links ]
16 S. Keeling, R. Stewart, C. Anderson and B. Robinson, Nickel and cobalt phytoextraction by the hyperaccumulator Berkheya coddii: implications for polymetallic phytomining and phytoremediation, Int. J. Phytoremediat, 2003, 5, 235-244. [ Links ]
17 L. L'Huillier, J. d'Auzac, M. Durand and N. Michaud-Ferrière, Nickel effects on two maize (Zea mays) cultivars: growth, structure, Ni concentration, and localization, Can. J. Bot., 1996, 74, 1547-1554. [ Links ]
18 A. Gramlich, A.B. Moradi, B.H. Robinson, A. Kaestner and R. Schulin, Dimethylglyoxime (DMG) staining for semi-quantitative mapping of Ni in plant tissue, Environ. Exp. Bot., 2011, 71, 232-240. [ Links ]
19 J. Mesjasz-Przybylowicz and W. Przybylowicz, PIXE and metal hyperaccumulation: from soil to plants and insects, X-Ray Spectrom., 2011,40, 181-185. [ Links ]
20 J.S. Angle, A.J. Baker, S.N. Whiting and R.L. Chaney, Soil moisture effects on uptake of metals by Thlaspi, Alyssum, and Berkheya, Plant Soil, 2003, 256, 325-332. [ Links ]
21 J. Mesjasz-Przybylowicz, W. Przybylowicz and C. Pineda, Nuclear microprobe studies of elemental distribution in apical leaves of the Ni hyperaccumulator Berkheya coddii, S. Afr. J. Sci., 2001, 97, 591-593. [ Links ]
22 A.B. Moradi, S. Swoboda, B. Robinson, T. Prohaska, A. Kaestner, S.E. Oswald, W.W. Wenzel and R. Schulin, Mapping of nickel in root cross-sections of the hyperaccumulator plant Berkheya coddii using laser ablation ICP-MS, Environ. Exp. Bot., 2010, 69, 24-31. [ Links ]
23 A. Moradi, S. Oswald, J. Nordmeyer-Massner, K. Pruessmann, B. Robinson and R. Schulin, Analysis of nickel concentration profiles around the roots of the hyperaccumulator plant Berkheya coddii using MRI and numerical simulations, Plant Soil, 2010, 328, 291-302. [ Links ]
24 B. Robinson, E. Lombi, F. Zhao and S. McGrath, Uptake and distribution of nickel and other metals in the hyperaccumulator Berkheya coddii, New Phytol., 2003,158, 279-285. [ Links ]
25 R.S. Boyd, M.A. Davis, M.A. Wall and K. Balkwill, Host-herbivore studies of Stenoscepa sp. (Orthoptera: Pyrgomorphidae), ahigh-Niherbivore of the South African Ni hyperaccumulator Berkheya coddii (Asteraceae), Insect Sci., 2007,14, 133-143. [ Links ]
26 K. Slatter, Nickel Accumulation and Tolerance in Berkheya coddii and its Application in Phytoremediation, M.Sc thesis, University of Natal, Pietermaritzburg, 1998. [ Links ]
27 L. Pillay, Uptake and Distribution in the Ni Hyperaccumulator Berkheya coddii, M.Sc thesis, University of Natal (Howard College), Durban, 2005. [ Links ]
28 B.K. Pramanik, Seasonal fluctuations of some sucrose metabolizing enzymes and sugar andorganic acid contents in broccoli, Asian J. Plant Sci. (Pakistan), 2004. [ Links ]
29 Waters, Accq.Fluor Reagent Kit, http://www.waters.com/webasset-s/cms/support/docs/wat0052881.pdf - 28 March [ Links ]
30 N. Verbruggen and C. Hermans, Proline accumulation in plants: a review, Amino Acids, 2008, 35, 753-759. [ Links ]
31 P. Hare and W. Cress, Metabolic implications of stress-induced proline accumulation in plants, Plant Growth Regul., 1997, 21,79-102. [ Links ]
32 P.K. Kishor, S. Sangam, R. Amrutha, P.S. Laxmi, K. Naidu, K. Rao, S. Rao, K. Reddy, P. Theriappan and N. Sreenivasulu, Regulation of proline biosynthesis, degradation, uptake and transport in higher plants: its implications in plant growth and abiotic stress tolerance, Curr. Sci., 2005, 88, 424-438. [ Links ]
33 S.S. Sharma and K.-J. Dietz, The significance of amino acids and amino acid-derived molecules in plant responses and adaptation to heavy metal stress, J. Exp. Bot., 2006, 57, 711-726. [ Links ]
34 K.H. Richau, A.D. Kozhevnikova, I.V. Seregin, R. Vooijs, P.L. Koevoets, J.A.C. Smith, V.B. Ivanov and H. Schat, Chelation by histidine inhibits the vacuolar sequestration of nickel in roots of the hyperaccumulator Thlaspi caerulescens, New Phytol., 2009, 183, 106-116. [ Links ]
35 a) R. Muralla, C. Sweeney, A. Stepansky, T. Leustek and D. Meinke, Genetic dissection of histidine biosynthesis in Arabidopsis, Plant Physiol., 2007, 144, 890-903; [ Links ] b) D. Bikard, D. Patel, C. Le Metté, V. Giorgi, C. Camilleri, M. J. Bennett and O. Loudet, Divergent evolution of duplicate genes leads to genetic incompatibilities within A. thaliana, Science, 2009, 323, 623-626. [ Links ]
36 F. Nasibi, T. Heidari, Z. Asrar and H. Mansoori, Effect of arginine pre-treatment on nickel accumulation and alleviation of the oxidative stress in Hyoscyamus niger, J. Soil Sci. Plant Nutr., 2013, 13,680-689. [ Links ]
37 a) T. Kato, Major nitrogen compounds transported in xylem vessels from roots to top in citrus trees, Physiol. Plantarum, 1981, 52, 275-279; [ Links ] b) M. Aidar, S. Schmidt, G. Moss, G. Stewart and C. Joly, Nitrogen use strategies of neotropical rainforest trees in threatened Atlantic Forest, Plant Cell Environ., 2003, 26, 389-399; [ Links ] c) P. Escher, M. Eiblmeier, I. Hetzger and H. Rennenberg, Spatial and seasonal variation in amino compounds in the xylem sap of a mistletoe (Viscum album) and its hosts (Populus spp. and Abies alba), Tree Physiol., 2004, 24, 639-650; [ Links ] d) P. Millard, R. Wendler, G. Grassi, G.-A. Grelet and M. Tagliavini, Translocation of nitrogen in the xylem of field-grown cherry and poplar trees during remobilization, Tree Physiol., 2006, 26,527-536; [ Links ] e) M. Nahm, T. Holst, A. Matzarakis, H. Mayer, H. Rennenberg and A. Geßler, Soluble N compound profiles and concentrations in European beech (Fagus sylvatica L.) are influenced by local climate and thinning, Eur. J. For. Res., 2006, 125, 1-14. [ Links ]
38 J. Gifford, Amino acid utilization in seeds of Loblolly pine during germination and early seedling growth (I. Arginine and arginase activity), Plant Physiol., 1997, 113, 1125-1135. [ Links ]
39 S. Groeber, W. Przybylowicz, G. Echevarria, E. Montarges-Pelletier, A. Barnabas and J. Mesjasz-Przybylowicz, Fate of nickel and calcium in seedlings of the hyperaccumulator Berkheya coddii during germination, Biol. Plant, 2015, 59, 560-569. [ Links ]
40 A. Leopold, F.I. Scott, W. Klein and K. Ramstad, Chelidonic acid and its effects on plant growth, Physiol. Plantarum, 1952, 5, 85-90. [ Links ]
41 M. Fainerman-Melnikova, J.K. Clegg, A.A. Pakchung, P. Jensen and R. Codd, Structural diversity of complexes between Cu (II) or Ni (II) and endocyclic oxygen- or nitrogen-containing ligands: synthesis, X-ray structure determinations and circular dichroism spectra, CrystEngComm., 2010, 12, 4217-4225. [ Links ]
42 N.P. Bhatia, K.B. Walsh and A.J. Baker, Detection and quantification of ligands involved in nickel detoxification in a herbaceous Ni hyperaccumulator Stackhousia tryonii Bailey, J. Exp. Bot., 2005, 56, 1343-1349. [ Links ]
Received 18 May 2016
Revised 12 August 2016
Accepted 12 August 2016
* To whom correspondence should be addressed. E-mail: letitia.pillay@gmail.com














