Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Chemistry
On-line version ISSN 1996-840X
Print version ISSN 0379-4350
S.Afr.j.chem. (Online) vol.68 Durban 2015
http://dx.doi.org/10.17159/0379-4350/2015/v68a31
RESEARCH ARTICLE
Adsorption of Mercury(II) onto activated carbons derived from Theobroma cacao pod husk
Charles M. KedeI, II; Peter P. NdibewuI, *; Makonga M. KalumbaI; Nikolay A. PanichevI; Horace M. NgomoIII; Joseph M. KetchaIII
IDepartment of Chemistry, Tshwane University of Technology, P.O. Box 56208, Arcadia, Pretoria, 0007 South Africa
IIDepartment of Chemistry, Faculty of Science, University of Douala, P.O. Box 24175, Douala, Cameroon
IIIDepartment of Chemistry, Faculty of Science, University of Yaoundé I, P.O. Box 812, Yaoundé, Cameroon
ABSTRACT
Activated carbon prepared from Theobroma cacao pod husk was used as a low-costbiosorbent for the removal of mercury(II) from aqueous solutions. The influence of pH and contact time on the adsorption was investigated by conducting a series of batch adsorption experiments. The equilibrium data were fitted to the Langmuir and Freundlich isotherm models. The Langmuir isotherm was found to best describe the experimental data. The amount adsorbed increased with increasing temperature. The maximum adsorption capacity of mercury was found to be 105 ng g-1 for an initial mercury concentration of 100 ppb. The kinetics of adsorption were modelled by means of the Lagergren-first-order and pseudo-second-order models. The pseudo-second-order model was found to explain the adsorption kinetics most effectively. It was also found that pore diffusion played an important role in the adsorption, and intra-particle diffusion was the rate-limiting step during the first 30 min. A FTIR study revealed that the carbonyl and sulfur functional groups present on the surface of the adsorbing material were involved in chemical interaction with mercury(II).
Keywords: Mercury, thermal desorption, adsorption, activated carbon, Theobroma cacao, pod husk
1. Introduction
Mercury (Hg) is one of the most harmful pollutants and it has become widespread in the environment mainly as a result of anthropogenic activities.1-2 Mercury has no beneficial biological function and its presence in living organisms is associated with cancer, birth defects and other undesirable outcomes.3 Mercury enters the human body mainly through sea food, drinking water and inhalation of polluted air.4 Therefore, according to the United States Environmental Protection Agency (USEPA),5 the tolerance limit for Hg2+ for drinking water is 1 L-1.Hg2+ causes damage to the central nervous system and chromosomes, impairment of pulmonary function and kidneys, chest pain and dyspnoea.6 Methylmercury (often written as methyl mercury) is an organometallic cation with the formula CH3Hg+ and is a serious bioaccumulative environmental toxicant.7 The harmful effects of methylmercury include the contamination of fish,7 an important source of protein around the world, especially for growing children. Hence, it is essential to remove Hg2+ from aqueous solution before its transport and cycling into the environment.
Conventional methods for removing Hg2+ from aqueous solutions include sulfide precipitation, ion exchange, alum and iron coagulation, adsorption on activated carbons and reverse osmosis.8 However, Hg2+ removal costs for most of these methods are still very high. This has led to the intensification of research focussed on new materials and technologies that will provide for an effective and economic removal of Hg2+ from aqueous solutions. Recent developments have centred on the investigation of non-conventional adsorbents like bentonite,9 modified forms of palm shell,10 and natural and aluminium pillared clays11 reported to be used with success to some extent for the removal of Hg2+ from aqueous solutions including waste Fe(III)/Cr(III) hydroxide.
Besides the economic implications, the use of agricultural by-products has many environmental advantages.12 In the cocoa-producing regions of the world (e.g. Cameroon, the Ivory Coast and Ghana in Africa and Brazil in the Amazon) agricultural wastes such as Theobroma cocoa pod husk are often disposed of under unsuitable conditions. These then rot, generating bad smells (unhygienic) with considerable negative impacts on the landscape or provide favourable environments for microorganisms to flourish which eventually find their way into drinking water streams. Consequently, the use of Theobroma cocoa pod husk for the purpose of removing Hg2+ from aqueous solutions presents multiple advantages from an environmental point of view. Firstly, since it is disposed of as a waste, its use would add value to an agricultural by-product. Secondly, the importance of effective and economic removal of Hg2+ cannot be overemphasized here as it is a global problem. Thirdly, and finally, Theobroma cocoa pod husk may be considered as a renewable resource because it can be replenished continuously, while the cocoa beans would continue to be traded as an important cash crop in many developing countries.
Cocoa pod husk represents between 70 and 75 % of the whole mass of the cocoa fruit, i.e. 700 to 750 kg of waste can be generated from a ton of cocoa fruit. Nevertheless, various techniques have been developed as alternative methods of disposal while creating valuable products, e.g. food antioxidants,13 dietary fibres,14 and animal feed.15 Cocoa pod husk possesses suitable characteristics making it a good precursor for the preparation of activated carbons owing to its cellulosic and hemicellulosic content.16
We reported from our preliminary findings that carbonized Theobroma cacao pod husk is effective for the treatment of dye in wastewater and the removal of As, Cd, Cr, Hg and Ni.17 Other authors have also reported the successful removal of organics, including pesticides from aqueous solutions.18 The objective of this study was to investigate the feasibility of using carbonized Theobroma cacao pod husk for the removal of Hg2+ from water by adsorption.
2. Experimental
2.1. Instrumentation
Mercury measurements from water samples were carried out after adsorption on the sorbent with a Model RA-915+ Zeeman mercury analyzer (Lumex, St. Petersburg, Russia) fitted with a PYRO-915 attachment. The working principal of the instrument is based on the thermal desorption of Hg from samples placed in two-stage pyrolysis tubes heated to 750 and 800 °C. When a known amount of sample is placed in the first preheated tube, Hg vapour, together with smoke formed after combustion of the organic materials in the matrix, are transported to the second tube with the analytical cell preheated to 800 °C. A multi-path analytical cell with a total optical length of 0.4 m allows detection of Hg (in pg) because background absorption is eliminated by the high-frequency Zeeman correction system. The concentration of Hg in the sample is determined from the integrated analytical signal, by using the built-in calibration curve, plotted with different certified reference materials (CRMs). The PYRO-915 attachment enhances Hg determination in samples with complex matrices, such as soils, sediments, oil products and foodstuff, by using the pyrolysis technique without sample pretreatment.19,20,21-22 Real-time measurements are made with visualization of the process on a computer display. With no chemical sample pretreatment and without addition of chemical modifiers, the risk of contamination is minimized.
2.2. Reagents and Reference Materials
All the chemicals used were of analytical reagent grade. Deionized doubly distilled (DDD) water was used throughout the experimental studies. Mercury standard solutions (10 mgL-1, Atomic Spectroscopy Standard, PerkinElmer, Inc, USA) were prepared by progressive dilution of a 10 mg L-1 mercury stock solution with DDD water. ACS reagent grade HCl, NaOH and buffer solutions (Merck, Germany) were used to adjust the solution pH to the required value.
The Theobroma cacao pod husks were obtained from a farm in Mbele, in the Central region of Cameroon (4°10'0.02"N and 11° 32'00"E), Africa. Primary treated municipal wastewater (MWW) samples were collected from the central municipal processing plant in Pretoria, South Africa (25.7256°S and 28.2439°E). They were filtered through Whatman paper #4 (USA) to remove suspended particles and then stored in a laboratory fridge at 4 °C until analysis within one week.
2.3. Preparation of Activated Carbon
The preparation of activated carbon was carried out according to the procedure recommended by Martinez et al.23The solid residue of Theobroma cacao was manually chosen, cleaned with deionized water, dried at 100 °C for 24 h, ground and passed through a sieve to obtain samples of 1-2.5 mm particle size. This raw material was treated with ZnCl2 and ZnCl2/FeCl3 at two different concentrations (50 and 75 %, m/m), (ZnCl2, ZnCl2/FeCl3 solution/char, m/m) and pyrolyzed in a tubular oven (Lindberg Blue), at three different temperatures (350, 450 and 550 °C). These ratios were selected from previous investigations24 which showed that the adsorption capacity increases remarkably with increasing ZnCL/char ratio. After the activation, the excess ZnCl2 and ZnCl2/FeCl3 were removed with a 0.1 M solution of hydrochloric acid (HCl), and then the product was washed with hot distilled water until a neutral pH was reached to obtain the sample for sorption measurements.
2.4. Characterization of Activated Carbons
The prepared activated carbons (ACs) from Theobroma cacao pod husk were examined with a scanning electron microscope (SEM) coupled to an X-ray (EDX) analyzer. N2-physisorption experiments (adsorption isotherms for pore size distribution (PSD) and surface area) were conducted with a Micromeritics ASAP 2010 (USA) surface area and porosity analyzer. Analyses of adsorption and desorption of nitrogen were conducted at 195 °C. Before the experiments, the sample was degassed at 200 °C overnight. Total surface areas were determined by applying the BET equation.25 External surface areas were determined by applying the t-plot model between the range 7-9 A.26 Pore size distributions were determined using both BJH methods.
Field emission scanning electron microscopy (FE-SEM) and energy-dispersive X-ray (EDX) analysis were conducted to observe the surface morphology of the ACs. The samples were gold coated to improve their conductivity to obtain good images. Elemental analysis (EA) for the carbon (C), nitrogen (N), oxygen (O), sulfur (S) and metal content of the various samples was carried out with the aid of the energy dispersive-X-ray device. The instrument used to obtain SEM images of the samples and EDS spectra was a JEOL JSM-7600F field emission scanning electron microscope, 800 mm2, X-Max coupled to a silicon drift energy dispersive X-ray detector (SDD) (Oxford Instruments Ltd, UK).
2.5. Batch Adsorption Studies
The adsorbate stock solution of 1000 mg L-1 Hg2+ was prepared from HgCl2 in distilled water. This stock solution was diluted as required to obtain standard solutions containing 10-100 ng L-1 Hg2+. Batch mode adsorption studies were carried out with 0.05 g of adsorbent and 50 mL of Hg2+ solution of a desired concentration in 100 mL conical flasks and were agitated at 160 rev min-1 for predetermined time intervals at room temperature in a mechanical shaker. The removal kinetics of Hg2+ were investigated by taking samples of the solution after the desired contact time (20-120 min) and the adsorbent was analyzed for its Hg2+ concentration with a RA-915+ Zeeman mercury analyzer.
2.6. Data Treatment
2.6.1. Isotherm Models
The sorption equilibrium data for Hg2+ on activated carbons were analyzed by means of the Freundlich and Langmuir isotherm models.27 The Freundlich isotherm equation x/m = kfC1/n can be written in the linear form as given in Equation 1.
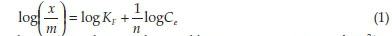
where x/m and Ce are the equilibrium concentrations of Hg2+ in the adsorbed and liquid phases in ng g-1, and KFand n are the Freundlich constants that are related to the sorption capacity and intensity, respectively. The Freundlich constants Kf and n can be calculated from the slope and intercept of the linear plot of log (x/m) against logCe.
The Langmuir sorption isotherm equation 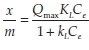 linearization becomes:
linearization becomes:

where Qmax is the maximum adsorption capacity (ng g-1) when all adsorption sites are occupied, Ce (ng L-1) is the equilibrium concentration of mercury, Qeis the equilibrium adsorption capacity and the Langmuir constant KL (dm3 ng-1) is derived from the ratio of the adsorption rate constant to the desorption rate constant.
2.6.2. Kinetics Models
Kinetics adsorption data were fitted to the pseudo-first-order kinetics model.28

where qe and qt refer to the amount of Hg2+ adsorbed (ng g-1)at equilibrium and at any time, t (min), respectively, and k1is the rate constant for pseudo-first-order sorption (min-1). Integration of Equation 3 for the boundary conditions t = 0 to t and qt = 0 to qt, gives Equation 4:

Equation 4 is rearranged to give Equation 5:

The slope of a plot of log(qe -qt) versus t was used to determine the first-order rate constant, k1. In many cases, the first-order equation of Lagergren28 does not fit well for the whole range of contact times and is generally applicable over the initial stage of the adsorption processes.29 Kinetics data were further treated with the pseudo-second-order kinetic model.29 The pseudo-second-order equation is also based on the sorption capacity of the solid phase. Contrary to the other model, it predicts the behaviour over the whole range of adsorption and is in agreement with an adsorption mechanism involving the rate-controlling step. The differential equation is given by Equation 6:

where k2is the rate constant for pseudo-second-order adsorption (g ng-1 min-1). Integrating Equation 6 for the boundary conditions t = 0 to t and qt= 0 to qt, gives Equation 7:

This is the integrated rate law for a pseudo-second-order reaction. Equation 7 can be rearranged to obtain a linear form (Equation 8):

The slope and intercept of a plot of t/qt versus t were used to calculate the second-order rate constant, k2. This model is more likely to predict the behaviour over the whole range of adsorption and is in agreement with the chemisorption mechanism being the rate-controlling step.30
2.6.3. Adsorption Mechanism
In order to gain insight into the mechanism and rate-controlling step affecting the kinetics of adsorption, the kinetics experimental results were fitted to the Weber and Morris intra-particle diffusion model,31 in which the rate of intra-particle diffusion is a function of t1/2and can be defined according to Equation 9:

where rpis the particle radius, Dtis the effective diffusivity of solutes within the particle, and kw is the intra-particle diffusion rate. Values of kwcan be obtained by linearizing the curve q = f (t1/2). Such types of plots may present multi-linearity, implying that the overall adsorption process may be controlled by one or more steps, such as film or external diffusion, intra-particle diffusion and a chemical reaction on the pore surface, or a combination of more than one step. Weber and Morris31 reported that if intra-particle diffusion was involved in the adsorption process, then a plot of the square root of time versus the adsorption amount would result in a linear relationship, and that the intra-particle diffusion would be the controlling step if this line passes through the origin.
3. Results and Discussion
3.1. Characterization of Activated Carbons
In this work, Theobroma cacao pod husk (GAC0), an industrial effluent abundantly available from the Theobroma bean processing plants, was utilized as a feedstock for preparation of activated carbon (GAC8: T = 550 °C, 75 % Zn for 3 h), GAC1 (T = 450 °C, 50 % Zn for 3 h) and GAC7 (T = 550 °C, 50 % Zn/Fe for 3h)via microwave-induced 50 and 75 % (w/w) of ZnCl2 and FeCl3 activation
3.1.1. Surface Areas and Pore Sizes
The N2 adsorption isotherms at 77 K for activated carbons produced from the Theobroma cacoa by chemical activation are presented in Fig. 1. All the samples gave type II isotherms, characteristic of microporous materials. The adsorption isotherms show adsorption-desorption hysteresis, indicating the presence of mesopores. Besides contributing significantly to the adsorption of the adsorbate, mesopores could be attributed to the interaction of functional groups at the surface of char which favours the evolution of molecules during the heating process of activation, then probably creating external pores.32 The overall porosity development determined by N2 adsorption at 77 K by the BET and BJH methods are shown in Table 1. It shows that all the samples have surface areas between 357.36 and 586.93 m2 g-1 with highly developed microporosity.
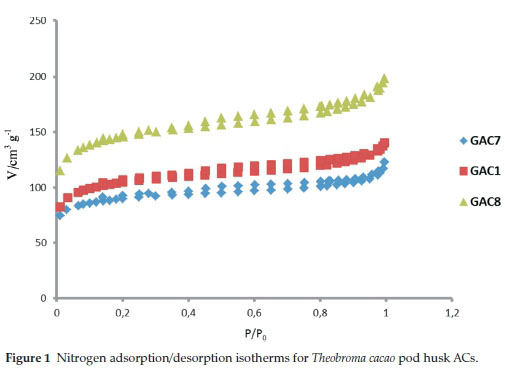
3.1.2. Functionalization
The Fourier transform infrared (FTIR) spectra of the activated carbons are shown in Fig. 2. The absorption at 2958 cm-1 indicates the presence of C-H groups.33 The peaks appearing at 1542.1 and 1504.6 cm-1 are as a result of carbonyl groups (C=O) while the bands appearing at 1057.7,1128.7 and 1162 cm-1 are ascribed to the formation of sulfur functional groups like a highly conjugated S = O.
3.1.3. Surface Morphology
To observe the morphology of the prepared AC samples, SEM images are shown in Fig. 3. Fig. 3A shows the micrograph for Theobroma cacao pod husk employed as the raw material for the preparation of the activated carbons. The material presents a surface morphology in the form of plates, possibly with a low specific surface area. The micrographs shown in Fig. 3B, C, D correspond to the materials collected after the activation process with ZnCl2 and FeCl3, with the salt mixture of 50 % Zn, 75 % Zn and 75 % Zn/Fe, respectively, for GAC1, GAC8 and GAC7. These images show that the morphology of the samples has changed after the activation process; suggesting that surface craters were produced, which contributes to the increase of the surface area after the activation. From this analysis, it is clear that the presence of zinc and iron species in the preparation of the samples produces 'craters' with thinner walls on the surfaces, thereby creating a weaker structure.
3.2. Batch Adsorption Studies
3.2.1. Effect of Contact Time
Figure 4 shows the effect of contact time on the removal of Hg2+. Experimental studies were carried out with varying adsorbents (GAC1,GAC7 and GAC8) with 100 ng L-1 of the initial mercury ion concentration. Equilibrium adsorption was established within 60 min for the metal ion. These data are important because equilibrium time is one of the parameters for economical wastewater treatment plant application in agreement with other authors.18 According to these results, the agitation time was fixed at 2 h for the rest of the batch experiments to make sure that equilibrium was attained.
3.2.2. Effect of pH
Figure 5 presents the effect of initial pH on the removal of Hg2+ by the GACs. It can be shown by speciation calculations that in the presence of Cl- the predominant species at pH > 6.0 are Hg(OH)2 and HgCl2, while at pH < 6.0 it is HgCl2.34 The formation of HgCl2 has been found to decrease the Hg2+ sorption onto a commercial FS-400 GAC.18 Accordingly, the adsorption of Hg(II) decreased when the pH was lowered from 7.0 to 2.0. On increasing the pH from 6, the adsorption capacity increased and became constant over the pH range of 8.0-12.0. This implies that Hg(OH)2 species may be retained in the micropores of carbon particles by chemisorption involving surface complexes.18 Functional groups, such as CxOH+, are assumed to be present on the surface of the carbons. When Hg2+ is present in the solution, the following surface complexes may be formed:
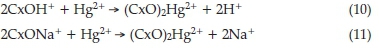
3.2.3. Equilibrium Isotherms
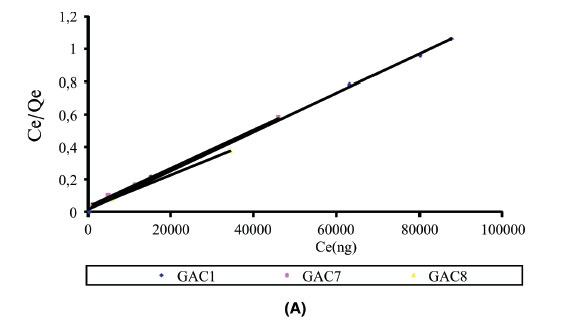
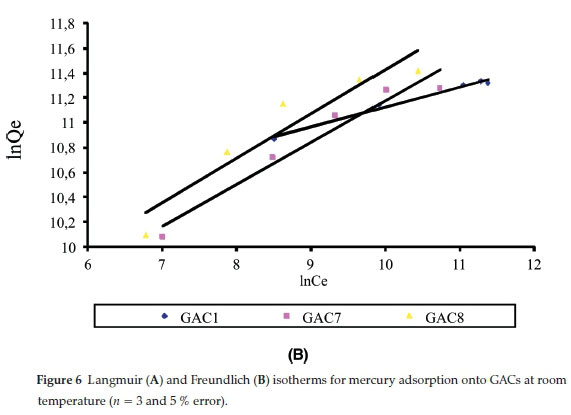
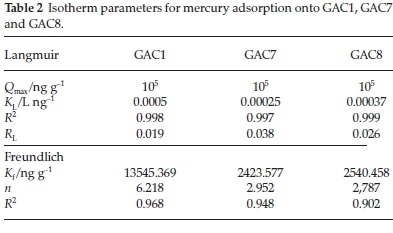
Adsorption isotherms provide information on the nature of the solute-surface interaction as well as the specific relation between the concentration of adsorbate and its degree of accumulation onto the adsorbent surface at constant temperature. These equilibrium experiments were performed at room temperature with an adsorbent mass of 0.05 g and an adsorbate concentration (C0) ranging from 25-125 ng L1. In order to understand the adsorption mechanism of mercury onto activated carbon, two adsorption isotherm models, Langmuir and Freundlich, were used to fit the experimental data. The isotherm parameters were determined by non-linear regression with Origin version 7.0, a Microsoft Windows-based statistical software. The adsorption isotherms obtained for the various GACs are illustrated in Fig. 6A and Fig. 6B. The values of the parameters and the correlation coefficients obtained for the different adsorbents are listed in Table 2. It is well known that the Langmuir model is usually used with an ideal assumption of a monolayer adsorption surface.27 The value of the correlation coefficients (R2) for the two models for different GACs show that the best fit was obtained with the Langmuir model. The essential characteristics of the Langmuir isotherm can be expressed in terms of a dimensionless constant separation factor or equilibrium parameter, RL, which is defined as RL=-where KLis 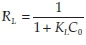 the Langmuir constant and C0 is the initial concentration of Hg2+. The RL value indicates the shape of the isotherm as follows: RL > 1, unfavourable; RL = 1, linear; 0 < RL < 1, favourable; and RL= 0, irreversible. According to Mckay et al.,29 RLvalues between 0 and 1 indicate favourable adsorption. The RLvalues for Hg2+ were 0.019, 0.038 and 0.026 for GAC1, GAC7 and GAC8, respectively, for 100 ng L-1. Hence, the adsorption of the Hg2+ on GACs seems to be favourable.
the Langmuir constant and C0 is the initial concentration of Hg2+. The RL value indicates the shape of the isotherm as follows: RL > 1, unfavourable; RL = 1, linear; 0 < RL < 1, favourable; and RL= 0, irreversible. According to Mckay et al.,29 RLvalues between 0 and 1 indicate favourable adsorption. The RLvalues for Hg2+ were 0.019, 0.038 and 0.026 for GAC1, GAC7 and GAC8, respectively, for 100 ng L-1. Hence, the adsorption of the Hg2+ on GACs seems to be favourable.
3.2.4. Kinetics Studies
In order to investigate the kinetics of adsorption of mercury, the Lagergren-first-order model28 and Ho's pseudo-second-order model30 were used. The values of the parameters and the correlation coefficients obtained by using non-linear regression with Origin version 7.0 at room temperatures are listed in Table 3. The fit of the experimental data to the kinetics models is illustrated in Fig. 7. It was found that Ho's pseudo-second-order model gave the highest values of the correlation coefficients and predicted qemore accurately than the other model investigated. Therefore, Ho's pseudo-second-order model could be used for the prediction of the kinetics of adsorption of mercury on the activated carbon.
3.2.5. Adsorption Mechanism
Figure 8 shows the intra-particle diffusion plot for the adsorption of mercury on the three activated carbons (GAC1, GAC7 and GAC8). From Fig. 8, it can be seen that the plots for GAC1, GAC7 and GAC8 show similar features; they have one linear segment followed by a plateau. In the first 60 min, the first linear portion did not pass through the origin, implying that the intra-particle diffusion did not play an important role and did not control the rate of the adsorption. After 60 min, the regression was nearly linear but did not pass through the origin, suggesting that intra-particle diffusion was not the sole rate-controlling step in this stage. The kwvalue evaluated from the first linear parts of these curves for GAC1, GAC7 and GAC8 were 13829,12123 and 13974 (ng g-1 min-0.5), respectively. The kwvalue increased with increasing impregnation of ZnCl2, which showed that the adsorption rate increased with increasing impregnation of ZnCl2. Comparing plots of GAC1, GAC7 and GAC8, the plot of GAC8 had an added curved portion in the first 60 min, showing that film diffusion played an important pole in this stage.31
4. Conclusions
The present investigation shows that the Theobroma cacao activated carbon was an effective and low-cost adsorbent for the removal of mercury(II) from dilute aqueous solution. The surface areas of the prepared activated carbons were relatively high with large pore volumes and were found to be microporous. The maximum adsorption capacity of mercury was found to be 105 ng g-1 for GACs for an initial mercury concentration of 100 ng L-1. The Langmuir and Freundlich isotherm equations were used to interpret the adsorption phenomenon of the adsorbate. The Langmuir isotherm was found to best describe the experimental data. The kinetics of adsorption was modelled with the Lagergren-first-order and pseudo-second-order equations, and the pseudo-second-order equation was found to explain the adsorption kinetics most effectively. Carbonyl and sulfur functional groups present on the surface of the adsorbents were involved in chemical interaction with mercury as revealed from the FTIR spectra. It was also found that pore diffusion played an important role in the adsorption.
Acknowledgements
This research was supported by the Universities of Yaoundé I and Douala, both in Cameroon. Appreciation is expressed to the Academy of Sciences for the Developing World (TWAS) in Trieste, Italy, for financial assistance to the main author to travel to the host institution, the Tshwane University of Technology (TUT), in Pretoria, South Africa, working under Profs R.I. McCrindle and P.P. Ndibewu. Finally, the National Research Foundation (NRF) of South Africa is thanked for having provided funds to the host institution (TUT) necessary for the procurement of chemicals used throughout the entire project.
References
1 E.G. Pacyna, J.M. Pacyna, J. Fudala, J.E. Strzelecka, S. Hlawicska and D. Panasiuk, Mercury emissions to the atmosphere from anthropogenic sources in Europe in 2000 and their scenarios until 2020, Sci. Total Environ., 2006,370, 147-156. [ Links ]
2 A. Kirby, I. Rucevska, V Yemelin, C. Cooke, O. Simonett, V. Novikov and G. Hughes, UNEP Global Mercury Assessment: Sources, Emissions, Releases and Environmental Transport, United Nations Environment Protection (UNEP), Report, 2013. [ Links ]
3 http://www.unep.org/PDF/PressReleases/GlobalMercuryAssessment2013.pdf [accessed 19 August 2013].
4 A. Bhan and N.N. Sarkar, Mercury in the environment: effect on health and reproduction, Rev. Environ. Health, 2005,20, 39-43. [ Links ]
5 J.C. Clifton II, Mercury exposure and public health, Pediatr. Clin. North Am., 2007, 54, 237-245. [ Links ]
6 U.S. EPA (U.S. Environmental Protection Agency). http://www.ep-a.gov/mercury/ [accessed 10 October 2013]. [ Links ]
7 M.H.G. Berntssen, A. Aatland and R. D. Handy, Chronic dietary mercury exposure causes oxidative stress, brain lesions, and altered behaviour in Atlantic salmon (Salmo salar) parr, Aquatic Toxicol., 2003, 65, 55-72. [ Links ]
6 WHO, Environmental Health Criteria 118, Inorganic Mercury, World Health Organization, Geneva, 1991, pp. 68-112. [ Links ]
9 J.H. Cai and C.Q. Jia, Mercury removal from aqueous solution using coke-derived sulfur-impregnated activated carbons, Ind. Eng. Chem. Res., 2010,49, 2716-2721. [ Links ]
10 T. Viraraghavan and A. Kapoor, Adsorption of mercury from wastewater by bentonite, Appl. Clay Sci., 1994, 9, 31-49. [ Links ]
11 S. Kushwaha, G. Sreelatha and P. Padmaja, Physical and chemical modified forms of palm shell: preparation, characterization and preliminary assessment as adsorbents, J. Porous. Mater., 2013, 20, 21-36. [ Links ]
12 M. Eloussaief, A. Sdiri and M. Benzina, Modelling the adsorption of mercury onto natural and aluminium pillared clays, Environ. Sci. Pollut. Res., 2013,20, 469-479. [ Links ]
13 A. Bhatnagara and M. Sillanpää, Utilization of agro-industrial and municipal waste materials as potential adsorbents for water treatment: a review, Chem. Eng. J., 2010, 157, 277-296. [ Links ]
14 A. Azizah, N. Nikruslawati and T.T. Swee, Extraction and characterization of antioxidant from cocoa by-products, Food Chem., 1999, 64, 199-202. [ Links ]
15 R. Redgwell, V. Trovato, S. Merinat, D. Curti and S. Hediger, Dietary fibre in cocoa shell: characterisation of component polysaccharides, Food Chem, 2003,81, 103-112. [ Links ]
16 E. Aregheore, Chemical evaluation and digestibility of cocoa (Theobroma cacao) by products fed to goats, Trop Anim. Health Pro., 2002, 34, 339-348. [ Links ]
17 A. Oladayo, Proximate composition of some agricultural wastes in Nigeria and their potential use in activated carbon production, J. Appl. Sci. Environ. Manag., 2010, 14, 55-58. [ Links ]
18 A. Basu, M.S. Rahaman, S. Mustafiz and M.R. Islam. Batch studies of lead adsorption from a multi-component aqueous solution onto Atlantic cod fish scale (Gadus morhua), J. Environ. Eng. Sci., 2007, 6, 455-462. [ Links ]
19 K. Kadirvelu, M. Kavipriya, C. Karthika, M. Radhika, N. Vennilamani and S. Pattabhi, Utilization of various agricultural wastes for activated carbon preparation and application for the removal of dyes and metal ions from aqueous solutions, Bioresource Technol., 2003,87, 129-132. [ Links ]
20 C. Bin, W Xiaoru and F.S.C. Lee, Pyrolysis coupled with atomic absorption spectrometry for the determination of mercury in Chinese medicinal materials, Anal. Chim. Acta., 2001,447, 161-169. [ Links ]
21 N. Panichev, M.M. Kalumba and K.L. Mandiwana, Solid phase extraction of trace amount of mercury from natural waters on silver and gold nanoparticles, Anal. Chim. Acta., 2014, 813, 56-62. [ Links ]
22 N.A. Panichev, Panichev and S.E. Panicheva, Determination of total mercury in fish and sea products by direct thermal decomposition atomic absorption spectrometry, Food Chem., 2015,166, 432-441. [ Links ]
23 S. Sholupov, S. Pogarev, V. Ryzhov, N. Moshynov and A. Stroganov, Zeeman atomic absorption spectrometer RA-915+ for direct determination of mercury in air and complex matrix samples. Fuel Process Technol, 2004, 85, 473-485. [ Links ]
24 M.L. Martinez, M.M. Torres, C.A. Guzman and D.M. Maestri, Preparation and characteristics of activated carbon from olive stones and walnut shells, Indust. Crops Products, 2006, 23, 23-28. [ Links ]
25 L.C.A. Oliveira, E. Pereiraa, I. R. Guimaraesa, A. Valloneb, M. Pereirac, J. P. Mesquitac and K. Sapag, Preparation of activated carbons from coffee husks utilizing FeCl3 and ZnCl2 as activating agents, J. Hazard Mater., 2009,165, 87-94. [ Links ]
26 C.O. Ania, B. Cabal, J.B. ParraandJ.J. Pis, Importance of hydrophobic character of activated carbons on the removal of naphthalene from the aqueous phase, Adsorp. Sci., 2007. [ Links ]
27 B.C. Lippens, & J.H. Deboer, Studies on pore systems in catalysts V The t-plot method. J. catal., 1965, 4, 319-323. [ Links ]
28 C.M. Kede, M.A. Etoh, P.P. Ndibewu, H.M. Ngomo and P.M. Ghogomu, Equilibria and kinetic studies on the adsorption of cadmium onto Cameroonian wetland clays. British J. Applied Sci. Technol., 2014, 4(7), 1070-1088. [ Links ]
29 S. Lagergren, About the theory of so-called adsorption of soluble substances, Kungl. Svenska vetakad. handl., 1898, 24, 1-39. [ Links ]
30 Y.S. Ho and G. McKay, The sorption of Lead(II) on peat, Water Res., 1999, 33, 578-584. [ Links ]
31 Y.S. Ho and G. McKay, Pseudo-second order model for sorption processes, Process Biochem., 1999, 34, 451-465. [ Links ]
32 W.J. Weber and J.C. Morris, Kinetics of adsorption on carbon from solution, J. Sanitary Eng. Div. Proceed. Am. Soc. Civil. Eng., 1963, 89, 31-59. [ Links ]
33 R.N. Abdoul, B.B. Loura, B. Abdelaziz and J.M. Ketcha, Modified composite activated carbon derived from post-consumer plastics and lignocellulosic materials; Amer. Chem. Sci. J., 2013, 3(1), 24-33. [ Links ]
34 G. Socrates, Infrared and Raman Characteristic Group Frequencies: Tables and Charts, 3rd edn., John Wiley & Sons, West Sussex, United Kingdom, 2001. [ Links ]
34 D. Mohan, V.K. Gupta, S.K. Srivastava and S. Chander, Kinetics of mercury adsorption from wastewater using activated carbon derived from fertilizer waste, Colloids Surfaces A: Physicochem. Eng. Aspects, 2001,177, 169-181. [ Links ]
Received 3 September 2013
Revised 18 April 2015
Accepted 7 August 2015
* To whom correspondence should be addressed. E-mail chacdw@upe.ac.za





![Polyaniline/SiO2 catalyzed one-pot synthesis of tetrahydrobenzo[b]pyran derivatives](/img/en/next.gif)