Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Chemistry
On-line version ISSN 1996-840X
Print version ISSN 0379-4350
S.Afr.j.chem. (Online) vol.68 Durban 2015
http://dx.doi.org/10.17159/0379-4350/2015/v68a7
RESEARCH ARTICLE
Kinetic and thermodynamic studies of the adsorption of Cr(VI) onto some selected local adsorbents
Elsay Mekonnen; Menberu Yitbarek; Tesfaye Refera Soreta*
Department of Chemistry, College of Natural Sciences, Jimma University, P.O. Box 378, Jimma, Ethiopia
ABSTRACT
In this study, locally available biomasses obtained from avocado kernel seeds (AKS), Juniperus procera sawdust (JPS) and papaya peels (PP) were investigated as adsorbents for the removal of Cr(VI). The study employed a batch method to investigate the efficiency of the adsorbents. Parameters that influence adsorption such as: pH, adsorbent dose, initial concentration of Cr(VI), contact time and temperature have been studied. Optimum parameters for the adsorption of 5 mg L-1 Cr(VI) were: pH:1, adsorbent dose: 0.5 g, contact time: 160 min and temperature: 313 K. The equilibrium data was best fitted to the Freundlich adsorption isotherm model. The results of kinetic models showed that the pseudo-second-order kinetic model was found to correlate well to the experimental data. The results of thermodynamic parameters showed that the adsorption process is feasible. The positive values of the entropy change suggest increased randomness at solid-liquid interfaces during the adsorption. The efficiencies of the adsorbents towards the removal of chromium from real samples were also examined and were found to remove chromium from waste water.
Keywords: Adsorption, chromium (VI), avocado kernel seed, papaya peel, Juniperus procera sawdust, local adsorbent, adsorption kinetic, Thermodynamic of adsorption.
1. Introduction
Chromium and its compounds are toxic when introduced into natural water from a variety of industrial wastes. The major sources are from dyeing, electroplating, leather tanning, metal processing, paint and pigments, and the textile and steel fabrication industries1. These industries may release large quantities of toxic metal wastewater effluents above the permissible limit. The permissible limit for the discharge of Cr(VI) into inland surface water in India2 and US3 is 0.1 mg L-1 and in potable water it is 0.05 mg L-1.
In general, a wide range of processes have been reported to remove Cr(VI) from water and wastewater such as chemical precipitation,4 ion-exchange5 and electrochemical methods.6 The application of such methods is however costly, lead to toxic sludge generation7 and is unaffordable for large-scale treatment of wastewater.8
The adsorption capacity of Cr(VI) depends on the types of biomaterials; coconut shell9 had 80 % removal and wheat bran10 removes chromium at pH 2. The rice bran as an adsorbent was used11 to remove the metal ions from aqueous solutions with sorption capacity of 12.341 mg g-1. Adsorption capacities of some other adsorbents of food waste such as cooked waste tea (30.39 mg g-1)12 and potato peel waste13 (8.012 mg g-1) were also reported. Along with this, agro-waste such as sunflower stems,14 was investigated for the removal of Cr(VI) with an adsorption technique. Pre-boiled sunflower stem and formaldehyde-treated sunflower stem have 4.9 and 3.6 mg g-1 adsorption capacity and 81.7 and 76.5 % removal efficiency of Cr(VI) respectively. Adsorption isotherms of chromium ions in aqueous solution on a granular activated carbon (GAC) and on a char of South African coal (CSAC) were reported. The CSAC shows a smaller adsorption capacity near 0.3 mg g-1 which slightly decreases by increasing pH and salinity.15 A variety of natural plant biomass like bael fruit16(Aegle marmelos coma) shell had shown 17.27 mg g-1 adsorption capacity. The low-cost agricultural waste by-products such as sugarcane bagasse,17, rice husk18 and neem bark19 for the elimination of heavy metals from wastewater have been investigated by various researchers.
In the present study, locally available biomasses obtained from avocado kernel seeds, Juniperus procera sawdust and papaya peels were investigated as adsorbents for the removal of Cr(VI). The adsorption of Cr(VI) from aqueous solutions and real samples under different kinetic and equilibrium conditions has been investigated in detail.
2. Materials and Methods
2.1 Chemicals
The chemicals used for this study were: sulphuric acid, H2SO4 (98 %, UNI-CHEM, Germany), hydrochloric acid, HCl (37 %, Riedel-deHaën, Germany), potassium hydroxide, KOH (90 %, BDH, England), potassium dichromate, K2Cr2O7 (99.5 %, FINKEM, England), glacial acetic acid, CH3COOH (≥99.99 %, Riedel-deHaën, Germany), sodium acetate, CH3COONa (≥98 %, Riedel-deHaën, Germany), ammonia, NH3 (23-25 %, E. Merck Ltd (India), ammonium chloride, NH4Cl (≥99.5 %, Riedel-deHaën, Germany) and methylene blue, C16H18N3ClS.3H2O (≥96 %, America Cyanamid). All chemicals used in this study were analytical reagent grade and were used without further purification.
2.2. Equipment
pH meter (HANNA instruments, pH 211), atomic absorption spectroscopy (AAS) (Model analytik Jena Nov AA 300, Germany) and UV-visible spectrophotometer (DR 5000, Hach, USA) were used.
2.3. Methods
2.3.1. Preparation of Stock Solution
A stock solution of chromium (VI) with a concentration of 1000 mg L-1 was prepared by dissolving 2.829 g of potassium dichromate (K2Cr2O7) in 1000 mL distilled water. The working solution was prepared by diluting the stock solution with distilled water to give the appropriate concentration (10 mg L-1) of the solution.
2.3.2. Preparation of Adsorbents
The adsorbents were prepared by collecting Juniperus procera sawdust (JPS) from woodwork shops (latitude 7°39'N, longitude 36°50'E), avocado kernel seeds (AKS) and papaya peels (PP) from local market area (latitude 7°40'N, longitude 36°50'E) and cutting them to small pieces, followed by washing in distilled water and drying for three days in open air. The prepared adsorbents were further dried in an oven at 105 °C until the mass of the adsorbents remain constant (approximate time: for PP 7 h, AKS 26 h, JPS 0.5 h). Then the adsorbents were gound and the resulting powders were sieved by the standard test sieve of 150, μm mesh size for the adsorption study. Different physical characteristics of the adsorbents were determined and are reported in Table 1. The surface area of the adsorbents were determined by the methylene blue adsorption method20 and the point of zero charge was determined by the solid addition method.21

2.3.3. Determination of Concentration of Chromium by Atomic Absorption Spectroscopy (AAS)
Flame atomic absorption spectroscopy was used for the analysis of chromium at the optimized parameters at wavelength of 357.9 nm, slit width of 0.2 nm, current 5 mA, burner height 8 mm.
2.3.4. Determination of Adsorption Efficiency
The adsorption efficiency studies were carried out after adjusting the pH values before and after adding a measured mass of the adsorbent of interest into 30 mL of the metal ion solution and shaken in thermostatic water bath shaker at the speed of 200 rpm. Depending on the parameters under study, the pH, adsorbent dose, initial metal ion concentration, contact time or temperature was varied to study the adsorption efficiencies. The residue was filtered after the time of adsorption had been completed. The final concentration of chromium left in the filtrates was analyzed using AAS. Then the adsorption efficiency (%), the amount of Cr(VI) adsorbed at equilibrium, qe (mg g-1) and at time t (min), qt mg g-1), were calculated according to the equations:22
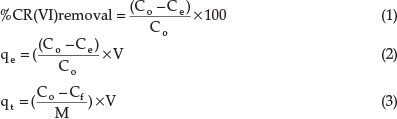
where Co is the initial concentration (mg L-1, i.e. concentration ofCr(VI) ionbefore adsorption), Ce is the equilibriumconcentra-tion (mgL-1, i.e. concentration ofCr(VI) ion after adsorption), Cf is the finalconcentration (mgL-1 ofCr(VI) ion in solution attime t in min), V is volume of solution in L and M is the mass of the adsorbents (g).
2.3.5. Effect of pH
The study of effect of pH on the adsorption of the metal ion was performed bycontacting0.5 gofthe adsorbents with 30 mL of 10 mg L-1 concentration of Cr(VI) ion solution within buffer solution ranging from pH 1 to 9. The pH of each solution was adjusted to the desire values by dropwise addition of0.1 M HCl or 0.1 M KOH, both before and after adsorbents of 0.5 g were added to 30 mL of working solution. The flasks containing the mixture were left in a thermostatic water bath shaker at 200 rpm and 25 °C for 180 min. The biomass was removed from the solution by filtration. The residual metal ion concentration left in the solutions was analyzed. Then the optimum pH was determined as the pH with the highest adsorption efficiency of Cr(VI) ion.
2.3.6. Effect of Adsorbent Dose
The adsorption of Cr(VI) ion by adsorbents of avocado kernel seeds, papaya peels and saw dust were investigated using the following adsorbent doses: 0.1, 0.2, 0.3, 0.4, 0.5, 0.6 and 0.7 g in 30 mL of10 mgL-1 Cr(VI) by keepingother variables constant (at optimal pH of 1, shaker speed = 200 rpm, contact time = 180 min and temperature = 25 °C). After adsorption time was completed the adsorbents were removed from the solution and the concentration of residual metal ion in each solution was determined using AAS.
2.3.7. Effect of Initial Chromium (VI) Concentration
Batch adsorption study was made to determine the effect of initial chromium (VI) concentration on the adsorption efficiency. Various chromium concentrations (5,10,20,30,40, and 50 mg L-1) of 30 mL were studied at the optimum conditions: pH of 1, adsorbent dose (0.5 g), shaking speed (200 rpm), contact time (180 min) and temperature (25 °C). After adsorption time was completed, the adsorbents were removed from the solution and the concentration of residual metal ion in each solution was determined using AAS.
2.3.8. Effect of Contact Time
The adsorption of Cr(VI) ions by the adsorbents were studied at various adsorption durations (10, 40, 70, 100, 130, 160 and 180 min). This was done by weighing 0.5 g of each of the adsorbents into the conical flasks containing 30 mL of 5 mg L-1 of Cr(VI) ion solutions at the optimized pH and the flasks were shaken in thermostatic water bath shaker at 200 rpm (25 °C) for various periods of time.
2.3.9. Effect of Temperature
The batch adsorption process was studied at different temperatures (20 to 45 °C) to investigate the effect of temperature on the adsorption process by keeping other parameters constant (pH of 1, adsorbent doses: 0.5 g and 5 mg L-1 of Cr(VI) ion) at 200 rpm for 160 min.
2.3.10. Preparation of Real Sample
Effluent at discharge point from Mojo tannery (which is a private leather tanning industry in Mojo, Oromia, Ethiopia) was collected. To study the efficiencies of the adsorbents for the removal of chromium from the effluent which had pH 4.95 and 4085 mg L-1 initial chromium concentration, 30 mL of effluent was measured and added into 50 mL conical flasks and the pH was adjusted to pH 1 by dropwise addition of 0.1 M HCl before and after addition of 0.5 g of all kinds of adsorbents within buffer solution. The flasks were then shaken in a thermostatic water bath shaker at the speed of 200 rpm and temperature of 40 °C for 160 min. The solutions were then filtered and prepared for analysis using AAS. The initial chromium concentration in the effluent was very high (4085 mg L-1) and was beyond the calibration curve limit and hence was determined after dilution of 100, μL of the real sample in to 50 mL (500-fold dilution).
2.3.11. Statistical Analysis
The average of triplicate AAS measurements was considered during the determination of Cr(VI) ion using multipoint external standardization method by AAS. The curve-fitting tests for data points for isotherm, thermodynamic and kinetic studies were performed using Microcal Origin 8.0 software.
3. Results and Discussion
3.1. Physical Characteristics of Adsorbents
Table 1 summarizes the physical characteristics of the adsorbents used in this report.
3.2. Effect of pH
Solution pH is one of the most important parameters that affects adsorption of metal ions by influencing the surface properties of the adsorbents and ionic forms of the chromium solutions.23 As it is shown in Fig. 1, maximum adsorption was observed at pH 1 for all adsorbents (i.e., sawdust = 98.18 %, avocado kernel seed = 96.35 % and papaya peel = 94.58 %).
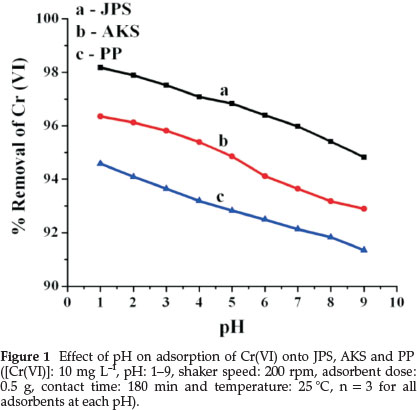
It was found that adsorption efficiencies of the adsorbents were slightly decreased from lower pH values to higher ones. More adsorption at acidic pH indicates that an increase in H+ ions on the adsorbents surface results in electrostatic attraction between positively charged adsorbents surface and chromate ions because HCrO4- is the dominant anionic form of Cr(VI) at lower pH.24
At pH 1, the chromium ions exist in the form of H2CrO4, while in the pH range of 1-6 different forms of chromium ions such as Cr2O72-, HCrO4-,Cr3O102-,Cr4O132- coexist, of which HCrO4-predominates. As pH increases this form shifts to CrO42- and Cr2O72-. Chromium exists in different oxidation states and the stability of these forms depends upon the pH of the system.25 Thus it was found that removal of Cr(VI) ions from solution was pH dependent and in view of these observations, the value of pH 1 was taken as the optimum pH for further studies.
3.3. Effect of Adsorbent Dose
Adsorbent dose variation experiment is one of the significant parts of the study that determines the capacity of adsorbents for a given initial concentration of Cr(VI) at the operating circumstances. Thus the experiments were carried out by varying adsorbent doses only in the range 0.1-0.7 g at 25 °C. The effect of adsorbent dose on the adsorption of chromium (VI) by those adsorbents is shown in Fig. 2.
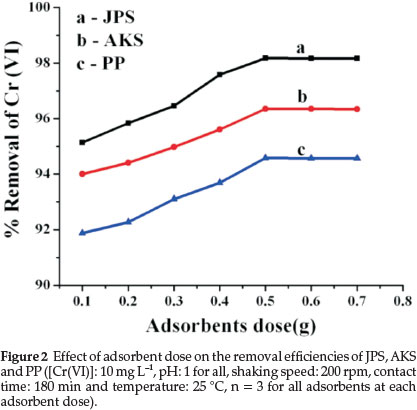
As illustrated in Fig. 2, the Cr(VI) removal efficiencies increases with increase in adsorbent dose and reach a saturation level at higher doses (i.e. starting from 0.5 g of the adsorbents) for all adsorbents that can be related to an increase in contact surface area of adsorbent particles.
Adsorbent doses beyond the steady state do not improve adsorption due to availability of excess adsorption sites than that of sorbets,26 assuming that the number of adsorption sites per unit mass of adsorbents remains constant. At lower dosage of adsorbents, there are insufficient active sites that the adsorbate can easily occupy. However, at higher dosage, active sorption sites are sufficiently available for the adsorbate to occupy. Similar results have been reported in the study on adsorption of Cr(VI) and Ni (II).24,27
3.4. Effect of Initial Chromium (VI) Concentration
The uptake of chromium on the adsorbents was studied at optimal pH values, the adsorbent dose of 0.5 g (in 30 mL solution), shaking speed (200 rpm), contact time (180 min) and temperature of 25 °C. The initial chromium concentrations studied were (5, 10, 20, 30, 40 and 50 mg L-1) and the percentage of sorption is given in Fig. 3, which showed that the percentage of Cr(VI) increases with decreasing adsorbate concentration.
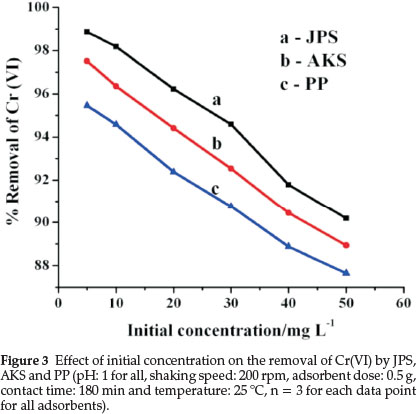
As shown in Fig. 3, the percentage of Cr(VI) ions adsorption decreased with increasing initial concentration from 5-50 mg L-1. This is because at lower concentration there are sufficient active sites that the sorbet (Cr(VI)) can easily occupy. However, at higher concentrations, active sorption sites are not sufficiently available for the adsorbate to occupy. Hence, Cr(VI) ions were not completely adsorbed from solutions due to the saturation of binding sites. In addition, the adsorbed negatively charged Cr(VI) ion electrostatically repel the incoming sorbet ions resulting in decrease of the adsorption percentage.20 These results are consistent with other reports.28
3.5. Effect of Contact Time
Figure 4 shows that chromium uptake was found to increase with increase in contact time and maximum percentage of adsorption occurred at 160 min where-after, it became constant.
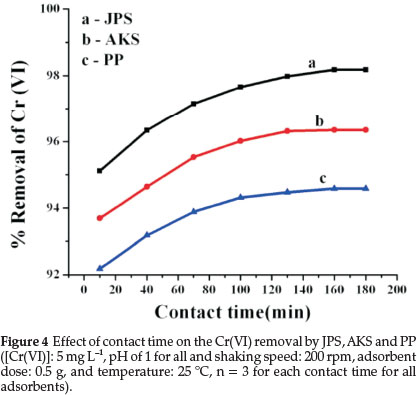
The initial rapid rate of adsorption may be due to the availability of the vacant active surfaces of the adsorbents for anionic Cr(VI) species in the solution. The later slow adsorption rate could be due to the electrostatic hindrance caused by already adsorbed adsorbate species and the slow pore diffusion of the ions.29
In general, the initial rate of adsorption is fast, and then a slower adsorption would follow as the available adsorption sites are slowly decreased. This is due to the fact that a large number of unoccupied surface sites are available for adsorption during the initial stage and after ascend of time the remaining unoccupied surface sites are difficult to be occupied due to repulsive forces between the solute molecules on the solid and bulk phases.30 Then from the results obtained the optimum contact time for adsorption onto adsorbents was fixed to be 160 min since the adsorption removal became constant after this contact time.
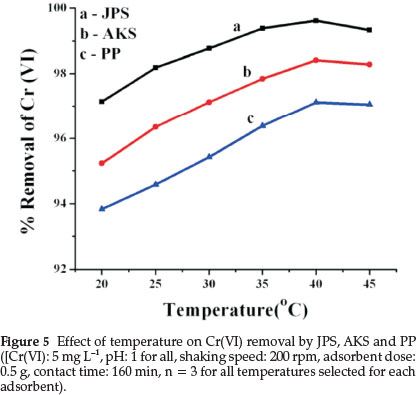
3.6. Effect of Temperature
Temperature is an important parameter that determines the thermodynamic of adsorption process.
As presented in Fig. 5, the percentage removal of Cr(VI) slightly increased with an increase of temperature from 20 °C to 40 °C and then decreased beyond 40 °C when the other conditions were kept constant. The enhancement in the adsorption efficiencies for some range of temperature may be due to the chemical interaction between adsorbate and adsorbents, creation of some new adsorption sites or the increased rate of intraparticle diffusion of Cr(VI) ions into the pores of the adsorbents. The decrease in percentage of adsorption at temperatures higher than 40 °C may be due to desorption caused by an increase of the thermal energy that may induces higher mobility of the adsorbate causing desorption.31,32
3.7. Adsorption Isotherms
Several models have been used to describe experimental data for adsorption isotherms. However, among these, the Langmuir and Freundlich isotherms are the most common models used by several researchers.33 According to the Langmuir isotherm, adsorption occurs at homogeneous sites and forms a monolayer. The linear forms of the Langmuir isotherm equation is given as:

where qe(mgg-1) isthe amountof Cr(VI) adsorbed atequilibrium and Ce(mgL-1) is the equilibriumconcentrationofthe adsorbate. Qm and KLare Langmuir constants related to the maximum adsorption capacity (mg g-1) and energy of adsorption (L mg-1), respectively. The essential characteristics of the Langmuir equation can be expressed in terms of a dimensionless factor, RL which is given as:

where KL is Langmuir constant and Co is the highest initial Cr(VI) ion concentration (mg L-1). The value of RL provides information as whether the adsorption is irreversible (RL= 0), favourable (0 < RL< 1), linear (RL= 1) or unfavourable (RL> 1).33
The Freundlich isotherm gives the relationship between equilibrium liquid and solid phase capacity based on the multilayer adsorption properties consisting of heterogeneous surface of the adsorbent.34 The linear forms of the Freundlich isotherm is:

where qeis the amount of Cr(VI) ion adsorbed (mg g-1) atequilibrium, Kf and n are the Freundlich constant, 1/n is the heterogeneity factor which is related to the capacity and intensity of the adsorption and Ce is the equilibrium concentration (mg L-1).35
The present study attempted to analyze the above mentioned isotherm parameters at 25 °C and the correlation coefficient R2 were calculated by fitting the experimental equilibrium data for Cr(VI) ion on all adsorbents using both Langmuir and Freundlich isotherms, which are presented in Table 2. The results clearly showed that the adsorption of Cr(VI) on adsorbents fits the Freundlich model best. The fact that the Freundlich model is a good fit suggests that physical adsorption as well as a heterogeneous distribution of active sites on the adsorbents surface exists.35
3.8. Adsorption Kinetic Studies
In order to evaluate the kinetic parameters, pseudo-first order and pseudo-second-order models were tested to analyze the adsorption kinetics.36,37
The pseudo-first-order equation is expressed as:
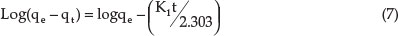
The pseudo-second-order equation is expressed as:

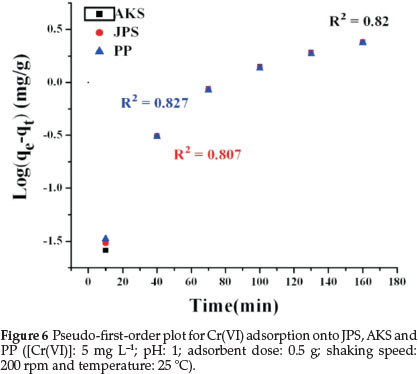
where qe is the mass of metal adsorbed at equilibrium (mg g-1), qtis the mass of metal at time t (min), K1 the first-order reaction rate constant of adsorption (min-1) and K2 the pseudo-second-order rate constant of adsorption (mg g-1 min-1).The values of K1 and qe were calculated from the slope and intercept of the plot of log (qe-qt) versus t while the values of K2 and qe were evaluated from the intercept and slope of a plot of t/qt versus t, respectively.37
Both adsorption kinetic models have been used to understand the adsorption kinetics and the correlation coefficient, R2, were calculated from these plots. These pseudo-first-order and pseudo-second-order models were calculated by using equations (7) and (8). However, the correlation coefficient, R2, values showed that the pseudo-second-order model fits better (Fig. 7 and Table 3) to the experimental data than the pseudo-first-order model (Fig. 6 and Table 3).
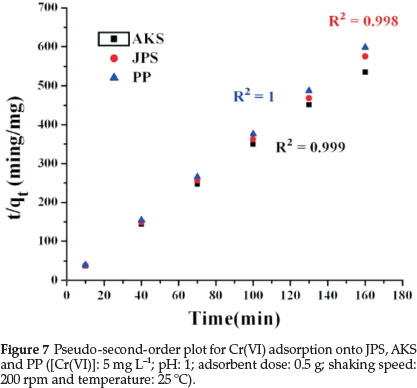
3.9. Thermodynamic Study
The thermodynamic parameters that help us to understand the nature of the adsorption of Cr(VI) ion on adsorbents are the standard change in Gibbs free energy (ΔG°), the standard change in entropy (ΔS°), and the standard change in enthalpy (ΔH°). The enthalpy change (ΔH°) from 25 °C to 45 °C was computed from the following equation:38

where ΔH" (KJ mol-1) and ΔS"( KJ mol-1 K-1) were calculated from the slope and intercept of the linear plot of lnKc versus 1/T. The equilibrium constant Kc can be evaluated at each temperature using the relationship:

where CAe and Ce is the amount of Cr(VI) ion adsorbed on solid phase at equilibrium and the equilibrium concentration (i.e. concentration of Cr(VI) left in solution after adsorption) in mg L-1, respectively. The calculated values of thermodynamic parameters are listed in Table 4.
The negative values of the standard Gibb's energy change (ΔGo) indicate the feasibility and spontaneous nature of the adsorption process. The positive values of the standard entropy change (ΔSo) shows the increased randomness at solid-liquid interfaces during the adsorption of metal ions onto all adsorbents could be due to some structural changes in the adsorbents.31,39
3.10. Adsorption Efficiencies of Adsorbents on Real Sample
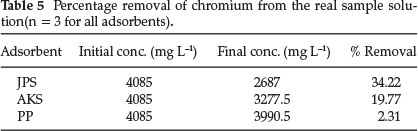
The efficiencies of the adsorbents toward the removal of chromium was tested using wastewater which at pH 4.95 and 4085 mg L-1 total chromium concentration, collected from effluent of Mojo Tannery (latitude 8°59'N, longitude 39°11'E). Adsorption experiments on this real sample were carried out after adjusting the pH to 1 (i.e. the optimum pH for JPS, AKS, and PP) and without any dilution to the water samples.
As the total chromium concentration in the effluent was high, the adsorption analysis by AAS were carried out after diluting the effluent to 500 times to bring the concentration of chromium within the linear calibration range. As presented in Table 5, the percentage removal by adsorptions seems small. This could be due to very high concentration of Cr(VI) in the effluent (beyond the optimized Cr(VI) dose) that may cause saturation of the adsorbents surface area and - due to co-adsorption of different forms of chromium ions and other ions present in the effluent.
3.11. Comparison of Chromium (VI) Adsorption with Various Adsorbents
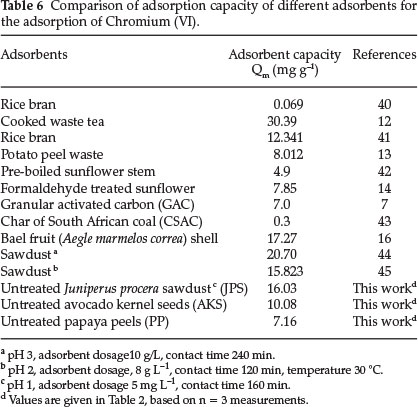
The adsorptive capacities of the adsorbents used in this work have been compared with those of others reported in the literature and the values of adsorption capacity as presented in Table 6. The experimental data of the present investigation were compared with reported values. Results of our investigation revealed that the adsorbents have higher adsorption capacity than rice bran, potato peel waste, sunflower and activated carbon (Table 6). The difference in adsorption capacity of the saw dust in this report to those previously reported could be due to the type of wood from which sawdust was obtained and the difference in experimental conditions for the study of adsorption of Cr(VI).
4. Conclusion
In this study, adsorption efficiencies of various low-cost locally available untreated adsorbents (JPS, AKS, and PP) toward the removal of Cr(VI) in aqueous solution and tannery waste effluent were investigated. The adsorption of Cr(VI) was found to be dependent on the pH of the system, the best results being obtained at pH 1.
The adsorption equilibrium data obtained for removal of Cr(VI) ion onto the adsorbents studied showed best fit to the Freundlich isotherm. The adsorption kinetic data fitted a pseudo-second-order model well. Thermodynamic studies predicted that the adsorption is feasible, spontaneous and exothermic in nature at a temperature of 20 °C, 25 °C, 30 °C, 35 °C, 40 °C and 45 °C. Afollow-up study is recommended to determine the concentration of different chromium species in the effluent and the effectiveness of such kind of adsorbents for the removal of chromium by adsorption.
Acknowledgement
The authors gratefully acknowledge the School of Graduate Studies and Department of Chemistry of Jimma University, Ethiopia, for providing financial support and facilities required to carry out this work.
References
1 K. Al-Sou'od, Adsorption kinetics for the removal of hexavalent chromium using low cost materials, Res. J. Chem. Environ, 2013,17,25-30. [ Links ]
2 Indian Standard, Drinking Water-Specification (first revision), IS 10500, 1991. [ Links ]
3 EPA, (Environmental Protection Agency) Environmental Pollution Control Alternatives, 1990, EPA/625/5-90/025, EPA/625/4-89/023, Cincinnati, US. [ Links ]
4 J. Kanagaraj, N.K. Chandra Babu and A.B. Mandal, Recovery and reuse of chromium from chrome tanning waste water aiming towards zero discharge of pollution, J. Cleaner Prod., 2008, 16, 1807-1813. [ Links ]
5 S.A. Cavaco, S. Fernandes, M.M. Quina and L.M. Ferreira, Removal of chromium from electroplating industry effluents by ion exchange resins, J. Hazard. Mater., 2007, 144, 634-638. [ Links ]
6 Y.X. Liu, D.X. Yuan, J.M. Yan, Q.L. Li and T. Ouyang, Electrochemical removal of chromium from aqueous solutions using electrodes of stainless steel nets coated with single wall carbon nanotubes, J. Hazard. Mater., 2011,186, 473-480. [ Links ]
7 M. Gholipour, H. Hashemipour and M. Mollashahi, Hexavalent chromium removal from aqueous solution via adsorption on granular activated carbon: adsorption, desorption, modeling and simulation studies, J. Eng. Appl. Sci, 2011, 6, 10-18. [ Links ]
8 R. Souag, D. Touaibia, B. Benayada and A. Boucenna, Adsorption of heavy metals (Cd, Zn and Pb) from water using keratin powder prepared from Algerien sheep hoofs, Europ. J. Sci. Res., 2009 35, 416-425. [ Links ]
9 G.H. Pino, L.M. Souza de Mesquita, M.L. Torem and G.A. Saavedra Pinto, Biosorption of cadmium by green coconut shell powder, Miner. Eng., 2006, 19, 380-387. [ Links ]
10 K.K. Singh, S.H. Hasan, M. Talat, V.K. Singh and S.K. Gangwar, Removal of Cr(VI) from aqueous solutions using wheat bran, Chem. Eng. J., 2009, 151, 113-121. [ Links ]
11 B. Singha and S.K. Das, Biosorption of Cr(VI) ions from aqueous solutions: kinetics, equilibrium, thermodynamics and desorption studies, Coll. Surf., B, 84, 221-232. [ Links ]
12 S. Dhanakumar, G. Solaraj, R. Mohanraj and S. Pattabhi, Removal of Cr(VI) from aqueous solution by adsorption using cooked tea dust, Indian J. Sci. Technol. 2007,1, 1-6. [ Links ]
13 M. A. Abdullah and A.G. Devi Prasad, Kinetic and equilibrium studies for the biosorption of Cr(VI) from aqueous solutions by potato peel waste, Int. J. Chem. Eng. Res., 2009,1, 51-62. [ Links ]
14 M. Jain, V.K. Garg and K. Kadirvelu, Chromium removal from aqueous system and industrial wastewater by agricultural wastes, Biorem. J., 2013, 17, 30-39. [ Links ]
15 P.K. Ghosh, Hexavalent chromium [Cr(VI)] removal by acid modified waste activated carbons, J. Hazard. Mater., 2009, 171, 116-122. [ Links ]
16. J. Anandkumar and B. Mandal, Removal of Cr(VI) from aqueous solution using bael fruit (Aegle marmelos correa) shell as an adsorbent, J. Hazard. Mater., 2009,168, 633-640. [ Links ]
17. D. Mohan and K.P. Singh, Single and multi-component adsorption of cadmium and zinc using activated carbon derived from bagasse - an agricultural waste. Water Res., 2002, 36, 2304-2318. [ Links ]
18. M. Ajmal, R.A.K. Rao, S. Anwar, J. Ahmad and R. Ahmad, Adsorption studies on rice husk: removal and recovery of Cd (II) from waste-water, Bioresour. Technol., 2003, 86, 147-149. [ Links ]
19 S. Ayub, S.I. Ali and N.A. Khan, Efficiency evaluation of neem (Azadirachta indica) bark in treatment of industrial wastewater, Environ. Pollut. Control. J., 2001, 4, 34-38 [ Links ]
20 J.C. Santamarina, K.A. Klein, Y.H. Wang and E. Prencke, Specific surface: determination and relevance, Can. Geotech.J., 2002,39,233-241. [ Links ]
21 V.C. Srivastava, I.D. Mall and I.M. Mishra, Characterization of mesoporous rice husk ash (RHA) and adsorption kinetics of metal ions from aqueous solution onto RHA, J. Hazard. Mater., 2006, 134, 257-267. [ Links ]
22 a) B. Alyuz and S. Veli, Kinetics and equilibrium studies for the removal of nickel and zinc from aqueous solutions by ion exchange resins, J. Hazard. Mater., 2009, 167, 482-488; [ Links ] b) B. Das, N.K. Mondal, P. Roy and S. Chattaraj, Equilibrium, kinetic and thermodynamic study on chromium (vi) removal from aqueous solution using Pistia stratiotes biomass, Chem. Sci. Trans., 2013, 2, 85-104. [ Links ]
23 B.V. Babu and S. Gupta, Adsorption of Cr(VI) using activated neem leaves: kinetic studies. Adsorption, 2008 14, 85-92. [ Links ]
24 A.A. Attia, S.A. Khedr and S.A. Elkholy, Adsorption of chromium ion (VI) by acid activated carbon, Braz. J. Chem. Eng., 2010 27, 183-193. [ Links ]
25 M. Nameni, M.R.A. Moghadam and M. Arami, Adsorption of hexavalent chromium from aqueous solutions by wheat bran, Int. J. Environ. Sci. Technol., 2008, 5, 161-168. [ Links ]
26 R. Gong, Y. Ding, H. Liu, Q. Chen and Z. Liu, Lead biosorption and desorption by intact and pretreated spirulina maximum biomass, Chemosphere, 2005, 58 125-130. [ Links ]
27 S.S. Gupta and K.G. Bhattacharyya, Adsorption of Ni (II) onto clays. J. Colloid Interface Sci., 2006, 295 21-32. [ Links ]
28 N. K. Hamadi, X. D. Chen, M.M. Farid and M.G.Q. Lu, Adsorption kinetics for the removal of Chromium (VI) from aqueous solution by adsorbents derived from used tyres and sawdust, Chem. Eng. J., 2001, 84, 95-101. [ Links ]
29 T. Karthikeyan, S. Rajgopal and L.R. Miranda, Chromium (VI) adsorption from aqueous solution by Hevea brasilinesis sawdust activated carbon, J. Hazard. Mater., 2005, 124, 192-199. [ Links ]
30 M. Danish, R. Hashim, M. Rafatullah, O. Sulaiman, A. Ahmad and G. Govind, Adsorption of Pb(II) Ions from aqueous solutions by date bead carbon activated with ZnCl2, Clean-Soil, Air, Water, 2011, 39, 392-399. [ Links ]
31 S.A. Wanees, A.M.M. Ahmed, M.S. Adam and M.A. Mohamed, Adsorption studies on the removal of hexavalent chromium-contaminated wastewater using activated carbon and bentonite, Chem. J., 2012, 02, 95-105. [ Links ]
32 S.S. Baral, S.N. Das and P. Rath, Hexavalent chromium removal from aqueous solution by adsorption on treated sawdust, Biochem. Eng. J., 2006, 31, 216-222. [ Links ]
33 P. K. Malik, Dye removal from wastewater using activated carbon developed from sawdust: adsorption equilibrium and kinetics, J. Hazard. Mater., 2004, 113, 81-88. [ Links ]
34 J.C. Igwe and A.A. Abia, Adsorption kinetics and intra-particulate diffusivities for bioremediation of Co (II), Fe (II) and Cu (II) ions from waste water using modified and unmodified maize cob, Int. J. Phys. Sci., 2007, 2, 119-127. [ Links ]
35 M. Roulia and A.A. Vassiliadis, Sorption characterization of a cationic dye, Microporous Mesoporous Mater., 2008,116, 732-740. [ Links ]
36 S. Chatterjee and S.H. Woo, The removal of nitrate from aqueous solutions by chitosan hydrogel beads, J. Hazard. Mater., 2009, 164, 1012-1018. [ Links ]
37 H. Demiral, I. Demiral, F. Tumsek and B. Karabacakoglu, Adsorption of chromium (VI) from aqueous solution by activated carbon derived from olive bagasse and applicability of different adsorption models, Chem. Eng. J., 2008, 144, 188-196. [ Links ]
38 P.K. Baskaran, B.R. Venkatraman, M. Hema and S. Arivoli, Adsorption studies of copper ion by low cost activated carbon, J. Chem. Pharm. Res., 2010, 2, 642-655. [ Links ]
39 I. Khazaei, M. Aliabadi and H.T. Hamed Mosavian, Use of agricultural waste for removal of Cr(VI) from aqueous solution, Iranian J. Chem. Eng., 2011, 8 1-13. [ Links ]
40 N.H. Hsu, S.L. Wang, Y.H. Liao, S.T. Huang, Y.M. Tzou and Y.M. Huang, Removal of hexavalent chromium from acidic aqueous solutions using rice straw-derived carbon, J. Hazard. Mater., 2009, 171, 1066-1070. [ Links ]
41 B. Singha and S.K. Das, Biosorption of Cr(VI) ions from aqueous solutions: kinetics, equilibrium, thermodynamics and desorption studies, Coll. Surf., B, 2011, 84, 221-232. [ Links ]
42 M. Jain, V.K. Garg and K. Kadirvelu, Chromium (VI) removal from aqueous system using Helianthus annuus (sunflower) stem waste, J. Hazard. Mater., 2009,162, 365-372. [ Links ]
43 F. Di Natale, A. Lancia, A. Molino and D. Musmarra, Removal of chromium ions form aqueous solutions by adsorption on activated carbon and char, J. Hazard. Mater., 2007 145 381-390. [ Links ]
44 A.K. Bhattacharya, T.K. Naiya, S.N. Mandal andS.K. Das, Adsorption, kinetics and equilibrium studies on removal of Cr(VI) from aqueous solutions using different low-cost adsorbents, Chem. Eng. J., 2008,137, 529-541. [ Links ]
45 M. Dakiky, M. Khamis, A. Manassra and M. Mer'eb, Selective adsorption of chromium (VI) in industrial wastewater using low-cost abundantly available adsorbents, Adv. Environ. Res., 2002, 6, 533-540. [ Links ]
Received 19 September 2014
Revised 15 December 2014
Accepted 18 December 2015
* To whom correspondence should be addressed. E-mail: tesfaye.refera@ju.edu.et














