Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
South African Journal of Chemistry
versión On-line ISSN 1996-840X
versión impresa ISSN 0379-4350
S.Afr.j.chem. (Online) vol.68 Durban 2015
http://dx.doi.org/10.17159/0379-4350/2015/v68a3
RESEARCH ARTICLE
http://dx.doi.org/10.17159 /0379-4350/2015/v68a3
A solvent-free synthesis of polyhydroquinolines via Hantzsch multicomponent condensation catalyzed by nanomagnetic-supported sulfonic acid
Somayeh Otokesh; Nadiya Koukabi*; Eskandar Kolvari; Ali Amoozadeh*; Masoumeh Malmir; Saeede Azhari
Department of Organic Chemistry, Faculty of Chemistry, Semnan University, Semnan, Iran
ABSTRACT
A simple and efficient procedure for the synthesis of polyhydroquinolines was developed, involving a one-pot four-component Hantzsch condensation of aromatic aldehydes, 1,3-cyclohexanediones, alkyl acetoacetate and ammonium acetate in the presence of a catalytic amount of nanomagnetic-supported sulfonic acid under solvent-free conditions. The method offers several advantages including high yields, short reaction times, a simple work-up procedure and catalyst reusability for several runs. Furthermore, easy isolation of the catalyst from the reaction mixture was enabled by use of an external magnet.
Keywords: Nanomagnetic-supported sulfonic acid, multicomponent reaction, solvent-free conditions, heterocyclic compound, Hantzsch reaction.
1. Introduction
Since the beginning of this century, different sciences have seen a rapid increase due to interest in materials at the nano-scale. Nanomaterials have attracted attention because of physical, electronic and magnetic properties. The field of nanomagnetic particles is a subset of nanomaterials. The applications for these materials are very diverse such as metal ion separations,1 enzyme immobilization,2 magnetic resonance imaging (MRI),3 drug delivery and catalysis.4 In chemistry, nanomagnetic catalysts have emerged as one of the most useful heterogeneous catalysts due to their numerous applications in organic synthesis.5 These catalysts have been studied in various significant protocols in organic chemistry because they are robust, inexpensive and readily available. Also, they can be easily prepared from their available metal salts and most importantly can be recycled for several runs without any loss of selectivity and activity.6
In the context of green chemistry, the design and development of organic synthesis performed through multicomponent reactions (MCRs) have become a significant area of research in organic chemistry since such processes improve atom economy, efficiency and convergence.7 Therefore, MCRs are often useful alternatives to sequential multistep synthesis. The synergistic use of nanomagnetic particles and MCRs allows efficient synthesis of diverse nitrogen-containing heterocycles. One of the most prominent methods to prepare these compounds is the Hantzsch condensation reaction providing polyhydroquinolines. The polyhydroquinoline moiety is a fertile source of biologically and pharmacologically important molecules such as vasodilator, bronchodilator, anti-atherosclerotic, hepto-protective, anti-tumor, anti-mutagenic, geroprotective, anti-diabetic agents, HIV protease inhibition and most importantly as calcium channel blockers.8-15 All mentioned cases demonstrate clearly the remarkable potential of the polyhydroquinoline derivatives as a source of valuable drugs.16,17 In continuation of our investigation on the use of nano-γ-Fe2O3-SO3H as catalyst for MCRs and our interest in synthesis of heterocycles containing a nitrogen atom,18,19 we report an efficient and facile synthesis of hexahydroquinolines under solvent-free conditions (Scheme 1).
2. Experimental
2.1. General
All chemicals were purchased from Merck, Fluka or Across companies and used without any further purification. Nano-g-Fe2O3-SO3H was prepared with the reported method.20 Melting points were recorded on electro thermal 9100 apparatus and are uncorrected. NMR spectra were recorded with a Bruker Avance spectrometer (1H NMR 300,400 MHz and 13C NMR 75,100 MHz) in pure deuteriated chloroform and DMSO with tetramethylsilane (TMS) as the internal standard. The IR spectra were recorded on a Perkin-Elmer model 783 spectrophotometer (Waltham, MA, USA). UV-Vis spectra were obtained as ethanol solutions (10-5 M) on a Shimadzu UV-1650PC spectrophotometer.
2.2. General Method for the Synthesis of Polyhydroquinolines (5a-o)
A mixture of aromatic aldehyde (1 mmol), alkyl acetoacetate (1 mmol), 1,3-cyclohexanedione (1 mmol), ammonium acetate (1.1 mmol) and nano-γ-Fe2O3-SO3H (0.031 g) was heated at 60 °C. After completion of the reaction (monitored by TLC), the mixture was cooled to room temperature and triturated with hot ethanol (5 mL). In the presence of a magnetic stirrer bar, nano-γ-Fe2O3-SO3H moved on to the stirrer bar steadily and the reaction mixture turned clear within 10 s. The catalyst was isolated by simple decantation. After evaporation of the solvent, the crude product was recrystallized from EtOH/H2O to give a pure product.
2.3. Spectral Data of Some Representative Compounds
Ethyl 2-Methyl-5-oxo-4-phenyl-4,6,7,8-tetrahydro-1H-quinoline-3-carboxylate (5j) (90 %) as a light cream solid.; (recrystallized from EtOH/H2O, TLC- n-hexane:ethyl acetate, 8:2, Rf = 0.14); M.p. 240-242 °C, vmax(KBr) 3296 (NH), 1641 (C=O) (acid), 1608 (C=O) (ketone), 1488 (OC2H5) (ester) cm-1; δ H (300 MHz, CDCl3) 1.17 (t,3H, CH3-CH2-O-C=O), 1.59 (m, 2H, CH2,7 H), 1.97 (m, 3H, CH3), 2.30-2.46 (m, 4H, 6,8 H), 4.05 (q, 2H, -O-CH2-CH3), 5.01 (s, 1H,4 H), 5.94 (s, 1H, NH), 7.08-7.31 (m, 5H, 2', 3', 4', 5', 6' H) ppm. UV (λmaxin EtOH): 378 nm.
Ethyl 4-(p-Methoxyphenyl)-2-methyl-5-oxo-7-phenyl-4/6/7/8-tetra-hydro-1H-quinoline-3-carboxylate (51) (96 %) as a white solid.; (recrystallized from EtOH/H2O, TLC- n-hexane:ethyl acetate, 8:2, Rf = 0.12); M.p. 236-238 °C, v max(KBr) 3280 (NH), 1689 (C=O) (acid), 1606 (C=O) (ketone), 1479 (OC2H5) (ester), 1222 (OCH3) (ether) cm-1; δh/ ppm (300 MHz, CDO3) 1.27 (m, 3H, CH3-CH2-O-C=O), 2.23-2.37 (m, 3H, CH3), 2.38-2.52 (m, 5H, 6.7.8 H), 3.71 (s, 3H, CH3-O-Ph), 4.07 (m, 2H, -O-CH2-CH3), 5.05 (m, 1H, 4 H), 6.71-7.12 (m, 4H, 2', 3', 5', 6' H), 6.96 (s, 1H, NH), 7.17-7.29 (m, 5H, 2",3",4",5",6" H); δc/ppm (75 MHz, CDCl3) 195.2 (C-5), 167.5 (C=OOC2H5), 157.8 (C-4'), 150 (C-2), 149.5 (C-1a), 143.4 (C-1"), 142.6 (C-1'), 139.7 (C-2' and C-6'), 139.3 (C-3" and C-5"),128.8 (C-2"and C-6"), 127 (C-4"), 126.6 (C-3' and C-5'), 113.2 (C-5a), 106.2 (C-3), 59.8 (O-CH2-CH3), 55.1 (O-CH3), 39.5 (C-6), 38.8 (C-4), 35.8 (C-8), 34.4 (C-7), 19.1 (1C, CH3 ), 14.2 (1C, CH3-CH2O). UV (λmax in EtOH): 224 nm; 417.19 (100.0 %), 418.20 (28.6 %),419.20 (4.8 %) m/z: 417 (100),418 (28.6 %) and419 (4.8 %), (Found: C,74.82; H, 6.83; N, 3.37 %. Calcd. for C26H27NO4(417.19); C,74.80; H,6.52; N 3.35 %).
Ethyl 2-Methyl-5-oxo-4/7-diphenyl-4/6/7/8-tetrahydro-1H-quino-line-3-carboxylate (5n) (99 %) as a white solid; (recrystallized from EtOHHO, TLC- n-hexane:ethyl acetate, 8:2, Rf = 0.13); M.p. 213-215 °C, vmax(KBr) 3276 (NH), 1701 (C=O) (acid), 1606 (C=O) (ketone), 1487 (OC2H5) (ester), cm-1; Oh (400 MHz, DMSO-d6) 1.14 (t, 3H, CH3-CH2-O-C=O), 2.32 (s, 3H, CH3), 2.35 (dd, 1H, 8 H), 2.5 (d, 1H, 8 H), 2.59 (m, 1H, 7 H), 2.68 (dd, 1H, 6 H), 2.79 (dd, 1H, 6 H), 3.17(s, 1H, NH), 4.0 (q, 2H, -O-CH2-CH3), 4.98 (s, 1H, 4 H), 7.11 (m, 1H, 4' H), 7.19-7.24 (q, 5H, 2', 4', 6', 2", 6" H), 7.3-7.36 (m, 4H, 3', 5', 3", 5" H), 9.21(s, 1H, NH) ppm;δC(100 MHz, DMSO-d6) 194.4 (C-5), 167.3 (C=OOC2H5), 151.1 (C-2), 148.1 (C-1a), 145.3 (C-1"), 143.9 (C-1'), 128.9 (C-2' and C-6'), 128.3 (C-3' and C-5'), 127.9 (C-3" and C-5"),127.4 (C-2" and C-6"), 127 (C-4"), 126.2 (C-4'), 111.2 (C-5a), 104.2 (C-3), 59.5 (O-CH2-CH3), 44.4 (C-6), 38.8 (C-4), 36.2 (C-8), 34 (C-7), 18.7 (1C, CH3), 14.6 (1C, CH3-CH2O),ppm.UV (λmax in EtOH): 361 nm; 387.18 (100.0 %), 388.19 (27.2 %), 389.19 (4.6 %) m/z: 417 (100), 418 (27.2 %) and 419 (4.6 %), (Found: C,77.47; H, 6.52; N, 3.63; O, 12.37 %. Calcd. for C25H25NO3 (387.18); C,77.47; H,6.52; N 3.63; O, 12.37 %).
Ethyl 4-(p-chlorophenyl)-2-methyl-5-oxo-7-phenyl-4, 6, 7, 8-tetrahydro-1H-quinoline-3-carboxylate (5o) (98 %) as a white solid; (recrystallized from EtOH/H2O, TLC- n-hexane:ethyl acetate, 8:2, Rf = 0.12); M.p. 190-192 °C, vmax(KBr) 3274 (NH), 1701(C=O) (acid), 1606 (C=O) (ketone), 1487 (OC2H5) (ester), 848 (C-Cl) cm-1; δh (400 MHz, DMSO-d6) 1.12 (t, 3H, CH3-CH2-O-C=O), 2.3 (t, 1H, 8 H), 2.31 (s, 3H, CH3), 2.5 (t, 1H, 8 H), 2.54-2.59 (7,1H, 8 H), 2.59-2.66 (m, 1H, 6 H), 2.74-2.82 (m, 1H, 6 H),), 3.17 (t, 1H, NH), 3.98 (q, 2H, -O-CH2-CH3),4.90 (d, 1H, 4 H), 7.22-7.28 (m, 5H, 2',6', 2", 4", 6" H), 7.31-7.33 (m, 4H, 3', 5', 3", 5" H), 9.21 (d, 1H, NH)ppm;dC(100 MHz, DMSO-d6) 194.2 (C-5), 167.1 (C=OOC2H5), 151.3 (C-2), 150.8 (C-1a), 147.1 (C-1"), 145.7 (C-1'), 143.8 (C-4'), 130.7 (C-2' and C-6'), 129.8 (C-3' and C-5'), 128.8 (C-3" and C-5"), 128.1 (C-2" and C-6"), 127.1 (C-4"), 111.0 (C-5a), 103.6 (C-3), 44.3 (O-CH2-CH3), 43.7 (C-6), 38.8 (C-4), 36.0 (C-8), 33.7 (C-7), 18.7 (1C, CH3), 14.5 (1C, CH3-CH2O) ppm. UV (λmax in EtOH): 363 nm; 421.14 (100.0 %), 422.14 (32.3 %), 423.15 (27.2 %) m/z: 421 (100 %), 422 (32.3 %) and 423 (27.2 %), (Found: C,71.19; H, 5.71; N, 3.32; Cl, 8.40; O, 11.38 %. Calcd. for C25H24ClNO3 (421.11); C,71.19; H,5.71; N, 3.32; Cl, 8.40; O, 11.38 %).
3. Results and Discussion
Hammett acidity function (H0) can be used to effectively express the ability and acidity strength of an acid in organic solvents.21 This method using 4-nitro aniline with Hammett indicators is a simple quantitative method, which can be obtained from the relative intensities of the absorption band by UV-visible spectrometer. The acidity of the solution can be calculated by using the following Hammett equation in the form of H0, the acidity function as given:
H0 = pK(I)aq + log[I]s/[HI+]s
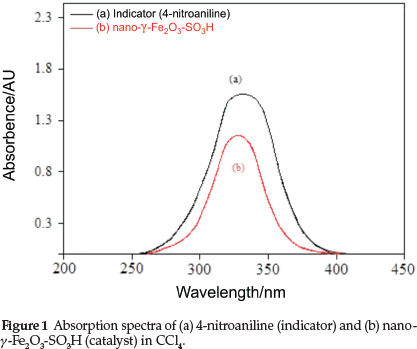
where [I] represents the indicator base, pK(I)aq is the protonation constant of the dye in aqueous solution (for example the pK(I)aq value of 4-nitroaniline is 0.99), which can be obtained from many references, [I]s and [HI+]s are respectively the molar concentrations of solvated non-protonated and protonated forms of the indicator. According to the Beer-Lambert law, the value of [I]s/[IH+]scan be determined and calculated from UV-visible spectrum measurements. In the present experiment, 4-nitroaniline was chosen as the basic indicator and CCl4 was chosen as the solvent because of its aprotic nature. A dilute solution (10-4 M) of 4-nitroaniline in CCl4 was prepared and used as a stock solution throughout the experiments. The maximal absorbance of the non-protonated form of 4-nitroaniline was observed at 331 nm in CCL. As shown in Fig. 1, the absorbance of the non-protonated form of the indicator in nano-γ-Fe2O3-SO3H was weak compared to the sample of the indicator in CCl4, which indicated that the indicator was partially in the form of [IH+]. The results obtained are listed in Table 1, which shows the acidity strength of nano-γ-Fe2O3-SO3H.
First, in order to optimize the conditions, the reaction of benzaldehyde, ethyl acetoacetate, 5,5-dimethyl-1,3-cyclohex-anedione and ammonium acetate was chosen as a model system under thermal conditions. The results listed in Table 2 show that nanomagnetic-supported sulfonic acid is the best catalyst affording the highest yield (Table 2, Entry 10), while both bulk-Fe2O3 and bulk-Fe2O3-SO3H provide lower yields (Table 2, Entries 4, 6). This is due to its higher surface areas and surface vacancies which are responsible for the excellent catalytic activity. Finally, for comparison, iron chloride salts, FeCl2.4H2O and FeCl3.6H2O, were also investigated for synthesis of polyhydro-quinoline. The results showed that the conversions were trace amounts (Table 2, Entries 2,3). By screening different amounts of nano-γ-Fe2O3-SO3H, we found that product 5a was obtained in yields ranging from 40 to 98 (Table 2, Entries 7-11). Thus, the best amount of catalyst was 0.031 g (8 mol%) (Table 2, Entry 10).
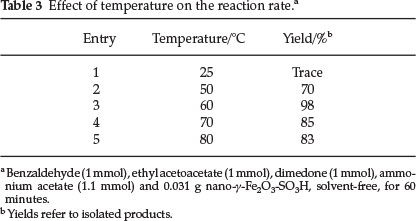
In the following study of the model reaction, we examined the influence of various temperatures on the reaction rate as well as yields of products. As indicated in Table 3, the best yield was obtained at 60 °C (Table 3 Entry 3), so we considered it as optimum temperature. Higher temperatures decreased the total yield by producing byproduct as shown by TLC (Table 3, Entries 4, 5).
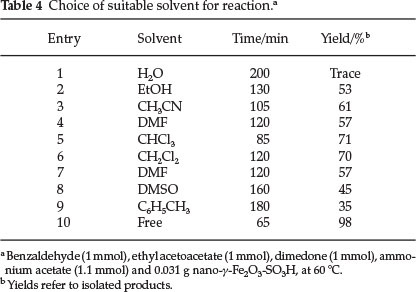
The choice of solvent was crucial. To this end, the reaction was carried out with different solvents (Table 4). Fortunately, when the reaction was carried out in solvent-free condition, good yields and short reactions times were achieved which was consistent with context of 'sustainable chemistry', concerning green chemistry aspects; 'the best solvent is no solvent at all'.
In order to show the merit of method in comparison with other reported results in the literature for similar reactions, we compared the results of nano-γ-Fe2O3-SO3H with other catalysts used for the synthesis of polyhydroquinoline derivatives in Table 5. This method has the following advantages: Magnetic separation can eliminate expensive centrifugation (compared to methods using nano catalysts without magnetic properties22), nano-sized magnetic particles have large specific surface area so adsorption capacity is high, for this reason, the reaction rate increased leading to energy savings. In contrast to other methods,23-34 the products are obtained in a shorter time. These results clearly indicate that nano-γ-Fe2O3-SO3H is an efficient, magnetic and reusable acidic catalyst, and the present method is found to be very effective for the synthesis of polyhydroquinoline derivatives.
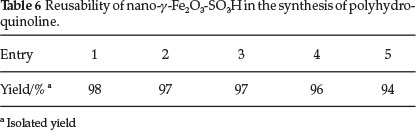
To determine the recyclability of the catalyst, the same model reaction was again studied under optimized conditions. The results are shown in Table 6. It is important to mention that our new conditions also provide a fast and facile work-up; after completion of the reaction, the mixture was triturated with hot ethanol. Within a few seconds, after stirring was stopped, the reaction mixture turned clear and the catalyst was deposited on the magnetic bar, which was easily removed with an external magnet, after being washed with acetone and dried in air, the nano-γ-Fe2O3-SO3H catalyst could be used at least five times without significant loss of activity. This efficient recyclability is most probably due to bound covalently sulfonic groups (-SO3H) to magnetic nanoparticles. However, the slight reduction of catalytic activity of the catalyst after recycling is probably due to the blockage of active sites on the catalyst surface (Fig. 2).
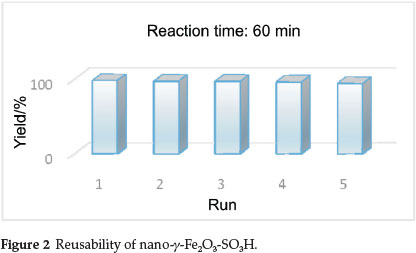
In order to evaluate the generality of our new conditions, we studied the reaction of aromatic aldehydes with electron-donating and electron-withdrawing groups, ethyl acetoacetate and different 1,3-cyclohexanediones in the presence of ammonium acetate and 0.031 g of nano-γ-Fe2O3-SO3H under obtained conditions. The results are summarized in Table 7. In all cases, the reactions were clean and rapid. The nature and location of substituent groups in the aromatic ring has been shown not to have much effect on the formation of the final product and afford the expected products in high yields.
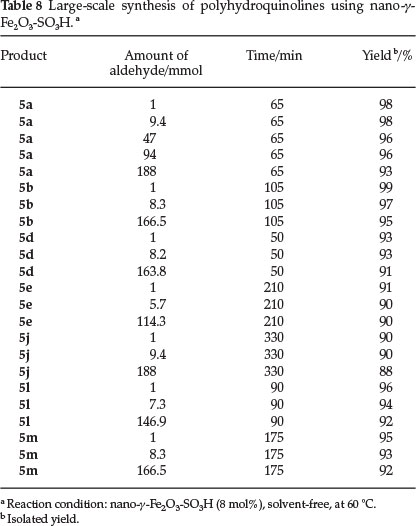
The methodology was extended for large-scale synthesis of polyhydroquinolines. First, the reaction was done using benzaldehyde, ethyl acetoacetate, 5,5-dimethyl-1,3-cyclohex-anedione on 1 mmol scale (Table 8, Entry 1). Then, the same reaction was expanded with different amounts of benzaldehyde such as 1 g (9.4 mmol), 5 g (47 mmol), 10 g (94 mmol) and 20 g (188 mmol) without affecting the yield of the product 5a (Table 8, Entries 2-5). While scaling up the reaction, it was found that 8 mol% of the catalyst is sufficient to promote the reaction effectively without significantly affectingthe rate of the reaction. This procedure was also applied to several other substituted benz-aldehydes and the products 5b, 5d, 5e, 5j, 5l and 5m were also synthesized (Table 8, Entries 6-23). Finally, the methodology could successfully be used for the synthesis of polyhydroquinolines on a large scale.
4. Conclusion
In conclusion, we have developed a simple new catalytic method for the synthesis of polyhydroquinoline derivatives via one-pot, four-component Hantzsch condensation of 1,3-cyclo-hexanedione, aryl aldehydes, alkyl acetoacetate and ammonium acetate in the presence of nanomagnetic-supported sulfonic acid as an efficient, reusable, and green heterogeneous catalyst under solvent-free conditions. Our new method offers several advantages including short reaction times, the use of mild reaction conditions, avoiding the use of harmful acids and involving a simple work-up procedure.
Supplementary material
The IR, MS and NMR spectra for the novel compounds (5l, 5n and 5o) are presented as supplementary material.
Acknowledgement
The authors gratefully acknowledge Semnan University Research Council for financial supporting this work.
References
1 T. Sen, A. Sebastianelli and I.J. Bruce, J. Am. Chem. Soc., 2006, 128, 7130-7131. [ Links ]
2 C. Song, L. Sheng and X. Zhang, Appl. Microbiol. Biotechnol., 2012, 96, 123-132. [ Links ]
3 R.S. Chaughule, S. Purushotham and R.V Ramanujan, Proc. Natl. Acad. Sci., India, Section A: Phys. Sci., 2012, 82, 257-268. [ Links ]
4 A. Andrade, R. Ferreira, J. Fabris and R. Domingues, Coating Nano-magnetic Particles for Biomedical Applications, 2011, Intech, DOI: 10.5772/19519. [ Links ]
5 M. Gawande, A. Rathi, P. Branco and R. Varma, Appl. Sci., 2013, 3, 656-674. [ Links ]
6 R.B.N. Baig and R. S. Varma, Chem. Commun., 2013,49, 752-770. [ Links ]
7 M. Jose Climent, A. Corma and S. Iborra, RSCAdvances, 2012,2,16-58. [ Links ]
8 A. Kumar, S. Sharma, VD. Tripathi, R.A. Maurya, S.P. Srivastava, G. Bhatia, A.K. Tamrakar and A.K. Srivastava, Bioorg. Med. Chem., 2010, 18, 41384148. [ Links ]
9 K. Turhan, S.A. Ozturkcan, Z. Turgut, M. Karadayi, A. Aslan and M. Gulluce, Toxicol. Ind. Health, 2012, 28, 605-613. [ Links ]
10 V. Gupta and U. Misra, Med. Chem. Res., 2008,17, 437-444. [ Links ]
11 B. Bülbül, G.S. Öztürk, M. Vural, R. §im§ek, Y. Sarioglu, A. Linden, M. Ülgen and C. Șafak, Eur. J. Med. Chem., 2009, 44, 2052-2058. [ Links ]
12 M.S. Al-Said, M.M. Ghorab, M.S. Al-Dosari and M.M. Hamed, Eur. J. Med. Chem., 2011,46, 201-207. [ Links ]
13 J. Mielcarek, I. Khmelinskii, M. Sikorski, H. Stefaniak, J. Photochem. Photobiol. A., 2007,192, 197-203. [ Links ]
14 H. Abu-Melha, Spectrochim. Acta Part A: Molecular and Biomolecular Spectroscopy., 2013, 113, 115-122. [ Links ]
15 C. Safak and R. Simsek, Mini Rev. Med. Chem, 2006. 6, 747-755. [ Links ]
16 N.K.Ladani,D.C.Mungra,M.P.PatelandR.G.Patel,Chin. Chem.Lett, 2011, 22, 1407-1410. [ Links ]
17 N. Edraki, A.R. Mehdipour, M. Khoshneviszadeh and R. Miri, Drug Discov. Today, 2009, 14, 1058-1066. [ Links ]
18 E. Kolvari, N. Koukabi and O. Armandpour, Tetrahedron, 2014, 70, 1383-1386. [ Links ]
19 N. Koukabi, E. Kolvari, A. Khazaei, M.A. Zolfigol, B. Shirmardi-ShaghasemiandH.Khavasi, R.Chem.Commun., 2011,47,9230-9232. [ Links ]
20 N. Koukabi, E. Kolvari, M.A. Zolfigol, A. Khazaei, B. Shirmardi-Shaghasemiand B. Fasahati,Adv. Synth. Catal.,2012,354,2001-2008. [ Links ]
21 H. Xing, T. Wang, Z. Zhou and Y. Dai, J. Mol. Catal. A: Chem., 2007,264, 53-59. [ Links ]
22 M. Tajbakhsh, E. Alaee, H. Alinezhad, M. Khanian, F. Jahani, S. Khaksar, P. Rezaee and M. Tajbakhsh, Chin. J. Catal., 2012, 33, 1517-1522. [ Links ]
23 J. Safari, S.H. Banitaba and S. Dehghankhalili. Chin. J. Catal., 2011,32, 1850-1855. [ Links ]
24 S.R. Cherkupally and R. Mekala, Chem. Pharm. Bull., 2008, 56, 1002-1004. [ Links ]
25 D.R. Patil, M.B. Deshmukh, S.M. Salunkhe, D.K. Salunkhe, G.B. Kolekar and P.V Anbhule. Der. Pharma. Chemica, 2010, 2, 342-348. [ Links ]
26 C.S. Reddy and M. Raghu, Indian J. Chem., Sect. B. 2008, 47B, 1578-1582. [ Links ]
27 L.M. Wang, J. Sheng, L. Zhang, J.W. Han, Z. Fan, H. Tian and C.T. Qian, Tetrahedron, 2005, 61, 1539-1543. [ Links ]
28 J.L. Donelson, R.A. Gibbs and S.K. De, J. Mol. Catal. A: Chem., 2006, 256, 309-311. [ Links ]
29 C.S. Reddy and M. Raghu, Chin. Chem. Lett., 2008,19, 775-779. [ Links ]
30 N.N. Karade, V.H. Budhewara, S.V. Shindeb and W.N. Jadhavb, Lett. Org. Chem., 2007,4,16-19. [ Links ]
31 J.-J. Xia and K.-H. Zhang, Molecules, 2012,17, 5339-5345. [ Links ]
32 E.MosaddeghandA.Hassankhani, Arabian J. Chem., 2012,5,315-318. [ Links ]
33 M. Arsalan, C. Faydali, M. Zengin, M. Kucukislamoglu and H. Demirhan, Turk. J. Chem., 2009, 33, 769-774. [ Links ]
34 M. Hong, C. Cai and W.-B. Yi, J. Fluorine Chem., 2010,131, 111-114. [ Links ]
35 S.S. Mansoor, K. Aswin, K. Logaiya and S.P.N. Sudhan. J. Saudi Chem. Soc., 2012, in press. [ Links ]
36 M.M. Kurbanova, E.Z. Huseynov, A.V. Gurbanov, A.M. Maharramov and S.W. Ng, Acta Crystallogr., Sect. E., 2012, 68, 2233-2233. [ Links ]
37 G.M. Ziaran, A.R. Badiei, Y. Khaniania and M. Haddadpour, Iran. J. Chem. Chem. Eng., 2010, 29, 1-10. [ Links ]
38 S.B. Sapkal, K.F. Shelke, B.B. Shingate and M.S. Shingare, Tetrahedron Lett., 2009, 50, 1754-1756. [ Links ]
39 M. M. Kurbanova, E. Z. Huseynov, A. V. Gurbanov, A. M. Mahar-ramov and R. Kia, Acta Crystallogr., Sect. E., 2013, 69, 541-541. [ Links ]
40 P. Rao, R. Kumari and S. Oruganti, Chem. Biol. Interf., 2013, 3, 38-49. [ Links ]
41 A. Kumar and R.A. Maurya, Tetrahedron Lett, 2007, 48, 3887-3890. [ Links ]
Received 18 September 2014
Revised 17 November 2014
Accepted 18 November 2014
* To whom correspondence should be addressed. E-mail: koukabi@gmail.com / aamozadeh@semnan.ac.ir














