Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Chemistry
On-line version ISSN 1996-840X
Print version ISSN 0379-4350
S.Afr.j.chem. (Online) vol.67 Durban Jan. 2014
RESEARCH ARTICLE
Synthesis and Spectroscopic, Thermal and Crystal Structure Studies of Hydrazinium Hydrogensuccinate
Thathan PremkumarI, II, *; Rajendran SelvakumarI; Nigam P. RathIII; Subbiah GovindarajanI, *
IDepartment of Chemistry, Bharathiar University, Coimbatore - 641 046, India
IIThe University College / Department of Chemistry, Sungkyunkwan University, Suwon 440-746, South Korea
IIIDepartment of Chemistry and Biochemistry and Center for Nanoscience, University of Missouri, St. Louis, MO 63121, USA
ABSTRACT
The mono-hydrazinium salt of succinic acid (hydrazinium hydrogensuccinate salt) was prepared by reacting hydrazine hydrate and succinic acid. Its spectral, thermal and structural properties have been determined. The salt, N2H5+.C4H5O4-, is a molecular salt containing discrete hydrazinium cations and hydrogen succinate anions. The crystal packing consists of infinite chains of anions and cations that are alternatively linked through O-H···N and bifurcated N-H···O bonds and this chain is connected to adjacent antiparallel chains by N-H···O hydrogen bonding. The adjacent chains are cross linked by N-H···O bonds from the donor N2H5+ ions, thus forming a three-dimensional network. The salt undergoes melting followed by decomposition to give gaseous products.
Keywords: Hydrazine, succinic acid, hydrazinium hydrogensuccinate, crystal structure, thermal studies.
1. Introduction
Dibasic acids are known to form N2H5HA, (N2H5)2Aand N2H5HA.H2A type salts (H2A = dibasic acid) with hydrazine. Though, a number of hydrazinium salts of the former two types with different dibasic acids have been reported,1-6 there appear to be very few reports available in the literature on the hydrazinium salts of the third type, viz.N2H5HA.H2A4. However, such salts of oxalic acid with ammonium7 and potassium7,8 ions and dipicolinic acid with sodium9,10 ions have been studied in detail, including their crystal structures. The preparation of hydrazinium salts has become a subject of recent interest due to their wide use as additives in propellants, drugs to treat cancer and Hodgkin's disease and explosives.1 They have also been utilized as ligands for the preparation of metal hydrazinium/ hydrazine complexes.11-13 Some of these salts are also used as flame-retardants14,15 and proton conductors.16 The preparation and thermal behaviour of some of these salts have recently been reported from our laboratory with a few aliphatic2,4 and aromatic5 carboxylic acids. However, the crystal structure of hydrazinium salts of aliphatic and aromatic carboxylic acids have not been reported. Although crystal structures of hydrazinium salts of some heteroaromatic carboxylic acids have been reported,17-19 there appears to be only one report20 available up to now in the Cambridge Structural Database (CSD)21 on the hydrazonium salt of the type N2H6A. Recently, for the first time, we reported two polymorphic modifications (forms A and B) of the N2H6(HA)2 type salt (Fig. 1b) formed by hydrazine with 3,5-pyrazole-dicarboxylic acid (Fig. 1a).22

Hydrazine forms two types of salts with oxalic acid (H2C2O4), viz.N2H4-H2C2O4 and 2N2H4-H2C2O4. Pratt and Richards23 studied these compounds with 1H NMR and have proposed that these compounds could be formulated as N2H5-HC2O4 and (N2H5)2C2O4, respectively. The structure of the first compound was extensively studied by X-ray24,25 and neutron26 diffraction methods and found to consist of chains of N2H5+ and HC2O4-ions. The chains are cross-linked by N-H-O bonds from the N2H5+ ions, thus forming a three-dimensional network. Although much has been reported on the N2H5HC2O4 salt, the crystal structure of the corresponding hydrazinium salt of succinic acid [HOOC(CH2)2COOH], N2H5(HOOC(CH2)2COO), has not been determined. In this paper we present the single crystal X-ray diffraction structure ofthe salt, N2H5(HOOC(CH2)2COO) (Fig. 2) along with its spectral and thermal properties.
2. Experimental
2.1. Preparation of Hydrazinium Hydrogensuccinate, N2H5(HOOC-(CH2)2-COO)
Hydrazinium hydrogensuccinate was prepared by mixing an aqueous solution of 10 % hydrazine hydrate (2 mL, 4 mmol) in 20 mL of distilled water and a 20 mL aqueous solution containing 0.476 g (4 mmol) succinic acid. The resulting clear solution was concentrated on a water-bath to about 20 mL and kept at room temperature for crystallization. The colourless crystals obtained after two days were separated and washed with dry ethanol, and air dried (yield: 85 %). Elemental analysis: C (%): 31.28 (Cald. 31.97); H (%): 5.90 (6.66); N (%): 17.93 (18.65); and % hydrazine: 21.08 (Cald. 21.31).
2.2. Physico-chemical Characterization
The hydrazine content of the salt was determined volumetri-cally by using a standard (0.025 M) KIO3 solution under Andrews' condition.27 IR spectra were recorded as KBr pellets with a Shimadzu FTIR 8000 spectrophotometer in the 4000-400 cm-1 range. Elemental analyses were performed on a Perkin-Elmer 240 B CHN analyser. Simultaneous TG-DTA measurements were carried out by using a Diamond TG/DTA thermal analyzer. The experiments were carried out in air with platinum cups as sample holders containing 5-10 mg of the samples at a heating rate of 10 °C min-1, up to 830 °C.
2.3. Single Crystal X-ray Diffraction Analysis
Preliminary examination and data collection were performed with a Bruker SMART Charge Coupled Device (CCD) Detector system single crystal X-ray diffractometer at 218 K using graphite monochromated Mo Κα radiation (λ = 0.71073 Å) equipped with a sealed tube X-ray source. The collected frames were integrated using an orientation matrix determined from the narrow frame scans. SMART and SAINT software packages28 were used for data collection and data integration. Analysis of the integrated data did not show any decay. Final cell constants were determined by a global refinement of xyz centroids. Collected data were corrected for systematic errors using SADABS29 based on the Laue symmetry using equivalent reflections. Structure solution and refinements were carried out by using the SHELXTL software package.30 The structure was solved by direct methods and refined successfully in the triclinic space group P 1. Full matrix least-squares refinement was carried out by minimizing Σ w(F02 - FC2)2. The non-hydrogen atoms were refined anisotropically to convergence. The hydrogen atoms were located and refined freely.
3. Results and Discussion
The analytical data for the hydrazinium hydrogensuccinate crystals (Fig. 2) agree with the proposed formula for the salt. The title compound is soluble in water and stable in air. The melting point of the salt was found to be 137-138 °C.
It is interesting to note that oxalic and succinic acids both form mono-hydrazinium and dihydrazinium salts, whereas malonic acid only forms the mono-hydrazinium salt, viz. N2H5(HOOC-CH2-COO). Sulfoacetic acid did not yield any expected hydrazinium salt. The reasons are very difficult to generalize.
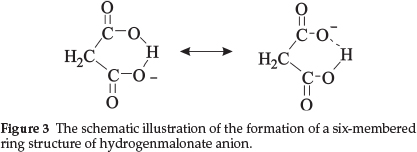
As reported,6 oxalic acid forms both salts. The hydrogenoxalate anion which is represented as HOOC-COO- has a strong electron withdrawing inductive effect (-I effect) due to the -OC-COO- group that makes the O-H bond more labile. This leads to the formation of a dihydrazinium salt. In the case of the hydrogenmalonate anion, the O-H bond is stabilized by the formation of a six-membered ring anion due to strong intramolecular hydrogen bonding as shown in Fig. 3 and as a result a proton cannot be abstracted by a weak base like N2H4 which prevents the formation of the dihydrazinium salt. In the case of the hydrogensuccinate anion, the intramolecular hydrogen bonding is not preferred due to an increase in size of the ring. Also the -I effect of the carboxylate group makes the O-H bond more labile. This helps to form the dihydrazinium salt.
3.1. Infrared Spectra
The IR spectra of succinic acid and its hydrazinium salt are given in Figs 4 and 5, respectively for comparison. The IR spectra of succinic acid and its monohydrazinium salt show absorption frequencies in the region 3465-3385 cm-1 due to O-H stretching of the free COOH group. The N-N stretching frequency of the free N2H5+ ion is known to occur in the 990-960 cm-1 region.1 The observed N-N stretching frequency at 965 cm-1 (Fig. 5) clearly reveals the presence of the N2H5+ ion in the salt. The bands at 1630 and 1410 cm-1, for the hydrazinium salt, are assigned to asymmetric and symmetric stretching frequencies of the carboxylate ions, respectively. The free carboxyl groups of the acid and salt show the free carbonyl asymmetric stretching frequencies in the region 1715-1705 cm-1.
3.2. Thermal Analysis
The simultaneous TG and DTA data of the hydrazinium hydrogensuccinate salt are shown in Fig. 6. The DTA of the hydrazinium hydrogensuccinate salt shows two endotherms (~135 and ~168 °C); the first sharp endotherm at 135 °C is attributed to melting (no mass loss observed in TG), whereas the second endotherm at 168 °C may be assigned to dehydra-zination of the melt to form succinic acid as an intermediate which further undergoes exothermic (multiplets) decomposition to give gaseous products similar to that of other simple salts.5 The decomposition of the melt is initiated at 168 °C and 99.9 % loss was observed by 600 °C. This is observed as a continuous decomposition in TG.
3.3. Single Crystal X-ray Diffraction Studies
Results of the single crystal X-ray diffraction studies for N2H5(HOOC(CH2)2COO) are presented in Tables 1-3.


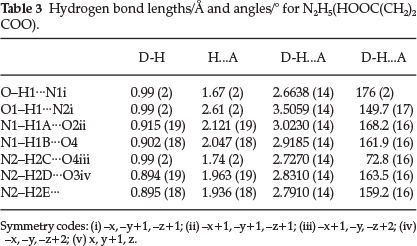
The crystal structure is built up of N2H5+ cations and hydrogen succinate anions, HOOC(CH2)2COO-, which are joined by hydrogen bonds. The structure of N2H5+(HOOC(CH2)2COO)- is shown in Fig. 7. The N-N distance of the N2H5+ ion is 1.4535 (14) A which is in agreement with the value reported for N2 H5HC2O424,25 in which the N-N bond length is 1.443 (5) A. The C-O and C-C bond lengths (Table 2) of the carboxylate groups are in agreement with the corresponding bond distances reported for hydrazinium hydrogenoxalate.24,25
The crystal structure is stabilized through N-H···Ό and O-H···O hydrogen bonds. The crystal packing is shown in Fig. 8. The packing consists of infinite chains of anions and cations that are alternatively linked through O-H···N and bifurcated N-H···O (O1-H1···N1, N2-H2C···O3 and N2-H2C···O4) hydrogen bonding with bond lengths of 2.6638 (14) - 3.2191 (14) A and this chain is connected to adjacent antiparallel chains by N1-H1B···O4 hydrogen bonding. These antiparallel chains run across the bc plane. Further, the adjacent antiparallel chain pairs are linked through N2-H2E···O3 bonding to form a two- dimensional sheet-like structure. These sheets are stacked along the a axis through N1-H1A-O2 and N2-H2D-O3 interactions furnishing a three-dimensional network structure. In simple salts, generally the hydrogen of the carboxylate adopts head to tail O-H-O hydrogen bonding leading to one-dimensional chains. Surprisingly, this is absent in this case, whereas the same is effectively present in hydrazinium hydrogen oxalate.24 The OH and NH hydrogen atoms form H-bonds with bond distances D-H = 0.894 (19) to 0.99 (2) A and H-A = 1.67 (2) to 2.61 (2) A. The hydrazinium nitrogen N2 is bonded to the hydrogens H2C, H2D and H2E with NH distances of 0.99(2), 0.894 (19) and 0.895 (18) A (Table 3), respectively. Elongation of one of the three NH bonds (N2-H2C bond) is most likely due to the fact that it is trans to a strong O-H---N hydrogen bond (torsion angle (O1)H1-N1-N2-H2C = 179.6 °).
4. Conclusions
The reaction of succinic acid with hydrazine hydrate gives the hydrazinium hydrogensuccinate salt. The salt undergoes melting followed by decomposition to give gaseous products.
The crystal structure of hydrazinium hydrogensuccinate has been determined by means of X-ray diffraction. The structure contains N2H5+ cations and hydrogen succinate anions, HOOC(CH2)2COO-, which are joined by hydrogen bonds. The crystal structure is stabilized through N-H···O and O-H···O hydrogen bonds. It has been observed that the hydrazinium nitrogen N2 is bonded to the hydrogens H2C, H2D and H2E with NH distances of 0.99 (2), 0.894 (19) and 0.895 (18) A, respectively. Elongation of one of the three NH bonds (N2-H2C bond) is most likely due to the fact that it is trans to a strong O-H···N hydrogen bond (torsion angle (O1)H1···N1-N2-H2C = 179.6 °).
Acknowledgements
Funding from the National Science Foundation (USA) for purchase of the X-ray diffractometer (MRI, CHE-0420497) is acknowledged.
References
1 E.W. Schmidt, Hydrazine and its derivatives - Preparation, Properties and Applications, Wiley Interscience, New York, 1984. [ Links ]
2 S. Yasodhai and S. Govindarajan, Thermochim. Acta, 1999, 338, 113-123. [ Links ]
3 S. Govindarajan, S.U.N. Banu, N. Saravanan and B.N. Sivasankar, Proc. Indian. Acad. Sci. (Chem. Sci), 1995,107, 559-565. [ Links ]
4 S. Yasodhai and S. Govindarajan, J. Therm. Anal., 2000, 62, 737-745. [ Links ]
5 K. Kuppusamy, B.N. Sivasankar and S. Govindarajan, Thermochim. Acta, 1995, 259, 251-262. [ Links ]
6 D. Gajapathy, S. Govindarajan and K.C. Patil, Thermochim. Acta, 1983, 60, 87-92. [ Links ]
7 M. Currie, J.C. Speakman and N.A. Curry, J. Chem. Soc. (A), 1967, 1862-1869. [ Links ]
8 D.J. Hass, Acta Cryst, 1964,17, 1511-1516. [ Links ]
9 K. Browning, K.A. Abbound and G.J. Palenik, J. Chem. Crystallogr., 1995, 25, 851-855. [ Links ]
10 P. Laine, A. Gourdon and J.-P. Launay, Inorg. Chem., 1995, 34, 5129-1537. [ Links ]
11 S. Govindarajan, K.C. Patil, M.D. Poojary and H. Manohar, Inorg. Chim. Acta, 1986,120, 103-107 [ Links ]
12 S. Govindarajan, K.C. Patil, H. Manohar and P.E. Werner, J. Chem. Soc. Dalton Trans., 1986, 119-123. [ Links ]
13 S. Yasodhai, T. Sivakumar and S. Govindarajan, Thermochim. Acta, 1999, 338, 57-65. [ Links ]
14 K.C. Patil, J.P. Vittal and C.C. Patel, Thermochim. Acta, 1981, 43, 213-219. [ Links ]
15 K.C. Patil, J.P. Vittal and C.C. Patel, J. Fire Retard. Chem., 1980, 7, 3-8. [ Links ]
16 S. Chandra and N. Singh, J. Phys. C: Solid State Phys, 1983, 16, 3081-3097. [ Links ]
17 W. Starosta and J. Leciejewicz, Acta Cryst. E, 2007, 63, o3734 (Suppl. 1-7). [ Links ]
18 W. StarostaandJ. Leciejewicz, Acta Cryst. E, 2008,64, o46 (Suppl. 1-3). [ Links ]
19 W. Starosta and J. Leciejewicz, The Open Cryst. J., 2008, 1, 31-36. [ Links ]
20 M. Delfino, J.C. Jacco, P.S. Gentile and D.D. Bray, J. Solid State Chem., 1977, 21, 243-251. [ Links ]
21 F.H. Allen and O. Kennard, 3D search and research using the Cambridge Structural Database, Chem. Des. Autom. News, 1993, 8, 31. [ Links ]
22 V.S.S. Kumar, T. Premkumar, N.P. Rath and S. Govindarajan, Indian J. Chem. B, 2007,46, 141-147. [ Links ]
23 I.L. Pratt and R.E. Richards, Trans. Faraday Soc, 1953, 49, 744-751. [ Links ]
24 N.B.K. Ahmed, R. Liminga and I. Olovsson, Acta Chem. Scand., 1968, 22, 88-96. [ Links ]
25 J.O. Thomas, Acta Cryst. B, 1973, 29, 1767-1776. [ Links ]
26 A. Nilsson, R. Liminga and I. Olovsson, Acta Chem. Scand., 1968, 22, 719-731. [ Links ]
27 A.I. Vogel, A Text Book of Quantitative Inorganic Analysis, 4th edn., Longman, London, 1986. [ Links ]
28 Bruker Analytical X-Ray; APEX2 and SAINT;Bruker AXS Inc., Madison, Wisconsin, USA, 2004. [ Links ]
29 R.H. Blessing, Acta Cryst. A, 1995, 51, 33-38. [ Links ]
30 G.M. Sheldrick, Acta Cryst. A, 2008, 64, 112-122. [ Links ]
Received 14 May 2013
Revised 7 March 2014
Accepted 25 March 2014
* Authors for correspondence. E-mail: TP: thathanpremkumar@gmail.com / SG:drsgovind@yahoo.co.in














