Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Chemistry
On-line version ISSN 1996-840X
Print version ISSN 0379-4350
S.Afr.j.chem. (Online) vol.67 Durban Jan. 2014
RESEARCH ARTICLE
Conformational Comparison of Cyclic α3β Tetrapeptide vs. α3β Pentapeptide
Sachin A. PawarI; Amit M. JabgundeII; Glenn E. M. MaguireI; Dilip D. DhavaleII; Thavendran GovenderI; Hendrik G. KrugerI, *; Fernando AlbericioI, III, IV, *
ICatalysis and Peptide Research Unit, School of Health Sciences, University of KwaZulu-Natal, South Africa
IIGarware Research Centre, Department of Chemistry, University of Pune, Pune-411007 India
IIIInstitute for Research in Biomedicine and CIBER BBN, 08028-Barcelona
IVDepartment of Organic Chemistry, University of Barcelona, Barcelona 08028, Spain
ABSTRACT
The γ-turn inducing sugar β-amino acid plays an important role in the formation of well-defined secondary structures of cyclic tetra- and pentapeptide. Two tetra- and one novel pentapeptide were synthesized from a sugar β-amino acid and the conformations of the compounds were established by NMR spectroscopy (EASY-ROESY) and compared with CD spectroscopic data. Although the γ-turn conformation was dominant for both tetra- and pentapeptide according to NMR spectroscopic analysis in DMSO, the pentapeptide exhibited the presence of a second conformation. CD spectroscopic results in methanol showed that the tetrapeptides have a γ-turn conformation, while the pentapeptide has a random structure.
Keywords: : Cyclic peptide, tetrapeptide, pentapeptide, conformational analysis, NMR spectroscopy, CD spectroscopy.
1. Introduction
Self-organization and self-assembly of peptides are emerging as a new approach to answer relevant questions in a broad number of areas such as biomedicine and biomaterials, amongst others.1-4 Peptide self-assembly originates from non-covalent and reversible interactions, such as hydrogen bonds, hydrophobic, Van der Waals and aromatic p-stacking interactions.1-4
Cyclic peptides derived from sugar b-amino acids are known to form biologically significant secondary structures. These macromolecules do not only form helices5 and turns6 but also self-assembled materials such as nanotubes.7 The self-assembly and ordered arrangements of sugar b-amino acid-based cyclic peptides have been studied with many different spectroscopic techniques.5-8 Cyclic sugar b-amino acid peptides also exhibit medicinal applications such as chemotherapeutic,9 anti-HIV,10-12 anti-proliferative13,14 and apoptotic agents.13-14
There are some reports in the literature of cyclic pentapeptide containing residues which induce more than one conformation such as b-turn and g-turn within the same peptide.15,16 As part of an ongoing project on hydroxyl propyl amine-based glycopep-tides as anti HIV agents,12,17 several cyclic glycopeptides were synthesized.12,17 γ-Turn structures are defined as peptides where the amide CO of the ith residue forms a hydrogen bond with the NH of the i + 2th residue.18
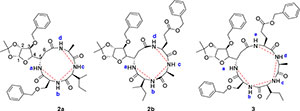
Herein we report a comparative conformational analysis of cyclic tetra- and pentapeptide (see Fig. 1). We have optimized the solution phase cyclization of linear glycopeptides for the effective synthesis of the cyclic peptides12 from L-ido sugar β-amino acid (1). The conformation of cyclic tetra- (2)12 and the novel pentapeptide (3) were studied by NMR spectroscopy, using EASY-ROESY experiments and CD spectroscopy.
2. Materials and Methods
Melting points were measured using a Stuart Scientific Melting Point Apparatus SMP3. Optical rotation was measured using a Perkin Elmer Instruments model 341 polarimeter with sodium light (589.0 nm). Infrared spectra (IR) were measured using a Perkin Elmer Precisely Spectrometer100 FT-IR spectrometer. All 1H, 13C COSY, HSQC, HMBC and ROESY spectra were recorded on a 9.4T Bruker AVANCE III 600 MHz instrument at room temperature (298 K) with DMSO as an internal standard using a sample concentration in the range of 20 (mg mL-1). High-resolution mass spectroscopic analysis was performed on a Bruker MicroTOF QII mass spectrometer in positive mode with an internal calibration. The compound was purified using semi-preparative HPLC on a Shimadzu Instrument LC-8A (Ace C18 150 mm x 21.2 mm x 5 microns) with a UV/VIS detector SPD-M20A (215 nm and 254 nm) and automated fraction collector FRC-10a with 15 mL min-1 flow rate. Analytical analysis of the compounds was performed on a liquid chromatograph mass spectrometer LCMS2020 (Waters Xbridge C18 150 mm x 4.6 mm x 5 microns) coupled to a UV detector (215 nm and 254 nm) and a Shimadzu mass spectrophotometer in the positive mode. The chemicals DIPEA, DMF, EtOH and TFA were purchased from Sigma-Aldrich Company and CTC resin, HCTU, HOBt and EDC were purchased from GL Biochem Ltd. (Shanghai).
CD spectroscopy: CD spectra were recorded at 298 K on a Jasco J-815 spectropolarimeter by using 0.1 cm path length quartz cells. The CD data are averages of four scans, collected at 0.1 nm intervals between 190 and 290 nm with a scanning speed of 50 nm min-1. The peptide solutions were prepared at concentrations of 0.1 mm in CH3OH. Ellipticity is reported as mean residue ellipticity [q], with approximate errors of 10 % at 220 nm.
3. Results and Discussion
3.1. Synthesis of Cyclic Peptides
Peptide 3 was synthesized by a combination of solid-phase using CTC-resin for the elongation of the linear peptide and in solution for the last cyclization step (coupling reagents EDC and HOBt), followed by cleavage from the resin (Scheme 1). The peptide was obtained with an overall yield of 85 %, a purity of >95 % and used without further purification.
3.2. Conformational Analysis with NMR Spectroscopy
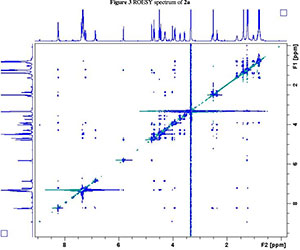
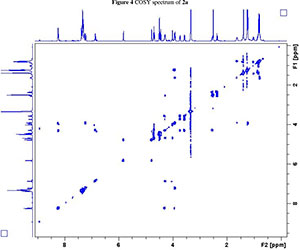
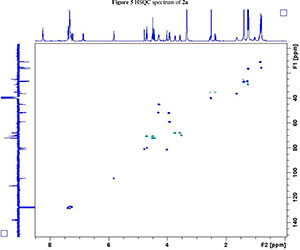
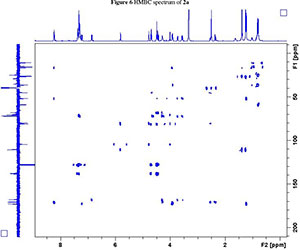
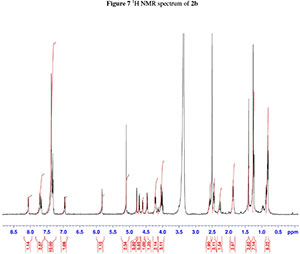
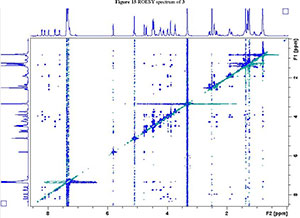
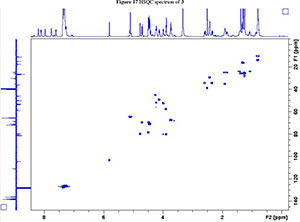
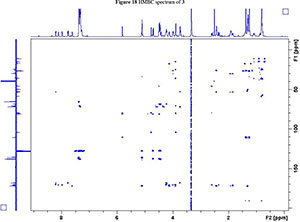
NMR spectroscopy studies of 2a, 2b, and 3 were carried out in DMSO, and the signal assignments were made with the aid of 2-D COSY, HSQC, HMBC and EASY ROESY NMR spectroscopy experiments. A previous report on cyclic sugar /3-amino acid tetrapeptides conformations, concluded that cyclic tetrapeptides containing an L-ido sugar /3-amino acid (1) form g-turn structures.6 The important differences observed between peptide 2a, 2b and 3 from the NMR spectroscopy studies are (a) significant deshielding of proton NHa in peptide 3 compared to 2a and 2b, (see Fig. 2) and (b) lowering of the JNHe-CaH value for the Glu residue in peptide 3. In our opinion, this could indicate the existence of another conformation along with the g-turn induced by the L-ido sugar /3-amino acid. Table 1 shows the complete comparison of chemical shifts (d) and coupling constants (J) for cyclic peptides 2a, 2b and 3.
The dihedral angle j (C'-N-Ca-C') is related to 3JNHCaH through the Karplus curve, small values of 3JNH-CaH (<4 Hz) indicates smaller j values (±60°) and large 3JNH-CaH (>8 Hz) specifies j values of ±120°.19 The existence of similar JNH-CaH constants for sugar amino acid residues in 2a, 2b and 3 suggest a nearly equal angle (j) at the corresponding residue. The low JNH-CaH value (5.1 Hz) of Glu in 3 indicates a smaller j value for this segment.19 This clearly reveals the presence of an additional secondary arrangement apart from the g-turn (3 → 1) in peptide 3. According to literature6 the observed NOE interactions of the amide protons in peptide 3 (see Fig. 2b) for the sugar /3-amino acid supports a g-turn structure defined by the angle between NH-Ser CαH, Ser NH-Ile CαH, Ile NH-Ala CαH and Ala NH-GluCαH (red dotted lines in Fig. 2). Possible hydrogen-bonding interaction for NH of Glu causes lowering of JNH-CaH and deshielding of the chemical shift value with respect to the NH of the other amino acid in peptide 3. This interaction is potentially the result of another conformation.
3.3. CD Analysis
The CD spectra of the cyclic tetrapeptides 2a and 2b in methanol displayed minima at ~225 nm, which is characteristic for g-turns in cyclic peptides.12,20-22 Subsequently the CD spectra of 3 showed minima at ~208 nm in MeOH as shown in Fig. 3 suggesting a loss of g-turn character. This result suggests that 3 is more flexible than the corresponding tetrapeptides.
According to literature for cyclic peptides, it was observed that for tetra-,21-24 penta-25 and hexapeptides25 that the g-turn character of these peptides can be lost upon changing the solvent from aprotic (acetonitrile dicholoromethane, chloroform) to protic solvents (methanol, water).21-27 For example, if the CD spectrum is recorded in dichloromethane, then γ-turns in cyclic peptides are observed (minima of about 225 nm in CD spectra). When the protic character of the solvent is systematically increased by addition of a protic solvent, then the minima in CD spectra gradually decrease to about 205 nm.21
These observations from literature appear to suggest that the tetra-glycopeptides reported herein are quite rigid, since the CD spectroscopic analysis in methanol, still reveal γ-turn character. However, the cyclic penta-glycopeptide seems to have lost this character in polar solvent according to CD spectra in methanol. When these penta-glycopeptide are analyzed with NMR spec-troscopy in an aprotic solvent such as DMSO, then a γ-turn character of the peptides is observed. It is important to point out that the Karplus relationship (3J values) used for characterization of the j values is quite crude.28 For example, a typical experimental 3J value of 2-4 Hz is associated with a dihedral angle of about 60° while a value of 5-8 Hz represents a 180° dihedral angle. According to Karplus theoretical J-values of 6.0,1.7, 2.2 and 9.2 Hz are 30°, 60°, 120° and 180° respectively.
4. Conclusions
The novel cyclic pentapeptide containing an L-ido 3-amino acid, which is a known turn inducing residue, was synthesized by a combination of solid-phase and solution phase. Comparison of NMR and CD spectroscopic data suggests that the expansion of the cyclic tetrapeptide with one additional residue leads to a more flexible conformation, rendering a peptide with both a gamma and random conformation. CD spectroscopic analysis of the latter in a protic solvent indicates a loss of the g-turn character, while it is retained for the tetrapeptide. These results should be taken into account when designing turn inducer building block containing cyclopeptides for different biological targets.
5. Experimental
5.1. Procedure for the Synthesis of Compound 4
(6S,9S,12S,15S)-6-{(S)-3-amino-3-[(3aR,5R,6S,6aR)-6-(benzyl-oxy)-2,2-dimethyltetrahydrofuro[2,3-rf][1,3]dioxol-5-yl]propan-amido}-15-(benzyloxymethyl)-12-sec-butyl-9-methyl-3,7,10, 13-tetraoxo-1-phenyl-2-oxa-8,11,14-triazahexadecan-16-oic acid (4)
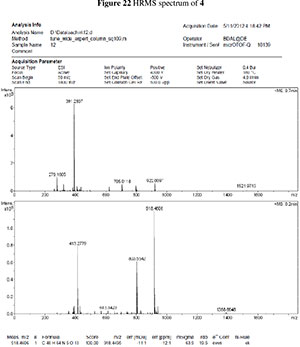
The peptide 4 was synthesized manually on an activated29 2-chlorotrityl chloride resin (1 g) which had been swelled in dry DCM for 30 min. The first amino acid Fmoc-Ser-OH was coupled to the resin using dry DCM (10 mL) and DIPEA (6.65 mmol) for 2 h under a N2 atmosphere. Resin substitution was 0.6 mol g-1 as determined by the Fmoc UV assay. The Fmoc group of serine was removed using 20 % piperidine in DMF (3 x 10 mL) for 30 min. Subsequently, amino acids Fmoc-Ile-OH, Fmoc-Ala-OH, and Fmoc-Glu-OH (0.20 mM) were coupled using DIPEA (1 mM) and HCTU (0.50 mM) in DMF. The Fmoc group of glutamic acid was removed using 20 % piperidine in DMF (3 x 10 mL) for 30 min and finally sugar amino acid 1 was coupled using DIPEA (1 mM) and HCTU (0.50 mM) in DMF. Standard washing procedures were used. Cleavage from the resin was performed, using a cleavage mixture of 5 % TFA in DCM (3 x 10 mL) for 30 min. The crude compound was then purified using semi-preparative HPLC on a Shimadzu Instrument LC-8A (Ace C18 150 mm x 21.2 mm x 5 mm) with an UV/VIS detector SPD-M20A (215 nm and 254 nm) and automated fraction collector FRC-10a with 15 mL min-1 flow rate and two buffer system water and MeOH containing 0.1 % HCOOH with method RP-HPLC: 5 % MeOH /95 % H2O/0.1 % HCOOH to 95 % MeOH /5 % H2O/0.1 % HCOOH in 35 min. Compound 4 was obtained in 64 % yield as a white solid, mp 171 °C. Elution time = 24.5 min. [a]D20 - 10.0 (c 0.1 mg mL-1, MeOH); vmax (neat)/cm-1 3277, 2928, 1625, 1526; 1H NMR (400 MHz, DMSO-d6,) 8.50(s, 1H), 8.28(s, 1H), 7.86-7.91(m, 2H), 7.28-7.34 (m, 15H), 5.89(s, 1H), 5.07(s, 2H), 4.80(s, 1H), 4.64(d, J = 10.0 Hz, 1H), 4.22-4.46(m, 8H), 3.91-4.04 (m, 3H), 3.68 (d, J = 13.6 Hz, 1H), 2.35-2.40 (m, 4H), 1.92(s, 1H), 1.75-.81(m,2H),1.40(m,1H),1.37(s,3H),1.25(m,3H),1.19(d,J= 5.2 Hz, 3H), 1.06 (s, 1H), 0.75-0.86(m, 6H) 13C NMR (100 MHz, DMSO-d6,) 172.2, 171.8, 170.6, 170.3, 169.4, 138.3, 137.5, 136.1, 128.3, 128.2, 128.0, 127.9, 127.8, 127.6, 127.4, 127.2, 110.9, 104.3, 81.2,80.6,72.0,70.6,70.0,65.4,56.9,53.2,51.5,48.2,47.2,36.8,30.0, 27.6, 26.5, 26.1, 24.0,17.7,15.2,11.1. HRMS (ESI+) m/z calcd. for C48H64N5O13; 918.4495; found [M+H] 918.4606
5.2. Synthesis of Compound 3
Benzyl 3-{(2S,5S,8S,11S,14S)-14-[(3aR,5R,6S,6aR)-6-(benzyloxy)-2,2-dimethyltetrahydrofuro[2,3-rf][1,3]dioxol-5-yl]-11-(benzyloxymethyl)-8-sec-butyl-5-methyl-3,6,9,12,16-penta-oxo-1,4,7,10,13-pentaazacyclohexadecan-2-yl}propanoate (3)
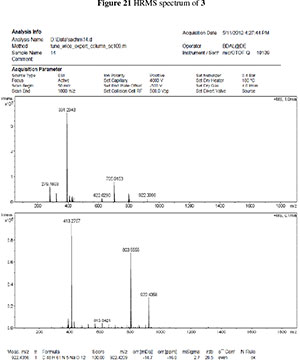
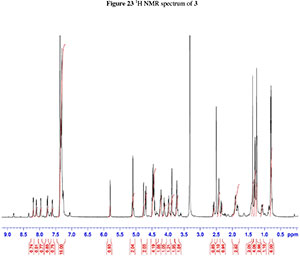
To a stirred solution of 4 in dry DCM, HOBt (5 eq.) and EDC (5 eq.) were added at 0 °C. The reaction mixture was then stirred at RT for 24 h. The reaction mixture was diluted with DCM and washed with 1M HCl, organic layer dried on Na2SO4, filtered off, and evaporated to dryness. The crude cyclic peptide was purified with silica-gel column chromatography using 2 % MeOH in DCM in 85 % yield as a white solid, mp 171 °C. Rf = 0.4 (5:95 MeOH:DCM) [a]D2010 (c 0.1 mg mL-1, MeOH); vmx (neat)/cm-1 3281, 2926, 1734, 1639, 1650, 1626, 1531; 1H NMR (400 MHz, DMSO-d6,) 8.19(d, J = 6.4 Hz, 1H), 8.10(d, J = 7.9 Hz, 1H), 7.96(d, J = 5.1 Hz, 1H), 7.75(d, J = 8.8 Hz, 1h), 7.60(d, J = 6.9 Hz, 1H), 7.26-7.36(m, 12H), 5.80(d, J = 3.0 Hz, 1H), 5.09 (d, J = 2.9 Hz, 2h), 4.76(d, J = 3.1 Hz, 1H), 4.69 (d, J = 11.2 Hz, 1H), 4.41-4.49(m, 4h), 4.20^.24(m, 2H), 4.12 (t, J = 7.9 Hz, 1H), 3.98(d, J = 7.9 Hz, 1h), 3.88-3.90(m, 2H), 3.71-3.73(m, 2H), 2.58 (d, J = 12.5 Hz, 1H), 2.41-2.42 (m, 2H), 2.34 (d, J = 14.3 Hz, 1H), 1.82-1.91 (m, 3h), 1.40(m, 1H), 1.37 (s, 3H), 1.29 (d, J = 7.0 Hz, 4H), 1.24 (m, 3h), 1.06-1.08 (m, 1H), 0.75-0.80(m, 6H) 13C NMR (100 MHz, DMSO-d6,) 172.1, 172.0, 170.8, 170.7, 170.3, 168.2, 138.1, 137.6, 136.0, 128.4, 128.2, 128.0, 127.9, 127.7, 127.6, 127.5, 127.4, 127.3, 110.7, 104.2, 81.1, 80.9, 73.3, 69.7, 61.9, 57.1, 52.0, 48.3, 47.5, 36.6, 27.5, 26.1, 26.1, 24.1,17.8,15.3,11.1. HRMS (ESI+) m/z calcd. for C48H61N5NaO12; 922.4209; found [M+Na] 922.4356
Acknowledgements
We thank the National Research Foundation, South Africa (Project No. UID-6982, Department of Science and Technology), Government of India (Project No. INT/SAFR/P3 (2)/2009) for financial support under the Indo-South Africa collaborative research programme, University of KwaZulu-Natal, Pune University and the Centre for High Performance Computing (www.chpc.ac.za), South Africa, for computational support.
Supplementary Material
All copies of the 1H, 13C and HRMS spectra of all compounds, and the 2D NMR spectra of 2a, 2b and 3, are given in the online supplement.
Abbreviations
CD, circular dichroism; CTC, 2-chlorotrityl chloride; DCM, dichloromethane; DIPEA, N-ethyl-N-isopropylpropan-2-amine; DMF, N,N-dimethylformamide; EDC, 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide; EASY ROESY, Efficient Adiabatic Symmetrized Rotating frame Overhause Effect Spec-troscopy, EtOH, ethanol; HCTU, 2-(6-chloro-1H-benzotriazol-1-yl)-N,N,N'N'-tetramethyluronium hexafluorophosphate; HOBt, N-hydroxybenzotriazol; HCOOH, formic acid; HIV, human immunodeficiency virus; MeOH, methanol; HPLC, high performance liquid chromatography; TFA, trifluoroacetic acid.
References
1 G.M. Whitesides, J.P. Mathias and C.T. Seto, Science, 1991, 254, 1312. [ Links ]
2 G.M. Whitesides and B. Grzybowski, Science, 2002, 295, 2418. [ Links ]
3 J.M. Lehn, Science, 2002, 295, 2400. [ Links ]
4 R. Fairman and K.S. Akerfeldt, Curr. Opin. Struct. Biol. 2005,15, 453. [ Links ]
5 S.A.W. Gruner, V. Truffault, G. Voll, E. Locardi, M. Stockle and H. Kessler, Chem. Eur. J., 2002, 8, 4366. [ Links ]
6 A. Sharma, S. Sharma, R.P. Tripathi and R.S. Ampapathi, J. Org. Chem., 2012, 77, 2001. [ Links ]
7 B. Jagannadh, M.S. Reddy, C.L. Rao, A. Prabhakar, B. Jagadeesh and S. Chandrasekhar, Chem. Commun., 2006, 4847. [ Links ]
8 E.G. vonRoedern, E. Lohof, G. Hessler, M. Hoffmann and H. Kessler, J. Am. Chem. Soc., 1996,118, 10156. [ Links ]
9 S. Chandrasekhar, C.L. Rao, M. Seenaiah, P. Naresh, B. Jagadeesh, D. Manjeera, A. Sarkar and M.P. Bhadra, J. Org. Chem., 2009, 74, 401. [ Links ]
10 E.N. Prabhakaran, V. Rajesh, S. Dubey and J. Iqbal, Tetrahedron Lett., 2000, 42, 339. [ Links ]
11 E.N. Prabhakaran, I.N. Rao, A. Boruah and J. Iqbal, J. Org. Chem., 2002, 67, 8247. [ Links ]
12 S.A. Pawar, A.M. Jabgunde, G.E.M. Maguire, H.G. Kruger, Y. Sayed, M.E.S. Soliman, D.D. Dhavale and T. Govender, Eur. J. Med. Chem., 2013, 60, 144. [ Links ]
13 S.A.W. Gruner, G. Keri, R. Schwab, A. Venetianer and H. Kessler, Org. Lett., 2001, 3, 3723. [ Links ]
14 T.K. Chakraborty, P. Srinivasu, S. Tapadar and B.K. Mohan, J. Chem. Sci., 2004,116, 187. [ Links ]
15 L.G. Pease and C. Watson, J. Am. Chem. Soc., 1978,100, 1279. [ Links ]
16 I.L. Karle, J. Am. Chem. Soc., 1978,100, 1286. [ Links ]
17 S.A. Pawar, A.M. Jabgunde, P. Govender, G.E.M. Maguire, H.G. Kruger, R. Parboosing, M.E. S. Soliman, Y. Sayed, D.D. Dhavale and T. Govender, Eur. J. Med. Chem., 2012, 53, 13. [ Links ]
18 L. Belvisi, C. Gennari, A. Mielgo, D. Potenza and C. Scolastico, Eur. J. Org. Chem., 1999, 389. [ Links ]
19 S. Raghothama, J. Indian Inst. Sci., 2010, 90:1, 145. [ Links ]
20 V. Madison, M. Atreyi, C.M. Deber and E.R. Blout, J. Am. Chem. Soc., 1974, 96, 6725. [ Links ]
21 M. Tamaki, M. Yabu, T. Saeki, A. Kaneko, S. Akabori and I. Mura-matsu, Bull. Chem. Soc. Jpn., 1999, 72, 733. [ Links ]
22 C. Spezzacatena, A. Pepe, L.M. Green, L.B. Sandberg, B. Bochicchio and A.M. Tamburro, Eur. J. Org. Chem., 2005, 1644. [ Links ]
23 D.H. Rich and R.D. Jasensky, J. Am. Chem. Soc., 1980, 102, 1112. [ Links ]
24 E.Vass,Z.Majer,K. KohalmyandM. Hollosi, Chirality, 2010,22,762. [ Links ]
25 M. Malesevic, Z. Majer, E. Vass, T. Huber, U. Strijowski, M. Hollosi and N. Sewald, Int. J. Pept. Res. Ther., 2006, 12, 165. [ Links ]
26 E. Vass, M. Kurz, R.K. Konat and M. Hollosi, Spectrochim. Acta Part A Mol. Biomol. Spectrosc., 1998, 54, 773. [ Links ]
27 V. Madison and K.D. Kopple, J. Am. Chem. Soc., 1980,102, 4855. [ Links ]
28 M. Karplus, J. Chem. Phys., 1959, 30, 11. [ Links ]
29 M. Harre, K. Nickisch and U. Tilstam, React. Funct. Polym., 1999,41,111. [ Links ]
Received 27 January 2014
Revised 11 April 2014
Accepted 23 April 2014
*Authors for correspondence. E-mail: kruger@ukzn.ac.za (G.K.) / albericio@irbbarcelona.org (F.A.)
Appendix
Table of Contents
1 Table 1: 1H NMR Chemical Shifts (δ, ppm) and Coupling Constants (J, Hz) of glycopeptides 2a, 2b and 3
2. 1H, 13C, COSY, ROESY, HSQC and HMBC Spectra's of compound 2a, 2b and 3
3. HRMS spectras of 3 and 4
Material and Methods
High resolution mass spectroscopic analysis was performed on a Bruker MicroTOF QII mass spectrometer in positive mode with an internal calibration. All 1H, 13C COSY, HSQC, HMBC spectra were recorded on a 9.4T Bruker AVANCE III 600 MHz instrument at room temperature (298K) with DMSO as an internal standard using a sample concentration in the range of 20 (mg/ml) and all ROESY Experiments were performed at room temperature (298K). Samples were dissolved in 450 µ! of deuterated DMSO-d6 and measured at 25 °C.. Processing and assignments were carried out using the Topspin 2.3 software from Bruker Karlsruhe.