Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
South African Journal of Animal Science
versão On-line ISSN 2221-4062
versão impressa ISSN 0375-1589
S. Afr. j. anim. sci. vol.52 no.1 Pretoria 2022
http://dx.doi.org/10.4314/sajas.v52i1.5
ARTICLES
Feeding Nigella sativa oil to broilers affects their performance, serum constituents and cecum microbiota
A.M. SaiedI; A.I. AttiaI; M.S. El-KholyI; A.G. EL NagarII, #; F.M. RedaI
IPoultry Department, Faculty of Agriculture, Zagazig University, Zagazig 44511, Egypt
IIDepartment of Animal Production, Faculty of Agriculture at Moshtohor, Benha University, 13736, Egypt
ABSTRACT
The objective of this study was to assess the effect of supplementation of Nigella sativa oil (NSO) as a growth promoter in broiler diets. A total of 300 unsexed one-day-old Arbor Acre broiler chicks were randomly divided into five treatment groups (60 chicks/group). Each group was divided into five replicates with 12 chicks per replicate. Group 1 was fed on the basal diet with no additives (control). Group 2 was fed the basal diet supplemented with oxytetracycline (OTC) at the level of 50 ppm. Groups 3, 4 and 5 were fed the basal diet supplemented with 500, 1000 and 1500 ppm NSO, respectively. Birds fed on 1500 ppm NSO diet showed the heaviest body weight at 14, 28 and 42 days of age. Feed conversion ratio was significantly improved at all ages with the inclusions of OTC and NSO generally superior to the control group. The relative weight of spleen was increased significantly by the addition of NSO at various levels. However, relative weights of thymus and bursa were not altered by the treatments. Total bacterial count, total yeast and moulds count, E. coli and salmonella spp. counts were reduced significantly, whereas the total lactic acid bacteria count was increased in OTC and NSO groups compared with control. Nigella sativa oil could be used in broiler chicken feeds as a natural alternative to antibiotic growth promoters to improve gut health and consequently growth performance.
Keywords: growth, feed conversion, immunity, organ weight
Introduction
In recent decades, the attention of poultry breeders has been on the use of growth promoters to improve performance. As a result, antibiotics were used widely in the poultry industry (Attia & Al-Harthi, 2015). Antibiotics had the desired effects of improving growth performance, but increased resistance of harmful bacteria to antibiotics (Kim et al., 2011; Hassanpour et al., 2013). After the European Union banned the use of antibiotics as growth promoters in animal feeds in January 2006, the quest for natural growth promoters (Saleh, 2014) became critical (Toghyani et al., 2010).
The seeds and oil of Nigella sativa (black cumin) have long been used to treat physiological disorders (Attia & Al-Harthi, 2015). Nigella sativa is a medicinal plant that could be used as a natural feed substitute in poultry and livestock production to increase efficiency and minimize the risk of antibiotic-resistant bacteria to humans and animals (Doyle, 2001). There is little information in the literature about the effects of NSO on performance and efficiency, carcass traits, and haemato-biochemical and immunological values of broiler chickens, with contradictory findings, depending on the concentration of NSO (Toghyani et al., 2010; Durrani et al., 2007). Hermes et al. (2009) found that adding 0.5% and 1% NSO to the broiler diet improved growth, white blood cells (WBCs), packed cell volume (PCV), and feed conversion ratio (FCR) significantly, whereas triglycerides and alanine aminotransferase (ALT) were decreased significantly compared with the untreated control group. Therefore, this study was conducted to evaluate the impact of NSO as a natural alternative to antibiotic growth promoters on growth performance, carcass traits, haemato-biochemical parameters, immune function, antioxidant status, and cecal microbiota in broiler chicks.
Materials and Methods
The current study was conducted at a private poultry farm in San-El-Hagar city, Sharkia Governorate, Egypt, according to the guidelines of the Advisory Committee on the Ethics of Animal Experiments, Poultry Department, Zagazig University, Egypt. The laboratory analyses, experiments and protocols were handled in accordance with the ethical standards as stated in the guidelines represented by the Committee of Animal Care and Welfare, Benha University, Egypt, Ethical Approval No. 2020-4.
A total of 300 unsexed one-day-old Arbor Acre broiler chicks were randomly divided into five treatment groups each of 60 chicks. Each group was split into five replicates, each containing 12 chicks. The replicates were placed in separate pens (100 χ 120 cm). As a control, the first group was fed only the basal diet with no supplements. The second group was given the basal diet plus 50 ppm oxytetracycline (OTC). The third, fourth, and fifth groups were given the basal diet plus 500, 1000, and 1500 ppm Nigella sativa oil, respectively. The NSO was purchased from El Hawag Company for Natural Oils, Cairo, Egypt. The basal diet was created to meet the nutritional requirements of broiler chicks from hatch to 42 days old (NRC, 1994).
Table 1 shows the composition and chemical analysis of the basal diet. All of the birds were reared under the same management and sanitary conditions. A total of 23 hours of light per day were provided by an artificial light source. Feed and water were freely available. The experimental period extended for 42 days (1 -42 days old). At 1, 14, 28, and 42 days old, chicks were weighed and feed intake (FI) was measured biweekly. The feed conversion ratio (g feed/g gain) and bodyweight gain were calculated.
Five birds from each treatment were chosen randomly to represent all replicates, weighed, and slaughtered according to Islamic procedure on day 42. Blood samples were obtained in heparinized tubes for laboratorial analyses. The liver, gizzard, heart, and lymphoid organs (thymus, bursa of Fabricius, and spleen) were all weighed individually in grams. The weights of the total giblets (liver, heart, and gizzard) and of the dressing (carcass + total giblets) were calculated.
Non-enzymatic colorimetric methods were used to determine total protein, albumin, aspartate aminotransferase (AST), alanine aminotransferase (ALT), and creatinine. Enzymatic colorimetric methods were used to assess glucose, triglycerides, total cholesterol, high density lipoprotein (HDL) cholesterol, urea, and uric acid. Serum globulin was measured by subtracting serum albumin from serum total protein. The albumin/globulin ratio (A/G ratio) was determined by dividing serum albumin by serum globulin. Serum very low-density lipoprotein (VLDL) cholesterol was calculated by dividing triglycerides by 5. Low density lipoprotein (LDL) cholesterol was calculated by subtracting serum HDL plus VLDL cholesterol from total cholesterol (Friedewald et al., 1972).
Immunoglobulin IgG, IgM, and IgA were measured in plasma using sandwich ELISA kits based on modifications of Bianchi et al. (1995). The colorimetric method was used to assess plasma levels of superoxide dismutase (SOD), reduced glutathione (GSH), total antioxidant potential (TAC), and malonaldehyde (MDA) according to Nishikimi et al. (1972), Beutler (1963), Koracevic et al. (2001), and Ohkawa et al. (1979), respectively. The activity of glutathione peroxidase (GPx) was measured with an ultraviolet system (Paglia & Valentine, 1967).
Cecum contents (10 g) were obtained from five birds per treatment and transferred separately to 250 mL Erlenmeyer flasks containing 90 mL sterile peptone (0.1% peptone) and saline solution (0.85% NaCl) and blended thoroughly. The total bacterial count (TBC), total yeasts and moulds count (TYMC), E. coli, Salmonella and lactic acid bacteria count were recorded according to Xia et al. (2004) and Reda et al. (2020a).
Prior to the analysis, data were checked for normality, and all percentages and data on the cecum microbiota count were transformed using logarithmic and arcsine transformations. Data were analysed using one-way analysis of variance according to this statistical model:
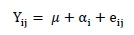
where: Yij = is the observation,
μ = is the overall mean,
αI = is the fixed effect of the ith treatment (I = 1,2,3,4, and 5), and
eij = random error.
Duncan's multiple range test (Duncan, 1955) checked the significance (P <0.05) of the differences between treatment means. Statistical analyses were performed with SAS software (SAS, version 9.4., SAS Institute Inc., Cary, North Carolina, USA).
Results and Discussion
Chicks fed a diet supplemented with 1500 ppm NSO had higher LBW and BWG at 14 days old (P< 0.01) than the control and the other groups (Table 2). Live bodyweight at 28 and 42 days old and BWG through 28 - 42 and 1 - 42 days old were (P <0.01) higher in chicks given 50 ppm OTC, and 1000 and 1500 ppm NSO compared with the control and 500 ppm NSO. Bodyweight gain was significantly higher in the groups treated with 1000 and 1500 NSO between 14 and 28 days old. Thus, broiler chicks fed a diet supplemented with 1500 ppm NSO outperformed all other treatments possibly because of the presence of unsaturated fatty acids and especially primary essential fatty acids, which are thought to be necessary for growth (Üstun et al., 1990). The present findings agreed with those of Halle et al. (1999), who assessed the effects of diets supplemented with various levels of black cumin essential oil (0.1 or 1 g/kg) or oilseed (10 or 50 g/kg) and found that growth was increased in one experiment, but no positive effects of these levels were found in a second experiment. According to Hermes et al. (2009), the inclusion of NSO at 0.5% and 1% improved broiler growth and reduced mortality rates in hot climates. Denli et al. (2004) found that feeding quails on diets supplemented with 60 mg/kg of Nigella essential oil improved BWG and feed efficiency. In addition, Saleh (2014) reported that feeding NSO at 1 ml/kg increased BWG compared with the control and antibiotic (avilamycin) groups. According to Attia and Al-Harthi (2015), adding NSO at 0.5 or 1.5 g/kg broiler diet may be used as an alternative growth promoter that is as efficient as or is better than zinc bacitracin.
Feed intake was significantly lower in NSO groups across 28 - 42 and 1 - 42 days old compared with the control. However, it had no impact between the ages of 1 and 14 days and 14 and 18 days. The addition of OTC or NSO at various levels improved FCR substantially over all experimental periods (1 - 14, 14 - 28, 28 - 42, and 1 - 42 days old). In general, chicks fed 1000 ppm NSO had a higher FCR and consumed less feed than the other groups and the control. Similarly, Saleh (2014) found that Nigella seed oil at 1 ml/kg diet had no significant effect on feed consumption in broiler chicks at 1- 21 days, but improved FI and FCR at 1-42 days. Osman (2002) stated that broiler chicks fed a diet containing 1 g NSO had the lowest FI (P <0.01), followed by those fed 0.5 g/kg feed. The levels of Nigella seed oil, except for 0.5 g/kg feed, improved FCR significantly compared with the control through all periods. Conversely, Attia and Al-Harthi (2015) found that FI and FCR were not affected substantially by supplementation of antibiotic and NSO levels in broiler chick diets.
The positive impact of NSO on feed utilization efficiency because of its antioxidant properties and phenolic compounds could account for the improvements in FCR. In addition, the active ingredients (nigellone and melatonin) had a nutrient-diversifying effect and worked together to increase nutrient utilization (El-Dakhakhny et al., 2002; Abu-Al-Basal 2011; Ali et al., 2012). The antibacterial and antifungal properties of NSO also could protect against hepatotoxicity and improve nutrient utilisation (Rathee, 1982). The NS could also induce thyroid hormone secretion indirectly and directly via the pituitary gland (Mohammed & Al-Suwaiegh, 2016) and thus boost the metabolic rate, resulting in better amino acid use (More et al., 1980).
The additions of 50 ppm OTC and 1500 ppm NSO resulted in substantially higher relative weights of liver, giblets, and dressing percentage compared with the control (Table 3). Supplementation of 50 ppm OTC and 1000 and 1500 ppm NSO resulted in significantly higher gizzard relative weight (Table 3). However, carcass and heart percentages were not altered significantly by the treatments. Changes in carcass composition could be attributed to improvements in BWG and FCR. Other studies revealed that essential oils inhibit pathogens in the digestive system and increase FCR and carcass yield (Alçiçek et al., 2003; Giannenas et al., 2003). The current findings were consistent with those of Osman (2002), who found that when broiler chicks were fed a diet supplemented with 1 g NSO/kg, the dressing percentage increased from 68.92% to 72.78%. Hermes et al. (2009) also found that dietary inclusion of NSO at 0.5% and 1% increased the relative weight of caecum and liver of broiler chicks.
Table 4 summarises the effects of OTC and NSO levels on plasma glucose, total protein, and its fractions (albumin and globulin) in broiler chick diets at 42 days old. The findings showed that plasma glucose levels, total protein, and globulin concentrations differed significantly between groups. However, when the treatments were compared with the control, plasma albumin concentration and A/G ratio were not affected. Birds that received 1000 and 1500 ppm NSO had substantially reduced blood glucose compared with control. Plasma total protein and globulin levels were substantially higher in chicks given a 1000 ppm NSO diet supplement. El-Soud et al. (2000) also found that birds fed diets containing 1% or 2% NSO had higher total protein, albumin, and globulin levels at all ages compared with control chicks. In birds fed diets containing NSO, Osman (2002) and Hermes et al. (2009) recorded small increases in plasma total protein. Saleh (2014) found that when broilers were fed NSO instead of avilamycin or a control diet, plasma total protein, albumin, and the A/G ratio all increased, but plasma glucose decreased. Attia and Al-Harthi (2015) also reported that plasma total protein and albumin levels were significantly lower in chicks fed diets containing 0.5 and 1.5 g NSO/kg compared with those fed zinc bacitracin.
An enzyme induced by NSO to increase liver synthetic activity may be responsible for the increase in total protein levels (Al-Jishi & Hozaifa, 2003). Furthermore, the increase in total protein and globulin may be owing to the immunostimulating effect of NSO (Aqel, 1993), which contains macro and micro mineral elements that aid in the growth process, boost the immune system, and boost total protein and albumin levels.
The addition of OTC and NSO to the broiler chick diet decreased plasma total cholesterol, triglycerides, LDL, and VLDL concentrations significantly (Table 4). Chicks fed a diet with 1000 ppm NSO had the lowest values, whereas chicks fed the control diet had the highest values. The concentrations of HDL, which was significantly higher in the plasma of chicks fed the antibiotic diet and NSO, showed the opposite pattern. Hermes et al. (2009) stated that NSO at 0.5% and 1% reduced total cholesterol and triglyceride significantly compared with control. Saleh (2014) found that dietary supplementation with NSO decreased plasma triglyceride, total cholesterol, and LDL levels while increasing plasma HDL levels significantly. Moreover, Osman (2002) found that feeding an NSO diet to birds resulted in a substantial increase in plasma cholesterol concentration. The content of unsaturated fatty acids, which may stimulate cholesterol excretion into the intestine and oxidation of cholesterol to bile acids (Tollba & Hassan, 2003), may explain the substantial reduction in cholesterol and triglyceride concentrations by feeding diets supplemented with NSO (El-Dakhakhny et al., 2000).
Plasma AST concentration was reduced significantly with OTC and NSO compared with the control (Table 5). Chicks fed 1500 ppm NSO had the lowest AST value compared with the control and other groups,. However, the addition of OTC and NSO to broiler clicks diets had no effect on plasma ALT, creatinine, urea, and uric acid concentrations. Hermes et al. (2009) found that feeding birds diets containing 0.5% and 1% NSO decreased the amount of AST and ALT significantly compared with the control. Saleh (2014) found that feeding broilers NSO reduced AST and ALT activities, which are indices of liver function. Furthermore, Attia and Al-Harthi (2015) found that plasma levels of ALT were higher in the 1.5 g NSO/kg group than the control and the 0.5 g and 1.0 g NSO/kg groups. Additionally, AST was higher in chicks fed 1 g NSO/kg diet compared with the other groups. The urea levels were significantly higher in chicks fed a 1.5 g/kg diet than in the other groups, but lower in chicks fed zinc bacitracin or 1 g NSO/kg diets than in the control (Attia & Al-Harthi, 2015).
Table 6 shows the effect of OTC and NSO levels on the relative weight of lymphoid organs. The relative weight of the spleen was increased significantly by NSO supplementation at various amounts. At 42 days old, there were no significant differences in the relative weight of the thymus and bursa of broiler chicks because of OTC and NSO supplementation. However, there was a numerical increase in the relative weights of the thymus and bursa in the OTC and NSO groups compared with the control. The increase of relative weights of lymphoid organs in broiler chicks fed diets containing NSO may be attributed to its role in activating the functions of these organs under normal temperature conditions (Radwan, 2003). The current findings are consistent with those of Osman (2002), who found that 1.0 g NSO/kg feed had a major effect on spleen weight compared with the control. Moreover, Hermes et al. (2009) found that birds fed NSO diets had significantly heavier spleens than control birds, and birds fed 1% NSO/kg feed had the highest relative spleen weight at 42 days old.
Table 6 shows the effect of dietary OTC and NSO supplementation on immune indices and antioxidant status in broiler chicks. There were no significant differences in average IgM values between trial groups. Groups treated with OTC and NSO had significantly higher IgA and IgG levels compared with the control and chicks fed a diet supplemented with 1000 ppm NSO had the highest IgA levels. This effect of NSO may be because NSO contains 67 constituents capable of inducing pharmacological and beneficial effects against bacteria, including staphylococcus and E. coli (Hanafy & Hatem, 1991). Antioxidant, antibacterial, and anti-inflammatory properties of black cumin seed active components have been shown to have beneficial effects on immunity and organs (Al Jabre et al., 2003; Al-Saleh et al., 2006; Arslan et al., 2005). Soliman et al. (2017) found a substantial increase in IgG and IgA in birds fed Nigella sativa Linn., but no significant changes in IgM in the treated groups or the control. Similarly, Al-Mufarrej (2014) found that supplementing broiler chicks with Nigella sativa Linn. seeds at 1% and 1.4% improved immune responsiveness. Table 6 shows that SOD, TAC, and GSH levels were significantly higher in chicks fed OTC and NSO diets than in control chicks. Plasma MAD levels were significantly lower in groups fed OTC and NSO diets relative to the control group, except for the 500 ppm NSO diet. Various supplements, on the other hand, had no major effect on GPx. The antioxidant activity of NSO is well known (M El-Dakhakhny et al., 2002; Mahmoud et al., 2002). Attia and Al-Harthi (2015) found that broiler chicks fed NSO diets had substantially higher SOD levels than the control group. In addition, the 1.5% NSO group had slightly higher GSH levels than the other groups.
Table 7 shows the treatment effects on the intestinal microflora of broilers at the end of the experiment (42 days old). Total bacterial count, TYMC, E. coli, and salmonella spp. counts were significantly lower in all groups relative to control. Supplementation with OTC and NSO improved total lactic acid bacteria count in all groups in contrast to the control. There is scientific evidence that herb extracts can reduce the number of pathogenic bacteria in poultry guts (Reda et al., 2020b). The seeds of Nigella sativa have antibacterial properties against a wide range of bacteria, including gram-positive and gram-negative bacteria (Kumar & Berwal, 1998). In terms of its bioactive components, thymoquinone has antimicrobial activity against Pseudomonas aeruginosa, Salmonella typhi and other bacteria, and antifungal, antiviral, and antiparasitic activity against a wide range of gram-positive and gram-negative bacteria, viruses, fungi, Candida spp. and parasites (Forouzanfar et al., 2014; Ratz-Lyko et al., 2014).
Alam et al. (2010) found that an ethanol extract of Nigeíía sativa was highly effective against B. subtiíis and S. aureus. The total inhibition of E. coli growth rate necessitated a high concentration of Nigeíía sativa extract. These findings suggest that black cumin seeds may have antibacterial activity against bacteria that are antibiotic resistant. Nigeíía sativa oil has also been shown to have antifungal activity against Aspergiííus species, curvula lunata, pathogenic yeast, and Candida albicans (Agarwal et al., 1979; Hanafy & Hatem, 1991). Controlling the gut microflora could affect broiler growth performance positively (Gunal et al., 2006). The added calories from NSO might increase bile flow rate, resulting in enhanced emulsification and stimulating pancreatic lipases which promotes fat digestion and fat-soluble vitamin absorption (Mahmoud et al., 2002). In addition, bioactive compounds in NSO, such as P. cymene, thymol, dithymoquinone, thymoquinone, and carvacrol, may thus improve feed digestibility and nutrient absorption by directly stimulating digestive enzymes (Nasir, 2009; Salam et al., 2013). The antioxidant properties and phenolic compounds in NSO have also been shown to have positive effect on feed utilization, which could explain the improvement in growth performance (Attia & Al-Harthi, 2015).
Conclusions
Dietary supplementation of NSO up to 1500 ppm in broiler diets had positive effects on growth and carcass characteristics, immunological parameters, antioxidant status and cecum microbiota. Thus, itmay be used as a natural alternative to antibiotic growth promoters in broiler chicken diets to encourage gut health and thus enhance growth performance.
Acknowledgements
This work was supported by the Poultry Department, Faculty of Agriculture, Zagazig University, Egypt. All the authors of the manuscript thank and acknowledge their universities and institutes. The English revision of the manuscript by Mr. Roderick Cantlay-Hollis is also acknowledged.
Authors' Contributions
AMS, MSE, AGE and FMR collected the data. AGE and FMR conducted the statistical analyses. AMS, AIA, MSE, AGE and FMR collaborated in interpreting the results, wrote the initial draft of this manuscript, and finalized the manuscript. AIA, and FMR, developed the original hypothesis and designed the experiments. The authors have read and approved the manuscript.
Conflict of Interest Declaration
The authors declare that they have no competing interests.
References
Abu-Al-Basal, M.A., 2011. Influence of Nigeíía sativa fixed oil on some blood parameters and histopathology of skin in staphylococcal-infected BALB/c mice. Pak. J. Biol. Sci. 14, 1038-1046. DOI: 10.3923/pjbs.2011.1038.1046 [ Links ]
Agarwal, R., Kharya, M.D. & Shrivastava, R., 1979. Pharmacological studies of essential oil and unsaponifiable matter of seeds of Nigella sativa. Indian J. Pharm. Sci. 41, 248. [ Links ]
AL-Douri, A. & Al-kazaz, S., 2010. The effect of Nigella sativa oil (black seed) on the healing of chemically induced oral ulcer in rabbit (experimental study). Al-Rafidain Dent. J. 10, 151-157. https://doi.org/10.33899/rden.2010.45402 [ Links ]
Al-Jishi, S.A. & Hozaifa, B.A., 2003. Effect of Nigella sativa on blood hemostatic function in rats. J. Ethnopharmacol. 85, 7-14. DOI: 10.1016/s0378-8741(02)00356-2 [ Links ]
Al-Mufarrej, S.I., 2014. Immune-responsiveness and performance of broiler chickens fed black cumin (Nigella sativa L.) powder. J. Saudi Soc. Agric. Sci. 13, 75-80. https://doi.org/10.1016/jjssas.2013.01.006 [ Links ]
Al-Saleh, I.A., Billedo, G. & El-Doush, I.I., 2006. Levels of selenium, d dl-a-tocopherol, dl-y-tocopherol, all-trans-retinol, thymoquinone and thymol in different brands of Nigella sativa seeds. J. Food Compos. Anal. 19, 167-175. https://doi.org/10.1016/jjfca.2005.04.011 [ Links ]
Alam, M.M., Yasmin, M., Nessa, J. & Ahsan, C.R., 2010. Antibacterial activity of chloroform and ethanol extracts of black cumin seeds (Nigella sativa) against multi-drug resistant human pathogens under laboratory conditions. J. Med. Plants Res. 4, 1901-1905. DOI: 10.5897/JMPR10.324 [ Links ]
Alcicek, A., Bozkurt, M. & Cabuk, M., 2003. The effect of an essential oil combination derived from selected herbs growing wild in Turkey on broiler performance. South African J. Anim. Sci. 33, 89-94. https://doi.org/10.4314/sajas.v33i2.3761 [ Links ]
Ali, M.A., Sayeed, M.A., Alam, M.S., Yeasmin, M.S., Khan, A.M. & Muhamad, 2012. Characteristics of oils and nutrient contents of Nigella sativa Linn. and Trigonella foenum-graecum seeds. Bull. Chem. Soc. Ethiop. 26, 55-64. DOI: 10.4314/bcse.v26i1.6 [ Links ]
Al Jabre, S., Al Akloby, O.M. & Al Qurashi, A.R., 2003. Thymoquinone, an active principle of Nigella sativa, inhibited Aspergillus niger. Pakistan J. Med. Res. 42, 185-191. [ Links ]
Aqel, M.B., 1993. Effects of Nigella sativa seeds on intestinal smooth muscle. Int. J. Pharmacogn. 31, 55-60. https://doi.org/10.3109/13880209309082918 [ Links ]
Arslan, S.O., Gelir, E., Armutcu, F., Coskun, O., Gurel, A., Sayan, H. & Celik, I.L., 2005. The protective effect of thymoquinone on ethanol-induced acute gastric damage in the rat. Nutr. Res. 25, 673-680. https://doi.org/10.1016/j.nutres.2005.06.004 [ Links ]
Attia, Y.A. & Al-Harthi, M.A., 2015. Schwarzkümmelsamenöl als Alternative zu antibiotischen Leistungsförderern bei Masthühnern. Eur. Poult. Sci. 79, 1-12. DOI: 10.1399/eps.2015.84 [ Links ]
Beutler, E. 1963. Improved method for the determination of blood glutathione. J. Lab. Clin. Med. 61, 882-888. [ Links ]
Bianchi, A.T.J., Moonen-Leusen, H.W.M., Van der Heijden, P.J. & Bokhout, B.A., 1995. The use of a double antibody sandwich ELISA and monoclonal antibodies for the assessment of porcine IgM, IgG and IgA concentrations. Vet. Immunol. Immunopathol. 44, 309-317. https://doi.org/10.1016/0165-2427(94)05307-E [ Links ]
Denli, M., Okan, F. & Uluocak, A.N., 2004. Effect of dietary black seed (Nigella sativa L.) extract supplementation on laying performance and egg quality of quail (Coturnix coturnix japonica). J. Appl. Anim. Res. 26, 73-76. https://doi.org/10.1080/09712119.2004.9706511 [ Links ]
Doyle, M.E., 2001. Alternatives to antibiotic use for growth promotion in animal husbandry. Food Res. Inst. University of Wisconsin-Madison. Pp.1-17. [ Links ]
Duncan, D.B., 1955. Multiple range and multiple F tests. Biometrics 11, 1-42. [ Links ]
Durrani, F.R., Chand, N., Zaka, K., Sultan, A., Khattak, F. M. & Durrani, Z., 2007. Effect of different levels of feed added black seed (Nigella sativa L.) on the performance of broiler chicks. Pakistan J. Biol. Sci. 10, 4164-4167. https://doi.org/10.3923/pjbs.2007.4164.4167 [ Links ]
El-Dakhakhny, M., Madi, N.J., Lembert, N. & Ammon, H.P.T., 2002. Nigella sativa oil, nigellone and derived thymoquinone inhibit synthesis of 5-lipoxygenase products in polymorphonuclear leukocytes from rats. J. Ethnopharmacol. 81, 161-164. https://doi.org/10.1016/S0378-8741(02)00051-X [ Links ]
El-Dakhakhny, M., Mady, N.I. & Halim, M.A. 2000. Nigella sativa L. oil protects against induced hepatotoxicity and improves serum lipid profile in rats. Arzneimittelforschung, 50, 832-836. DOI: 10.1055/s-0031-1300297 [ Links ]
El-Soud, S.B.A., 2000. Studies on some biological and immunological aspects in Japanese quail fed diets containing some Nigella sativa seed preperations. Egypt. Poult. Sci. J. 20, 757-776. [ Links ]
Forouzanfar, F., Bazzaz, B.S.F. & Hosseinzadeh, H., 2014. Black cumin (Nigella sativa) and its constituent (thymoquinone): a review on antimicrobial effects. Iran. J. Basic Med. Sci. 17, 929. [ Links ]
Friedewald, W.T., Levy, R.I. & Fredrickson, D.S., 1972. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin. Chem. 18, 499-502. https://doi.org/10.1093/clinchem/18.6.499 [ Links ]
Giannenas, I., Florou-Paneri, P., Papazahariadou, M., Christaki, E., Botsoglou, N.A. & Spais, A.B., 2003. Effect of dietary supplementation with oregano essential oil on performance of broilers after experimental infection with Eimeria tenella. Arch. Anim. Nutr. 57, 99-106. https://doi.org/10.1080/0003942031000107299 [ Links ]
Gunal, M., Yayli, G., Kaya, O., Karahan, N. & Sulak, O., 2006. The effects of antibiotic growth promoter, probiotic or organic acid supplementation on performance, intestinal microflora and tissue of broilers. Int. J. Poult. Sci. 5, 149-155. https://doi.org/10.3923/ijps.2006.149.155 [ Links ]
Halle, I., Thomann, R. & Flachowsky, G., 1999. Vitamine und Zusatzstoffe in der ernahrung von Mensch und Tier; 7.Pages 469-472 in Symposium Jena/Thuringen, Germany. [ Links ]
Hanafy, M.S.M. & Hatem, M. E., 1991. Studies on the antimicrobial activity of Nigella sativa seed (black cumin). J. Ethnopharmacol. 34, 275-278. https://doi.org/10.1016/0378-8741(91)90047-H [ Links ]
Hassanpour, H., Moghaddam, A.K.Z., Khosravi, M. & Mayahi, M., 2013. Effects of synbiotic on the intestinal morphology and humoral immune response in broiler chickens. Livest. Sci. 153, 116-122. https://doi.org/10.1016/j.livsci.2013.02.004 [ Links ]
Hermes, I.H., Attia, F.A.M., Ibrahim, K.A., & El-Nesr, S.S., 2009. Effect of dietary Nigella sativa L. on productive performance and nutrients utilization of broiler chicks raised under summer conditions of Egypt. Egypt. Poult. Sci. J. 29, 145-172. [ Links ]
Kim, J.D., Nhut, T.M., Hai, T.N. & Ra, C.S., 2011. Effect of dietary essential oils on growth, feed utilization and meat yields of white leg shrimp L. vannamei. Asian-Australasian J. Anim. Sci. 24, 1136-1141. https://doi.org/10.5713/ajas.2011.11006 [ Links ]
Koracevic, D., Koracevic, G., Djordjevic, V., Andrejevic, S. & Cosic, V., 2001. Method for the measurement of antioxidant activity in human fluids. J. Clin. Pathol. 54, 356-361. https://doi.org/10.1136/jcp.54.5.356 [ Links ]
Kumar, M. & Berwal, J.S., 1998. Sensitivity of food pathogens to garlic (Allium sativum). J. Appl. Microbiol. 84, 213-215. https://doi.org/10.1046/j.1365-2672.1998.00327.x [ Links ]
Mahmoud, M.R., El-Abhar, H.S. & Saleh, S., 2002. The effect of Nigella sativa oil against the liver damage induced by Schistosoma mansoni infection in mice. J. Ethnopharmacol. 79, 1-11. https://doi.org/10.1016/S0378-8741(01)00310-5 [ Links ]
Mohammed, A.A. & Al-Suwaiegh, S.B., 2016. Effects of Nigella sativa on mammals' health and production. Adv. Anim. Vet. Sci 4, 630-636. DOI: 10.14737/journal.aavs/2016/4.12.630.636 [ Links ]
More, T., Rai, A. K., & Manohar, S., 1980. Note on the effect of thermal exposure on body fluid composition of different breeds and breed crosses of sheep. Indian J. Anim. Sci. 50, 207-209. [ Links ]
Nasir, Z., 2009. Comparison of effects of Echinacea purpurea juices and Nigella sativa seeds on performance, some blood parameters, carcass and meat quality of broilers. Ph.D. thesis, University of Hohenheim, Stuttgart-Hohenheim, Germany. [ Links ]
NRC, 1994. Nutrient requirements of poultry: Ninth revised edition. The National Academies Press. Washington DC. https://doi.org/10.17226/2114 [ Links ]
Nishikimi, M., Rao, N.A. & Yagi, K., 1972. The occurrence of superoxide anion in the reaction of reduced phenazine methosulfate and molecular oxygen. Biochem. Biophys. Res. Commun. 46, 849-854. https://doi.org/10.1016/S0006-291X(72)80218-3 [ Links ]
Ohkawa, H., Ohishi, N. & Yagi, K., 1979. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 95, 351-358. https://doi.org/10.1016/0003-2697(79)90738-3 [ Links ]
Osman, M., 2002. Beneficial effects of black seed oil inclusion in broiler diet on performance and carcass characteristics. Egypt. J. Poult. Sci. 22, 839-853. [ Links ]
Paglia, D.E. & Valentine, W.N., 1967. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J. Lab. Clin. Med. 70, 158-169. DOI: 10.5555/uri:pii:0022214367900765 [ Links ]
Radwan, N.L., 2003. Effect of using some medicinal plants on performance and immunity of broiler chicks. M.Sc. thesis, Cairo University, Egypt. [ Links ]
Rathee, P.S., 1982. Antimicrobial activity of essential oil, fixed oil and unsaponifiable matter of (Nigella sativa L.). Indian J. Pharmacol. Sci. 44, 8-10. [ Links ]
Ratz-tyko, A., Herman, A., Arct, J. & Pytkowska, K., 2014. Evaluation of antioxidant and antimicrobial activities of Oenothera biennis, Borago officinalis, and Nigella sativa seedcake extracts. Food Sci. Biotechnol. 23, 1029-1036. https://doi.org/10.1007/s10068-014-0140-2 [ Links ]
Reda, F.M., Alagawany, M., Mahmoud, H.K., Mahgoub, S.A. & Elnesr, S.S., 2020a. Use of red pepper oil in quail diets and its effect on performance, carcass measurements, intestinal microbiota, antioxidant indices, immunity and blood constituents. Animal 14, 1025-1033. https://doi.org/10.1017/S1751731119002891 [ Links ]
Reda, F.M., El-Saadony, M.T., Elnesr, S.S., Alagawany, M. & Tufarelli, V., 2020b. Effect of dietary supplementation of biological curcumin nanoparticles on growth and carcass traits, antioxidant status, immunity and caecal microbiota of Japanese quails. Animals 10, 5, 754. https://doi.org/10.3390/ani10050754 [ Links ]
Salam, S., Sunarti, D. & Isroli, I., 2013. Physiological responses of blood and immune organs of broiler chicken fed dietary black cumin powder (Nigella sativa) during dry seasons. J. Indones. Trop. Anim. Agric. 38, 185-191. https://doi.org/10.14710/jitaa.38.3.185-191 [ Links ]
Saleh, A., 2014. Nigella seed oil as alternative to avilamycin antibiotic in broiler chicken diets. S. Afr. J. Anim. Sci. 44, 254. https://doi.org/10.4314/sajas.v44i37 [ Links ]
Soliman, E.S., Hamad, R.T. & Ahmed, A., 2017. Prophylactic and immune modulatory influences of Nigella sativa Linn. in broilers exposed to biological challenge. Vet. world 10, 1447. DOI: 10.14202/vetworld.2017.1447-1455 [ Links ]
Toghyani, M., Toghyani, M., Gheisari, A., Ghalamkari, G. & Mohammadrezaei, M., 2010. Growth performance, serum biochemistry and blood hematology of broiler chicks fed different levels of black seed (Nigella sativa) and peppermint (Mentha piperita). Livest. Sci. 129, 173-178. https://doi.org/10.1016/j.livsci.2010.01.021 [ Links ]
Tollba, A.A.H. & Hassan, M.S.H., 2003. Using some natural additives to improve physiological and productive performance of broiler chicks under high temperature conditions. 2-Black cumin (Nigella sativa) or garlic (Allium sativum). Egypt. Poult. Sci. 23, 327-340. [ Links ]
Üstun, G., Kent, L., Cekin, N. & Civelekoglu, H., 1990. Investigation of the technological properties of Nigella sativa (black cumin) seed oil. J. Am. Oil Chem. Soc. 67, 958-960. [ Links ]
Xia, M.S., Hu, C.H. & Xu, Z.R., 2004. Effects of copper-bearing montmorillonite on growth performance, digestive enzyme activities, and intestinal microflora and morphology of male broilers. Poult. Sci. 83, 1868-1875https://doi.org/10.1093/ps/83.11.1868 [ Links ]
Submitted 15 November 2021
Accepted 5 December 2021
Published 30 January 2022
# Corresponding author: ayman.elnagar@fagr.bu.edu.eg














