Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Animal Science
On-line version ISSN 2221-4062
Print version ISSN 0375-1589
S. Afr. j. anim. sci. vol.50 n.5 Pretoria 2020
http://dx.doi.org/10.4314/sajas.v50i5.13
ARTICLE
Effects of vitamin E, an oil blend and L-Arginine on breast meat from broiler chickens
J. KhatunI, V; T. C. LohI, II, #; H. L. FooIII, IV; H. AkitI; R. MohamadIII; N. ShazaliI
IDepartment of Animal Science, Faculty of Agriculture, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
IIInstitute of Tropical Agriculture and Food Security, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
IIIDepartment of Bioprocess Technology, Faculty of Biotechnology and Biomolecular Sciences, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
IVInstitute of Bioscience, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
VDepartment of Animal Science and Nutrition, Chittagong Veterinary and Animal Sciences University, Chittagong, Bangladesh
ABSTRACT
The study examined the effects of supplementing vitamin E on the fatty acid profile and breast meat quality of broilers fed diets containing an oil blend and L-Arginine. Two hundred sixteen Cobb 500 one-day-old broilers were randomly allocated to six treatments, namely T1: 6% palm oil (control); T2: blend of 4% palm oil and 2% sunflower oil + 0.25% L-Arginine (positive control); T3: T2 with 20 mg/kg vitamin E added; T4: T2 with 50 mg/kg vitamin E added; T5: T2 with 100 mg/kg vitamin E added; and T6: T2 supplemented with 150 mg/kg vitamin E. Relative to T1, the other diets increased growth rate and improved feed conversion ratio (FCR) similarly. Linoleic, arachidonic, and polyunsaturated fatty acids were greater and palmitic and saturated fatty acids were lower in the meat of birds fed T2-T6 relative to T1. Supplementation of vitamin E reduced drip loss (DL) and increased redness, tenderness, free thiol content, and the oxidative stability of meat during storage at 4 °C. Regardless of diet, free thiol, redness and Warner-Bratzler shear force (WBSF) value decreased significantly, whereas the carbonyl content, thiobarbituric acid-reactive substances (TBARS), and DL of breast meat increased over the ageing period. In conclusion, relative to T1, the other diets were effective in improving growth performance and meat quality during post-mortem storage.
Key words: fatty acid profile, free thiol, lipid oxidation, palm oil, protein oxidation, sunflower oil
Introduction
Dietary fat and oils are often added to the diet of rapidly growing birds to fulfil their energy requirements and thus improve their productivity at low cost (Abdulla et al., 2015). Diets that contain unsaturated fatty acids (UFAs) have greater metabolizable energy values owing to better intestinal absorption compared with those that contain saturated fats (Tancharoenrat et al., 2012). Further, a high intake of saturated fatty acids (SFAs) and cholesterol is associated with increased occurrence of human diseases, especially cardiovascular disease (Houston et al., 2011). Therefore, supplementation of unsaturated fats could be used to alter the fatty acid profile of chicken meat to make it a functional food. Health benefits would be anticipated from feeding diets that contained a combination of palm oil (PO) and sunflower oil (SO), which would lead to increased polyunsaturated fatty acids (PUFAs) and decreased fat and cholesterol contents in the meat (Khatun et al., 2017).
Feeding unsaturated fats to poultry, particularly PUFAs, can enhance lipid oxidation and affect meat flavour and colour adversely (Abdulla et al., 2015), reduce performance (Zhong et al., 2014), and induce oxidative challenges that affect their health. These harmful effects can be overcome with supplemental antioxidant such as vitamin E, which may also increase the shelf life of the meat (Li et al., 2009).
L-Arginine is an essential amino acid for chickens and has an active role in protein synthesis (Tan et al. 2009), tissue growth, and enhancement of immune response (Emadi et al., 2010). It has also been found to be effective in reducing body fat in chickens (Fouad et al., 2013; Khatun et al., 2019). Thus, it was hypothesized that supplementation of vitamin E to diets that contained a blend of PO and SO and L-Arginine would increase growth performance, have favourable effects on the fatty acid profile of breast muscle, improve meat quality, and increase shelf life. The objective of this study was to evaluate the effects of feeding an oil blend, L-Arginine, and vitamin E on the fatty acid profile, meat quality, lipid and protein oxidation of breast muscle of broiler chickens.
Materials and Methods
The experiment followed the guidelines sanctioned by the Institutional Animal Care and Use Committee of Research Policy at Universiti Putra Malaysia (UPM/IACUC/AUP-R081/2016). Two hundred and sixteen Cobb 500 one-day-old broiler chicks were randomly assigned to one of six treatments in a completely randomized design. Each chick was weighed and wing-banded. There were six replicates of each treatment with six chicks per replication. The experimental diets consisted of T1: 6% palm oil (control); T2: a blend of 4% palm oil and 2% sunflower oil + 0.25% L-Arginine (positive control); T3: T2+ 20 mg/kg vitamin E; T4: T2 + 50 mg/kg vitamin E: T5: T2 + 100 mg/kg vitamin E; and T6: T2 + 150 mg/kg vitamin E. Arginine was added in the form of L-Arginine monohydrochloride (99.6% L-Arg) (HerbstoreUSA,Walnut, California, USA) and vitamin E as a-tocopherol acetate (Adisseo Asia Pacific Pte., Ltd., Singapore). All diets were iso-caloric and iso-nitrogenous and wereformulated according to the nutrient requirements for broilers using FeedLIVE software (FeedLIVE 1.52, Bangkok, Thailand). The feeding trial lasted six weeks. The starter diets (Table 1) were offered on days 1 to 21. Their nutritional composition is presented in Table 2. The finisher diets (Table 3) were offered on days 22 to 42. Their nutritional composition is presented in Table 4.
At 42 days old, 12 birds per treatment (two replicates) were randomly selected and slaughtered to sample the breast muscle. These samples were cut into five portions (about 80 to 100 g) and each portion was allocated to one of five ageing periods, that is, 0, 1, 5, 7, and 10 days post-mortem. The day 0 samples were snap frozen in liquid nitrogen immediately after collection and stored at -80 °C. The samples for the subsequent ageing periods were vacuum-packed and refrigerated (4 °C). After completion of the ageing periods, the muscle samples were taken from the refrigerator and cut into sub samples. Then, the sub samples were kept at -20 °C to analyse meat quality or at -80 °C to determine their fatty acid profile, lipid oxidation, vitamin E, thiol and carbonyl contents.
The fatty acids were extracted from breast muscle samples (1g) using a mixture of chloroform and methanol at a ratio of 2: 1 (v/v) (Folch et al., 1957, in Khatun et al., 2017). Determination of a-tocopherol from the meat samples followed the method of Rutkowski and Grzegorczyk (2007) using an appropriate standard, a blank sample, and the meat samples. The absorbance values of the test samples and standard were measured against the blank at 539 nm wavelength using a spectrophotometer. The a-tocopherol content was calculated with this formula:

where: Cs = concentration of standard,
Ax = absorbance of test sample, and
As = absorbance of standard.
The pH of breast meat at the post-mortem ageing periods was determined with a portable pH meter (Mettler Toledo, AG 8603, Switzerland). The pH reading was measured in triplicate for each sample and an average of three readings was used to calculate each sample. Drip loss and cooking loss (CL) were measured by calculating the percentage of weight loss, relative to the initial weight (Honikel, 1998) (see also Khatun et al., 2018) using this equation:
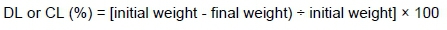
Meat tenderness was measured with the sample that had been used to determine CL. Each sample was kept vacuum-packed in a sealed bag to avoid evaporation and stored overnight at 4 °C. The following day, each sample was cut into at least three sub samples (cuboid) 1 cm * 1 cm * 2 cm with the long axis aligned with the muscle fibre direction. Each sub sample was sheared perpendicular to the muscle fibres by the TA.HDplus® texture analyser with a Volodkevitch blade set (Texture Technologies Inc., Hamilton, Massachusetts, USA) (Cavitt et al. 2004). Warner-Bratzler shear force (WBSF) values were reported as the average of all sub sample values, as kg unit. Meat samples were taken from the -80 °C freezer and thawed overnight at 4 °C. The colour measurements were conducted using a colour flex spectrophotometer (Hunter Lab Reston, VA, USA) according to AMSA (2012).
Lipid oxidation in muscle samples was measured with the TBARS method (Lynch & Frei, 1993). The malondialdehyde (MDA) value was calculated from a standard curve of 1, 1, 3, 3-tetraethoxypropane, and expressed as mg MDA/kg meat.
Myofibril proteins were extracted according to Morzel et al. (2006) with minor modifications (Sabow et al., 2016). Protein concentration was assessed according to Bradford (1976). The standard solutions for protein quantification were prepared with bovine serum albumin. The protein concentration of each sample was determined according to a standard curve of protein concentration against absorbance at 595 nm wavelength. The free thiol concentration was estimated based on Ellman's method using 2, 2-dithiobis (5-nitropyridine) (DTNP) (Winterbourn, 1990) with slight modifications (Morzel et al., 2006). The stock solution containing 4 mg of myofibrillar proteins was dissolved mixed with 3 ml of 100 mM phosphate buffer at pH 8 containing 8 M urea. About 30 μl of 10 mM DTNP was added, followed by incubation for one hour at room temperature. Absorbance at 386 nm was measured with a spectrophotometer (Spectronic Instruments, ThermoFisher Scientific Inc., Waltham, Massachusetts, USA) against a blank buffer without protein. The absorbance value of the blank was subtracted, and thiol concentration was estimated using an absorption coefficient of 14,000 M-1 cm-1. The results were expressed as nmol of free thiol per mg of protein.
Carbonyl content was determined according to Oliver et al (1987) with slight modification (Botsoglou et al., 2012). Carbonyl concentration was calculated from the absorption of supernatant from the sample and the control tubes at 370 nm. The results were expressed as nmol of carbonyl per mg of protein based on an absorption coefficient of 21.0 mM-1 cm-1.
The data for fatty acid profile were analysed statistically using PROC GLM of SAS (SAS Institute Inc., Cary North Carolina, USA). The differences in the meat quality parameters because of the post-mortem ageing period were determined through repeated measure analysis again using the GLM procedure. The means were separated using the Duncan's multiple range test. A P-value of less than 0.05 was considered significant.
Results and Discussion
Bodyweight and bodyweight gain were greater in T2-T6 than in T1. Moreover, FCR was greater (P <0.05) in chickens fed T1 than those fed the other treatments. However, there were no significant differences (P >0.05) in bodyweight gain and FCR between the chickens fed the various levels of vitamin E and those fed the positive control diet. Feed intake was not affected by the treatments over the rearing periods. The performances of the broiler chickens fed the dietary treatments are shown in Table 5.
The breast meat of T2--T6 had a lower (P <0.05) percentage of palmitic and total SFA and a greater (P <0.05) percentage of linoleic acid, total UFA, total PUFA, UFA to SFA ratio, and PUFA to SFA ratio compared with T1. The addition of various levels of a-tocopherol had no effect on muscle fatty acid composition except for eicosapentaenoic acid. The effects of oil blend, L-Arginine, and a-tocopherol acetate supplementation on the fatty acid composition of the breast muscle in broiler chickens are presented in Table 6.
Vitamin E supplementation significantly affected the a-tocopherol content of the breast muscle at various periods of post-mortem ageing in broiler chickens fed diets differing in oil blend and L-Arginine The a-tocopherol concentration in the meat rose (P <0.05) with increasing levels of vitamin E in the diet, particularly with the inclusion of 50 mg/kg vitamin E or more in the diet (T4, T5 and T6). The a-tocopherol concentration decreased with the increased duration of post-mortem ageing.
On days 5 and 7 post mortem, the treatments influenced (P <0.05) the DL of breast muscle in broiler chickens. Drip loss in T4, T5 and T6 was lower (P <0.05) than in the other treatments. Irrespective of treatment, DL increased with post-mortem ageing, whereas no further changes of DL were observed from five to seven post-mortem storage days for T4 to T6. No significant differences (P >0.05) were observed in the CL of breast muscle among the treatments and post-mortem ageing periods. The DL, CL, pH, WBSF values, and colour coordinates of breast muscle of broiler chickens fed various treatments are presented in Table 7.
Supplementation of vitamin E in the broiler diet increased the tenderness of meat. On day 1 postmortem, the WBSF values in T4, T5 and T6 diets were lower (P <0.05) than T1 and T2 diets. The WBSF value in T3 birds was intermediate did not differ (P >0.05) from either T1 and T2 or T4, T5 and T6. On day 5 post-mortem, WBSF values in birds fed the T4, T5, and T6 diets were lower (P <0.05) than the other treatments. On day 7 post-mortem, treatments had no effect (P >0.05) on WBSF values. Regardless of treatment, WBSF decreased significantly with increased post-mortem ageing. The WBSF value of breast muscle on days 5 and 7 did not differ (P >0.05) in T4, T5 and T6.
Treatments had no effect (P >0.05) on the pH values of the breast muscle at any post-mortem ageing period. The meat pH values on day 0 post-mortem were higher than day 1 post mortem across treatments. No changes were observed in the meat pH values on days 1, 5, and 7 post-mortem in the birds fed the T4, T5 and T6 diets.
Dietary treatment had no effect (P >0.05) on the lightness (L*) and yellowness (b*) values of the breast muscle at any post-mortem ageing period. On days 1 and 5 post-mortem, the a* value rose (P <0.05) as the level of vitamin E increased in diet. The a* values decreased (P <0.05) as post-mortem ageing increased.
Thiobarbituric acid-reactive substance values rose (P <0.05) across all treatments as the post-mortem ageing period increased. Vitamin E (P <0.05) influenced lipid peroxidation (TBARS value) in all the postmortem ageing periods except on days 0 and 1 post-mortem. At 5, 7, and 10 days post-mortem, TBARS values were lower in the chicken fed the T4, T5, and T6 diets than the other treatments.
On days 0, 1 and 5 post-mortem, treatment had no effect on the meat free thiol content. On days 7 and 10 post-mortem, the free thiol content in T4, T5, and T6 was greater (P <0.05) compared with the other treatments. Irrespective of treatment, thiol content reduced (P <0.05) with post-mortem ageing. Dietary treatment had no effect on the meat carbonyl content at any post-mortem ageing period. Regardless of treatment, carbonyl content increased with post-mortem ageing. The effects of treatments and post-mortem ageing period on lipid oxidation, free thiol content and carbonyl content in breast muscle of broiler chickens are shown in Table 8.
The increased bodyweight and bodyweight gain in T2--T6 could be attributed to the greater degree of unsaturation of the oil blend compared with the single oil in the control diet. Moreover, L-Arginine is a substrate that is involved in biosynthesis of protein, polyamines, glutamine, proline, and ornithine which are crucial to animal growth (Khajali & Wideman, 2010). Chickens fed the oil blend and L-Arginine increased bodyweight significantly compared with the control diet. These results were consistent with those of Emadi et al. (2011) and Wu et al. (2011), who reported that L-Arginine increased the growth performance of chickens and ducks, respectively
The fatty acid composition of chicken muscle is attributed to the synthesis of de novo fatty acid from protein and carbohydrate precursors and fatty acids. In the current study, supplementation of the oil blend and L-Arginine reduced the concentrations of Palmitic acid and total SFA, and increased the concentrations of palmitoleic acid, linoleic acid, eicosapentaenoic acid, and total UFA in breast muscle. These results were consistent with those of Velasco et al. (2010) and Ma et al. (2015), who observed a similar pattern of fatty acid profile changes in the meat of chicken and pigs, respectively. However, vitamin E supplementation did not affect the meat fatty acid profile in the current study. This observation is consistent with that of Zduhczyk et al. (2011), who reported that meat fatty acid compositions were altered by fat type, but not by vitamin E, when diet was supplemented with vitamin E and various oil sources.
In the current study, meat a-tocopherol concentration increased with the rising level of supplementation in the broiler diet. Skrivan et al. (2010) also observed greater levels of a-tocopherol in the breast and thigh muscle with increased levels of a-tocopherol in a broiler diet. Giraudeau et al. (2013), suggested that the increased a-tocopherol concentration in meat could protect the cell membrane from oxidative damage caused by unsaturated phospholipids. However, as post-mortem ageing continued, the a-tocopherol concentration decreased, possibly because of the interruption of the antioxidant defence system and augmented lipid oxidation with increased post-mortem storage time. Adeyemi et al. (2015) similarly reported decreased vitamin E content with time in post-mortem-aged chevon. Water-holding capacity (WHC) of meat is a vital index for assessing meat quality. Vitamin E is soluble in fat and plays a vital role in upholding the integrity of the cell membrane (Khan et al., 2011), which can improve WHC and reduce fluid loss from the muscle. In the current study, vitamin E supplementation reduced DL in the breast muscle of broiler chickens. Li et al. (2009) also found that vitamin E supplementation (100 to 200 mg/kg) reduced DL in the thigh and breast muscles. As expected, DL increased over the post-mortem ageing period in the current study. This could be attributed to the disruption of collagen and myofibrillar proteins during ageing, which causes the myofibrillar proteins to lose their WHC (Abdulla et al., 2017). Karami et al. (2011) also observed increased DL over the post-mortem ageing period in chevon. The lack of significant trend over time in DL from the breast muscle of broiler chickens fed the diets supplemented with vitamin E might be the result of a protective effect of vitamin E on the cell membrane.
A combination of fluid and soluble substances is lost from the meat during cooking (Lawrie & Ledward, 2006). Like Zhang et al. (2011), vitamin E had no effect on CL in the current study. Irrespective of treatment, post-mortem aging time had no effect on CL. This could be because of the similar meat pH values that were observed over the post-mortem ageing period since pH has previously been shown to be associated with CL (Dalle Zotte et al., 1995). The current findings were consistent with those of Zhang et al. (2013b), who reported no significant differences in the ultimate pH value of broiler breast meat following supplementation with various levels of vitamin E.
Dietary supplementation of vitamin E influenced the WBSF values of broiler breast meat. Zhang et al. (2013b) also found that vitamin E decreased meat WBSF and increased WHC. The WBSF decreased with post-mortem ageing in the current study as expected, perhaps because of the weakening of myofibrillar structure by endogenous muscle proteinases during ageing. This finding was in tandem with those results reported by Abdulla et al. (2017), who found reduced WBSF values of broiler breast meat as the postmortem ageing period increased.
Breast meat pH was not affected by vitamin E supplementation at any post-mortem ageing period. This could be because of similar management practices and slaughter processes during the experiment. Irrespective of treatment, breast meat pH declined at day 1 post mortem compared with day 0 post mortem. This decrease might be because of anaerobic glycolysis in the post-mortem muscle, which is required to convert muscle glycogen to lactic acid. A pH value of chicken meat lower than 5.7 at day 1 post mortem is considered poor quality because of low WHC (Corzo et al., 2009). In the present study, the meat pH values on day 1 post-mortem for all treatments were within the normal range (Abdulla et al., 2015). Across all treatments the pH values of the meat differed according to then number of days post mortem. This suggests a stability of muscle oxidative status induced by vitamin E. However, the pH value of meat from chickens that were not supplemented with vitamin E (T1, T2) or had a low level of vitamin E (T3) increased at day 7 post mortem. This could be because of increased meat alkalization caused by spoilage organisms (Abdulla et al., 2015).
During slaughter, the antioxidant level of muscle plays an important part in determining colour stability (Ponnampalam et al., 2012). In the present study, increased vitamin E in the diets enhanced a* values and conserved the colour of meat by reducing lipid oxidation, which indirectly delayed myoglobin oxidation. Similar findings were reported by Zhang et al. (2013b), Li et al. (2003), and Santé-Lhoutellier et al. (2008).
Lipid oxidation affects the colour, flavour and nutritional value of meat. The high meat TBARS values observed for the birds fed T2 could be attributed to the lack of antioxidant and relatively higher amount of PUFA in meat from this group. However, significant reductions in TBARS values were observed in T4, T5 and T6 compared with T1 and T2. This might be attributed to the increased content of a-tocopherol in the meat of birds supplemented with vitamin E compared with the other groups. These findings are consistent with those of Giraudeau et al. (2013) and Coetzee and Hoffman (2001).
A TBARS value greater than 1.5 mg/kg meat is associated with an objectionable aroma owing to lipid oxidation and it is not suitable for further storage (Buckley & Morrissey, 1992; Coetzee & Hoffman, 2001), In the present study, TBARS values on days 0, 5, 7 and 10 post mortem were consistently less than 1.5 mg/kg in the meat from broilers fed T3 - T6. The implication of the current observation was that supplementation of at least 50 mg/kg vitamin E with oil blend and L-Arginine improved the shelf life of meat between 7 and 10 days of refrigeration (4 °C). On the other hand, the meat from the non-supplemented group should not be stored more than five days at 4 °C. These observations corroborate the findings of Voljc et al. (2011) that vitamin E supplementation can markedly decrease lipid oxidation and extend meat shelf life. Nonetheless, a detailed microbiological analysis is needed to substantiate this assertion.
In the current study, the increased meat thiol content observed in T4 - T6 on days 7 and 10 post mortem was in line with the findings of Batifoulier et al. (2002), who showed that supplementation of vitamin E with 6% soybean oil protected the free thiol groups from oxidation in turkey muscle compared with those fed the control diet. In the present study, the free thiol concentration in meat decreased over the postmortem ageing period consistent with results from Sabow et al. (2015) in a study of chevon. This could be because of the oxidation of the amino acid side chain (Lund et al., 2011 or a decrease in solubility and other catalytic activities, which enhanced the aggregation and degradation of protein (Nieto et al., 2013).
Carbonyl is a product of oxidized proteins and it is considered a useful indicator of protein oxidation in food (Zhang et al., 2013a). However, carbonyl is only one group of amino acid, which should not be considered representative of the whole protein oxidation phenomena. In the current study, treatments had no effect on the meat carbonyl content. Similarly, dietary supplementation of vitamin E was found to have no effect on carbonyl content in turkey meat (Batifoulier et al., 2002). However, the meat carbonyl content increased over the post-mortem ageing period, a result which is similar to the observed increase in carbonyl content in goat meat that was aged 7 days (Adeyemi et al., 2015).
Conclusion
The diets T2 - T6 produced increased growth and improved the meat quality in broiler chickens. Vitamin E decreased meat DL, increased redness, improved tenderness, and increased the lipid oxidative stability of meat during post-mortem ageing. However, these diets had no effect on the meat fatty acid profile relative to T1. In addition, vitamin E increased the free thiol content of meat on day 7 post mortem. No differences in the meat quality, lipid oxidation and thiol content were attributable to the level of vitamin E supplementation. Thus, supplementation of vitamin E at 50 mg/kg with an oil blend and L-Arginine could be used to improve growth, meat quality and meat shelf life.
Acknowledgements
The authors are thankful to the Organization for Women in Science and Technology for the Developing World (OWSD), Trieste, Italy, for awarding a PhD fellowship to the first author.
Authors' Contributions
JK conducted the experiment and analysed the meat samples; JK, TCL, HA and HLF designed and supervised the research; JK, RM and NS collected data and evaluated the carcasses. JK drafted the manuscript; JK, TCL, HLF, HA and RM reviewed and finalized the writing.
Conflict of Interest Declaration
No potential conflict of interest was reported by the authors.
References
Abdulla, N.R., Loh, T.C., Akit, H., Sazili, A.Q., Foo, H.L., Mohamad, R., Rahim, R,A., Ebrahimi, M. & Sabow, A.B., 2015. Fatty acid profile, cholesterol and oxidative status in broiler chicken breast meat fed different oil sources and calcium levels. S. Afr. J. Anim. Sci. 45,153-163. DOI: 10.4314/sajas.v45i2.6 [ Links ]
Abdulla, N.R., Zamri, A.N.M., Sabow, A.B., Kareem, K., Nurhazirah, S., Foo, H.L. & Sazili, A.Q., Loh, T.C., 2017. Physico-chemical properties of breast muscle in broiler chickens fed probiotics, antibiotics or antibiotic-probiotic mix. J. Appl. Anim. Res. 45(1), 64-70. DOI: 10.1080/09712119.2015.1124330 [ Links ]
Adeyemi, K.D., Sabowm, A.B., Shittu, R.M., Karim, R. & Sazili, A.Q., 2015. Influence of dietary canola oil and palm oil blend and refrigerated storage on fatty acids, myofibrillar proteins, chemical composition, antioxidant profile and quality attributes of semimembranosus muscle in goats. J. Anim. Sci. Biotech. 6,1-13. DOI: 10.1186/s40104-015-0050-z [ Links ]
AMSA (American Meat Science Association)., 2012. AMSA meat color measurement guidelines. American Meat Science Association, Champaign, IL. [ Links ]
AOAC, 1984. Official methods of analysis. 14th ed. Association of Official Analytical Chemists, Inc., Arlington, Virginia, USA. [ Links ]
Batifoulier, F., Mercier, Y., Gatellier, P. & Renerrem, M., 2002. Influence of vitamin E on lipid and protein oxidation induced by H2O2-activated MetMb in microsomal membranes from turkey muscle. Meat Sci. 61,389-395. DOI: 10.1016/S0309-1740(01 )00209-1 [ Links ]
Botsoglou, E., Govaris, A., Ambrosiadis, I. & Fletouris, D., 2012. Lipid and protein oxidation of a-linolenic acid-enriched pork during refrigerated storage as influenced by diet supplementation with olive leaves (Olea europea L.) or a- tocopheryl acetate. Meat Sci. 92, 525-532. DOI: 10.1016/j.meatsci.2012.05.022 [ Links ]
Bradford, M.M., 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analyt. Biochem. 72, 248-254. DOI: 10.1006/abio.1976.9999 [ Links ]
Buckley, D.J. & Morrissey, P.A., 1992. Animal production highlights. In: Vitamin E and meat quality. Hoffmann-La Roche Ltd., Basel, Switzerland. Pp. 24-27. [ Links ]
Cavitt, L.C., Youm, G.W., Meullenet, J.F., Owens, C.M. & Xiong, R., 2004. Prediction of poultry meat tenderness using razor blade shear, Allo Kramer shear and sarcomere length. J. Food Sci. 69,11-15. [ Links ]
Coetzee, G.J.M. & Hoffman, L.C., 2001. Effect of dietary vitamin E on the performance of broilers and quality of broiler meat during refrigerated and frozen storage. S. Afr. J. Anim. Sci. 31,158-173. DOI: 10.4314/sajas.v31i3.3799 [ Links ]
Corzo, A., Schilling, M.W., Loar, R.E., Jackson, V., Kin., S. & Radhakrishnan, V., 2009. The effects of feeding distillers dried grains with solubles on broiler meat quality. Poult. Sci. 88, 432-439. DOI: 10.3382/ps.2008-00406. [ Links ]
Dalle Zotte, A., Parigi Bini, R., Xiccato, G. & Simionato S., 1995. Proprietatecnologiche e sensorialidella carne di coniglio. Rivista di Coniglicoltura 32, 33-39. [ Links ]
Emadi, M., Jahanshiri, F., Azizi Jalalian, F., Kaveh, K., Bejo, M.H., Ideris, A., Assumaidaee, A.A. & Alimon, A.R., 2010. Immunostimulatory effects of arginine in broiler chickens challenged with vaccine strain of infectious bursal disease virus. J. Anim. and Vet. Advan. 9, 594-600. [ Links ]
Emadi, M., Jahanshiri, F., Kaveh, K., Hair-Bejo, M., Ideris, A. & Alimon, A.R., 2011. Nutrition and immunity: The effects of the combination of arginine and tryptophan on growth performance, serum parameters and immune response in broiler chickens challenged with infectious bursal disease vaccine. Avian Pathology 40, 63-72. [ Links ]
Folch, J., Lees, M. & Sloane-Stanley, G., 1957. A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. and Chem. 226, 497-509. [ Links ]
Fouad, A.M., El-Senousey, H.K., Yang, X.J. & Yao, J.H., 2013. Dietary L-Arginine supplementation reduces abdominal fat content by modulating lipid metabolism in broiler chickens. Anim. 7, 1239-1245. DOI: 10.1017/S1751731113000347 [ Links ]
Giraudeau, M.K., Sweazea, K., Butler, M.W. & McGraw, K.J., 2013. Effects of carotenoid and vitamin E supplementation on oxidative stress and plumage coloration in house finches (Haemorhous mexicanus). Comp. Biochem. Physiol. Part A. 166, 406-413. DOI: 10.1016/j.cbpa.2013.07.014 [ Links ]
Honikel, K.O., 1998. Reference methods for the assessment of physical characteristics of meat. Meat Sci. 49, 447-457. DOI: 10.1016/S0309-1740(98)00034-5 [ Links ]
Houston, D.K., Ding, J., Lee, J.S., Garcia, M., Kanaya, A.M., Tylavsky, F.A., Newman, A.B., Visser, M. & Kritchevsky, S.B., 2011. Dietary fat and cholesterol and risk of cardiovascular disease in older adults: The health ABC study. Nutr., Met. and Cardio. Dis. 21, 430-437. DOI: 10.1016/j.numecd.2009.11.007 [ Links ]
Karami, M., Alimon, A.R., Sazili, A.Q., Goh, Y.M. & Ivan, M., 2011. Effects of dietary antioxidants on the quality, fatty acid profile, and lipid oxidation of Longissimus muscle in Kacang goat with ageing time. Meat Sci. 88, 102-108. DOI: 10.1016/j.meatsci.2010.12.009 [ Links ]
Hhajali, F. & Wideman, R., 2010. Dietary arginine: Metabolic, environmental, immunological and physiological interrelationships. World's Poult. Sci. J. 66(4), 751-766. doi:10.1017/S0043933910000711 [ Links ]
Khan, R.U., Naz, S., Nikousefat, Z., Tufarelli, V., Javdani, M., Rana, N. & Laudadio, V., 2011. Effect of vitamin E in heat-stressed poultry. World's Poult. Sci. J. 67, 469-478. DOI: 10.1017/S0043933911000511 [ Links ]
Khatun, J., Loh, T.C., Akit, H., Foo, H.L. & Mohamad, R., 2017. Fatty acid composition, fat deposition, lipogenic gene expression and performance of broiler fed diet supplemented with different sources of oil. Anim. Sci. J. 88(9), 4061413. DOI: 10.1111/asj.12775. [ Links ]
Khatun, J., Loh, T.C., Akit, H., Foo, H.L. & Mohamad, R., 2018. Influence of different sources of oil on performance, meat quality, gut morphology, ileal digestibility and serum lipid profile in broilers. J. Appl. Anim. Res. 46(1), 479-485, DOI:10.1080/09712119.2017.1337580. [ Links ]
Khatun, J., Loh, T.C., Akit, H., Foo, H.L. & Mohamad, R. & Kareem. K.Y. 2019. Dietary supplementation with L-Arginine and combinations of different oil sources beneficially regulates body fat deposition, lipogenic gene expression, growth performance and carcass yield in broiler chickens. Anim. Prod. Sci. DOI: https://doi.org/10.1071/AN19205 [ Links ]
Lawrie, R. & Ledward, D., 2006. Lawrie's meat science. Woodhead, Cambridge. [ Links ]
Li, T.S., Han, R.L., Xing, Y.G., Sun, Y.L., Wang., B.Y. & Cao, F., 2003. Effects of dietary vitamin E level on performance and meat quality of Luxi yellow broilers. Acta Zoonutrimenta Sinic. 15, 44-48. [ Links ]
Li, W.J., Zhao, G.P., Chen, J.L., Zheng, M.Q. & Wen, J., 2009. Influence of dietary vitamin E supplementation on meat quality traits and gene expression related to lipid metabolism in the Beijing-you chicken. Brit. Poult. Sci. 50, 188-198. DOI: 10.1080/00071660902755409 [ Links ]
Lund, M.N., Heinonen, M., Baron, C.P. & Estevez, M., 2011. Protein oxidation in muscle foods: A review. Mol. Nutr. Food Res. 55, 83-95. DOI: 10.1002/mnfr.201000453 [ Links ]
Lynch, S.M. & Frei, B., 1993. Mechanisms of copper-and iron-dependent oxidative modification of human low density lipoprotein. J. Lipid Res. 34, 1745-1753. [ Links ]
Ma, X., Zheng, C., Hu, Y., Wang, L., Yang, X. & Jiang, Z., 2015. Dietary L-Arginine supplementation affects the skeletal longissimus muscle proteome in finishing pigs. PLOS ONE 10 (1), e0117294. DOI: 10.1371/journal.pone.0117294 [ Links ]
Morzel, M., Gatellier, P., Sayd, T., Renerre, M. & Laville, E., 2006. Chemical oxidation decreases proteolytic susceptibility of skeletal muscle myofibrillar proteins. Meat Sci. 73, 536-543. DOI: 10.1016/j.meatsci.2006.02.005 [ Links ]
Nieto, G., Jongberg, S., Andersen, M.L. & Skibsted, L.H. 2013. Thiol oxidation and protein cross-link formation during chill storage of pork patties added essential oil of oregano, rosemary, or garlic. Meat Science 95(2), 177-184. [ Links ]
Oliver, C.N., Anh, B.W., Moerman, E.J., Goldstein, S. & Stadtman, E.R., 1987. Age-related changes in oxidized proteins. J. Biol. Chem. 262, 5488-549. [ Links ]
Ponnampalam, E.N., Butler, K.L., McDonagh, M.B., Jacobs, J.L. & Hopkins, D.L., 2012. Relationship between muscle antioxidant status, forms of iron, polyunsaturated fatty acids and functionality (retail colour) of meat in lambs. Meat Sci. 90, 297-303. DOI: 10.1016/j.meatsci.2011.07.014 [ Links ]
Rutkowski, M. & Grzegorczyk, K., 2007. Modifications of spectrophotometric methods for antioxidative vitamins determination convenient in analytic practice. Acta Sci. Pol. Technol. Aliment. 6, 17-28. [ Links ]
Sabow, A.B. 2016. Physiological stress responses and meat quality characteristics in goats subjected to different stunning and slaughter methods. PhD thesis, Dept. of Animal Science, University Putra Malaysia. [ Links ]
Sabow, A.B., Sazili, A.Q., Zulkifli, I., Goh, Y.M., Kadir Mohd Zainal Abidin, A.B. & Adeyemi, K.D., 2015. Physico-chemical characteristics of longissimus lumborum muscle in goats subjected to halal slaughter and anesthesia (halothane) pre-slaughter. Anim. Sci. J. 86(12), 981-91. DOI: 10.1111/asj.12385 [ Links ]
Santé-Lhoutellier, V., Engel, E. & Gatellier, P., 2008. Assessment of the influence of diet on lamb meat oxidation. Food Chem. 109, 573-579. DOI: 10.1016/j.foodchem.2007.11.081 [ Links ]
Skrivan, M., Dlouhá, G., Englmaierová, M. & Cervinková, K., 2010. Effects of different levels of dietary supplemental caprylic acid and vitamin E on performance, breast muscle vitamin E and A, and oxidative stability in broilers. Czech. J. Anim. Sci. 55, 167-173. [ Links ]
Tan, B., Yin, Y., Liu, Z., Li, X., Xu, H., Kong, X., Huang, R., Tang, W., Shinzato, I., Smith, S.B. & Wu, G., 2009. Dietary L- Arginine supplementation increases muscle gain and reduces body fat mass in growing finishing pigs. Amino Acids 37, 169-175. DOI: 10.1007/s00726-008-0148-0 [ Links ]
Tancharoenrat, P., Ravindran, V., Zaefarian, F. & Ravindran, G., 2014. Digestion of fat and fatty acids along the gastrointestinal tract of broiler chickens. Poult. Sci. 93, 371-379. DOI: 10.3382/ps.2013-03344 [ Links ]
Velasco, S., Ortiz, L.T., Alzueta, C., Rebole, A., Trevino, J. & Rodriguez, M.L., 2010. Effect of inulin supplementation and dietary fat source on performance, blood serum metabolites, liver lipids, abdominal fat deposition, and tissue fatty acid composition in broiler chickens. Poult. Sci. 89, 1651-1662. DOI: 10.3382/ps.2010-00687 [ Links ]
Voljc, M., Frankic, T., Levart, A., Nemec, M. & Salobir, J., 2011. Evaluation of different vitamin E recommendations and bioactivity of a-tocopherol isomers in broiler nutrition by measuring oxidative stress in vivo and the oxidative stability of meat. Poult Sci. 90, 1478-1488. DOI: 10.3382/ps.2010-01223 [ Links ]
Winterbourn, C.C., 1990. Oxidative reactions of hemoglobin. Meth. in Enzymol. 186, 265-272. [ Links ]
Zdunczyk, Z., Gruzauskas, R., Semaskaite, A., Juskiewicz, J., Raceviciute-Stupeliene, A. & Wroblewska, M., 2011. Fatty acid profile of breast muscle of broiler chickens fed diets with different levels of selenium and vitamin E. Arch. Geflügelk. 75 (4), 264-267. [ Links ]
Zhang, W., Xiao, S. & Ahn, D.U., 2013a. Protein oxidation: Basic principles and implications for meat quality. Crit. Rev. Food Sci. Nutr. 53, 1191-1201. DOI: 10.1080/10408398.2011.577540 [ Links ]
Zhang, W., Xiao, S., Lee, E.J. & Ahn, D.U., 2011. Effects of dietary oxidation on the quality of broiler breast meat. Anim. Indust. Rep. 657, 48. [ Links ]
Zhang, Y., Shan, A.., Jiang, W., Bi. C. & Li, Z., 2013b. The effect of vitamin E on growth performance and meat quality in broilers given diets containing distillers' dried grain with soluble (DDGS). Brit. Poult. Sci. 54, 138-143. DOI: 10.1080/00071668.2012.757578 [ Links ]
Zhong, X., Gao, S., Wang, J.J., Dong, L., Huang, J., Zhang, L.L. & Wang, T., 2014. Effects of linseed oil and palm oil on growth performance, tibia fatty acid and biomarkers of bone metabolism in broilers. Brit. Poultry Sci. 55, 335-342. DOI: 10.1080/00071668.2014.891097 [ Links ]
Submitted 20 April 2020
Accepted 15 July 2020
Published 14 November 2020
# Corresponding author: tcloh@upm.edu.my














