Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Animal Science
On-line version ISSN 2221-4062
Print version ISSN 0375-1589
S. Afr. j. anim. sci. vol.44 n.2 Pretoria Feb. 2014
Estimation of genetic parameters for body measurements and their association with yearling liveweight in the Makuie sheep breed
S. JafariI, II, #, ##; A. HashemiI
IDepartment of Animal Science, Faculty of Agriculture, Urmia University, Urmia, Iran
IIWest Azerbaijan Jihad-Agriculture Organization, Makuie Sheep Breeding Station, Maku City, Iran
ABSTRACT
The main objective of this study was to estimate the genetic parameters for body measurement and yearling live bodyweight traits in the Makuie sheep breed. The data set consisted of 2 144 lambs from 122 sires and 984 dams recorded in 21 years from 1989 to 2009 at the Makuie Sheep Breeding and Raising Station (MSBS). The traits that were studied were height at withers, height at rump, body length, heart girth, leg circumference and yearling weight. The analyses were carried out based on an animal model with the restricted maximum likelihood (REML) approach using a derivate-free (DF) algorithm. Six animal models were fitted, differentiated by including or excluding maternal effects, and with and without covariance between maternal and direct genetic effects. Ignoring the maternal effects, the direct heritability was estimated as 0.20, 0.24, 0.10, 0.14, 0.02 and 0.36 for height at wither, height at rump, body length, heart girth, leg circumference and yearling weight, respectively. The estimates were slightly higher when maternal effects, genetic or environmental, were ignored in the analyses. Using bivariate analysis, the estimates for additive genetic correlations ranged from 0.56 to 0.81 among the studied traits. A log-likelihood ratio test (LRT) was used to select the most appropriate univariate model for each trait. Based on LRT, the direct additive genetic and maternal permanent environmental effects were regarded as an important source of variation of the studied traits. The estimates of genetic parameters can be considered a basis for calculating selection indices for body measurements, as well as revealing their association with yearling bodyweight traits.
Keywords: (Co) variance components, direct and maternal effects, heritability
Introduction
The Makuie sheep is a native breed of Azerbaijan province (Iran) and is also found in Turkey, where it is called the White Karaman. Its total population is estimated at 2.7 million (Abbasi & Ghafouri-Kesbi, 2011). It has been adapted to cold and highland environments (Safari, 1986). Makuie is a medium-sized (ewe: 45 -48 kg, ram: 51 - 53 kg) fat-tailed sheep breed. The common body colour is white with black rings around the eyes, nose and knees (Saadatnoori & Syahmansoor, 1981). It is a multipurpose breed, producing milk, wool and mutton. However, the main purpose of flock holders is mutton production owing to the high economic and religious values of sheep meat in Iran. The breed is raised in extensive or semi-intensive systems (Jafari et al., 2012).
Regarding the importance of this breed in the regional economy, the Makuie Sheep Breeding and Raising Station (MSBS) was established in Maku city by the Agricultural Ministry in 1986. The main purpose was the protection and improvement of the breed (Safari, 1986).
Meat yield is a complex polygenic trait that is highly affected by non-genetic and genetic factors. Biometric characteristics or linear measurements with simple genetic controls could be used as indirect criteria in many domestic animal species to help meat-yield improvement. Body and weight measurements describe more completely an individual or population than the conventional methods of weighing and grading (Salako, 2006).
Using measurement criteria, breeders are able to identify early and late maturing animals with different sizes (Brown et al., 1973). It will be helpful to identify appropriate animals at an earlier growth stage for election and prediction of mature ranking. Body measurement traits and related selection indices not only help breeders to evaluate animal weight, but could be used as functional indicators in animal production (Salako, 2006).The age at puberty is influenced by breed, genetic selection, nutrition, and season of birth, as well as body size.
Lambs with bigger bodies tend to mature at an earlier age. Breeders can use biometric traits as a criterion in selection programmes. The present study focused on estimating genetic parameters of body measurements with six models and their correlations with yearling liveweight in the Makuie sheep breed.
Material and Methods
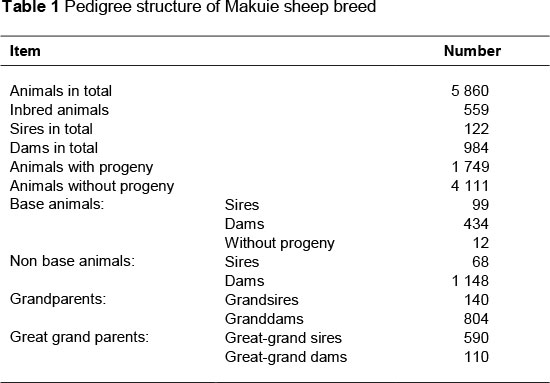
The body measurement data of 2 144 lambs from 122 sires and 984 dams recorded in 21 years from 1989 to 2009 at MSBS were used in the present study (Table 1). The data included height at withers, height at rump, body length, heart girth, leg circumference and yearling weight. Height at withers measures the distance from a platform on which the animal stands to the withers. The measurement is best made with a special measuring stick on two arms, one held vertically, and the other at right angles, so that it slides firmly up and down to record the height. Height at rump is the distance from the surface of a platform to the rump using a measuring stick as described for height at withers. Body length refers to the distance from the first cervical vertebra to the base of the tail where it joins the body. Heart girth is a circumferential measure taken around the chest just behind the front legs and withers with a tape measure. Leg circumference refers to the circumference of the rear legs. The midpoint between the hock and pin bone on the right rear leg is used to measure leg circumference, using a tape measure.
Variance and covariance components were estimated based on an animal model with the restricted maximum likelihood (REML) approach, using a derivate-free (DF) algorithm (Meyer, 1989). The year of birth, sex, birth type and age of dam had 21, 2, 3 and 6 levels, respectively, and were regarded as fixed effects. Six different animal models that differentiated by including or excluding maternal effects were fitted for each trait, with and without covariance between maternal and direct genetic effects, as described by Meyer (1992). The linear forms of six models were:
Model I: Yijklm = µ+ YRi+ SXj+ BTk+ AD1+ ANm+ eijklm
Model II: Yijklm = µ+ YR+ SXj+ BTk + ADl+ ANm+ PEn+ eijklm
Model III: Yjklmn = µ+ YR+ SXj+BTk+ ADl+ ANm+ Mn+ eijklmn(ram=0)
Model IV: Yijklmn = µ+ YRi+ SXj+ BTk+ ADl+ ANm+ Mn+ eijklmn(ram ≠0)
Model V: Yijklm = µ+ YRi+ SXj+ BTk+ ADl+ ANm+ PEn+ Mo + eijklmn(ram=0)
Model VI: Yijklm = µ+ YRi+ SXj+ BTk+ ADl+ ANm+ PEn+ Mo + eijklmn(ram ≠0)
where Yijk = each observation on underlying trait belongs to its appropriate group; µ = overall mean of population; YRi = fixed effect of year i; SXj = fixed effect of sex j; BTk = fixed effect of birth type k; ADl = fixed effect of age of dam l; ANm = individual additive genetic effect of animal m; PEn = random effect of permanent maternal environment in n levels (n = number of maternal levels for each trait ); Mn = maternal genetic effect; eijkl= residual random effect of observation ijk.
Depending on the fitted model, the direct additive genetic variance (σ2a), maternal genetic variance (σ2m), permanent environmental variance (σ2pe) and residual variance (σ2e) are considered the main sources of random variance. The direct heritability (h2a), maternal heritability (h2) and variance due to the permanent environment (c2) were estimated as σ2a/σ2p, σ2m/σ2 and σ2pe/σ2p, respectively, where σ2p was the phenotypic variation. Genetic covariance between direct and maternal genetic effects (σam) and consequent correlations between these effects (ram) were considered in models 4 and 6. Total heritability (h2t) was estimated as σ2a + 0.5σ2m + 1.5σam/σ2p (Willham, 1972).
Estimates of variance and covariance components and genetic parameters for height at withers, height at rump, body length, heart girth and leg circumference at 12 months of age and yearling weight and likelihood values for each analysis under the six models are summarized in Table 2. To choose the best model, the likelihood ratio test (LRT) was applied. This test was used to determine the significance between full and nested models. Statistical significance for models was set at P <0.05. The LRT value was calculated as -2ln (likelihood for full model) + 2ln (likelihood for nested model). If the LRT value was greater than a critical value from a X2 distribution with an appropriate degree of freedom equal to the difference in the number of (co)variance components fitted for the two models it can be concluded that the additional random effect had a significant effect on the model and the nested model was not better. When the differences were not significant, the nested model that had fewer parameters was chosen as the appropriate model.
Results and Discussion
Number of records, mean of traits, standard deviations and coefficient of variance according to different fixed effects are presented in Table 2. The overall mean value of the height at rump was, by 1.5 cm, higher than that of wither height. In mountainous conditions such as Maku region, the greater height of the rear part of the body can be proposed as a breed advantage. This advantage may promote the climbing ability of the animal, especially in the seasonal migration between valleys and high pastures. This finding is in accordance with the results of Cam et al. (2010) for the Turkish Karayaka sheep breed.
Low coefficients of variation (CVs) were observed for body measurements, and have been reported by other researchers (Fourie et al., 2002; Ermias & Rege, 2003; Alfolayan et al., 2006; Salako, 2006). The relatively low CV value estimates of the body measurements in the present study can be explained by the small differences among the animals of the population, greater uniformity of the traits, minor changes in these traits by environmental qualifications, better response to selection and other unknown factors.
Data structure from incorporating fixed effects on body measurement traits is presented in Table 2. The environmental factors showed a significant effect on body measurement traits in Makuie sheep.
All traits, except heart girth, were affected (P <0.001) by the sex of the animal. For height at withers, height at rump and body length, the males were bigger by 2, 2.21 and 1 cm, respectively, than the females. However, females were superior to males in leg circumference. Male and female did not have significant difference in heart girth. These results are similar to the reports of Kunene et al. (2007) for the Zulu sheep breed. Significant influences of sex factor on growth may be because of physiological characteristics and the endocrinal system, type and measure of hormone secretion, especially sexual hormones (Aghaali-Gamasaee et al., 2010).
A significant effect of birth type on body measurement traits has been reported in other breeds such as Muzaffarnagri sheep (Mandal et al, 2010). This could be the result of limited uterine space during pregnancy, limited capacity of ewes to provide more nutrients during pregnancy, more milk for lambs in the period from lambing to weaning and competition for milk suckling between multiple-birth lambs (Rajab et al., 1992). Significant influences of birth year on these traits can be explained by differences in management, food availability, disease, climatic condition (such as rate of rainfall, humidity and temperature, which affected the quality and quantity of pasture forage) and raising system in different years. The results for age of dam on height at rump, body length and heart girth were non-significant. Its effect on height at withers, leg circumference and yearling weight was significant (P <0.05). Younger ewes tend to produce smaller lambs. Primiparous ewes have not reached their mature weight and their growth is compromised because of foetal growth. This could affect lamb size and weight. It is well known that mothering ability, such as milk yield, increases with parity, as older ewes are usually larger and produce more milk. The same results have been reported by El Fadili et al. (2000) on the Moroccan Timahdit breed of sheep.
The most suitable models are shown in bold (Table 3). Table 3 shows that the significant model for analyzing of height at wither, height at rump, body length and leg circumference traits was Model 1. This suggested that the notable source of random variation for height at withers, height at rump, body length and leg circumference was direct additive genetic effects. According, the LRT at a probability level of 5% revealed that considering maternal effects when analysing the traits was pointless. The two other traits, heart girth and yearling weight, were affected significantly by the maternal environmental effect in addition to direct genetic effect. This findings revealed that heart girth and yearling weight were affected (P <0.05) by the maternal permanent environmental characters (so-called dam-lamb association) such as uterus environment, amount of milk production, milk composition and udder conditions (Khan et al., 2006). These results are in accordance with reports of Mandal et al. (2010) for body measurements traits in Muzzafarnagri sheep breed at 12 months of age. Permanent environmental maternal effect in model (Model 2) explained 2% to 16% of total phenotypic variance for body measurement traits and yearling weight.
Direct heritability estimates using Model I were 0.20, 0.24, 0.10, 0.14, 0.02 and 0.36 for height at withers, height at rump, body length, heart girth, leg circumference and yearling weight, respectively. The estimates were slightly higher when maternal effects, genetic or environmental, were ignored from the analyses. Direct heritability has been estimated at 0.17, 0.17, 0.11, 0.21 and 0.22 for height at withers, height at rump, body length, heart girth and yearling weight, respectively, by Abbasi & Ghafouri-kesbi (2011) in Makuie sheep, based on 10 years data.
Although, a negative ram is impossible from a biological perspective (Matiatis & Pollott, 2003), introducing a non-zero covariance between direct and maternal genetic effects in models (Models 4 and 6) yielded a negative correlation between these effects. Based on previous studies, the probable reason for a negative ram is poor environmental conditions, such as udder problems, non-sufficient nutrition and experimental conditions (Meyer, 1992).
Recent studies have shown that the data structure plays the major role in producing a negative correlation between direct and maternal genetic effects. A low progeny record per dam in data structure produces a negative ram (Matiatis & Pollott, 2003), whereas a high number of progeny records per dam produces a positive ram.
From a developmental perspective, some negative correlations provide checks and balances between direct and maternal effects for growth traits, whereas this condition could undermine species by becoming extreme in view of producing more and more milk (Cundiff, 1972).
Since the maternal genetic effect is heritable, the total heritability (h2t) of the studied traits in the present study may be somewhat higher than the estimates of direct heritability (h2a), unless the correlations between direct and maternal genetics are large and negative. Table 3 shows a direct-maternal genetic correlation of 0.00 to -0.98. Including the direct-maternal genetic covariance in the animal models (Models 4 and 6), the estimates of total heritability for all traits were lower than those of direct heritability estimates owing to the negative estimates of ram (Table 3). According to the h2t formula, when direct-maternal genetic covariance is negative, the presence of this covariance decreases total heritability and, in consequence, the potential response to selection (Wilson et al., 2005).
In Model 3, which included maternal genetics in addition to direct additive effect, the added random effect explained only 0.00% to 29% of random variation of the studied traits. However, the presence of maternal genetic was non-significant in affecting the total phenotypic variance.
Characteristics of the data structure of phenotypic, additive, permanent environmental and residual correlations are summarized in Table 4. Direct genetic correlations were generally higher than phenotypic correlations. The additive genetic correlation between studied traits, arising chiefly from pleiotropy, was positive in all cases (0.25 to 0.99), indicating that selection for a superior body measurement character would result in genetic improvement in the other traits that were studied (Gowane et al., 2011). Phenotypic correlations were estimated in the range of 0.32 to 0.90.
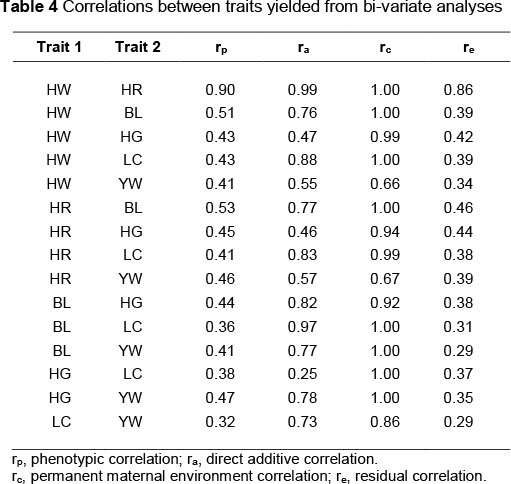
These results are nearly similar to reports on the Belgian bleu du Maine, Suffolk and Texel sheep (Janssens & Vandepitte, 2004) and Yankasa lambs (Yakubu, 2010). In accordance with the results reported for growth traits of the Tygerhoek Merino flock (Duguma et al., 2002), generally the estimates of maternal permanent environmental correlation were higher than the estimates of direct, phenotypic and residual correlations. Genetic (phenotypic) correlations between body measurements and yearling weight were estimated at moderate to high. The highest genetic (phenotypic) correlation was estimated between heart girth and yearling body liveweight. In other words, the heart girth is the most reliable character for the prediction of yearling body liveweight among the body measurements. In a selection programme based on body measurement, heart girth would be a suitable selection criterion, since it has relatively high genetic correlation with yearling weight.
Conclusion
Results obtained in this study indicated that moderate to high total heritability estimates for body measurements and yearling weight traits can be used successfully as guides to selection in programmes aimed at improvement of local sheep. It also revealed that a substantial variability among individuals in the Makuie sheep breed population. The estimated genetic parameters using six models could be proposed as an ethnic characteristic. The high additive genetic correlations among the body measurements indicated the importance of this effect in the similarity of traits. The high correlation between body measurements and yearling weight suggested that genetic progress in the body measurement traits and liveweight traits is possible at the same time.
Acknowledgement
All esteemed colleagues at Makuie Sheep Breeding Station, West Azerbaijan Jihad-Agricultural Organization and Shut City Jihad-Agricultural Management Office are appreciated for their sterling efforts in improving MSBS goals.
References
Abbasi, M.A. & Ghafouri-Kesbi, F., 2011. Genetic co (variance) components for body weight and body measurements in Makooei sheep. Asian-Austral. J. Anim. Sci. 24, 739-743. [ Links ]
Aghaali-Gamasaee, V., Hafezian, S.H., Ahmadi, A., Baneh, H., Farhadi, A. & Mohamadi, A., 2010. Estimation of genetic parameters for body weight at different ages in Mehraban sheep. Afr. J. Biotechnol. 9 (32), 5218-5223. [ Links ]
Alfolyan, R.A., Adeyinka, I.A. & Lakpini, C.A.M., 2006. The estimation of live weight from body measurement in Yankasa sheep. Czech. J. Anim. Sci. 51, 343-348. [ Links ]
Brown, J.E., Brown, C.J. & Butts, W.T., 1973. Evaluating relationships among immature measures of size, shape and performance of beef bulls. J. Anim. Sci. 36, 1010-1020. [ Links ]
Cam, M.A., Olfaz, M. & Soydan, E., 2010. Body measurements reflect body weights and carcass yield in Karayaka sheep. Asian J. Anim. Vet. Adv. 5 (2), 120-127. [ Links ]
Cundiff, L.V., 1972. The role of maternal effects in animal breeding: VIII Comparative aspect of maternal effects. J. Anim. Sci. 35, 1335-1337. [ Links ]
Duguma, G., Schoeman, S.J., Cloete, S.W.P. & Jordaan, G.F., 2002. Genetic parameter estimates of early growth traits in the Tygerhoek Merino flock. S. Afr. J. Anim. Sci. 32, 66-75. [ Links ]
El Fadili, M., Michaux, C., Detilleux, J. & Leroy, P.L., 2000. Genetic parameters for growth traits of the Moroccan Timahdit breed of sheep. Small. Rumin. Res. 37, 203-208. [ Links ]
Ermias, E. & Rege, J.E.O., 2003. Characteristic of live animal allometric measurements associated with body fat in fat- tailed sheep. Livest. Prod. Sci. 81, 271-281. [ Links ]
Fourie, P.J., Neser, F.W.C., Olivier, J.J. & Van der Westhuizen, C., 2002. Relationship between production performance, visual appraisal and body measurements of young Dorper rams. S. Afr. J. Anim. Sci. 32, 256-262. [ Links ]
Gowane, G.R., Chopra, A., Prince, L.L.L., Mishra, A.K. & Arora, A.L., 2011. Genetic analysis for growth traits of prolific Garole * Malpura (GM) sheep. Trop. Anim. Health. Prod. 43, 299-303. [ Links ]
Jafari, S., Hashemi, A., Manafiazer, G., Darvishzadeh, R., Razzagzadeh, S. & Farhadian, M., 2012. Genetic analysis of growth traits in Iranian Makuie sheep breed. Ital. J. Anim. Sci. 11, 98-102. [ Links ]
Janssens, S. & Vandepitte, W., 2004. Genetic parameters for body measurements and type traits in Belgian Bleu du Maine, Suffolk and Texel sheep. Small Rumin. Res. 54, 13-24. [ Links ]
Khan, H.M., Bhat, A.S., Singh, P.K. & Islam, R., 2006. Effect of dam lamb relationship on body weight gain of Coriedale lambs during pre-weaning period. Indian J. Anim. Res. 40 (1), 82-84. [ Links ]
Kunene, N., Nesamvuni, E.A. & Fossey, A.F., 2007. Characterization of Zulu (Nguni) sheep using linear body measurement and some environmental factors affecting these measurements. S. Afr. J. Anim. Sci. 37, 11-20. [ Links ]
Mandal, A., Dass, G., Rout, P.K. & Roy, R., 2010. Genetic parameters for direct and maternal effects on post-weaning body measurements of Muzaffarnagari sheep in India. Trop. Anim. Health. Prod. 10, 9752-9756. [ Links ]
Matiatis, N. & Pollott, G.E., 2003. The impact of data structure on genetic (co) variance components of early growth in sheep, estimated using an animal model with natural effects. J. Anim. Sci. 81, 101-108. [ Links ]
Meyer, K., 1989. Restricted maximum likelihood to estimate variance components for animal models with several random effects using a derivative-free algorithm. Genet. Sel. Evol. 21, 317-340. [ Links ]
Meyer, K., 1992. Variance components due to direct and maternal effects for growth traits of Australian beef cattle. Livest. Prod. Sci. 31, 179-204. [ Links ]
Rajab, M.H., Cartwright, I.C., Dahm, P.F. & Figueireda, E.A.P., 1992. Performance of three tropical hair sheep breeds. J. Anim. Sci. 70, 3351-3359. [ Links ]
Saadatnoori, M. & Siahmansoor, S., 1986. Principles of Sheep Industry. Third edition, published by Armagan Publication. pp. 101-105. (in Persian). [ Links ]
Safari, E., 1986. Report for identification of Makuie ecotype. Published by Agriculture Ministry of Iran. pp. 10-25. (in Persian). [ Links ]
Salako, A.E., 2006. Application of morphological indices in the assessment of type and function in sheep. Int. J. Morphol. 24 (1), 13-18. [ Links ]
Willham, R.L., 1972. The role of maternal effects in animal breeding: III. Biometrical aspects of maternal effects in animals. J. Anim. Sci. 35, 1288-1293. [ Links ]
Wilson, A.J., Coltman, D.W., Pemberton, J.M., Overall, A.D.J., Byrne, K.A. & Kruuk, L.E.B., 2005. Maternal genetic effects set the potential for evolution in a free-living vertebrate population. J. Evolution. Biol. 18, 405-414. [ Links ]
Yakubu, A., 2010. Path coefficient and path analysis of body weight and biometric traits in Yankasa lambs. Slovak J. Anim. Sci. 43, 17-25. [ Links ]
Received 4 December 2012
Accepted 5 March 2014
First published online 5 June 2014
# Corresponding author: shojaj@gmail.com
## West Azerbaijan Jihad-Agriculture Organization, Makuie Sheep Breeding Station, Maku City, Iran














