Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Animal Science
On-line version ISSN 2221-4062
Print version ISSN 0375-1589
S. Afr. j. anim. sci. vol.43 n.4 Pretoria 2013
Nutrient intake, digestibility and nitrogen retention in indigenous goats fed on Acacia nilotica fruits treated for condensed tannins
T. TshabalalaI, II, #; J.L.N. SikosanaIII; E. ChivandiI, IV
IDepartment of Livestock & Wildlife Management, Midlands State University, P. Bag 9055 Gweru, Zimbabwe
IISchool of Animal, Plant and Environmental Sciences, University of the Witwatersrand, P. Bag 3, Johannesburg, 2050 South Africa
IIIDepartment of Research and Specialist Services, Matopos Research Institute, P. Bag K5137, Bulawayo, Zimbabwe
IVSchool of Physiology, Faculty of Health Sciences, University of the Witwatersrand, 7 York Road, Johannesburg, South Africa
ABSTRACT
Polyethylene glycol (PEG 4000), Browse PlusTM (BP), wood ash (WA) and boiling water were evaluated as tannin deactivation methods on tannins in Acacia nilotica fruits. Twenty four Matebele goats of mean body mass, 38.7 ± 3.12 kg, were used in a digestibility and nitrogen balance trial. A completely randomized design was used, and the initial weight was used as a covariate in data analysis. The goats were randomly allocated to the treatments and were fed for 28 days on a basal diet of hay and A. nilotica fruits treated with PEG (MW 4000), BP, WA or boiling water. Acacia nilotica fruits had 4.39% catechin equivalents of condensed tannins, thus the amount of PEG, BP and WA used to treat the fruits was 1.5 parts per part of condensed tannin in the fruits. It was hypothesized that treating the fruits would deactivate the tannins in them and increase intake, digestibility and nitrogen retention. Treating the fruits for condensed tannins had no effect on hay and fruit intake. None of the treatments had an effect on the apparent digestibility of dry matter, organic matter, neutral and acid detergent fibre and nitrogen retention in the goats. The goats that were fed on the boiled fruits had a negative N retention of -1.03 g/day. It was concluded that none of the detannification methods improved feed intake, apparent nutrient digestibility and nitrogen retention. It is recommended that each detannification method should be tested on different types of tannins and at different concentrations before totally dismissing the treatments as not useful.
Keywords: Boiling, Browse Plus, pods, proanthocyanidins, PEG, wood ash
Introduction
Shortage of good-quality feed, especially during the long dry season in tropical and subtropical areas, has increased the need to provide supplementary feed to maintain livestock condition (Hove et al., 2001). However, there has been a drastic increase in the cost of feed in recent years owing to the economic hardships faced by most African countries. Few communal farmers can afford to buy supplementary feed for livestock, and some have resorted to collecting tree fruits and feeding them to livestock as protein supplements, especially during the dry season (Kindness et al., 1999; Sikosana et al., 2002). Tree fruits are especially valuable because they ripen and fall on the ground in the dry season when there is no green forage (Timberlake et al. , 1999). Many trees produce potentially nutritious fruits containing up to 200 g crude protein/kg (Tanner et al., 1990).
Trees of the genus Acacia, in particular Acacia nilotica, with yields of about 35 kg of fruit per tree per season, contribute to the bulk of tree fruits gathered for feeding goats (Timberlake et al., 1999). However, A. nilotica has been reported to contain condensed tannins that reduce browse utilization efficiency by herbivores (Tanner et al., 1990; Phale&Madibela, 2006; Nsahlai et al., 2011). Condensed tannins act as defensive mechanisms in plants against herbivory (Cooper & Owen-Smith, 1985). They (condensed tannins) are water-soluble polymeric phenolics that bind to proteins, preventing microbial attack on proteins (Zucker, 1983; Reed et al., 1990), and reduce microbial enzyme activity (Ngwa et al., 2003; Nsahlai et al., 2011) in the rumen. In ruminants, condensed tannins reduce forage intake (Tanner et al., 1990; Mueller-Harvey & Mcallan, 1992), digestibility and nitrogen retention (Tanner et al., 1990; Silanikove et al., 1997). While the negative effects of tannins on ruminant animal performance have been well documented (Tanner et al., 1990; Silanikove et al., 1997; Ngwa et al., 2003; Nsahlai et al., 2011), it has been noted that tannin-protein complexes are stable in the near-neutral pH environment of the rumen, but disintegrate on reaching the acid environment of the abomasum (Reed, 1995). In this way, tannins increase nitrogen flow to post-ruminal sites hence an increase in nitrogen retention and animal weight gains (Reed, 1995; Kaitho et al., 1997; Nsahlai et al., 1999). Detannification treatments could increase the feed value of these tanniferous forages. There are few reports on the effects of treatments on condensed tannins in A. nilotica fruits on the digestibility and animal performance.
Several methods have been used to alleviate the deleterious effects of tannins in browse trees, including the use of polyethylene glycol (PEG 4000), Browse PlusTM (BP), polyvinylpyrrolidone (PVP) and calcium hydroxide formulation (Cooper Pvt Ltd, Zimbabwe), wood ash (WA) and boiling in hot water. The tannin binding compounds, PEG and PVP, bind to tannins with a higher affinity than protein (Barry & Duncan, 1984; Waghorn, 1990; Silanikove et al., 2001; Priolo et al., 2005; Mlambo et al., 2007) thus increasing the availability of protein for utilization by browsers. The calcium hydroxide in BP provides an alkaline environment in which tannins undergo oxidation to inert forms (Mueller-Harvey & Mcallan, 1992). The alkaline nature of WA and heat from boiling water deactivates the tannin-protein bond in forages (Mukuru et al., 1992; Smith et al., 2005) hence releasing proteins for digestion and absorption by the ruminant. Despite this knowledge of the effects of these detannification agents in browse species, few attempts have been made to determine their efficacy on the fruits of A. nilotica and the subsequent impact of treating the fruits with detannification agents on goat performance.
While there is scientific evidence that goats produce proline-rich proteins (PRPs) that precipitate tannins and thus allow them to consume tanniniferous forages (Shimada, 2006), it has been noted that this adaptation comes about following chronic exposure to such forages. However, Hanovice-Ziony et al. (2010) concluded that salivary tannin-binding proteins did not have a major role in Mediterranean goats. Similarly, Makkar (2003) reported that despite being fed tannin-rich oak leaves, in winter, the saliva of goats did not contain PRPs. Interestingly, Salem et al. (2013) noted that chronic exposure, with 24-hour saliva samplings at days 1 - 2, 31 - 32 and 61 - 62 to quebracho tannins did not increase the protein concentration in parotid saliva of goats. Furthermore, many researchers have used goats in evaluating the efficacy of different detannification agents (Getachew et al. , 1998; Makkar, 2003). More importantly, goats are the predominant small ruminants in western Zimbabwe affected by shortages of nutritional adequate feed during the dry season. Despite their being considered "adapted" to tannins (Mlambo et al., 2004), due to the inadequate comprehension of the tannin adaptation mechanisms coupled with divergent views with regards to role of PRPs in goats' adaptation to tanniniferous forages, the mechanisms reported in literature might not hold under semi-arid conditions of western Zimbabwe, hence the use of goats in the study. The objective of the study was to determine the efficacy of WA, BP, PEG and boiling on the reduction of the effects of condensed tannins in A. nilotica fruits on feed intake and digestibility.
Materials and Methods
The study was conducted at Matopos Research Institute (28° 30 E; 20° 23 S; altitude: 1340 m asl), 30 km south of Bulawayo, Zimbabwe. Mean minimum and maximum annual temperatures are 21 °C and 29 °C, respectively. Average annual rainfall is 600 mm. The vegetation is characterized by thorny Acacia species, mostly A. karroo and A. nilotica with an understorey layer dominated by perennial grasses with occasional annuals (Ward et al., 1979).
The amount of fruits per treatment needed to last the experiment was estimated to be 40 kg. The amount of PEG given to animals was 1.5 parts per part of condensed tannin (Waghorn, 1990). The Vanillin-HCL method (Burns, 1971; Broadhurst & Jones, 1978) showed that A. nilotica fruits contained 4.39% catechin equivalent of proanthocyanidins (condensed tannins). This implied that 40 kg of A. nilotica fruits had approximately 1.756 kg of proanthocyanidins. Application of PEG was used as a standard for other detannification agents, which was 2.7 kg per 40 kg of fruits. A PEG solution was made through dissolving 2.7 kg of PEG in 20 L of distilled water, which was adequate to wet 40 kg of the fruits. This was followed by soaking 40 kg of the fruits in the solution for 15 min, which was sufficient time for wetting the fruits. Fruits were subsequently sun dried. The BP treatment of fruits was conducted in the same way as the PEG treatment. A BP solution was made by dissolving 2.7 kg of BP in 20 L of distilled water. Then the fruits were soaked in the solution for 15 min.
Wood ash (WA) powder was obtained by burning an A. nilotica tree. The WA solution was made by dissolving 2.7 kg WA in 20 L distilled water. Then fruits were soaked in the solution for 15 min. About 100 L of water was boiled and then 40 kg A. nilotica fruits were cooked for 15 min, and then sun dried. The control treatment comprised untreated A. nilotica fruits. All the fruits were subjected to post-treatment milling through a 14 mm sieve, while hay was milled to pass a 25 mm sieve.
Twenty-four castrated Matebele male goats, aged between 30 and 35 months, with an average initial average body mass of 38.7 kg (± 3.12), were used in the trial. Animals were randomly allocated to the five treatments, and each treatment had five replications except BP, which had four. At the beginning of the experiment, goats were dewormed using 8 mL Systemex liquid (active ingredient oxfendazole 2.265% m/v) (Cooper, Zimbabwe) and vaccinated against pulpy kidney with enterotoxaemia vaccine (Onderstepoort biological product, South Africa). Goats were penned individually in metabolism crates measuring 120 cm long, 54 cm wide and 90 cm high and were 90 cm above the ground. They were fed supplements daily at 07:00, followed by hay, which was fed ad libitum from 10:00. Animals had access to clean drinking water in the afternoon (14:00) for 20 min. The goats were allowed to adapt to the various diets and metabolism crates for 21 days and the subsequent seven days were used for sample collection.
Total output of faeces and urine from each animal were collected at 07:00 from the metabolism crates every morning during the collection period. Faeces were then weighed and 10% of the total faecal matter was bulked and thoroughly mixed for the entire collection period and stored in a freezer (-20 °C), awaiting chemical analyses. A measuring cylinder was used to determine daily urine volume and 20 mL 25% (v/v) sulphuric acid was mixed with urine to prevent nitrogen volatilization (Dube, 2003). Samples from urine, which was 10% of the total output, were bulked over the collection period and stored in a freezer at -20 °C, awaiting analysis. Fruit and hay intake for each day during the collection period were determined by subtracting the mass of the fruit and hay refusals from the mass offered before putting out feed for the next day.
Dry matter (DM) and organic matter (OM) for feeds and faecal matter were determined as described by the Association of Official Analytical Chemists (AOAC, 1999). Total nitrogen (N) in all samples was determined by the micro-Kjeldahl method (AOAC, 1999). Neutral detergent fibre (NDF) and acid detergent fibre (ADF) were analysed as outlined by (Van Soest et al., 1991). The apparent digestibility of DM, OM, NDF and ADF was calculated. The difference between N intake and N output in faeces and urine was a measure of N retention.
A one-way analysis of variance was done using the Genstat Statistical Package (Lawes Agricultural Trust, 2002) and means were separated using least significant difference (LSD) statistic. Initial weight of the goats was used as a covariate in testing the effect of the treatments on the digestibility of DM, OM, ADF and NDF and N-retention. The model used in the analysis of variance was:
Yijk = µ + Ti + (ßj )+ eijk; where:
Yijk = response variable of interest (nutrient intake, digestibility and nitrogen balance)
µ = overall mean common to all observations
Ti = fixed effect of the ith detannification treatment (i = 1, 2...4)
Bj = fixed effect of the individual goat induction body mass used as a covariate
eijk = random residual error.
Results
The Vanillin-HCL method showed the presence of proanthoscynadins (PA) in untreated A. nilotica (4.39% catechin equivalents). Hay had the highest fibre (NDF and ADF) and lowest nitrogen content compared with the various treated A. nilotica fruits (Table 1).
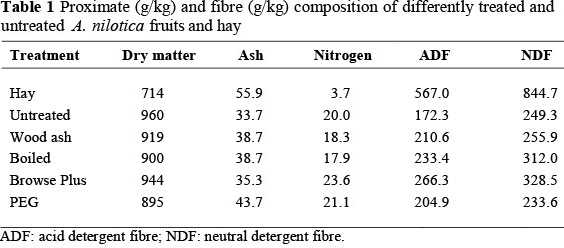
The quantity of hay and fruits supplements consumed by the goats was not different (P >0.05) between treatment groups (Table 2). Apparent digestibility coefficients of DM, OM, ADF and NDF were not different among treatments (P >0.05) (Table 2). The nitrogen content of faeces from goats consuming A. nilotica fruit subjected to the different detannification treatments was similar (P >0.05) (Table 2). While the nitrogen retention for goats that were subjected to A. nilotica fruits treated with WA, BP and PEG was similar (P >0.05). However, they were all higher than their counterparts that were given A. nilotica fruits subjected to boiling water (Table 2).
Discussion
Several studies have contended that it is possible to deactivate condensed tannins and increase the intake and digestibility of forage. For example, treating Leucaena leucocephala forage with BP was noted to increase its intake by 32% when fed to sheep (Majuva-Masafu & Linington, 2006). Treating Lotus pedunculatus with PEG was shown to deactivate the tannins and increase the intake of forage by sheep (Barry & Duncan, 1984). Mlambo et al. (2004) reported an increase in intake by over 50% when PEG was used to treat tannins in Dichrostachys cineria fruits. These studies are in contrast with findings in the present study, suggesting that tannins found in A. nilotica fruits did not respond to any of these treatments. A no response was also reported by Mlambo (2002) when he compared PEG-treated and alkali-treated fruits with untreated in an in-vitro fermentation experiment. He concluded that A. nilotica fruits contain less condensed tannins and more hydrolysable tannins and low molecular weight soluble phenolics, hence tannin deactivation methods were not effective. The standard error of deviation for supplement intake was high. This could be owing to the low intake of the boiled fruits, although not significant. The researchers suspect that boiling the fruits could have made them less palatable.
Nitrogen excretion is shifted from urine to faeces in feeds that are rich in phenolic compounds (Reed & Soller, 1987). A large proportion of N excreted by goats was in faeces, except for the goats fed fruits treated with boiling water where there was more N in urine (Table 2). Treating fruits with hot water could have resulted in an increased urinary N output in animals on boiled treatment, which led to a negative N retention. Although the results were not significant, negative retention could be a result of inadequate fermentable energy in the rumen, leading to an increase in urinary N (Smith et al., 2005). Negative N retention was also observed when goats were offered 200 g/day of untreated A. nilotica (Sikosana et al., 2002). These findings corroborate the boiled treatment in our study.
There are variations in types and quantities of tannins among forages (Madibela et al., 2006). Different tannins act differently, affecting the intake and digestibility of the forage (Silanikove et al., 2001). Lotus corniculatus and L. pedunculatus have been reported to have the same level of condensed tannin, but ruminants respond differently to them, perhaps because their tannins are chemically different (Min et al., 2003). In our study the tannins in A. nilotica did not respond to any of these treatments. There are studies that have reported that A. nilotica does contain condensed tannins, such as Tanner et al. (1990), Phale & Madibela (2006) and Nsahlai et al. (2011). In contrast to Phale & Madibela (2006) and Tanner et al. (1990) the researchers support the findings by Ayoub (1985) and Mueller-Harvey (2001) that A. nilotica contains hydrolysable tannins (catechin galletes) instead of condensed tannins. A study by Cooper & Owen-Smith (1985) confirmed that A. nilotica contains fewer condensed tannins, but more hydrolysable tannins that inactivate digestive enzymes. However, they pointed out that the effects of hydrolysable tannins are more pronounced on insect herbivores than in animals. The Vanillin-HCL method, which was used in this study to detect tannins, fails to distinguish between hydrolysable and condensed tannins (Mueller-Harvey, 2001). Zucker (1983) suggested that the role of hydrolysable tannins is to inactivate the digestive enzymes of herbivores. Little attention has been given to the way these hydrolysable tannins can be counteracted in ruminants.
Not all forages containing high tannin cause detrimental effects on animals: A. seyal was reported to increase growth rate in sheep (Reed et al., 1990) and A. brevispica was found to cause optimum N retention in sheep and goats (Woodward & Reed, 1997), despite their high tannin content.
Conclusion
Detannification of A. nilotica fruits using different methods did not improve intake and digestibility by goats. Treatment by boiling fruits led to negative nitrogen retention in goats. It is recommended that each detannification method should be tested on different types of tree species and on tannins at different levels before totally dismissing the potential of the treatments.
Acknowledgements
The authors are grateful to the Matopos Research Institute for the animals and facilities that were used in this study. The study was part of a funded project under the Livestock Production Programme (LPP) by the Department for International Development (DFID) (UK). The views expressed in this manuscript are not those of the sponsors.
References
AOAC, 1999. Official Methods of Analysis of AOAC International. Gaithersburg: AOAC International. [ Links ]
Ayoub, S.M.H., 1985. Flavanol molluscicides from the Sudan Acacias. Int. J. Crude Drug Res. 23, 87-90. [ Links ]
Barry, T.N. & Duncan, S.J., 1984. The role of condensed tannins in the nutritional value of Lotus pedunculatus for sheep. Part 1. Voluntary intake. Br. J. Nutr. 51, 485-491. [ Links ]
Broadhurst, R.B. & Jones, W.T., 1978. Analysis of condensed tannins using Acidified Vanillin. J. Sci. Food Agr. 29, 788-794. [ Links ]
Burns, R.E., 1971. Methods of evaluation of tannins in sorghum. Agron. J. 63, 511-512. [ Links ]
Cooper, S.M. & Owen-Smith, N., 1985. Condensed tannins deter feeding by browsing ruminants in a South African savanna. Oecologia 67, 142-146. [ Links ]
Dube, J.S., 2003. Variations in proanthocyanidin levels of provenances of six Acacia species and the Influence of proanthocyanidins on ruminant nutrition. PhD thesis. University of Zimbabwe, Harare, Zimbabwe. [ Links ]
Getachew, G., Makkar, H.P.S. & Becker, K., 1998. Tannin-binding agents to alleviate anti-microbial effects of tannins in the rumen. In: Proceedings of the Third Tannin Conference on Plant Polyphenols: Chemistry and Biology, Bend, OR, USA. [ Links ]
Hanovice-Ziony, M., Gollop, N., Landau, S.Y., Ungar, E.D., Muklada, H., Glasser, T.A., Perevolotsky, A. & Walker, J.W., 2010. No major role for binding by salivary proteins as a defense against dietary tannins in Mediterranean goats. J. Chem. Ecol. 36, 736-743. [ Links ]
Hove, L., Topps, J.H., Sibanda, S. & Ndlovu, L.R., 2001. Nutrient intake and utilisation by goats fed dried leaves of the shrub legumes Acacia angustissima, Calliandra calothyrsus and Leucaena leucocephala as supplements to native pasture hay. Anim. Feed Sci. Technol. 91, 95-106. [ Links ]
Kaitho, R.J., Umunna, N.N., Nsahlai, I.V., Tamminga, S. & Van Bruchem, J., 1997. Utilization of browse supplements with varying tannin levels by Ethiopian Menz sheep: 1. Intake, digestibility and live weight changes. Agrofor. Syst. 39, 145-159. [ Links ]
Kindness, H., Sikosana, J.L.N., Mlambo, V. & Morton, J.F., 1999. Socio-economic surveys of goat keeping in Matobo and Bubi districts. UK Natural Resources Institute. [ Links ]
Lawes Agricultural Trust 2002. Genstat version 6.1.0.205. Lawes Agricultural Trust (Rothamsted Experimental Station). Hertfordshire, England. [ Links ]
Madibela, O.R., Seitshiro, O. & Mochankana, M.E., 2006. Deactivation effects of polyethylene glycol (PEG) on in vitro dry matter digestibility of Colophospermum mopane (Mophane) and Acacia browse trees in Botswana. Pak. J. Nutr. 5, 343-347. [ Links ]
Majuva-Masafu, M.M. & Linington, M.J., 2006. The effect of Browse PlusTM on nutrient intake, rumen pH and digestibility of a sole diet of Leucaena leucocephala forage. Afr. J. Range Forage Sci. 23, 171-175. [ Links ]
Makkar, H.P.S., 2003. Effects and fate of tannins in ruminant animals, adaptation to tannins, and strategies to overcome detrimental effects of feeding tannin-rich feeds. Small Rumin. Res. 49, 241-256. [ Links ]
Min, B.R., Barry, T.N., Attwood, G.T. & McNabb, W.C., 2003. The effect of condensed tannins on the nutrition and health of ruminants fed fresh temperate forages: a review. Anim. Feed Sci. Technol. 106, 3-19. [ Links ]
Mlambo, V. 2002. Modifying the nutritional effects of tannins present in Acacia and other tree fruits offered as protein supplements to goats in Zimbabwe. PhD thesis, University of Reading, UK. [ Links ]
Mlambo, V., Smith, T., Owen, E., Mould, F.L., Sikosana, J.L.N. & Mueller-Harvey, I., 2004. Tanniniferous Dichrostachys cinerea fruits do not require detoxification for goat nutrition: in sacco and in vivo evaluations. Livest. Prod. Sci. 90, 135-144. [ Links ]
Mlambo, V., Sikosana, J.L.N., Mould, F.L., Smith, T., Owen, E. & Mueller-Harvey, I., 2007. The effectiveness of adapted rumen fluid versus PEG to ferment tannin-containing substrates in vitro. Anim. Feed Sci. Technol. 136, 128-136. [ Links ]
Mueller-Harvey, I., 2001. Analysis of hydrolysable tannins. Anim. Feed Sci.Technol. 11, 3-20. [ Links ]
Mueller-Harvey, I. & Mcallan, A.B., 1992. Tannins. Their biochemistry and nutritional properties. Adv. Plant Cell Biochem. Biotechnol. 1, 151-217. [ Links ]
Mukuru, S.Z., Butler, L.G., Rogler, J.C., Kireis, A.W., Ejeta, G., Axtell, J.D. & Mertz, E.T., 1992. Traditional Processing of high tannin sorghum grain in Uganda and its effect on tannin, protein in digestibility and rat growth. J. Sci. Food Agric. 40, 1172-1175. [ Links ]
Ngwa, A.T., Nsahlai, I.V. & Iji, P.A., 2003. Effect of feeding legume pods or alfalfa in combination with poor quality grass straw on microbial enzyme activity and production of VFA in the rumen of South African Merino sheep. Small Rumin. Res. 2003, 83-94. [ Links ]
Nsahlai, I.V., Umunna, N.N. & Osuji, P.O. 1999. Influence of feeding sheep on oilseed cake following the consumption of tanniferous feeds. Livest. Prod. Sci. 60, 59-69. [ Links ]
Nsahlai, I.V., Fon, F.N. & Basha, N.A.D., 2011. The effect of tannin with and without polyethylene glycol on in vitro gas production and microbial enzyme activity. S. Afr. J. Anim. Sci. 41, 337-344. [ Links ]
Phale, O. & Madibela, O.R., 2006. Concentration of soluble condensed tannins and neutral detergent fibre-bound tannins in fodder trees and forage crops in Botswana. J. Biol. Sci. 6, 320-323. [ Links ]
Priolo, M., Bella, M., Lanza, V., Galofaro, L., Biondi, D., Barbagallo, H., Ben Salem & Pennisi, P., 2005. Carcass and meat quality of lambs fed fresh sulla (Hedysarum coronarium L.) with or without polyethylene glycol or concentrate. Small Rumin. Res. 59, 281-288. [ Links ]
Reed, J.D., 1995. Nutritional toxicology of tannins and related polyphenols in forage legumes. J. Anim. Sci. 73, 1516-1528. [ Links ]
Reed, J.D. & Soller, H., 1987.Phenolics and nitrogen utilization in sheep fed browse. In: Herbivore Nutrition Research, 2nd International Symposium on the Nutrition of Herbivores. Ed: Rose, M., University of Queensland, Brisbane. pp. 47-48. [ Links ]
Reed, J.D., Soller, H. & Woodward, A., 1990. Fodder tree and straw diets for sheep: Intake, growth, digestibility and the effects of phenolics on nitrogen utilization. Anim. Feed Sci. Technol. 30, 39-50. [ Links ]
Salem, A.Z.M., López, S., Ranilla, M.J. & Gonzalez, J.S., 2013. Short- to medium-term effects of consumption of quebracho tannins on saliva production and composition in sheep and goats. J. Anim. Sci. 91, 1341-1349. [ Links ]
Shimada, T. 2006. Salivary proteins as defense against dietary tannins. J. Chem. Ecol. 32, 1149-1163. [ Links ]
Sikosana, J.L.N., Smith, T., Mlambo, V., Owen, E., Mueller-Harvey, I. & Mould, F., 2002. Acacia and other tree fruits as dry season feed supplements for goats. Natural Resources International Ltd. Aylesford, Kent, UK, pp. 69-75. [ Links ]
Silanikove, N., Gilboa, N. & Ntsan, Z., 1997. Interactions among tannins, supplementation and polyethylene glycol in goats given oak leaves: Effects on digestion and food intake. Anim. Sci. 64, 479-484. [ Links ]
Silanikove, N., Perevolotsky, A. & Provenza, F.D., 2001. Use of tannin- binding chemicals to assay for tannins and their negative postingestive effects in ruminants. Anim. Feed Sci.Technol. 91, 69-81. [ Links ]
Smith, T., Mlambo, V., Sikosana, J.L.N., Maphosa, V., Mueller-Harvey, I. & Owen, E., 2005. Dichrostachys cinerea and Acacia nilotica fruits as dry season feed supplements for goats in a semi-arid environment: Summary of a DFID funded project in Zimbabwe. Anim. Feed Sci. Technol. 122, 149-157. [ Links ]
Tanner, J.C., Reed, J.D. & Owen, E., 1990. The nutritive value of fruits (pods with seeds) from Acacia spp., compared with extracted noug (Guizotia Abyssinica) meals as supplements to maize stover for Ethiopian highland sheep. Anim. Prod. 51, 127-133. [ Links ]
Timberlake, J., Fagg, C. & Barnes, R., 1999. Field Guide to the Acacias of Zimbabwe. Harare, Zimbabwe: CBC Publishing Associates. [ Links ]
Van Soest, P.J., Robertson, J.B. & Lewis, B.A., 1991. Methods for dietary fiber, and nonstarch polysaccharides in relation to animal nutrition. J. Dairy Sci. 74, 3583-3597. [ Links ]
Waghorn, G.C., 1990. Beneficial effects of low concentration of condensed tannins in forages fed to ruminants. Proc. Aust. Soc. Anim. Prod. 18, 412-415. [ Links ]
Ward, H.K., Richardson, F.D., Denney, R.P. & Dye, P.J.W., 1979. Matopos Research Station: a perspective. Rhodesia Agr. J. 76, 5-18. [ Links ]
Woodward, A. & Reed, J.D., 1997. Nitrogen metabolism of sheep and goats consuming Acacia brevispica and Sesbania sesban. J. Anim. Sci. 75, 1130-1139. [ Links ]
Zucker, W.V., 1983. Tannins: Does structure determine function? An ecological perspective. Am. Nat. 121, 335-365. [ Links ]
Received 19 April 2012
Accepted 28 June 2013
First published online 18 December 2013
# Corresponding author: tshabalala@gmail.com














