Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Animal Science
On-line version ISSN 2221-4062
Print version ISSN 0375-1589
S. Afr. j. anim. sci. vol.42 n.2 Pretoria Jan. 2012
W.J. Steyn; N.H. Casey; C. Jansen van Rensburg#
Department of Animal and Wildlife sciences, Faculty of Natural and Agricultural Science University of Pretoria, South Africa 0002
ABSTRACT
The study tested the performance of intact male pigs from a selected genetic line subjected to differing feeding regimens and penning conditions. The trial was a 2 x 3 x 2 x 3 factorial design, consisting of winter and summer periods, three sire lines, two diets and three feeding regimens. The pigs were intact males grown over three phases, starter (25 to 50 kg); grower (51 to 80 kg) and finisher (81 to 105 kg).The pigs were randomly allocated to three feeding regimens, a controlled single feeding, ad libitum single feeding and ad libitum group feeding, with six animals per ad libitum group. This resulted in 96 pigs in six treatments with six replicates. The diets were high (HF) and low (LF) nutrient dense feeds, where the LF was 95% of the HF. Season affected growth; the winter animals had a significantly greater growth response, end-mass and average daily gain (ADG). The HF diet resulted in significant improved ADG, feed conversion ratio and protein deposition rate, especially during summer. However, end-mass, ADG and average protein deposition rates of controlled-fed pigs were significantly lower compared to the ad libitum group and single-fed animals. The hypothesis was affirmed that high-performing intact male pigs are sensitive to and affected by feeding regimens and penning conditions that will affect their production.
Keywords: Protein and lipid deposition, growth performance, ad libitum versus restricted feeding
Introduction
Modern pig production units aim to achieve high daily growth rates with minimal feeding requirements in order to reach specific target slaughter mass. Pigs have been intensively selected for various growth traits, i.e. high lean tissue deposition, resulting in pigs with higher maximum protein retention than the unimproved strains. To determine the amino acid requirements accurate estimates of whole body protein deposition is required. The rate of maximum protein deposition will determine the pigs' nutrient requirements for growth and its response to nutrient or management changes.
Pigs are widely recognised as social animals and physical closeness and interaction are important. However, the production environment and intense social interactions become stressors that can affect the animals' production performances. Pigs raised in commercial conditions are normally penned in groups, whereas in experimental studies the animals are frequently penned individually. Competition at the feeder, social facilitation and social stress are factors that may be responsible for the differences in feeding behaviour and production parameters between group-penned and individual-penned pigs. The social environment in group-penned pigs could affect feed intakes and consequently the production parameters of growing pigs compared to those penned individually. Growth performance is usually greater when pigs are penned individually than when they are penned in groups (Gonyou et al., 1992; Hacker et al., 1994; Bornett et al., 2000). In addition, group-penned pigs modify their feeding behaviour by eating less frequently, but consume more food once-off at a faster rate than pigs penned individually (De Haer & Merks, 1992).
Voluntary feed intakes (VFI) of pigs determine nutrient intake levels that impact on the efficiency of pork production. A simulation study indicated that the highest returns per pig per year were achieved when
VFI was just sufficient to meet requirements for maximum protein deposition (Kanis, 1995). The optimal feed intake minimises feed conversion ratio (FCR) and maximises lean meat growth. Voluntary feed intake is variable since environmental factors such as hot temperatures can affect it (Hyun et al, 1998). Depressed growth in summer may be due to the redirection of energy for maintenance requirements (Kouba et al., 2001) and/or a decline in VFI (Rinaldo et al., 2000). Animals in warm conditions have increased physical activities, such as respiratory hyperventilation, which are consistent with additional energy costs and higher maintenance requirements. Gous (2007) suggested that animals in a cooler environment can overconsume a marginally deficient feed and would therefore perform better than would animals in a hotter environment.
The hypothesis tested was that high-performing intact male pigs are significantly sensitive to and affected by feeding regimens and penning conditions that will affect their production negatively in an intensive production system located at 1 059 m altitude in a subtropical climatic region of South Africa.
The hypothesis was tested under three conditions, namely the effect of a high protein and energy diet on pigs' growth performances and carcass parameters when fed during winter and summer, the animals' performances under controlled versus ad libitum feeding with the composition of the feed having been optimised according to a prescribed growth model for maximum protein deposition and optimum feed conversion ratio, and group versus individually penned pigs.
Materials and Methods
The experiment was performed in a 2 x 3 x 2 x 3 factorial design, consisting of two periods, three sire lines, two diets and three feeding regimens. The periods were winter (Period 1), 6 June to 13 August, and summer (Period 2), 3 October to 10 December in the southern hemisphere.
The animals were male progeny born from 30 Topigs-40 sows that had been randomly inseminated with semen from three selected Topigs Tempo AI boars with performance indices >100, each representing a sire line. The pigs came from a high health unit with specific pathogen free (SPF) housing conditions. After 11 weeks, 32 boars per sire line were randomly selected (no ranking or other selection criteria was applied) to make provision for each period of the trial. They were taken to a SPF unit at the Hatfield Research Farm of the University of Pretoria, tagged and the mass recorded. Animals were allocated to either single-pens (3.5 m2 of area per pig) or group-pens (1.2 m2 of area per pig) according to their starting mass and particular sire line. Animals were handled only once a week for recording their mass and P2 fat measurements.
The animals were randomly allocated to three feeding regimens, namely controlled single feeding (CSF), ad libitum single feeding (ASF) and ad libitum group feeding (AGF). An ad libitum group comprised six animals. This made provision for six different treatments with six replicates. A total of 96 weaner pigs were randomly divided into the six treatments.
The diets were of high (HF) and low (LF) nutrient density. The LF diet was formulated at 95% of the digestible energy (DE), crude protein and amino acid levels of the HF diet. These were formulated using a matrix-type programme, Format International® (London, UK). Two different diets were formulated for each of the starter, grower and finisher phases. The ratio of energy to protein of the two diets was kept closely aligned.
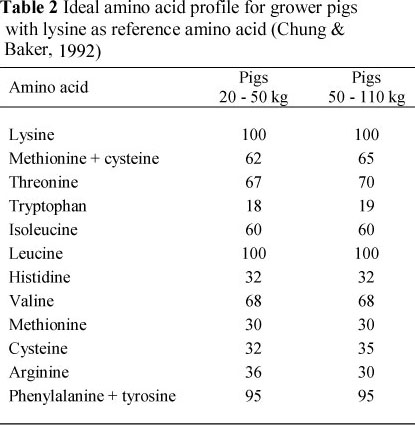
The digestible lysine requirements for the three phases were specifically recommended for the Tempo sire line by the Institute of Pig Genetics (IPG) (Beuningen, The Netherlands) (Table 1). The specifications of the other amino acids were derived by using the lysine requirements as reference amino acid and the amino acid profile proposed by Chung & Baker (1992) (Table 2). Amino acid requirements were expressed as apparent ileal digestible values. Diets were based on maize and sunflower/soya oilcake meal. Formulated nutrient specifications are shown in Table 3 and raw material composition in Table 4.
The controlled feed intake was calculated and adjusted every week according to the individual's mass in terms of the recommendations of the IPG model (Table 1). Feeding the production phase rations were according to mass: starter (25 kg to 50 kg); grower (51 kg to 80 kg) and finisher (81 kg to 105 kg).
Feed intake (FI) (kg) was recorded weekly as the difference between the total that had been added to the feeders and the orts. Average daily gain (ADG) was calculated from weekly mass gains. Feed conversation was determined as the ratio of FI/ADG.
Fat measurements were taken every week from 15 weeks of age to the end of the trial using the Renco fat-o-meter. Four measuring points were used to measure back fat thickness. Measuring points (1, 2, 3 and 4) are all of equal distance, 5 cm left of the spinal cord stretching from the shoulder (last point of the scapula) to the last rib. The Institute of Pig Genetics (IPG) calculated the protein and lipid deposition rates using a computerised model based on the principles of De Greef et al. (1994). Metabolisable energy (ME) intake was calculated as 0.96 times the measured digestible energy intake (Noblet & Henry, 1993). Energy retained in protein and lipid in the body were calculated by using average values for the energy content of protein as 23.7 MJ/kg and for lipids as 39.3 MJ/kg. Subsequently, the efficiency of ME utilisation was calculated for protein and lipid retention, respectively.
The animals were slaughtered at the completion of each period at a commercial abattoir. Hot and cold carcass mass, dressing percentage (DP) and lean meat percentage (LMP) of each pig were ascertained. Lean meat percentage was determined by measuring fat thickness and eye muscle thickness with a Hennessy Grading Probe on a hanging carcass between the second and third last ribs, 45 mm from the mid - back line.
The LMP was calculated as follows: LMP = 72.5114 - (0.4618 x fat thickness) + (0.057 x eye muscle thickness) (Visser, 2004).
Ambient minimum and maximum temperatures (°C) were recorded daily. The average minimum and maximum and standard deviation for the entire winter (Period 1) and summer (Period 2) were calculated.
Dry matter, ash, moisture, amino acids, crude fat (ether extract), crude fibre and crude protein were obtained according to AOAC (2000) procedures. Crude protein was determined by the Dumas method and amino acids by ultra-performance liquid chromatography (UPLC) Amino Acid Analysis Solution (Waters Corporation, Milford, Massachusetts). Crude fibre and neutral detergent fibre (NDF) were measured with the Fibre-Tech apparatus (Robertson & Van Soest, 1981) and acid detergent fibre (ADF) as described by Goering & Van Soest (1988). Gross energy content of the feed was determined with the MC-1000 Modular Bomb Calorimeter and starch according to the AOAC (1984) procedure. Calcium analysis was done by using the Perkin Elmer Atomic Spectrophotometer-2380 (Giron, 1973). Phosphorus analysis was done with the Spekol 1300 apparatus using the spectrophotometric method (AOAC, 2000).
The SAS (2009)®General Linear Models (GLM) procedure was applied to determine the significance of differences between groups, sire lines, diets, treatments, combinations of interactions over time and growth and carcass parameters. Least square means (LSM) and standard deviations (SD) were calculated for different groups, diets, treatments and interactions. Significance of difference (5%) between LSM was determined using the Fischer's test (Samuels, 1989). Starting mass was included as a covariant in the growth parameter analysis and was only significant in the final mass.
The statistical model used to analyse the data:
Yijk = μ + Ri + Fj + Pk + RFij + RPik +FPjk + RFPjk + e,k
Where μ = population mean of the appropriate trait;
Ri = effect of the ith feed regimen;
Fj = effect of the jth feed treatment;
Pk = effect of the kth period;
RFij = effect of the interaction of the ith feed regimen and jth feed treatment;
RPik = effect of the interaction of the ith feed regimen and the kth period;
FPjk = effect of the interaction of the jth feed treatment and the kth period;
RFPijk = effect of the interaction of the ith feed regimen, jth feed treatment and the kth period; eijk = random effects
The trial conformed to the requirements of the Animal Use and Care Committee of the University of Pretoria, reference number EC080125-003.
Results
No significant differences (P >0.05) were found between sire lines. These were then excluded from the analysis.
The average minimum and maximum temperatures (°C) and (SD) for the entire winter (Period 1) were 18 (1.8) and 24.2 (0.72) and summer (Period 2) 22 (0.7) and 30.5 (1.88), respectively.
Production parameters differed between periods (P <0.05) (Table 5). The data were corrected for starting mass and starting P2 values and differences (P <0.05) that occurred were due to season. Period had significant (P <0.05) effects on end mass, ADG, end P2 and protein deposition. Energy was used more efficiently (P <0.05) for lipid retention during summer than during the winter period (Table 5).
Pigs that received the HF feeds had a higher growth response in terms of end mass, ADG, feed FCR and average protein deposition rate than the pigs on the LF feeds (P <0.05) (Table 6).
The AGF pigs had lower end masses, ADG and feed intakes than ASF pigs (P <0.05) (Table 7). End mass, ADG and FI were corrected for season effects and dietary treatment. Although AGF pigs had a lower performance compared to ASF pigs, AGF pigs had a (P <0.05) better LMP (P <0.05) and there were no differences in FCR between these two regimens (P >0.05). The CSF pigs had lower end masses, ADG, feed intakes, end P2, average protein and lipid deposition rates and DP compared to ASF pigs (P <0.05). The CSF pigs also had better FCR, lean meat LMP and more efficient energy utilisation for protein retention compared to ASF pigs (P <0.05).
Discussion
The effect of high ambient temperature on the growth performance in pigs is well documented in the literature. The growth performance of pigs in tropical conditions is often regarded as low (Egbunike, 1986). Numerous studies have shown the negative effect of increasing temperature on VFI, daily mass gain and the retention of fat and energy (Quiniou et al, 2000; Rinaldo et al, 2000). The optimum temperature range for finisher pigs is between 10 °C and 23.9 °C (Myer & Bucklin, 2001). Temperature above 23.9 °C has negative effects on the animals' growth performances (Kouba et al., 2001). The high air temperature and relative high humidity during summer are characteristics of the tropical climate of South Africa.
The trial conducted over different seasons resulted in the winter animals having a greater growth response compared to the summer animals with their end mass and ADG being higher (P <0.05). Feed intake is the limiting factor influencing growth rate in tropical areas. Lower FI contributes to lower ADG and a decline in energy available for tissue deposition resulting in lower productivity. During the summer the animals did not have a lower FI compared to the winter period (P >0.05). Interestingly, this was inconsistent with data from various researchers (Nienaber & Hahn, 1983; Rinaldo & Le Dividich, 1991) who reported significant lower FI during the warmer seasons. Rinaldo et al. (2000) tested the effects of the tropical climate on VFI and performance of growing pigs. No depressive effects during the hot season on FCR were found, but there was a reduction in ADG and FI. The reduction in ADG was mainly attributed to a decline in feed consumption. First, this discrepancy may be due to the lower rate in the reduction of feed consumption during summer that could be related to the favourable night time temperature. The minimum temperatures during summer were within the range of thermo neutral temperatures of 20 - 22 °C. Second, the parental lines used for the cross-bred progeny were the Topigs 40 and Tempo. Both these lines are known for their adaptability and efficiency in the tropical conditions and were bred and selected for high VFI under high environmental temperatures. Body fatness can also influence how pigs react towards ambient temperature as leaner pigs are less insulated and less sensitive to high temperatures (Rinaldo et al., 2000).
The inferior growth performance of animals in summer may be due to less energy being directed towards lean tissue growth (P <0.05) and more energy towards lipid growth, that contributes less to weight gain than lean tissue. In fact, the summer-grown pigs showed a higher efficiency of energy utilisation for lipid retention (P <0.05), possibly because less energy was used for heat production to maintain body temperature (Rinaldo & Le Dividich, 1991).
Much research focused on the influence of energy concentration of the feed on VFI, growth performance and carcass characteristics. For this trial, dietary fibre was used to decrease the DE content of the low energy diets, resulting in more bulky feeds. Within a certain range of variation, dietary fibre content has no effect on growth performance provided energy density is adequate (Baird et al., 1975). Therefore, if the energy level is adequate, the pig can tolerate quite wide ranges of fibre in the diet. The effects of dietary fibre associated with bulkiness, i.e. gut fill, on retention time and nutrient availability are well documented (Shriver et al., 2003; Wilfart et al., 2007). Despite some negative impacts, farmers in tropical areas use fibrous crop by-products and forages extensively as alternatives to expensive cereals in pig diets. Although the use of fibrous ingredients in pig diets may not always be efficient in terms of an animal's performance, the economical asset of the operation is mostly to the producer's advantage (Ogle, 2006).
Beaulieu et al. (2009) showed how changes in DE concentration achieved through graded changes in diet composition affected the performance and carcass composition of growing pigs. An improvement (P <0.05) in ADG with increased DE was observed. Voluntary feed intake decreased (P <0.05) whereas FCR and daily energy intake improved (P <0.05) with increased DE content. In a commercial pig farm, the overall ADG remained unaffected by DE content although an improvement in growth up to 80 kg mass was observed with higher energy concentrations. These results supported previous studies where it was reported that the capacity of a pig for growth exceeded its ability to consume sufficient energy between 20 kg and 50 kg. In heavier animals, though, energy intake is not a limiting factor for growth (Campbell & Dunkin, 1990). Furthermore, dietary fibre is better digested by more mature pigs and therefore the energy contribution of dietary fibre increases as the animal matures (Noblet & Le Goff, 2001). It was therefore suggested to feed grower/finisher pigs a higher energy dense diet during the early stages of growth (20 kg to 50 kg) and less dense diets during the later stages (50 kg to 105 kg).
Results of this study showed an improvement in ADG, FCR and protein deposition rate (P <0.05) when animals were fed a higher energy content in their diet, despite similar feed intake values (P >0.05) between the two groups. Gut fill and subsequently DE intake might have been limiting factors for growth performance. Higher heat increment of fibrous material further decreased the available (net) energy of the low energy diets (Noblet & Le Goff, 2001). More energy was available for the animals that received the high energy diets, which resulted in faster protein deposition rates. Faster lean tissue growth improved FCR because of the higher efficiency of energy utilisation compared to that of lipid growth. The faster absolute growth rate resulted in heavier carcasses which improved FCR by offsetting the fixed cost of maintenance.
This is indicative of the animals' capabilities of utilising the additional energy available. Increased DE content improved animal performances, but not necessarily delivered the best economic results. The response to different DE concentrations is not easy to predict. Changes in energy concentration inevitably lead to changes in ingredients, making it difficult to distinguish ingredient effects from energy effects.
The increased concentration of pig herds, improved genetics and environmental constraints necessitated new feeding strategies for growing pigs. It is important to determine precisely the energy value of feeds and also to know the actual VFI curves for adapting feed supply to energy requirements of animals. The goal is that a pig should daily consume only enough nutrients for maximum growth potential or maximum lean deposition rate as established by its genotype (Knabe, 1996). Under these conditions optimal pig growth performance and nitrogen excretion will be achieved. Feeding different DE levels for various seasons of the year as well as for different age/mass groups and for a specific gender and genotype is an appropriate tool to optimise feed utilisation of grower/finisher pigs.
A growth model developed by IPG was used to determine the specific maintenance and production requirements for lysine for this line of grower pigs. The model is a mathematical model designed to accurately quantify the daily nutrient requirements of pigs based on inputs that affect performance. The goal was to improve the efficiency of lean pork production by deriving protein deposition and lipid deposition rates and by integrating current knowledge of genetic potential of specific lines and their nutrient intake levels. Feeding the diets according to the exact nutrient requirements of the specific genetic line and the specific daily feed allowance were determined, i.e. the optimal feed intake curve, which was then applied. Various factors throughout the trial could have had negative influences on the controlled FI animals' growth performances, i.e. genetic capabilities, health status, tropic environment, raw materials used and effects of different feeding patterns. CSF animals were very efficient in converting feed into tissue. However, overall growth performances were poor. The mean LMP of carcasses were high and lipid deposition rates were low, but no significant differences were observed between controlled FI and AGF animals. The controlled FI animals showed the best FCR (P <0.05) of the treatment groups, probably because of the favourable lean meat : lipid deposition. End mass, ADG and average protein deposition rates were lower compared to the AGF and ASF animals (P <0.05).
In grower/finisher research programmes most animals are tested in individual housing systems, whereas commercially grown pigs are kept in groups. De Haer & De Vries (1993) reported that this had a significant influence on growth rate, backfat thickness and most FI traits. The lower growth rate and backfat thickness in group pens are a result of lower VFI, lower digestibility and also a higher level of activity due to social interaction. Performance results from this trial confirmed this with differences shown in performance between group-penned and individual-penned pigs (P <0.05). ASF pigs showed a higher growth rate and final mass (P <0.05). No significant differences were observed between backfat thickness and FCR between the group and individual-penned animals.
The results show that controlled FI amounted to a restricted FI in that the animals were not afforded the opportunity to eat to satisfy their inherent needs, irrespective of the environmental effects. The CSF animals had slower growth rates, higher fat deposition rates and lower protein deposition rates. It can be concluded that the optimal feeding curve predicted for the specific genotype using the prescribed model was not necessarily the optimal feed curve for the type of pig, namely intact males of a genotype developed for lean meat growth and feed efficiency, used in the trial. Results indicated a deficiency in nutrient supply due to insufficient feed allowance. If an animal has a protein and energy deficiency the correction of either one usually assists the other (Ferguson & Theeruth, 2002). Various authors indicated that an animal's attempt to overcome protein deficiency will result in an increased VFI and a consequential increase in lipid deposition due to the over-consumption of energy (Ferguson & Gous, 1997; Ferguson et al., 2000).
Conclusion
The hypothesis was affirmed that high-performing intact male pigs are significantly sensitive to and affected by season, feeding regimen and penning conditions. Feeding of nutrient-dense diets during summer improved pig performance. Individual-penned pigs showed higher feed intakes, growth rates and final mass than the group-penned animals. Controlled feeding of the intact male pig according to a growth model developed by IPG in the Netherlands, resulted in slower growth rates, higher fat deposition rates and lower protein deposition rates. It can therefore be concluded that the optimal feed curve for pigs of the genotype used in this trial and in typical subtropical environmental conditions, differed from the prescribed model. Controlled feeding, however, resulted in an improvement in FCR which could mean that the IPG model may deliver better economic results.
Acknowledgements
The authors wish to acknowledge R.J. Coertze for his technical assistance, Topigs SA for assistance with acquiring the animals and the University of Pretoria for collateral support.
References
AOAC, 1984. Official method of analysis (14thed) Volume I. Association of Official Analytical Chemists, Inc. Maryland, USA. [ Links ]
AOAC, 2000. Official method of analysis (17thed) Volume I. Association of Official Analytical Chemists, Inc. Maryland, USA. [ Links ]
Baird, D.M., McCampbell, H.C. & Allison, J.R., 1975. Effects of levels of crude fibre, protein and bulk in the diets for finishing hogs. J. Anim. Sci. 41, 1039-1047. [ Links ]
Beaulieu, A.D., Williams, N.H. & Patience, J.F., 2009. Response to dietary digestible energy concentration in growing pigs fed cereal grain-based diets. J. Anim. Sci. 87, 965-976. [ Links ]
Bornett, H.L.I., Morgan, C.A., Lawrence, A.B. & Mann, J., 2000. The effect of group housing on feeding patterns and social behaviour of previously individually housed growing pigs. Appl. Anim. Behav. Sci. 70, 127-141. [ Links ]
Campbell, R.G. & Dunkin, A.C., 1990. Feeding growers and finishers. In: Pig Production in Australia. Eds Gardner, J.A.A., Dunkin, A.D. & Lloyd, L.C., Butterworths, Sydney, Australia. pp. 85-99. [ Links ]
Chung, T.K. & Baker, D.H., 1992. Ideal protein for swine and poultry. BioKyowa Technical Review4, Chesterield, NutriQuest, Inc. [ Links ]
De Greef, K.H., Verstegen, W.A., Kemp, B. & Van der Togt, P.L., 1994. The effect of body weight and energy intake on the composition of deposited tissue in pigs. Anim. Prod. 58, 263-270. [ Links ]
De Haer, L.C.M. & Merks, J.W.M., 1992. Patterns of daily food intake in growing pigs. Anim. Prod. 54, 95-104. [ Links ]
De Haer, L.C.M. & De Vries, A.G., 1993. Feed intake patterns of and feed digestibility in growing pigs housed individually or in groups. Livest. Prod. Sci. 33, 227-292. [ Links ]
Egbunike, G.N., 1986. Response of European pigs to improved management under the humid tropical climatic conditions in Ibadan: A Review. Pig News and Information 7, 315-324. [ Links ]
Ferguson, N.S. & Gous, R.M., 1997. The influence of heat production on voluntary food intake in growing pigs given protein-deficient diets. Anim. Sci. 64, 365-378. [ Links ]
Ferguson, N.S. & Theeruth, B.K., 2002. Protein and lipid deposition rates in growing pigs following a period of excess fattening. S. Afr. J. Anim. Sci. 32, 97. [ Links ]
Ferguson, N.S., Arnold, G.A., Lavers, G. & Gous, R.M., 2000. The response of growing pigs to amino acids as influenced by environmental temperature. 1 Threonine. Anim. Sci. 70, 287-297. [ Links ]
Giron, H.C., 1973. Atomic Absorption Newsletter 12, 28. Perkin Elmer Atomic Spectrophotometer. [ Links ]
Goering, H.K. & Van Soest, P.J., 1988. Forage Fiber Analyses (Apparatus, reagents, procedures and some applications). Agriculture Handbook No. 379. A.R.S., U.S. Dept. of Agric. ADSRI - JAN 1988. [ Links ]
Gonyou, H.W., Chapple, R.P. & Frank, G.R., 1992. Productivity, time budgets and social aspects of eating in pigs penned in groups of five or individually. Appl. Anim. Behav. Sci. 34, 291-301. [ Links ]
Gous, R.M., 2007. Predicting nutrient responses in poultry: Future challenges. Animal 1, 57-65. [ Links ]
Hacker, R.R., Du, Z. & D'arcy, C.J., 1994. Influence of penning type and feeding level on sexual behaviour and feet and leg soundness in boars. J. Anim. Sci. 72, 2531-2537. [ Links ]
Hyun, Y., Ellis, M., Riskowski, G. & Johnson, R.W., 1998. Growth performance of pigs subjected to multiple concurrent environmental stressors. J. Anim. Sci. 76, 721-727. [ Links ]
Kanis, E., 1995. Optimization of growth and body composition in pigs. In: Proc. 2nd Dummerstof Muscle Workshop, Rostock, Germany. pp. 191. [ Links ]
Kouba, M., Hermier, D. & Le Dividich, J., 2001. Influence of a high ambient temperature on lipid metabolism in the growing pig. J. Anim. Sci. 79, 81-87. [ Links ]
Knabe, D.A., 1996. Optimizing the protein nutrition of growing-finishing pigs. Anim. Feed Sci. Technol. 60, 331-341. [ Links ]
Myer, F. & Bucklin, R., 2001. Influence of hot-humid environment on growth performance and reproduction of swine. Website: http://edis.ifas.ufl.edu/AN107 Accessed May 30, 2007. [ Links ]
Nienaber, J.A. & Hahn, G.L., 1983. Performance of growing finishing pigs in response to the thermal environment. ASAE Mid-Central meeting, March 16, 1983, St Joseph, Missouri, MCR. pp. 81-137. [ Links ]
Noblet, J. & Henry, Y., 1993. Energy evaluation systems for pig diets: a review. Livest. Prod. Sci. 36, 121-141. [ Links ]
Noblet, J. & Le Goff, G., 2001. Effect of dietary fibre on the energy value of feeds for pigs. Anim. Feed Sci. Technol. 90, 35-52. [ Links ]
Ogle, B., 2006. Forages for pigs: nutritional, physiological and practical implications. In: Proceedings of the workshop-seminar on forages for pigs and rabbits. Eds Preston, T.R. & Ogle, B., August 21-24th 2006. [ Links ]
Quiniou, N., Dubois, S. & Noblet, J., 2000. Voluntary feed intake and feeding behaviour of group-housed growing pigs are affected by ambient temperature and body weight. Livest. Prod. Sci. 63, 245-253. [ Links ]
Rinaldo, D. & Le Dividich, J., 1991. Assessment of optimal temperature for performance and chemical body composition of growing pigs. Livest. Prod. Sci. 29, 61-75. [ Links ]
Rinaldo, D., Le Dividich, J. & Noblet, J., 2000. Adverse effects of tropical climate on voluntary feed intake and performance of growing pigs. Livest. Prod. Sci. 66, 223-234. [ Links ]
Robertson, J.B. & Van Soest, P.J., 1981. The Analysis of Dietary Fibre in Food. Eds James, W.P.T. & Theander, O., Dekker, New York. ADSRI-JAN 1988. [ Links ]
Samuels, M.L., 1989. Statistics for the Life Sciences. Collier MacMillian Publishers, London. [ Links ]
Shriver, J.A., Carter, S.D., Sutton, A.L., Richert, B.T., Senne, B.W. & Pettey, L.A., 2003. Effects of adding fiber sources to reduced-crude protein, amino acid-supplemented diets on nitrogen excretion, growth performance, and carcass traits of finishing pigs. J. Anim. Sci. 81, 492-502. [ Links ]
SAS, 2009. Statistical Analysis Systems user's guide: Statistics Version 12. SAS Institute Inc., Cary, North Carolina, USA. [ Links ]
Visser, D.P., 2004. Structuring of breeding objectives in the pork supply chain in South Africa. PhD thesis. University of Pretoria, South Africa. [ Links ]
Wilfart, A., Montagne, L., Simmins, P.H., Van Milgen, J. & Noblet, J., 2007. Sites of nutrient digestion in growing pigs: Effect of dietary fibre. J. Anim. Sci. 85, 976-983. [ Links ]
Copyright resides with the authors in terms of the Creative Commons Attribution 2.5 South African Licence. See: http://creativecommons.org/licenses/by/2.5/za Condition of use: The user may copy, distribute, transmit and adapt the work, but must recognise the authors and the South African Journal of Animal Science.
# Corresponding author: christine.jansenvanrensburg@up.ac.za














