Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
South African Journal of Animal Science
versão On-line ISSN 2221-4062
versão impressa ISSN 0375-1589
S. Afr. j. anim. sci. vol.42 no.2 Pretoria Jan. 2012
Pedigree analysis of an ostrich breeding flock
M.D. FairI, #; J.B. van WykI; S.W.P CloeteII, III
IDepartment of Animal, Wildlife and Grassland Sciences, University of the Free State, P.O. Box 339, Bloemfontein 930l, South Africa
IIDepartment of Animal Sciences, University of Stellenbosch, Matieland 7602, South Africa
IIIElsenburg Agricultural Development Institute, Private Bag X1, Elsenburg 7607, South Africa
ABSTRACT
Pedigree records, maintained from 1978 to 2005 at the Oudtshoorn Research Farm, South Africa, of 40 074 birds of a pair-breeding ostrich flock were used to estimate the effective number of founders (fe), the effective number of ancestors (/0), the effective population size and the effective genome equivalents (fg) under random mating, to assess the genetic variability present in the population. The average level of completeness of the pedigrees was high (99.3%) in the first generation, and the average level of inbreeding (F), calculated from the pedigrees, was 0.51%. The reference population was defined as the 39 784 birds hatched from 1990 to 2005. The estimated measures of variability were fg = 47.3, fe = 59 and fa = 58, with an fjfa ratio of 1.02. The numbers of ancestors responsible for 100%, 50% and 20% of the genes in the reference population were 254, 21 and 6, respectively. The largest individual contribution to the population hatched from 1990 to 2005 was from a male that was responsible for 4.85% of the genetic variability. The generation interval for the four selection pathways - calculated as the average age of parents when offspring that were kept for reproduction were born - were sire to son (7.74 ± 4.92), sire to daughter (7.77 ± 5.13), dam to son (7.50 ± 4.29) and dam to daughter (7.90 ± 4.92). The average generation interval of the reference population was 7.72 ± 4.79 years. The linear regressions of mean annual individual rate of inbreeding on year of birth for the two distinct periods 1995-2002 and 2003-2005 were 0.08% and -0.07% per year, respectively. The estimate of effective population size (Ne), computed via the increase in the individual rate of inbreeding, was 112.7. Estimates of Ne using the alternative methods of tracing the numbers of generations were 73.6, 177.4 and 95.3 for complete, maximum and equivalent complete generations. The results of this study indicated that the population under study was at an acceptable level of genetic variability.
Keywords: Effective number of founders and ancestors, effective population size, level and rate of inbreeding.
Introduction
Domestic animal diversity is an integral part of global biodiversity that requires sound management for sustainable use and future availability (FAO, 1992). The effective population size, computed from the rate of inbreeding of a population, is a measure of the genetic diversity and drift, and is used to characterize the risk of extinction of animal populations and genetic variability available for future selection (Wright, 1969; Duchev et al., 2006). The founder effect is a measure of the loss in genetic diversity caused through the formation of small groups of individuals that may remain isolated in a population. The founder effect contributes to genetic drift, which can play an important role in determining the genetic makeup of subsequent generations.
Inbreeding occurs when individual animals mate that are more closely related than they would be if mating were random (Falconer & Mackay, 1996). There is a direct relationship between an increase in inbreeding and the reduction of heterozygosity for a given locus in a closed, unselected and panmictic population of finite size (Wright, 1931). The common practice in the selection of livestock in which animals are selected by truncation on estimated breeding values across age classes results in increased genetic gains, but also increased rates of inbreeding. This poses the threat of inbreeding depression, which may hamper selection response and genetic diversity in the long run (Bijma et al., 2001). Inbreeding and the rate of inbreeding (AF) also have an effect on the effective population size (Ne). For example, the Ne based on AF among dairy cattle breeds in the US was reported to be 161, 61, 65, 39 and 30 for the Ayrshire, Brown Swiss, Guernsey, Holstein and Jersey populations, respectively (Weigel, 2001).
Earlier works reported numerous studies on the genetic parameters (Bunter & Cloete, 2004; Cloete et al. , 2006; 2008c) and selection responses (Cloete et al., 2008a) for the production traits of a pair-bred ostrich flock at Oudtshoorn Research Farm. However, no genetic variability analyses based on pedigree data have been done so far. It is known that the breeding structures in ostrich flocks are unlike those of other livestock, with confounding between random effects in a pair-bred population, as well as a very narrow male : female ratio (Cloete et al., 1998; 2008c). Awareness of the pedigree structure, of levels of inbreeding and of the effective population size of a local flock of ostriches can be used to avoid the possible loss of genetic variability and fitness (e.g. fertility) owing to inbreeding when formulating modern breeding programmes for optimum production.
The aim of this study was thus to investigate the pedigree structure, inbreeding levels, generation interval, effective population size and founder contributions of a well-documented ostrich research flock. Knowledge of these parameters could help the industry when formulating breeding programmes.
Material and Methods
The data of a pair-breeding ostrich flock (n = 40 074), maintained at Oudtshoorn Research Farm, South Africa, were used for this study. The data included records collected from 1978 to 2005 and edited where necessary for inconsistencies regarding pedigree. Each breeding pair was kept in a separate paddock to facilitate the identification of parentage of the hatched chicks. The general management of the breeding pairs, eggs and chicks has been described in detail (Van Schalkwyk et al., 1996; Cloete et al., 1998; Bunter, 2002; Cloete et al., 2006; 2008c).
The ENDOG software program (version 4.8) (Gutierrez & Goyache, 2005) was used for all genealogical analyses of the pedigree data. ENDOG is a population genetics computer program that conducts several demographic and genetic analyses on pedigree data to monitor the changes in genetic variability and population structure in a population.
Several parameters were calculated for assessment of the concentration of the origin of animals and genes. The effective number of founders (/e) was defined as the number of equally contributing founders that would be expected to produce the same genetic diversity in the population under study. For a given number of total founders, the more balanced their expected genetic contributions are, the higher the effective number of founders will be. The effective number of ancestors (/a) is the minimum number of ancestors (including founders and non-founders) that explains the complete diversity of the population (Boichard et al., 1997). The ratio of fe/fa (1.02; Table 1) can be used to evaluate the loss in genetic variability in the founders owing to bottlenecks between the base population and the reference population. This ratio is an indication of the importance of bottlenecks in the development of the population. The "founder genome equivalent" (fg) can be defined as the number of founders that would be expected to produce the same genetic diversity as in the population under study if the founders were equally represented and no loss of alleles occurred (Lacy, 1989). The number of founder genome equivalents accounts not only for unbalanced contributions of parents to the next generation (as /e and /a) and bottlenecks in the pedigree (as /a), but also for the random loss of genes from parents to their offspring; therefore, /g is always smaller than /e and /a, and decreases more rapidly over time. The degree to which the founder genome equivalent is smaller is an indication of the degree of random loss of alleles owing to drift (Lacy, 1989; Tahmoorespur & Sheikhloo, 2011). The parameter /g was obtained by the inverse of twice the average co-ancestry of the individuals included in a pre-defined reference population (Caballero & Toro, 2000). The reference population was defined as all animals with both parents known.

To assess the completeness of the pedigree, ENDOG computes these three traced generations for each animal in the pedigree:
(i) Fully traced (complete) generations, defined as those separating the progeny of the furthest generation, where the second-generation ancestors of the individual are known. Ancestors with both parents unknown were considered founders (generation 0).
(ii) Maximum number of generations traced, defined as number of generations separating the individual from its furthest ancestor.
(iii) Equivalent complete generations computed for the pedigree of each animal as the sum over all known ancestors of the term (1/2)n where n is the number of generations separating the animal from each known ancestor (Maignel et al., 1996; Boichard et al., 1997).
The inbreeding coefficient (Fi) for each animal in the dataset was calculated according to Meuwissen & Luo's (1992) method. Individual inbreeding coefficients were used to compute the individual rate of inbreeding (AFi) according to the methodology described by Gonzalez-Recio et al. (2007) and modified by Gutierrez et al. (2009). The individual rate of inbreeding is an alternative measure of inbreeding that is adjusted for the depth of the known pedigree. This coefficient corrects the cumulative inbreeding coefficient, F, according to the pedigree depth of each animal. It is not a measurement of inbreeding, but an indicator of the increment in inbreeding for each animal, regardless of the number of generations known in its pedigree. Therefore, the individual rate of inbreeding makes it possible to distinguish between two animals with the same inbreeding coefficient, but with a different number of generations over which inbreeding was accumulated (González-Recio et al., 2007; Gutierrez et al., 2009). Slow inbreeding allows natural and artificial selection to operate and to remove the less fit animals. Less inbreeding depression is thus expected among the individuals who accumulated the inbreeding over a larger number of generations (Van Wyk et al., 2009).
This coefficient should not be affected by a possible nonlinear increase of inbreeding over time and, thus, two animals with the same inbreeding coefficient could have different inbreeding depression effects, depending on the number of complete generations in their particular pedigrees (González-Recio et al., 2007).
The individual rate of inbreeding was calculated as 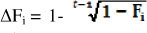 , where Fi is the individual coefficient of inbreeding of animal i and t is the "equivalent complete generations". The t was calculated with ENDOG ν 4.8 computer program (Gutierrez & Goyache, 2005). Using AFÍ5 ENDOG computes the 1 effective population size (Ne) as
, where Fi is the individual coefficient of inbreeding of animal i and t is the "equivalent complete generations". The t was calculated with ENDOG ν 4.8 computer program (Gutierrez & Goyache, 2005). Using AFÍ5 ENDOG computes the 1 effective population size (Ne) as 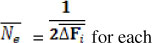 generation.
generation.
The average generation interval was calculated as the average age of the parents at the hatching of their selected progeny (James, 1977).
Trends were calculated for the average annual levels of inbreeding, as well as for the rate of inbreeding, using the regressions of applicable values on the year of hatch.
Results and Discussion
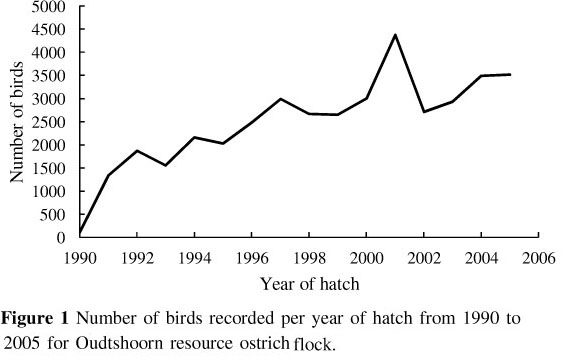
The trend of number of birds hatched per year is shown in Figure 1. A steep increase is evident from 1990 to 1992, after which the number of birds in the population increased at a more gradual rate.
Parameters characterizing the genetic variability of the ostrich flock at Oudtshoorn Research Farm are presented in Table 1. There were 253 equivalent founders (animals with one or more unknown parents) that contributed to the reference population in the original data set of 40 074 records. The effective number of founders for the reference population was 59, accounting for 24% of the total number of founders. The reference population consisted of 39 784 birds hatched between 1990 and 2005. The number of ancestors responsible for 100% of the variation in the reference group was 254. However, 50% of that variation was explained by only 21 animals, and 20% by 6 ancestors. The animal with the largest individual contribution to the genetic make-up of the birds born between 1990 and 2005 was a male that was responsible for 4.85% of the genetic variation.
The ratio offe/fa (1.02: close to unity; Table 1) indicated that the population has been stable in terms of numbers of effectively contributing ancestors and that bottlenecks have not played a significant role in the population. If the ratio is larger than one, bottlenecks have played a role in the formation of the population (S0rensen et al., 2005). The number of founder genome equivalents (fg) was 47.3, which is smaller than those estimated by /e and /a, as would have been expected, and indicated a small random loss of alleles owing to drift (Lacy, 1989; Tahmoorespur & Sheikhloo, 2011).
Pedigree completeness of the male and female lines up to three generations back is illustrated in Figure 2. The first ancestral generation, calculated from all animals in the data set, was 99.3% complete. The second generation was 69% and 74% complete. The completeness decreased to as low as 34% in the third generation.
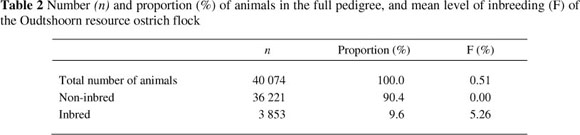
The numbers and proportions (%), relative to the full pedigree, as well as the mean level of inbreeding of the Oudtshoorn resource ostrich flock, is depicted in Table 2. When all the animals in the study were considered, the average inbreeding coefficient was 0.51%, compared with an average of 5.26% for the inbred animals. As shown in Table 2, only 9.6% of all the animals were inbred to some extent. No animals hatched before 1995 were inbred. The annual generation interval range of this flock (8.3 to 10.4) during this period was long (Fair et al., 2006) resulting in few replacements being added to the flock per annum. It was thus easy to avoid the mating of related birds.
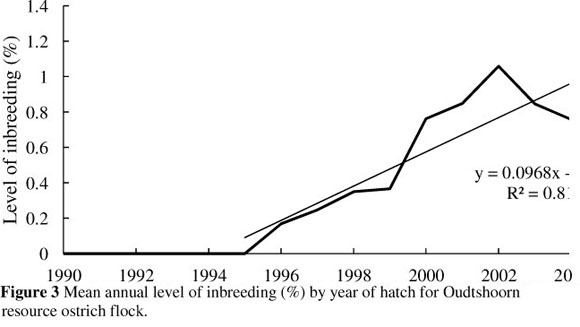
The trend depicting the average annual level of inbreeding for the ostrich flock under investigation is presented in Figure 3. The mean annual level of inbreeding was 0% from 1990 to 1995, rising steadily from 1996 to 2005 at a rate of 0.10% per annum to an average level of 1.2%. The increase in average annual inbreeding may be the result of a number of matings between close relatives in 2000 which resulted in 311 full sib, 363 half sib and 24 parent-offspring progeny. The current level of inbreeding in the flock is low and it should be possible to continue at relatively low levels, given sound mating management. It should, however, be kept in mind that inbreeding is likely to increase over time in any closed population of finite size.
The trend of the annual individual rate of inbreeding (AFi) for the ostrich flock under investigation is presented in Figure 4.
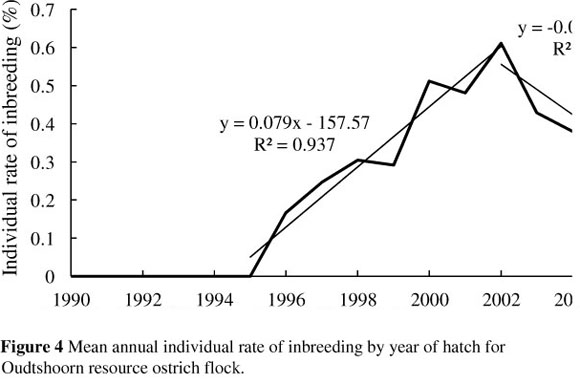
Similar to the trend in the average annual level of inbreeding, a sharp increase is noticeable in the individual rate of inbreeding from 1996 onwards. This was the stage at which the selection lines reported by Cloete et al. (2008b) were formed. Linear regressions of the individual rate of inbreeding on year of hatch resulted in estimates of the individual rate of inbreeding for the two distinct periods 1995 - 2002 and 2003 -2005 of 0.08% and -0.07% per year, respectively. The critical level of 0.5% per year suggested for animal breeding programmes (Nicholas, 1989) is exceeded by the rate that was observed over the first period. During this period, three separate breeding lines were established, comprising an unselected control line, a line selected for weight, and a line selected for chick output (Cloete et al., 2008a). Because the lines were represented by relatively few animals (5 - 7 new breeding pairs introduced per line per year), inbreeding could have been expected to accrue, as with any population of finite size. However, estimates of the individual rate of inbreeding declined from 2002 to below 0.5% in 2003, 2004 and 2005 at an average rate of -0.07% per year. The reduction in the rate of inbreeding coincides with the introduction of unrelated breeding birds to the flock at the beginning of the 2003 breeding season (Cloete et al., 2008b). Also, because the mating of related individuals was recognised by those involved in the flock husbandry at this stage, inbreeding was started to be managed during mating decisions in the flock. It should be noted that obtained AFi values are still subject to change owing to the relative shallowness of the analysed pedigrees (equivalent complete generations = 2.17), and more generations of pedigree data are needed to make proper use of this parameter. Cervantes et al. (2008) also reported that the trend of AFi values tended to become more stable with the increase in equivalent generations owing to the correction resulting from deeper pedigrees of the individuals.
The estimate of effective population size (Ne), computed via the individual rate of inbreeding (Gutierrez et al., 2008), for the current study was 112.7 animals, which is appreciably higher than the critical value of 50 animals suggested by the FAO (1998). Meuwissen (1999) stated that owing to mutation and drift, the critical Ne size should be between 50 and 100 animals. However, the method of calculating Ne can have a substantial effect on the outcome. In the current study, different Ne -values were obtained using three methods of tracing generations in the pedigrees of animals, that is, complete generations, maximum generations and equivalent complete generations, and yielded Ne -values of 73.6, 177.4 and 95.3 respectively. Furthermore, estimates of Ne are usually not constant and may change over time, given changes in average levels of inbreeding in the population, generation interval and the number of known parents and progeny per parent; that is, current estimates of Ne can decrease to below the critical value if the rate of inbreeding in the population should increase. Although the depth of the pedigrees and the levels of completeness of the pedigrees should be considered in making a comparison between different estimates of Ne, the estimated effective population size (112.7) of the ostrich resource flock at the Oudtshoorn Research Farm currently seems to be at acceptable levels.
Five forces are active in making the calculated Ne-values using AF,- different from the real population size. These forces are fluctuating population size; sex ratio of breeding animals; Poisson distribution of family (offspring) numbers; overlapping generations (Felsenstein, 1971); and spatial dispersion of the breeding population. Unlike dairy herds, where the ratio of sires to dams is low, the sex ratio of ostrich breeding animals was close to 1 : 1 for all years under consideration, as would be expected in a pair-bred population (Cloete et al., 1998; 2008b). Spatial dispersion does not play a role in the flock as each male is paired off with a single female and is kept in a paddock that restricts interbreeding with other birds.
The calculated generation interval for the four gamete pathways were sire to son (7.74 ± 4.92 years); sire to daughter (7.77 ± 5.13 years); dam to son (7.50 ± 4.29 years); and dam to daughter (7.90 ± 4.79 years). The average generation interval of the reference population was 7.72 ± 4.79 years. This value is high and may hamper ostrich breeders from making reasonable genetic progress in the selection of production and reproduction traits. The average age of female and male breeding birds, however, was lowered intentionally in later years (Cloete et al., 2006). Whereas breeding animals as old as 22 years were initially kept in the breeding flock, male and female breeding animals are now culled at 10 - 11 years to help shorten the generation interval. This strategy was prompted by observed age trends in reproductive fitness, as described by Cloete et al. (1998) and Bunter (2002).
More than 200 000 slaughter birds are produced annually in South Africa (Brand & Jordaan, 2011). This flock is therefore a miniscule sample of the total number of breeding ostriches found in the country. The effective population size of South African ostriches is probably large and varied compared with other domestic livestock populations (particularly dairy, beef and sheep).
Conclusions
Pedigree analysis was found to be useful in determining the status of genetic variability in the ostrich flock maintained at the Oudtshoorn Research Farm. The results showed a relatively low rate of inbreeding, resulting in a comparatively high effective population size. Inbreeding levels are currently low and manageable. However, the rather long generation interval of 7.7 years needs to be shortened to increase the rate of genetic improvement owing to more opportunities to select superior birds for economically important production and reproduction traits.
The individual rate of inbreeding that was obtained is still subject to change owing to the shallowness of the analysed pedigrees, and more generations of pedigree data is necessary to make proper use of this parameter. Follow-up studies are recommended for continued monitoring of the genetic variability in the flock and for the calculation of AFi parameters as more data become available. The higher the numbers of generations in the pedigree, the more stable the genetic variability parameters are likely to become. It is concluded that the population investigated demonstrated acceptable levels of genetic variability.
Acknowledgements
We express our sincerest gratitude to all those responsible for the maintenance and recording of the Oudtshoorn resource ostrich flock. The study was partially funded by the THRIP programme of the South African Department of Trade and Industry. The authors wish to thank the National Research Foundation for financial support of this research.
References
Bijma, P., Van Arendonk, J.A.M. & Woolliams, J.A., 2001. Predicting rates of inbreeding for livestock improvement schemes. J. Anim. Sci. 79, 840-853. [ Links ]
Boichard, D., Maignel, L. & Verrier, E., 1997. The value of using probabilities of gene origin to measure genetic variability in a population. Genet. Sel. Evol. 29, 5-23. [ Links ]
Brand, T.S. & Jordaan, J.W., 2011. The contribution of the South African ostrich industry to the national economy. Appl. Anim. Husb. Rur. Dev. 4, 1-7. Accessible at: http://www.sasas.co.za/aahrd/2011/1 (downloaded on 27 October 2011). [ Links ]
Bunter, K.L., 2002. The genetic analysis of reproduction and production traits recorded for farmed ostriches (Struthio camelus). PhD dissertation, University of New England, Australia: pp. 280. [ Links ]
Bunter, K.L. & Cloete, S.W.P., 2004. Genetic parameters for egg-, chick- and liveweight traits recorded in farmed ostriches (Struthio camelus). Livest. Prod. Sci. 91, 9-22. [ Links ]
Caballero, A. & Toro, M.A., 2000. Interrelations between effective population size and other pedigree tools for the management of conserved populations. Genet. Res., Camb. 75, 331-343. [ Links ]
Cervantes, I., Goyache, F., Molina, A., Valera, M. & Gutiérrez, J.P., 2008. Application of individual increase in inbreeding to estimate realised effective sizes from real pedigrees. J. Anim. Breed. Genet. 125, 301-310. [ Links ]
Cloete, S.W.P., Van Schalkwyk, S.J. & Brand, Z., 1998. Ostrich breeding progress towards a scientifically based-strategy. In 'Ratites in a competitive world. Proc. 2nd International scientific ratite conference, Oudtshoorn, South Africa, 21-25 September 1998'. Ed. Huchzermeyer, F.W. pp. 55-62. [ Links ]
Cloete, S.W.P., Bunter, K.L, Lambrechts, H., Swart, D. & Greyling, J.P.C., 2006. Variance components for live weight, body measurements and reproductive traits of pair-mated ostrich females. Br. Poult. Sci. 47, 147-158. [ Links ]
Cloete, S.W.P., Brand, Z., Bunter, K.L. & Malecki, I.A., 2008a. Direct responses in breeding values to selection of ostriches for liveweight and reproduction. Aust. J. Exp. Agric. 48, 1314-1319. [ Links ]
Cloete, S.W.P., Brand, M.M., Hoffman, L.C. & Muller, M.M., 2008b. Live weight and reproduction performance of Zimbabwean Blue and South African Black ostriches. S. Afr. J. Anim. Sci. 38, 65-73. [ Links ]
Cloete, S.W.P., Engelbrecht, A., Olivier, J.J. & Bunter, K.L., 2008c. Deriving a preliminary breeding objective for commercial ostriches: an overview. Aust. J. Exp. Agric. 48, 1247-1256. [ Links ]
Duchev, Z., Distl, O. & Groeneveld, E., 2006. Early warning system for loss of diversity in European livestock breeds. Archiv. Anim. Breed. 49, 521-531. [ Links ]
Fair, M.D., Van Wyk, J.B. & Cloete, S.W.P., 2006. Genetic parameters for monthly reproduction of ostrich females. Proc. 41st Congr. SA Soc. Anim. Sci. 49, (3-6 April 2006, Bloemfontein). [ Links ]
Falconer, D.S. & Mackay, T.F.C., 1996. Introduction to Quantitative genetics. Longman Group, Essex, UK. [ Links ]
FAO, 1992. United Nations Environment Programme, Convention on Biological Diversity, Environmental Law and Institutions Programme Activity Centre, 1-52. [ Links ]
FAO, 1998. Secondary Guidelines for Development of National Farm Animal Genetic Resources Management Plans. Page 63 in Management of Small Populations at Risk. Food Agric. Org. United Nations, Rome, Italy. [ Links ]
Felsenstein, J., 1971. Inbreeding and variance effective numbers in populations with overlapping generations. Genetics 68, 581-597. [ Links ]
González-Recio, O., López de Maturana, E. & Gutiérrez, J.P., 2007. Inbreeding depression on female fertility and calving ease in Spanish dairy cattle. J. Dairy Sci. 90, 744-5752. [ Links ]
Gutiérrez, J.P. & Goyache, F., 2005. A note on ENDOG: a computer programmes for analysing pedigree information. J. Anim. Breed. Genet. 122, 172-176. [ Links ]
Gutiérrez, J.P., Cervantes, I., Molina, A., Valera, M. & Goyache, F., 2008. Individual increase in inbreeding allows estimating effective sizes from pedigrees. Genet. Sel. Evol. 40, 359-378. [ Links ]
Gutiérrez, J.P., Cervantes, I. & Goyache, F., 2009. Improving the estimation of realized effective population sizes in farm animals. J. Anim. Breed. Genet. 126, 327-332. [ Links ]
James, J.W., 1977. A note on selection differentials and generation length when generations overlap. Anim. Prod. 24, 109-112. [ Links ]
Lacy, R.C., 1989. Analysis of founder representation in pedigrees: founder equivalents and founder genome equivalents. Zoo. Biol. 8, 111-123. [ Links ]
Maignel, L., Boichard, D. & Verrier, E., 1996. Genetic variability of French dairy breeds estimated from pedigree information. Interbull Bull. 14, 49-54. [ Links ]
Meuwissen, T.H.E., 1999. Operation of conservation schemes. In: Genebanks and the Conservation of Farm Animal Genetic Resources. Ed. Oldenbroek, J.K., DLO Inst. Anim. Sci. Health, Lelystad, The Netherlands. pp. 91-112. [ Links ]
Meuwissen, T.H.E. & Luo, Z., 1992. Computing inbreeding coefficients in large populations. Genet. Sel. Evol. 24, 305-313. [ Links ]
Nicholas, F.W., 1989. In: Evolution and Animal Breeding. Eds Hill, W.G. & Mackay, T.F.C., CAB Intl, Wallingford, UK. [ Links ]
S0rensen, A.C., S0rensen, M.K. & Berg, P., 2005. Inbreeding in Danish dairy cattle breeds. J. Dairy Sci. 88, 1865-1872. [ Links ]
Tahmoorespur, M. & Sheikhloo, M., 2011. Pedigree analysis of the closed nucleus of Iranian Baluchi sheep. Small Rumin. Res. 99, 1-6. [ Links ]
Van Schalkwyk, S.J., Cloete, S.W.P. & De Kock, J.A., 1996. Repeatability and phenotypic correlations for live weight and reproduction in commercial ostrich breeding pairs. Br. Poult. Sci. 37, 953-962. [ Links ]
Van Wyk, J.B., Fair, M.D. & Cloete, S.W.P., 2009. Case Study: The effect of inbreeding on the production and reproduction traits in the Elsenburg Dormer sheep stud. Livest. Sci. 120, 218-224 (Special issue on "Genetic resources"). [ Links ]
Weigel, K.A., 2001. Controlling inbreeding in modern breeding programs. J. Dairy Sci. 84 (E. Suppl.), E177-E184. [ Links ]
Wright, S., 1931. Evolution in Mendelian populations. Genetics 16, 97-159. [ Links ]
Wright, S., 1969. Evolution and the Genetics of Populations: The Theory of Gene Frequencies, Vol. 2. University of Chicago Press, Chicago, IL. [ Links ]
Copyright resides with the authors in terms of the Creative Commons Attribution 2.5 South African Licence. S ee: http://creativecommons.org/licenses/by/2.5/za Condition of use: The user may copy, distribute, transmit and adapt the work, but must recognise the authors and the South African Journal of Animal Science.
# Corresponding author: fairmd@ufs.ac.za














