Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
South African Journal of Animal Science
versão On-line ISSN 2221-4062
versão impressa ISSN 0375-1589
S. Afr. j. anim. sci. vol.42 no.2 Pretoria Jan. 2012
REVIEW
Animal factors affecting fatty acid composition of cow milk fat: a review
E. SamkováI, #; J. SpickaI; M. PesekI; T. PelikánováI; O. HanusII
IUniversity of South Bohemia, Faculty of Agriculture, Seské Budëjovice, Czech Republic
IIResearch Institute for Cattle Breeding, Rapotín, Czech Republic
ABSTRACT
The review summarizes literature dealing with the effects of animal factors (breed, cow individuality, parity and stage of lactation) on fatty acid (FA) composition of milk fat. Genetic parameters affecting the composition of the FAs in milk are reviewed and the possibilities for altering milk fat composition are discussed. Cow individuality and the stage of lactation appear to be the main animal factors affecting milk fat composition. Breed and parity affect the variability in FA composition to a limited extent. Some of these factors can be used effectively to alter milk fat composition. Polymorphism of the enzymes, stearoyl-CoA desaturase (SCD) and acyl-CoA-diacylglycerol acyltransferase (DGAT) can explain to some extent the variability among cows. The great individual differences, probably given by varying SCD activities, may be used in breeding programmes, supported by the heritability estimates determined for individual FAs. Effective results can also be achieved through the combined effect of several factors. For instance, the level of conjugated linoleic acid could be increased not only by feed factors, but also through thorough knowledge of rumen biohydrogenation or by cow selection using information on SCD and DGAT polymorphism. The animal factors that are discussed are closely related to milk yield, particularly fat content. Both parameters can change FA composition. Thus, it is necessary in breeding programmes to take these relationships into consideration, along with known genetic correlations.
Keywords: Breed, genetic correlations, heritability, milk and fat yield, parity, single nucleotide polymorphism, stage of lactation
Introduction
Interest in the chemical composition of animal fats has increased steadily since the first scientific reports were published on the negative effects of these fats on human health. The consumption of milk and often of other milk products has decreased owing to widespread reports on the hypercholesterolaemic effects of certain fatty acids (FAs) in humans. Such a situation has stimulated interest in research into altering milk fat (MF) composition.
Fatty acids, the most important component of MF, constitute about 90% of its weight. Over 95% of the FAs are bound in triacylglycerols, the remainder in mono- and diacylglycerols, phospholipids and cholesterol esters. Free FAs are present in small proportions. Fatty acids differ in chain length and degree of unsaturation, position and orientation of double bonds. Among the hundreds of FAs that have been identified in MF, only 15 occur at concentrations of 10 g per kg and higher. Saturated and unsaturated FAs constitute about 65% and 35% of the FAs, respectively (Jensen, 2002; Parodi, 2004).
The FAs in ruminant milk are synthesized (i) in the mammary gland (so-called de novo synthesis) from acetate and to a lesser extent from β?-hydroxybutyrate. The precursors are produced in the rumen from dietary polysaccharides. This is the origin of the FAs with shorter carbon chains (<C15 and a portion of C16), while (ii) about one half of the FAs (a portion of C16 and >C17; so-called preformed FAs) are synthesized from dietary lipids and adipose tissue reserves (Bauman et al., 2006; Nafikov & Beitz, 2007; Harvatine etal., 2009).
Trans-isomers of unsaturated FAs (TFA) originate from rumen bacterial biohydrogenation of cispolyunsaturated FAs obtained from the diet. They constitute up to 5% of all FAs (Precht & Molkentin, 2000; Glasser et al., 2008). Within this group, FAs designated cis-9, trans-11-C18:2, with the trivial name, rumenic acid (Kramer et al., 1998), and trans-10, cis-12-C18:2 are two main isoforms of the group of conjugated linoleic acids (CLAs). The CLAs are considered highly beneficial to human health (Gnádig et al., 2001) and thus desirable in milk products (Dhiman et al., 2005). Nevertheless, these two isoforms can have different effects on metabolism and cell functions. The majority of scientific reports attribute the beneficial properties of the CLA for human health without differentiation of the individual isoforms. However, few investigations have studied the effects of individual isoforms on human health, though some researchers (Rajakangas et al. , 2003) have attributed negative effects such as pro-carcinogenic effects (especially colon and prostate cancer) to the trans-10, cis-12 isomer in animals. Furthermore, most of these studies have been conducted on animal models, and recent studies have indicated that some effects observed in animals do not pertain to humans (Gebauer et al. , 2007).
Similarly, the odd- and branched-chain FAs (up to 4% of all FAs) in MF have been synthesized predominantly by specific rumen bacteria. The interest in these FAs lies in better understanding of rumen function, their anticarcinogenic activity, as well as their influence on the melting point of MF (for more information see Vlaeminck et al., 2006).
Fatty acid composition, their position in the molecules of triacylglycerols, as well as the proportion of saturated and unsaturated FAs, affect nutritional, sensory and technological properties of MF extensively. However, the perception of optimal FA composition can vary among nutritionists and dairy technologists. A high proportion of saturated FAs can improve fat stability against oxidation and thus diminish a potential for sensory defects. Nevertheless, such a composition has been perceived as undesirable from a nutritional point of view. Saturated FAs, particularly lauric, myristic and palmitic, and TFA have been assessed negatively as risk factors for cholesterolaemia, which, in turn, is considered a risk factor for cardiovascular diseases in humans (Mensink, 2005; Gebauer et al. , 2007). However, generalized recommendations to reduce the consumption of fatty dairy foods because of their high content of saturated FAs should be made with caution. Short-chain FAs reduce low-density lipoprotein (LDL)-cholesterol concentration, and the negative effects of ruminant TFA in comparison with industrial TFA are ambiguous (see review by German et al., 2009). Unlike other saturated FAs, stearic acid lowered LDL-cholesterol levels (Hunter et al., 2010). Some mono-(oleic acid) and polyunsaturated FAs (long-chain n-3 FAs) are known to possess substantial antiatherogenic and further positive health properties (Kris-Etherton et al., 2009; Lopez-Huertas, 2010; Siri-Tarino et al., 2010).
The proportion of nutritionally desirable FAs to detrimental ones can be altered, utilizing factors that effectively change MF composition. These factors are usually classified in two or three groups: feed, animal and environmental (Palmquist et al., 1993; Perdrix et al., 1996; Jensen, 2002). To date, the greatest attention has been focused on feed factors. Their application has enabled important essential changes in MF composition, especially following the feeding of oil seeds, and plant and fish oils. A more desirable composition of MF has also been reported for milk from cows on pasture (Chilliard et al., 2001; 2008; Kalac & Samková, 2010).
Over the past few years, numerous reports have been published on the effects of environmental conditions, especially heat stress (e.g. O'Brien et al., 2007; Wang et al., 2010).
In comparison with the abundance of studies on the effect of cow nutrition on FA composition, information on the effect of animal factors is considerably limited. Breed, stage of lactation and cow individuality are the most frequently studied animal factors affecting MF composition. These are assessed alone or in combination with other factors (Kelsey et al. , 2003; Secchiari et al. , 2003; Soyeurt et al. , 2006a; Samková, 2008; Stoop et al., 2009a).
The aim of this article is to review current knowledge of animal factors that can alter the FA composition of MF of cows.
2. Genetic parameters
In comparison with other milk constituents, FA composition is most amenable to change. Goddard (2001) reported that knowledge of genetics could be useful in altering MF composition. Methods to achieve this include choice of breed, traditional breeding programmes, including progeny testing, selection of bulls and cows based on specific genes, and transgenesis.
The application of animal factors to change in fat composition is dependent on genetic variability of individual FAs and their groups. Of special interest is the focus on genetic and phenotypic correlations related to milk production and proportion/production of FAs, as well as relations between individual FAs (see 'Milk and fat yield').
The results reviewed by Gibson (1991) showed that the coefficient of genetic variation was in the range of 0.05 - 0.2 for molar proportion of individual FAs in MF, and heritability estimates were moderate (ca. 0.3). These values were calculated from the results obtained from 254 dairy cows, daughters of 10 bulls (Renner & Kosmack, 1974). Higher heritability estimates (0.8 - 0.98) were observed in 25 twins, 15 dizygotic and 10 monozygotic twin pairs (Edwards et al. , 1973). Recently the results of genetic research have prompted increasing interest in genetic parameters dealing with FAs (Soyeurt et al. , 2007; Bobe et al. , 2008; Schennink et al., 2008; Soyeurt et al., 2008a; b; Arnould & Soyeurt, 2009; Mele et al., 2009). These authors proved the existence of genetic variability and determined moderate heritability coefficients based on the results from various countries and breeds, and of numerous animal populations and milk samples.
Stoop et al. (2008) estimated genetic parameters for individual FAs, expressed in g/100 g of fat. The data were derived from 1 918 cows (all over 87.5% Holstein-Friesian) and 101 bulls from 398 commercial herds in the Netherlands (Table 1). It was shown that estimates of heritability for the individual FAs and their groups are correlated with the length of the carbon chain: "de novo" synthetized FA (C4:0 to C14:0 and half of C16:0) had higher heritability estimates (0.31 - 0.54) than FAs originating from the diet and from body fat mobilization (LCFA and PUFA) (0.09 - 0.21) (Gibson, 1991; Stoop et al., 2008; Bastin et al., 2011).
However, heritability estimates for FAs expressed as g/100 g fat have different, mainly higher values than the heritabilities for FAs expressed as g/100 g milk. This suggests a considerable effect of milk yield, similar to the different values of heritability for FAs when expressed in g/day (Soyeurt et al. , 2007; Bobe et al. , 2008).
Some research groups (Schennink et al. , 2008; Mele et al. , 2009) have studied genetic variability of unsaturation indices (Table 1). These indices approximate the measurement of stearoyl-CoA desaturase (SCD, also known as A9-desaturase) activity. Several methods may be used to estimate SCD activity. The indices of unsaturation are defined as ratios of FAs dependent on this enzymatic activity: product of Δ9-desaturase to substrate Δ9-desaturase (e.g. Soyeurt et al., 2008b), substrate to product (e.g. Chouinard et al., 1999) and product to substrate + product (e.g. Kelsey et al. 2003; Schennink et al., 2008). Thus, the interpretation of indices had to be carried out with caution. For instance, although CLA is produced as an intermediate in the rumen biohydrogenation of linoleic acid, its major source in MF is endogenous synthesis by SCD in the mammary gland and other tissues from trans-11-C18:1 acid (vaccenic acid). In fact, vaccenic acid is produced as a rumen biohydrogenation intermediate from both linoleic and linolenic acids. Thus, knowledge on mammary synthesis of MF, rumen fermentation and dietary supply of lipids can be applied to alter MF composition (Lock & Garnsworthy, 2002; Lock & Bauman, 2004; Mosley et al. , 2006).
The initial reported relationships indicate a need for further research, which could elucidate the genetics of milk FA unsaturation level and clarify the interaction between genetics and feeding.
3. Breed
Most published papers dealing with the effect of cow breed have evaluated the response in FA composition following a change in feed composition (Carroll et al., 2006; Kliem et al., 2009; Ferlay et al., 2010). Some authors found inter-breed differences in MF composition, resulting in different technological properties with the potential to produce unique milk products (Auldist et al., 2004; De Marchi et al., 2008).
Two breeds, Holstein (Friesian) and Jersey, have been tested most frequently (Morales et al. , 2000; White et al. , 2001; Croissant et al. , 2007). Nevertheless, inter-breed differences in MF composition were reported in other breeds, such as Belgian Blue, Brown Swiss, Montbéliarde, Salers and Simmental (Agabriel et al., 2001; Moore et al., 2005; Soyeurt et al., 2006a; Barlowska et al., 2009), particularly in comparison with the Holstein. Numerous studies compared MF composition of indigenous and universally used breeds, including their crossbreds (Malacarne et al., 2001; Zegarska et al., 2001; Pesek et al., 2005; Talpur et al., 2006; Moioli et al., 2007; Palladino et al., 2010).
Comparative studies seem to indicate that dairy breeds with a high milk fat content often have a less desirable MF composition (higher levels of saturated and hypercholesterolaemic FAs, and a lower proportion of polyunsaturated FAs) than breeds with a lower milk yield or fat content. The fat composition produced by indigenous breeds, dual-purpose breeds and crossbreds appears to have a more desirable profile than imported dairy breeds (mostly Holstein). Such differences are apparent from Table 2. The proportions of FA groups that are important from a technological or nutritional point of view (including values of Δ9-desaturase indices) were calculated from mean proportions of the individual FAs, reported in the cited works.
To select these works, we took into consideration parameters of the experiments (e.g. number of cows, number of analysed samples, number of determined FAs, and type of diets) to ensure maximum comparability of the results. Cow nutrition was the most variable factor in these experiments. The diets varied in forage type (pasture, silage, hay) and in ratio of forage to concentrates. Thus, meta-analysis of data is problematic as various factors are involved in the experiments and the experimental conditions are often described incompletely.
Although the collected data were not all available in some cited publications, it is evident from Table 2 that the highest levels of hypercholesterolaemic and saturated FAs were observed in breeds characterized by a high milk fat content, such as the Ayrshire (51.9% and 74.7%) and Jersey (48.7% and 72.4%).
Data on statistically evaluated inter-breed differences in the proportions of individual FAs are presented in Table 3. It may seem somewhat surprising that differences between Holstein and Jersey breeds from several independent investigations are ambiguous for most of the FA profiles. Similar differences in statistical significance are apparent in two studies comparing the Holstein and Brown Swiss breeds.
These results support the opinion that the effect of breed explains only a limited proportion of the variability (Kelsey et al., 2003; Soyeurt et al., 2006a; Samková, 2008). Further reasons for inter-breed differences may be the low number of cows used in these experiments, resulting in different responses of cows on different diets. This suggestion is supported by the results of Ferlay et al. (2006). In three experiments with numerically nearly equal groups of Montbéliarde and Tarentaise cows they observed statistical significance differences in the proportion of certain FAs.
Mele et al. (2007) and Schennink et al. (2008) attributed differences in MF composition among breeds to varying activity of SCD (see Section 4).
4. Cow Individuality
Several papers have reported that the proportion of individual FAs in MF varied among dairy cows within a breed to a greater extent than inter-breed differences (Kelsey et al., 2003; Soyeurt et al., 2006a).
High variability in the composition of MF from cows fed the same diet enabled Bobe et al. (2003) to select animals with widely different FA compositions. The authors were successful in obtaining butter with a higher proportion of unsaturated FAs, resulting in better texture parameters: more spreadable, softer, and less adhesive. A further improvement was obtained from a combination of dairy cow selection and change in diet (Bobe et al. , 2007b).
The importance of the effect of cow individuality on MF composition was confirmed by Elgersma et al. (2006), who tested the responses of individual cows to changes in diets. They found that even if the patterns in response to the diet changes were similar, the concentration of CLA differed among cows.
The existence of great individual variability in several breeds, characterized by minimum and maximum values in FA composition, is apparent from Table 4. Ranges in nutritionally important FAs such as CLA, hypercholesterolaemic and monounsaturated FAs could be a major factor in altering the proportion of desirable and undesirable FAs.
Lock & Garnsworthy (2002) and Kelsey et al. (2003) explained the differences in CLA and monounsaturated FA proportions among individual cows by different SCD activity, similar to inter-breed variability. The SCD, as the key enzyme of mammary lipid metabolism, participates in the formation of the double bond in the cis-Δ9- position in a large spectrum of medium- and long-chain FAs. Variability in SCD activity is explained by single nucleotide polymorphism (SNP) in the SCD gene located on chromosome 26 (exon 5) (Mele et al., 2007; Schennink et al., 2008). SNP causes substitution (A293V) of valine (allele V) with alanine (allele A). Thus, there are three genotypes (VV, VA, and AA) with different distributions in breeds. The SCD allele A was associated with a higher proportion of monounsaturated FAs.
Kgwatalala et al. (2007) hypothesized that SNP in the SCD gene accounts for some differences between Canadian Holstein and Jersey cattle. While three SNPs (A702G, T762C, C878T) were identified in both breeds (44 and 48 cows, respectively), only one SNP (G435A) was unique to Holsteins. Thus, SNPs characterized four genetic variants in Holsteins, with only two variants in Jerseys.
Single nucleotide polymorphism (A293V) has been associated with some milk FAs in Italian HolsteinFriesian (Mele et al., 2007), Italian Brown (Conte et al., 2010), Piedmontese and Valdostana cattle (Moioli et al., 2007). The distribution of the SCD genotype in 297 Italian Holsteins was 0.27, 0.60, and 0.13 for AA, VA, and VV, respectively. The frequencies of alleles A and V were 0.57 and 0.43, respectively. Conte et al. (2010) found that the allele frequencies in 351 Italian Brown cows were 0.18 and 0.82, respectively. Moioli et al. (2007) found frequencies of allele A of 0.42 in 27 Piedmontese and 0.65 in 27 Valdostana cows. Schennink et al. (2008) reported a high frequency (0.73) of allele A in 1725 Dutch Holstein-Friesian cows.
Moreover, Milanesi et al. (2008) reported SNPs (A702G, T762C, C878T) in the SCD gene in 11 cattle breeds (in total, 336 animals), studied in Italy. High variability and differences across breeds showed an association to different selection goals (milk, meat, dual-purpose). Such results support the opinion (see Section 3) about milk production, for example milk yield and milk fat, of the individual breeds.
Acyl-CoA-diacylglycerol acyltransferase (DGAT), a key enzyme in triacylglycerol synthesis, may also play a significant role in changing saturated FAs into unsaturated ones. The gene polymorphism in the DGAT gene located on chromosome 14 (exon 8) may explain genetic variation in fat content, milk and fat yields (Hradecká et al., 2008) and it has also a strong effect on milk FA composition (Schennink et al., 2008). Dinucleotide polymorphism (K232A) causes replacement of lysine (allele K) with alanine (allele A). In comparison with allele K, the allele A of DGAT was associated with significantly lower indices of C10, C12, C14 and C16 acids and with significantly higher indices of C18 and CLA. The frequencies of allele A were 0.6 in 1713 Dutch Holstein-Friesian cows (Schennink et al., 2008) and 0.98 in 351 Italian Brown cows (Conte et al., 2010).
As reported by Schennink et al. (2008), genetic variance explained by DGAT polymorphism is lower (3% - 15%) than SCD polymorphism (6% - 52%). Genetic variance due to SCD polymorphism is higher (34% - 2%) for Δ9-desaturase indices of C10-C14 acids than for Δ9-desaturase indices of C18 acids (12% -15%). Relatively high genetic variance explained by SCD and DGAT polymorphism (31 % and 14%, respectively) for index C16 can be caused by the two above mentioned ways of C16-FAs formation (de novo and preformed).
Similarly, as in the inter-breed differences, varying enzymatic activities in individual cows may be affected by SCD and DGAT polymorphism. Thus, the selection of dairy cows could increase the proportio n of nutritionally required FAs.
Stoop et al. (2009b) reported that quantitative trait loci (QTL) might also participate in the FA phenotypic variance. QTL is a locus with genes controlling quantitative properties, linked for example with milk composition. However, more genes can be responsible for genetic variation in milk production traits (Goddard, 2001; Ordovas et al., 2008). Phenotypic variance explained by QTL was 3% - 8% and 4% - 13% for short- and medium-chain FAs, respectively, and 4% - 10% for FAs with long carbon chain and 3% - 8% for Δ9-desaturase indices (Schennink et al., 2009; Stoop et al., 2009b).
As evident from the last two sections, inter- and within-breed differences in MF composition do exist. Based on the recent state of knowledge, several factors may be involved, for example different milk (fat or protein) yields of the individual breeds, different activity of desaturases and genetic polymorphism. The expected discoveries of genetic polymorphism could hold great promise for future explanation of the principles of inter-breed differences and differences in FA formation.
Future knowledge on gene identification and genetic polymorphism can contribute to the elucidation of genetic variance and the process of FA biosynthesis.
5. Parity
Although data in the literature on the effect of parity (or age) on MF composition are limited, it is indisputable that this factor affects MF composition (Kelsey et al., 2003; Craninx et al., 2008; Samková, 2008; Soyeurt et al., 2008b). Most papers categorize cows into two groups, primiparous and multiparous. In experiments, which did not evaluate the factor of parity separately, both groups were present to balance the experimental design (Bargo et al., 2006; Ferlay et al., 2006; Mántysaari et al., 2007).
As the available data seem to indicate, primiparous cows produce MF with a higher proportion of unsaturated FAs and lower proportion of saturated FAs than cows in second and further lactations. For instance, Thomson et al. (2000) reported higher proportions of oleic acid and total unsaturated FAs in the fat of primiparous cows compared with multiparous ones. In a similar comparison, Craninx et al. (2008) observed significantly lower levels of palmitic acid and higher levels of stearic acid, oleic acid, VA and CLA in MF of primiparous cows.
The different MF composition from primiparous and multiparous cows can be partially explained by changing milk production and fat content during the individual lactations (Bradford & Allen, 2004). Miller et al. (2006) reported that the content of FA synthase in the mammary gland, participating in FA biosynthesis, was very low during the initial third of lactation and then gradually increased in primiparous cows. In multiparous animals the level of FA synthase in the early lactation was the same as that in the primiparous cows at the end of lactation.
Wathes et al. (2007) suggested that there are differences between primiparous and multiparous cows in the control of tissue mobilization that may promote nutrient partitioning into growth, as well as milk during the first lactation. Metabolic demands for milk production limit the deposition of preformed FAs to adipose tissue during the initial 90 days of lactation (Lake et al. , 2007).
6. Stage of Lactation
The effect of stage of lactation was studied more extensively than the role of parity. Lactation has often been divided into three periods: early (<100 days in milk), mid (100 - 200 days in milk) and late (>200 days in milk).
Milk sampled during these three periods (usually one sample per cow) was used for comparison of differences in FA composition during lactation (Barlowska et al., 2005; Garnsworthy et al., 2006; Mele et al. , 2007; 2009). Commonly, the highest sampling frequency has been during the early period (Kay et al. , 2005; Komprda et al. , 2005; Lake et al. , 2007), the period with the most significant changes in FA composition. Some authors took more than five samples during a lactation (Bernal-Santos et al. , 2003; Secchiari et al. , 2003; Craninx et al. , 2008; Samková, 2008).
The most extensive changes in MF composition within early lactation occur during the initial weeks and become less extensive from the eight week of lactation (Bernal-Santos et al. , 2003; Secchiari et al. , 2003; Kay et al. , 2005; Lake et al. , 2007). Nevertheless, Fearon et al. (2004) reported that during late-lactation cows produced MF containing a significantly higher proportion of unsaturated FAs than during mid-lactation.
Depending on the fat sources (de novo synthesis or preformed FAs) the changes in MF composition during the lactation may follow different patterns. As lactation progresses, the relative proportions of most de novo FAs (short- and medium-chain FAs) increase, whereas proportions of most preformed FAs (long-chain FAs) decrease (Palmquist et al., 1993; Secchiari et al., 2003; Kay et al., 2005; Komprda et al., 2005; Garnsworthy et al., 2006; Kgwatalala et al., 2009). In the case of C16:0, where only one half originates from de novo synthesis, the relationship follows the same pattern as that seen for the FA's synthesised completely de novo (Figure 1). Its content is the lowest during the initial days of lactation, while for C18:1 (Figure 2) it is at the highest level during this period. This is explained by the negative energy balance in dairy cows with an increased mobilization of long-chain FAs from adipose tissue reserves. Lake et al. (2007) reported that cows have a significant energy deficit during the initial 30 days of lactation in particular. The significant role of energy balance is emphasized by Stoop et al. (2009a). According to Bauman & Griinari (2003), the contribution of preformed FAs can vary from about 5% (when cows are in a good physiological state) to 20%.
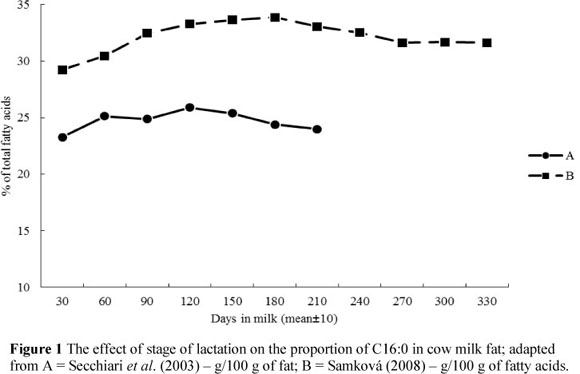
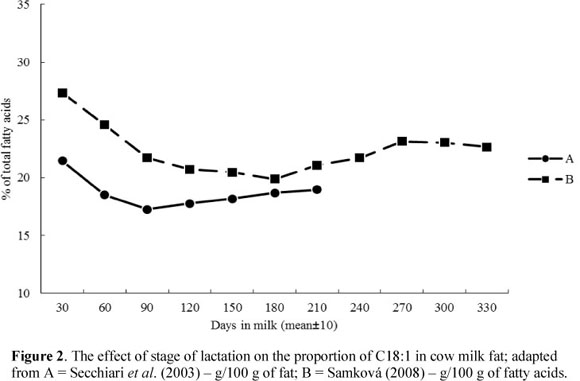
The odd- and branched-chain FAs with chain lengths of 14 and 15 carbon atoms followed the lactation curves of the short- and medium-chain FAs (increase in early lactation). In contrast, odd- and branched-chain FAs with a chain length of 17 carbon atoms follow the pattern of long-chain FAs, and showed a decrease during the early lactation period (Craninx et al., 2008). Levels of trans isomers of unsaturated FAs, including VA and CLA, were the lowest at early lactation and increased gradually (Figure 3). The highest proportions of VA and CLA were observed at the end of the lactation (Secchiari et al., 2003; Barlowska et al., 2005; Kay et al., 2005; Mele et al., 2007; Samková, 2008; Mele et al., 2009).
7. Milk and Fat Yield
Fatty acid composition has been related to milk production. Milk and fat yields are affected by individual animal factors such as breeds, individuality, parity, stage of lactation and milk production level. The relationships between the parameters of milk production (fat or protein contents, milk yield as well as fat and protein yields) and the FA proportion or FA yield appears to determine the understanding of the animal factor effects. Such relationships have been studied by numerous authors (e.g. Soyeurt et al., 2007; 2008b; Craninx et al., 2008; Schennink et al., 2008; Stoop et al., 2008).
An association between fat content and FA composition has been proven by Akerlind et al. (1999). They tested 48 Swedish Red and White cows selected for high or low milk fat content. Statistically significant differences were found mainly in proportions of FAs with carbon chains >C16, including palmitic (higher proportion in cows selected for high fat content), oleic, linoleic, linolenic acids and CLA (higher proportions in cows selected for low fat content). On the other hand, selection for milk yield decreased contents of milk protein and fat but had little effect on milk FA composition (Kay et al. , 2005; Bobe et al., 2007b).
Soyeurt et al. (2007) and Stoop et al. (2008) tested genetic correlations between milk yield, fat content, fat yield and FA composition and reported lower correlation between milk/fat yields and FA composition than between fat content and FAs (Table 5). However, Stoop et al. (2008) reported a relatively high correlation between milk yield and C16:0 (-0.50) and a moderate one between milk yield and C18:1 (+0.32). Nearly identical genetic correlations were reported in both the papers between fat content and FAs with the highest proportions of acids C16:0 (+0.60 and +0.65, respectively) and C18:1 (-0.78 and -0.63, respectively), supporting the perception that milk from breeds or individual cows with a high milk fat content have a nutritionally less desirable FA composition. The higher fat content was associated with a lower proportion of FAs >C18 and monounsaturated FAs as indicated by correlations of -0.72 and -0.22, respectively. Genetic correlations of fat content with C10 to C16 Δ9-desaturase indices were low but positive, whereas with C18 and CLA the indices were negative (Schennink et al. , 2008).
The positive genetic correlations observed by Soyeurt et al. (2008b) between the indices of C14, C16 and C18 (0.72; 0.62 and 0.97, respectively) and monounsaturated FAs showed that a proportion of the monounsaturated FAs is linked to SCD activity.
Several factors probably explain why the selection for milk production resulted in an increased proportion of de novo FAs to preformed FAs. For instance, relatively high genetic correlations were reported between individual FAs (Soyeurt et al., 2007; Stoop et al., 2008). Moderate genetic correlation coefficients were determined between milk yield and FAs and also between fat content and proportion of short- and medium-chain FAs. Furthermore, heritability estimates for these FAs seem to be higher than that for long-chain FAs.
Thus, it is important to pay attention to selection criteria because of their potential effects on various physiological processes (Veerkamp et al., 2003; Martin & Sauvant, 2007). Breeder associations usually use health and other parameters rather than milk yield (kg/d). It would be useful to take into consideration not only the main compositional parameters, fat and protein, but also FA composition or usage of gene-assisted selection as useful selection criteria. Furthermore, association with traits of other dairy cows, such as fertility and longevity, should be considered in the selection process.
The selection of individual cows according to their specific MF composition for particular milk products could be feasible if analytical methods to determine FA composition were available and cheaper than gas chromatography. Mid-infrared spectrometry analysis (Soyeurt et al., 2006b; Kaylegian et al., 2009) seems to be promising in this context. If FA composition could be used in a breeding programme, the ratio of saturated to unsaturated FAs or ratio of hypercholesterolaemic to unsaturated FAs seem to be acceptable selection criteria, though the use of the individual FAs would be too complicated.
Conclusions
Composition of cows' milk fat is influenced by numerous factors, including animal factors. Some of them can be utilized to improve the technological and nutritional properties of milk. It has been shown that FA composition can be affected to a large degree by cow individuality and stage of lactation, while breed and parity are factors of lower significance. Thus, desirable changes in fat composition can be achieved mainly through the factors of cow individuality and, to a lesser extent, breed. The utilization of these factors could be possible owing to the genetic variability in FA composition. The availability of data on genetic parameters (heritability, correlations) for the individual FAs and polymorphism of key enzymes SCD and DGAT can be used to achieve increased levels of nutritionally desirable FAs. Moreover, cows with increased SCD activity in the mammary gland could be selected for an increased production of monounsaturated FAs and CLA.
As the literature data indicate, processes in the rumen have extraordinary effects on FAs' proportion of milk fat. Thus, biochemical changes, especially biohydrogenation, should be studied in more detail.
It is necessary to keep in mind relationships of animal factor effects with milk production, resulting from genetic correlations between milk yield, fat content and proportions of FAs. Selection of cows for low fat content can result in a more desirable milk fat composition for human health, while selection for milk yield can affect the proportion of most individual FAs to a limited extent.
The results of the genetic research seem to hold promise for future efforts aimed at alteration of milk fat composition. Significant advances can be made by utilizing all available knowledge of genetic parameters and heritability concerning short- and medium-chain FAs and genetic polymorphism in medium-chain and unsaturated FAs.
Acknowledgement
The research was financially supported by the Ministry of Agriculture of the Czech Republic (Project MZe No. QH81210) and the Ministry of Education, Youth and Sports of the Czech Republic (Project MSM No. 6007665806). The helpful comments of anonymous reviewers are highly acknowledged.
References
Agabriel, C., Coulon, J.B., Journal, C. & Rancourt, B. de, 2001. Chemical composition of herd milk and farming system in the Massif Central. Prod. Anim. 14, 119-128 (in French, English abstract). [ Links ]
Ákerlind, M., Holtenius, K., Bertilsson, J. & Emanuelson, M., 1999. Milk composition and feed intake in dairy cows selected for high and low milk fat percentage. Livest. Prod. Sci. 59, 1-11. [ Links ]
Arnould, V.M.R. & Soyeurt, H., 2009. Genetic variability of milk fatty acids. J. Appl. Genet. 50, 29-39. [ Links ]
Auldist, M.J., Johnston, K.A., White, N.J., Fitzsimons, W.P., Boland, M.J., 2004. A comparison of the composition, coagulation characteristics and cheesemaking capacity of milk from Friesian and Jersey dairy cows. J. Dairy Res. 71, 51-57. [ Links ]
Bargo, F., Delahoy, J.E., Schroeder, G.F., Baumgard, L.H. & Muller, L.D., 2006. Supplementing total mixed rations with pasture increase the content of conjugated linoleic acid in milk. Anim. Feed Sci. Technol. 131, 226-240. [ Links ]
Barlowska, J., Litwinczuk, Z. & Topyla B., 2005. Physical-chemical parameters of milk from different cow breeds in spring-summer feeding period. Med. Weter. 61, 937-939 (in Polish, English abstract). [ Links ]
Barlowska, J., Grodzicki, T., Topyla, B. & Litwinczuk, Z., 2009. Physicochemical properties of milk fat from three breeds of cows during summer and winter feeding. Arch. Tierz.-Arch. Anim. Breed. 52, 356-363. [ Links ]
Bastin, C., Gengler, N. & Soyeurt, H., 2011. Phenotypic and genetic variability of production traits and milk fatty acid contents across days in milk for Walloon Holstein first-parity cows. J. Dairy Sci. 94, 4152-4163. [ Links ]
Bauman, D.E. & Griinari, J.M., 2003. Nutritional regulation of milk fat synthesis. Annu. Rev. Nutr. 23, 203-227. [ Links ]
Bauman, D.E., Mather, I.H., Wall, R.J. & Lock, A.L., 2006. Major advances associated with the biosynthesis of milk. J. Dairy Sci. 89, 1235-1243. [ Links ]
Bernal-Santos, G., Perfield II, J.W., Barbano, D.M., Bauman, D.E. & Overton, T.R., 2003. Production responses of dairy cows to dietary supplementation with conjugated linoleic acid (CLA) during the transition period and early lactation. J. Dairy Sci. 86, 3218-3228. [ Links ]
Bobe, G., Hammond, E.G., Freeman, A.E., Lindberg, G.L. & Beitz, D.C., 2003. Texture of butter from cows with different milk fatty acid compositions. J. Dairy Sci., 86, 3122-3127. [ Links ]
Bobe, G., Zimmerman, S., Hammond, E.G., Freeman, A.E., Porter, P.A., Luhman, C.M. & Beitz, D.C., 2007a. Butter composition and texture from cows with different milk fatty acid compositions fed fish oil or roasted soybeans. J. Dairy Sci. 90, 2596-2603. [ Links ]
Bobe, G., Lindberg, G.L., Freeman, A.E. & Beitz, D.C., 2007b. Short communication: Composition of milk protein and milk fatty acids is stable for cows differing in genetic merit for milk production. J. Dairy Sci. 90, 3955-3960. [ Links ]
Bobe, G., Bormann, J.A.M., Lindberg, G.L., Freeman, A.E. & Beitz, D.C., 2008. Short communication: Estimates of genetic variation of milk fatty acids in US Holstein cows. J. Dairy Sci. 91, 1209-1213. [ Links ]
Bradford, B.J. & Allen, M.S., 2004. Milk fat responses to a change in diet fermentability vary by production level in dairy cattle. J. Dairy Sci. 87, 3800-3807. [ Links ]
Caroll, S.M., DePeters, E.J., Taylor, S.J., Rosenberg, M., Perez-Monti, H. & Capps, V.A., 2006. Milk composition of Holstein, Jersey and Brown Swiss cows in response to increasing levels of dietary fat. Anim. Feed Sci. Technol. 131, 451-473. [ Links ]
Chilliard, Y., Ferlay, A. & Doreau, M., 2001. Effects of different types of forages, animal fat or marine oils in cow's diet on milk fat secretion and composition, especially conjugated linoleic acid (CLA) and polyunsaturated fatty acids. Livest. Prod. Sci. 70, 31-48. [ Links ]
Chilliard, Y., Glasser, F., Enjalbert, F., Ferlay, A. & Schmidely, P., 2008: Recent data on the effects of feeding factors on cow milk fatty acid composition. Sci. Aliments 28, 156-167. [ Links ]
Chouinard, P.Y., Corneau, L., Barbano, D.M., Metzger, L.E. & Bauman, D.E., 1999. Conjugated linoleic acids alter milk fatty acid composition and inhibit milk fat secretion in dairy cows. J. Nutr. 129, 1579-1584. [ Links ]
Conte, G., Mele, M., Chessa, S., Castiglioni, B., Serra, A., Pagnacco, G. & Secchiari, P., 2010. Diacylglycerol acyltransferase 1, stearoyl-CoA desaturase 1, and sterol regulatory element binding protein 1 gene polymorphisms and milk fatty acid composition in Italian Brown cattle. J. Dairy Sci. 93, 753-763. [ Links ]
Craninx, M., Steen, A., Van Laar, H., Van Nespen, T., Martin-Tereso, J., De Baets, B. & Fievez, V., 2008. Effect of lactation stage on the odd- and branched-chain milk fatty acids of dairy cattle under grazing and indoor conditions. J. Dairy Sci. 91, 2662-2677. [ Links ]
Croissant, A.E., Washburn, S.P., Dean, L.L. & Drake, M.A., 2007. Chemical properties and consumer perception of fluid milk from conventional and pasture-based production systems. J. Dairy Sci. 90, 4942-4953. [ Links ]
De Marchi, M., Bittante, G., Dal Zotto, R., Dalvit, C. & Cassandro, M., 2008. Effect of Holstein Friesian and Brown Swiss breeds on quality of milk and cheese. J. Dairy Sci. 91, 4092-4102. [ Links ]
Dhiman, T.R., Nam, S.H. & Ure, A.L., 2005. Factors affecting conjugated linoleic acid content in milk and meat. Crit. Rev. Food Sci. Nutr. 45, 463-482. [ Links ]
Drackley, J.K., Beaulieu, A.D. & Elliott, J.P., 2001. Responses of milk fat composition to dietary fat or nonstructural carbohydrates in Holstein and Jersey cows. J. Dairy Sci. 84, 1231-1237. [ Links ]
Edwards, R.A., King, J.W.B. & Yousef, I.M., 1973. Genetic variation in fatty-acid composition of cow milk. Anim. Prod. 16, 307-310. [ Links ]
Elgersma, A., Tamminga, S. & Ellen, G., 2006. Modifying milk composition through forage. Anim. Feed Sci. Technol. 131, 207-225. [ Links ]
Fearon, A.M., Mayne, C.S., Beattie, J.A.M. & Bruce, D.W., 2004. Effect of level of oil inclusion in the diet of dairy cows at pasture on animal performance and milk composition and properties. J. Sci. Food Agric. 84, 497-504. [ Links ]
Ferlay, A., Martin, B., Pradel, P., Coulon, J.B. & Chilliard, Y., 2006. Influence of grass-based diets on milk fatty acid composition and milk lipolytic system in Tarentaise and Montbéliarde cow breeds. J. Dairy Sci. 89, 4026-4041. [ Links ]
Ferlay, A., Martin, B., Lerch, S., Gobert, M., Pradel, P. & Chilliard, Y., 2010. Effects of supplementation of maize silage diets with extruded linseed, vitamin E and plant extracts rich in polyphenols, and morning vs. evening milking on milk fatty acid profiles in Holstein and Montbeliarde cows. Animal 4, 627-640. [ Links ]
Garnsworthy, P.C., Masson, L.L., Lock, A.L. & Mottram, T.T., 2006. Variation of milk citrate with stage of lactation and de novo fatty acid synthesis in dairy cows. J. Dairy Sci. 89, 1604-1612. [ Links ]
Gebauer, S.K., Psota, T.L. & Kris-Etherton, P.M., 2007. The diversity of health effects of individual trans fatty acid isomers. Lipids 42, 787-799. [ Links ]
German, J.B., Gibson, R.A., Krauss, R.M., Nestel, P., Lamarche, B., van Staveren, W.A., Steijns, J.M., de Groot, L., Lock, A.L. & Destaillats, F., 2009. A reappraisal of the impact of dairy foods and milk fat on cardiovascular disease risk. Eur. J. Nutr. 48, 191-203. [ Links ]
Gibson, J.P., 1991. The potential for genetic change in milk fat composition. J. Dairy Sci. 74, 3258-3266. [ Links ]
Glasser, F., Schmidely, R., Sauvant, D. & Doreau, M., 2008. Digestion of fatty acids in ruminants: a metaanalysis of flows and variation factors: 2. C18 fatty acids. Animal 2, 691-704. [ Links ]
Gnádig, S., Rickert, R., Sebedio, J.L. & Steinhart, H., 2001. Conjugated linoleic acid (CLA): physiological effects and production. Eur. J. Lipid Sci. Technol. 103, 56-61. [ Links ]
Goddard, M., 2001. Genetics to improve milk quality. Aust. J. Dairy Technol. 56, 166-170. [ Links ]
Harvatine, K.J., Boisclair, Y.R. & Bauman, D.E., 2009. Recent advances in the regulation of milk fat synthesis. Animal 3, 40-54. [ Links ]
Hradecká, E., Citek, J., Panicke, L., Rehout, V. & Hanusová, L., 2008. The relation of GH1, GHR and DGAT1 polymorphisms with estimated breeding values for milk production traits of German Holstein sires. Czech J. Anim. Sci. 53, 238-245. [ Links ]
Hunter, J.E., Zhang, J. & Kris-Etherton, P.M., 2010. Cardiovascular disease risk of dietary stearic acid compared with trans, other saturated, and unsaturated fatty acids: a systematic review. Am. J. Clin. Nutr. 91, 46-63. [ Links ]
Jensen, R.G., 2002. The composition of bovine milk lipids: January 1995 to December 2000. J. Dairy Sci. 85, 295-350. [ Links ]
Kalac, P. & Samková, E., 2010. The effects of feeding various forages on fatty acid composition of bovine milk fat: A review. Czech J. Anim. Sci. 55, 521-537. [ Links ]
Kay, J.K., Weber, W.J., Moore, C.E., Bauman, D.E., Hansen, L.B., Chester-Jones, H., Crooker, B.A. & Baumgard, L.H., 2005. Effects of week of lactation and genetic selection for milk yield on milk fatty acid composition in Holstein cows. J. Dairy Sci. 88, 3886-3893. [ Links ]
Kaylegian, K.E., Dwyer, D.A., Lynch, J.M., Bauman, D.E., Fleming, J.R. & Barbano, D.M., 2009. Impact of fatty acid composition on the accuracy of mid-infrared fat analysis of farm milks. J. Dairy Sci. 92, 2502-2513. [ Links ]
Kelsey, J.A., Corl, B.A., Collier, R.J. & Bauman, D.E., 2003. The effect of breed, parity, and stage of lactation on conjugated linoleic acid (CLA) in milk fat from dairy cows. J. Dairy Sci. 86, 2588-2597. [ Links ]
Kgwatalala, P.M., Ibeagha-Awemu, E.M., Hayes, J.F. & Zhao, X., 2007. Single nucleotide polymorphisms in the open reading frame of the stearoyl-CoA desaturase gene and resulting genetic variants in Canadian Holstein and Jersey cows. DNA Seq. 18, 357-362. [ Links ]
Kgwatalala, P.M., Ibeagha-Awemu, E.M., Mustafa, A.F. & Zhao, X., 2009. Stearoyl-CoA desaturase 1 genotype and stage of lactation influences milk fatty acid composition of Canadian Holstein cows. Anim. Genet. 40, 609-615. [ Links ]
Kliem, K.E., Aikman, P.C., Humphries, D.J., Morgan, R., Shingfield, K.J. & Givens, D.I., 2009. Effect of replacing calcium salts of palm oil distillate with extruded linseeds on milk fatty acid composition in Jersey and Holstein cows. Animal 3, 1754-1762. [ Links ]
Komprda, T., Dvorak, R., Fialová, M., Sustová, K. & Pechová, A., 2005. Fatty acid content in milk of dairy cows on a diet with high fat content derived from rapeseed. Czech J. Anim. Sci. 50, 311 -319. [ Links ]
Kramer, J.K.G., Parodi, P.W., Jensen, R.G., Mossoba, M.M., Yurawecz, M.P. & Adlof, R.O., 1998. Rumenic acid: A proposed common name for the major conjugated linoleic acid isomer found in natural products. Lipids 33, 835-835. [ Links ]
Kris-Etherton, P.M., Grieger, J.A. & Etherton, T.D., 2009. Dietary reference intakes for DHA and EPA. Prostaglandins Leukot. Essent. Fatty Acids 81, 99-104. [ Links ]
Lake, S.L., Weston, T.R., Scholljegerdes, E.J., Murrieta, C.M., Alexander, B.M., Rule, D.C., Moss, G.E. & Hess, B.W., 2007. Effects of postpartum dietary fat and body condition score at parturition on plasma, adipose tissue, and milk fatty acid composition of lactating beef cows. J. Anim. Sci. 85, 717-730. [ Links ]
Leiber, F., Kreuzer, M., Nigg, D., Wettstein, H.R., Richard, M. & Scheeder, L., 2005. A study on the causes for the elevated n-3 fatty acids in cows' milk of alpine origin. Lipids 40, 191-202. [ Links ]
Lock, A.L. & Garnsworthy, P.C., 2002. Independent effects of dietary linoleic and linolenic acids on the conjugated linoleic acid content of cow's milk. Anim. Sci. 74, 163-176. [ Links ]
Lock, A.L. & Bauman, D.E., 2004. Modifying milk fat composition of dairy cows to enhance fatty acids beneficial to human health. Lipids 39, 1197-1206. [ Links ]
Lopez-Huertas, E., 2010. Health effects of oleic acid and long chain omega-3 fatty acids (EPA and DHA) enriched milks. A review of intervention studies. Pharmacol. Res. 61, 200-207. [ Links ]
Malacarne, M., Summer, A., Formaggioni, P., Franceschi, P. & Mariani, P., 2001. Fatty acids composition of milk fat from four cattle breeds reared in the Parmigiano-Reggiano cheese production area. Ann. Fac. Med. Vet. - Universita di Parma 21, 249-259. (in Italian, English abstract) [ Links ]
Mántysaari, P., Khalili, H., Sariola, J. & Rantanen, A., 2007. Use of barley fibre and wet distillers' solubles as feedstuffs for Ayrshire dairy cows. Anim. Feed Sci. Technol. 135, 52-65. [ Links ]
Martin, O. & Sauvant, D., 2007. Dynamic model of the lactating dairy cow metabolism. Animal 1,1143-1166. [ Links ]
Mele, M., Conte, G., Castiglioni, B., Chessa, S., Macciotta, N.P.P., Serra, A., Buccioni, A., Pagnacco, G. & Secchiari, P., 2007. Stearoyl-coenzyme A desaturase gene polymorphism and milk fatty acid composition in Italian Holsteins. J. Dairy Sci. 90, 4458-4465. [ Links ]
Mele, M., Dal Zotto, R., Cassandro, M., Conte, G., Serra, A., Buccioni, A., Bittante, G. & Secchiari, P., 2009. Genetic parameters for conjugated linoleic acid, selected milk fatty acids, and milk fatty acid unsaturation of Italian Holstein-Friesian cows. J. Dairy Sci. 92, 392-400. [ Links ]
Mensink, R.P., 2005. Effects of stearic acid on plasma lipid and lipoproteins in humans. Lipids 40, 1201-1205. [ Links ]
Milanesi, E., Nicoloso, L. & Crepaldi, P., 2008. Stearoyl CoA desaturase (SCD) gene polymorphisms in Italian cattle breeds. J. Anim. Breed. Genet. 125, 63-67. [ Links ]
Miller, N., Delbecchi, L., Petitclerc, D., Wagner, G.F., Talbot, B.G. & Lacasse, P., 2006. Effect of stage of lactation and parity on mammary gland cell renewal. J. Dairy Sci. 89, 4669-4677. [ Links ]
Moioli, B., Contarini, G., Avalli, A., Catillo, G., Orru, L., De Matteis, G., Masoero, G. & Napolitano, F., 2007. Effect of stearoyl-coenzyme A desaturase polymorphism on fatty acid composition of milk. J. Dairy Sci. 90, 3553-3558. [ Links ]
Moore, C.E., Kay, J.K., Colllier, R.J., VanBaale, M.J. & Baumgard, L.H., 2005. Effect of supplemental conjugated linoleic acids on heat-stressed Brown Swiss and Holstein cows. J. Dairy Sci. 88, 1732-1740. [ Links ]
Morales, M.S., Palmquist, D.L. & Weiss, W.P., 2000. Milk fat composition of Holstein and Jersey cows with control or depleted copper status and fed whole soyabeans or tallow. J. Dairy Sci. 83, 2112-2119. [ Links ]
Mosley, E.E., Shafii, B., Moate, P.J. & McGuire, M.A., 2006. Cis-9, trans-11 conjugated linoleic acid is synthesized directly from vaccenic acid in lactating dairy cattle. J. Nutr. 136, 570 -575. [ Links ]
Nafikov, R.A. & Beitz, D.C., 2007. Carbohydrate and lipid metabolism in farm animals. J. Nutr. 137, 702-705. [ Links ]
O'Brien, M.D., Wheelock, J.B., La Noce, A.J., Rhoads, M.L., Rhoads, R.P., VanBaale, M.J., Collier, R.J. & Baumgard, L.H., 2007. Effects of heat stress vs. underfeeding on milk fatty acid composition. J. Anim. Sci. 85, 58-59. [ Links ]
Ordovas. L., Zaragoza, P., Altarriba, J. & Rodellar, C., 2008. Identification of 14 new single nucleotide polymorphisms in the bovine SLC27A1 gene and evaluation of their association with milk fat content. J. Dairy Res. 75, 129-134. [ Links ]
Palladino, R.A., Buckley, F., Prendiville, R., Murphy, J.J., Callan, J. & Kenny, D.A., 2010. A comparison between Holstein-Friesian and Jersey dairy cows and their F-1 hybrid on milk fatty acid composition under grazing conditions. J. Dairy Sci. 93, 2176-2184. [ Links ]
Palmquist, D.L., Beaulieu, A.D. & Barbano, D.M., 1993. Feed and animal factors influencing milk fat composition. J. Dairy Sci. 76, 1753-1771. [ Links ]
Parodi, P.W., 2004. Milk fat in human nutrition. Aust. J. Dairy Technol. 59, 3-59. [ Links ]
Perdrix, M.F., Sutter, F. & Wenk, C., 1996. The fatty acid composition of milk fat: factors of variation. Rev. Suisse Agric. 28, 71-76. (in French, English abstract) [ Links ]
Pesek, M., Spicka, J. & Samková, E., 2005. Comparison of fatty acid composition in milk fat of Czech Pied cattle and Holstein cattle. Czech J. Anim. Sci. 50, 122-128. [ Links ]
Pesek, M., Samková, E. & Spicka, J., 2006. Fatty acids and composition of their important groups in milk fat of Czech Pied cattle. Czech J. Anim. Sci. 51, 181-188. [ Links ]
Precht, D. & Molkentin, J., 2000. Frequency distributions of conjugated linoleic acid and trans fatty acids contents in European bovine milk fats. Milchwisseschaft 55, 687-691. [ Links ]
Rajakangas, J., Basu, S., Salminen, I. & Mutanen, M., 2003. Adenoma growth stimulation by the trans-10, cis-12 isomer of conjugated linoleic acid (CLA) is associated with changes in mucosal NF-kappa B and cyclin D1 protein levels in the Min mouse. J. Nutr. 133, 1943-1948. [ Links ]
Renner, E. & Kosmack, U., 1974. Genetische Aspekte zur Fettsáurenzusammensetzung des Milchfettes. 2. Fettsáurenmuster der Milch von Nachtkommenpopulationen. Züchtungskunde 46, 217-226. [ Links ]
Samková, E., 2008. The effects of selected factors on fatty acid composition in cow milk fat. Thesis, Mendel University of Agriculture and Forestry in Brno, Czech Republic. 186 pp. (in Czech, English summary). [ Links ]
Schennink, A., Heck, J.M.L., Bovenhuis, H., Visker, M.H.P.W., Van Valenberg, H.J.F. & Van Arendonk, J.A.M., 2008. Milk fatty acid unsaturation: Genetic parameters and effects of stearoyl-CoA desaturase (SCD1) and acyl CoA: diacylglycerol acyltransferase 1 (DGAT1). J. Dairy Sci. 91, 2135-2143. [ Links ]
Schennink, A., Stoop, W.M., Visker, M.H.P.W., Van der Poel, J.J., Bovenhuis, H. & Van Arendonk, J.A.M., 2009. Genome-wide scan for bovine milk-fat composition. II. Quantitative trait loci for long-chain fatty acids. J. Dairy Sci. 92, 4676-4682. [ Links ]
Secchiari, P., Mele, M., Serra, A., Buccioni, A., Paoletti, F. & Antongiovanni, M., 2003. Effect of breed, parity and stage of lactation on milk conjugated linoleic acid content in Italian Friesian and Reggiana cows. Ital. J. Anim. Sci. 2, 269-271. [ Links ]
Shingfield, K.J., Salo-Váánánen, P., Pahkala, E., Toivonen, V., Jaakkola, S., Piironen, V. & Huhtanen, P., 2005. Effect of forage conservation method, concentrate level and propylene glycol on the fatty acid composition and vitamin content of cows'mik. J. Dairy Res. 72, 349-361. [ Links ]
Siri-Tarino, P.W., Sun, Q., Hu, F.B. & Krauss, R.M., 2010. Saturated fat, carbohydrate, and cardiovascular disease. Am. J. Clin. Nutr. 91, 502-509. [ Links ]
Soyeurt, H., Dardenne, P., Gillon, A., Croquet, C., Vanderick, S., Mayeres, P., Bertozzi, C. & Gengler, N., 2006a. Variation in fatty acid contents of milk and milk fat within and across breeds. J. Dairy Sci. 89, 4858-4865. [ Links ]
Soyeurt, H., Dardenne, P., Dehareng, F., Lognay, G., Veselko, D., Marlier, M., Bertozzi, C., Mayeres, P. & Gengler, N., 2006b. Estimating fatty acid content in cow milk using Mid-infrared spectrometry. J. Dairy Sci. 89, 3690-3695. [ Links ]
Soyeurt, H., Gillon, A., Vanderick, S., Mayeres, P., Bertozzi, C. & Gengler, N., 2007. Estimation of heritability and genetic correlations for the major fatty acids in bovine milk. J. Dairy Sci. 90, 4435-4442. [ Links ]
Soyeurt, H., Dardenne, P., Dehareng, F., Bastin, C. & Gengler, N., 2008a. Genetic parameters of saturated and monounsaturated fatty acid content and the ratio of saturated to unsaturated fatty acids in bovine milk. J. Dairy Sci. 91, 3611-3626. [ Links ]
Soyeurt, H., Dehareng, F., Mayeres, P., Bertozzi, C. & Gengler, N., 2008b. Variation of Delta(9)-desaturase activity in dairy cattle. J. Dairy Sci. 91, 3211-3224. [ Links ]
Stoop, W.M., Van Arendonk, J.A.M., Heck, J.M.L., Van Valenberg, H.J.F. & Bovenhuis, H., 2008. Genetic parameters for major milk fatty acids and milk production traits of Dutch Holstein-Friesians. J. Dairy Sci. 91, 385-394. [ Links ]
Stoop, W.M., Bovenhuis, H., Heck, J.M.L. & Van Arendonk, J.A.M., 2009a. Effect of lactation stage and energy status on milk fat composition of Holstein-Friesian cows. J. Dairy Sci. 92, 1469-1478. [ Links ]
Stoop, W.M., Schennink, A., Visker, M.H.P.W., Mullaart, E., Van Arendonk, J.A.M. & Bovenhuis, H., 2009b. Genome-wide scan for bovine milk-fat composition. I. Quantitative trait loci for short- and medium-chain fatty acids. J. Dairy Sci. 92, 4664-4675. [ Links ]
Talpur, F.N., Bhanger, M.I. & Khuhawar, M.Y., 2006. Comparison of fatty acids and cholesterol content in the milk of Pakistani cow breeds. J. Food Compos. Anal. 19, 698-703. [ Links ]
Thomson, N.A., Van der Poel, W. & Peterson, S.W., 2000. Seasonal variation of the fatty acid composition of milk fat from Friesian cows grazing pasture. Proc. N. Z. Soc. Anim. Prod. 60, 314-317. [ Links ]
Veerkamp, R.F., Beerda, B. & Van der Lende, T., 2003. Effects of genetic selection for milk yield on energy balance, levels of hormones, and metabolites in lactating cattle, and possible links to reduced fertility's. Livest. Prod. Sci. 83, 257-275. [ Links ]
Vlaeminck, B., Fievez, V., Cabrita, A.R.J., Fonseca, A.J.M. & Dewhurst, R.J., 2006. Factors affecting odd-and branched-chain fatty acids in milk: A review. Anim. Feed Sci. Technol. 131, 389-417. [ Links ]
Wang, J.P., Bu, D.P., Wang, J.Q., Huo, X.K., Guo, T.J., Wei, H.Y., Zhou, L.Y., Rastani, R.R., Baumgard, L.H. & Li, F.D., 2010. Effect of saturated fatty acid supplementation on production and metabolism indices in heat-stressed mid-lactation dairy cows. J. Dairy Sci. 93, 4121-4127. [ Links ]
Wathes, D.C., Cheng, Z., Bourne, N., Taylor, V.J., Coffey, M.P. & Brotherstone, S., 2007. Differences between primiparous and multiparous dairy cows in the inter-relationships between metabolic traits, milk yield and body condition score in the periparturient period. Domest. Anim. Endocrinol. 33, 203-225. [ Links ]
White, S.L., Bertrand, J.A., Wade, M.R., Washburn, S.P., Green Jr., J.T. & Jenkins, T.C., 2001. Comparison of fatty acid content of milk from Jersey and Holstein cows consuming pasture or total mixed ration. J. Dairy Sci. 84, 2295-2301. [ Links ]
Zegarska, Z., Jaworski, J., Paszczyk, B., Charkiewicz, J. & Borejszo, Z., 2001. Fatty acid composition with emphasis on trans C18:1 isomers of milk fat from Lowland and Black and White and Polish Red cows. Pol. J. Food Nutr. Sci. 10, 41-44. [ Links ]
Copyright resides with the authors in terms of the Creative Commons Attribution 2.5 South African Licence. See: http://creativecommons.org/licenses/by/2.5/za Condition of use: The user may copy, distribute, transmit and ad apt the work, but must recognise the authors and the South African Journal of Animal Science.
# Corresponding author: samkova@zf.jcu.cz














