Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
South African Journal of Animal Science
versión On-line ISSN 2221-4062
versión impresa ISSN 0375-1589
S. Afr. j. anim. sci. vol.42 no.1 Pretoria ene. 2012
The performance of broilers on a feed depends on the feed protein content given previously
R.M. Gous#; G.C. Emmans; C. Fisher
Animal and Poultry Science, University of KwaZulu-Natal, Pietermaritzburg, South Africa Scottish Agricultural College, Bush Estate, Midlothian, EH26 0QE, Scotland
ABSTRACT
It has been proposed that all animals have an inherent relationship between body protein and lipid that can be described allometrically, and the hypothesis tested in the research reported here is that the animal will at all times attempt to retain this relationship. The test was accomplished by feeding broilers, of three genotypes and in two experiments, in such a way as to produce lean and fat birds that were then subjected to a range of dietary protein levels in a second feeding period, during which their performance was measured. Birds were initially offered one of two feeds with widely different protein to energy ratios until they reached a pre-defined liveweight, after which they were given one of two feed protein contents in Experiment 1 and four in Experiment 2. Their performance was monitored until a second pre-defined liveweight was reached, at which time they were killed for carcass analysis. The genotype selected to be lean, in Experiment 1, showed no response to protein level in the second period, whether they were fat or lean at the start. Conversely, the genetically fat birds showed some additional growth in males and additional efficiency in the females. Averaged across genotypes and sexes, birds initially in the fat state gained only 6.9 g lipid/d versus 13.5 g lipid/d for the nutritionally lean broilers. In Experiment 2, growth rate and feed conversion efficiency (FCE) were related directly to dietary protein content and were higher for those birds made nutritionally fat. Carcass lipid gain was lower for the initially fat birds on the three highest dietary protein treatments. All birds made fat at 880 g and 1000 g, by giving them a low protein feed, had a much reduced fat content in their subsequent gain, provided that the protein content of the feed used was sufficiently high, indicating that they were making use of the excessive lipid reserves as an energy source. The hypothesis tested cannot be rejected by the evidence presented.
Keywords: Fatness, broiler nutrition, dietary protein content, body lipid : protein ratio
Introduction
Experimental evidence indicates that pigs that are fatter than their desired level show a reduction in lipid gain if given a feed that is adequate in dietary protein (Kyriazakis & Emmans, 1991; Kyriazakis et al, 1991; Stamataris et al, 1991; Ferguson & Theeruth, 2002). It has been proposed by these authors that this response will continue until the degree of fatness has returned to levels similar to those observed in animals that have been unrestricted or that have followed normal growth. Ferguson & Theeruth (2002) presented supporting evidence in the literature for the proposition that although a maximum limit is set on the growth of body protein for a given degree of maturity, no such limit exists for lipid deposition. Emmans (1981) and Whittemore (1998) have alluded to body lipid reserves as being labile, fluctuating on account of feed level, previous feed offered, and environmental conditions.
When growing chickens are given ad libitum access to feeds of low first-limiting amino acid to energy ratio, they take more time and eat more feed energy to reach a given weight, and are also fatter at that weight (Jackson et al., 1982; Gous et al., 1990). Feeds that have these effects may be used in practice, either by accident or by design. The performance of chickens following such treatment is thus of practical interest, as well as theoretical. Although this is a general problem in growth theory, it has become more relevant with the development of growth simulation models, where the state of the animal at any stage of growth is seen as the consequence of the food and environment to which it has been subjected to that point, and which must then impact on subsequent food intake and chemical growth (Emmans, 1981). Eits et al. (2003) showed that responses of broilers to dietary protein depend on previous protein nutrition and sex, and as a result, suggested that protein levels in grower and finisher diets should be optimised simultaneously, not independently.
Any growth model may be seen, in large part, as proposing solutions to two problems: (i) What will be the rates of growth and feed intake in non-limiting nutritional and environmental conditions, and what are these conditions for a given genotype? (ii) What will be the rates of growth and feed intake if either the nutritional or environmental conditions are limiting? But there is a further problem, which has received little theoretical attention, despite the enormous amount of experimental literature (see reviews by Wilson & Osbourn, 1960; Donovan, 1984; Berge, 1991; Ryan et al., 1993 among others): (iii) Following a period in which potential growth is not achieved because of nutritional or environmental constraints, what will be the rates of growth and feed intake on the return to non-limiting conditions, and what are these conditions?
The main hypothesis that underlies the research reported here is that an animal will, at all times, attempt to restore the lipid : protein ratio at any given protein weight which is that set by its genotype. It will do this subject to maximizing its rate of protein growth. Were this not the case, the prediction of voluntary food intake by a bird or animal given ad libitum access to food would be more difficult, as this rule is central to Emmans' (1981) food intake theory in governing the amount of body lipid that might be utilised as an energy source. The fatness of an animal has been shown to affect its voluntary food intake (Foot, 1972; Sibbald & Rhind, 1997), so an accurate prediction of changes in body lipid content during growth is necessary for forecasting subsequent voluntary food intake.
Materials and Methods
Two experiments dealing with the influence of the state of a broiler on its response to protein were conducted at the Poultry Research Institute in Roslin, Scotland. In the first experiment, 100 chicks of two strains were reared in tier brooders from day old, before being transferred to single cages at about 900 g liveweight. At this weight, three or four birds of each sex of each genotype from each of the initial feeds (see below) were killed and their total bodies, including feathers, were chemically analysed. The experiment was of a factorial design with 2 x 2 x 2 x 2 = 16 treatments, with each replicated on three chickens housed individually (n = 48). The factors were strains, which were the lean and fat broiler lines developed by Whitehead (1990); sexes; feed protein contents of 124 (LP) g CP/kg or 310 (HP) g CP/kg, fed from day old to 900 g; and feed protein contents from 900 g liveweight, which were HP or MP (248 g CP/kg). For clarity, the design used can be viewed in Table 4.
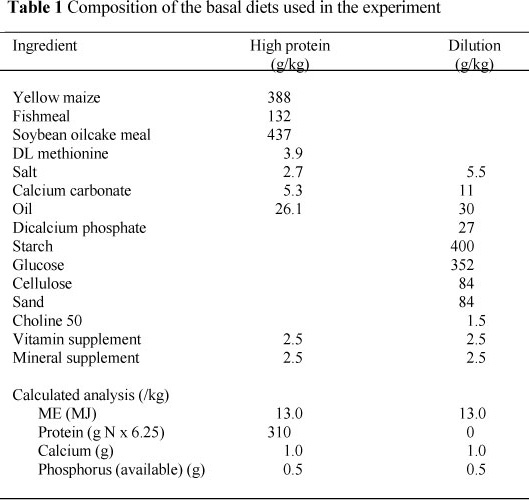
At the nominal (pre-defined) liveweight of 1750 g, all birds in the second period of the experiment were killed and their total bodies, including feathers, were chemically analysed. The birds were fed ad libitum throughout. From 900 g to 1750 g the birds were weighed and their feed intakes measured daily. The feeds used are shown in Table 1. LP contained 0.4 of HP and 0.6 of the dilution feed, while MP contained 0.8 of HP and 0.2 of the dilution feed.
In trial 2, Ross female broiler chicks were reared in tier brooders before being transferred to single cages at a nominal (pre-defined) liveweight of 1000 g. They were fed LP or HP up to this weight. At 1000 g, nine chicks from each of the two early feeds were killed and their total bodies analysed. A further 48 birds, 24 from each initial feed, were transferred to one of four feed protein contents, which were LP, MP and HP, as before, and LMP, which had 186 g CP/kg and was a mixture of 0.6 of HP and 0.4 of the dilution feed. Each of the 2 first feeds x 4 second feed treatments was replicated on six birds, of which two were killed at each of the nominal slaughter weights of 1500 and 2000 g. As with the chicks sampled at 1000 g, the total bodies of these sampled birds were chemically analysed. From 1000 g to the nominal slaughter weights, the birds were weighed and their feed intakes measured daily.
The temperature of the room in which the birds were kept during the second phase of each experiment was maintained at 22 °C to 25 °C.
The composition of the high protein feed (HP) is shown in Table 1. All other feeds used in the two phases of the two experiments were produced by blending HP with a protein-free diluent, also shown in Table 1. This procedure ensured that all feeds contained the same amino acid balance. The two basal feeds contained the same concentrations of energy, major and minor minerals, and vitamins.
Each carcass, including feathers, was minced in order to analyse the water, fat, protein and ash contents, using the AOAC (1990) procedures.
Keeping birds to 2500 g liveweight in cages led to some problems with leg weakness. Because some birds had to be culled for this reason, the full design of Experiment 2 was not met. The liveweight intervals used were 1000 - 1500 g and 1500 - 2000 g. The data were analysed using Minitab's general linear model (GLM), where all terms were fitted. The growth and intake variables analysed were as follows: Experiment 1, daily feed intake, daily gain and feed efficiency; Experiment 2, daily feed intake, daily gain and feed efficiency for the weight intervals 1000 - 1500 g and 1500 - 2000 g. Body composition data were analysed: Experiment 1, at 900 and 1800 g liveweight; Experiment 2, at 1000 g, 1500 g and 2000 g. Gains of the chemical components were analysed for the 900 - 1800 g interval for Experiment 1 and for the 1000 - 1500 g and 1500 - 2000 g intervals for Experiment 2.
Results
In the first experiment there were no significant differences between treatments in the liveweights at which the second period treatments were started. The mean value of 896 g was close to the target value of 900 g. The LP birds took 34.5 d to reach this weight from day old, which was longer (P <0.01) than the 27.1 d taken by the HP birds. There were no appreciable or significant effects of sex or genotype on the time taken.
The mean final slaughter weight was 1781 g, again close to the intended value of 1750 g. As there were no significant treatment effects in initial or final weight, all treatments were compared, as was intended, over the same liveweight range.
Rate of gain in the second period was faster (P <0.01) in the lean genotype, by 6.7 g/d (or 1.9 d less time), than in the fat, and in the males, which grew 13.2 g/d faster (or 4.3 d less time) than the females (Table 2). There were no significant effects of prior or current feed protein content on growth rate, and no interaction was significant. The growth of the birds from HP and from LP, averaged over all other factors, is illustrated in Figure 1. The absence of any compensatory growth is clear.
The main effects of genotype, prior feed protein content, sex and current dietary protein content on feed intake were all statistically significant (P <0.05), as shown in Table 2. The daily rate of feed consumption was higher for the fat genotype, for birds originally fed LP, for males, and for the birds subsequently fed MP (248 g CP/kg). Although there was no difference in feed intake by the fat line between those fed the HP and the MP feeds (126.8 vs. 127.0 g/d respectively), the difference in feed intake by birds of the lean line on the two feeds was large (107 vs. 127 g/d). This was the only interaction that was significant (P <0.05).
All of the main effects on FCE were statistically significant (P <0.01), with higher FCE values occurring in the lean line, the birds initially fed HP, the males, and those subsequently fed HP. The only first-order interaction that was significant was that between genetic line and the protein content of the feed given in the first period. There was virtually no difference in FCE between the birds of the fat line that had been given LP or HP in the first period (401 vs. 405 g gain/kg feed respectively). But the birds of the lean line that had been fed HP showed a considerably increased FCE (P <0.01) of 566, compared with a value of 445 g/kg for those of the same genotype that had been fed LP in the first period.
As was intended, the birds from LP contained more lipid (P <0.01) than those given HP (135 vs. 74 g lipid/kg) at the end of the first period, as shown in Table 3. The subsequent feeding treatments produced significant (P <0.01) differences in carcass composition at the end of the second period of the experiment. Water, ash and protein contents were higher for the lean line than for the fat line, and the lipid content was considerably lower, 111 vs. 181 g lipid/kg (Table 4). Females had higher lipid contents in their carcasses than males. The high protein diet in the second period resulted in carcasses with higher water and protein contents, but with a lower lipid content (134 g lipid/kg vs. 159) than broilers fed the medium protein feed.
From the point of view of this experiment, the most important result was the marked reduction in the rate of lipid gain in Phase 2 of the experiment for the birds made fat at 900 g by being fed LP. They gained only 6.9 g lipid/d compared with those from HP, which gained 13.5 g lipid/d (P <0.01). The lipid content in the gain was only 128 g/kg compared to 237 (P <0.01). Consequently, the birds previously fed LP contained less body lipid (135 vs. 158 g lipid/kg) than those from HP at the end of the trial.
In the second experiment, the time taken to reach the starting weight of 1000 g (Table 5) differed significantly (P <0.01) between the LP (30.5d) and the HP (24.7d) treatments. The mean weights at the start of the second period were very similar for the LP treatment, 1025 g, and the HP, 1038 g. Mortalities and culling resulted in few birds remaining for the period from 2000 g to 2500 g liveweight, so only the results of the two periods, from 1000 g to 1500 g and from 1500 g to 2000 g liveweight, are reported below.
The birds from LP grew faster (P <0.01) (Table 5) than those from HP in the 1000 to 1500 g liveweight interval. Growth rate was also related directly to dietary protein content in both periods. Only the birds from HP that were continued on HP did not follow the common trend; these birds grew more slowly, 55.0 g/d, than the birds on the next lowest protein content, 61.0 g/d. Statistically significant differences in growth rate on the second feed disappeared in the period 1500 g to 2000 g liveweight, although the trend remained (P <0.01) in those birds initially fed on LP.
Mean feed intake in the first liveweight interval increased linearly (114, 115, 119 and 121 g/d, respectively) as the protein content in the second feed declined (P <0.01) (Table 5), but this was not evident in the second period. Feed conversion efficiency in the period 1000 to 1500 g liveweight was higher (P <0.01) for birds that had been fed LP in the first period, and declined as the dietary protein content was reduced. Differences in FCE between those birds from LP and those from HP were no longer evident in the period 1500 -2000 g liveweight, although significant differences remained between birds on feeds of the different protein contents (Table 5).
At the end of the initial period the LP birds had 154, and the HP birds 74 g lipid/kg carcass (Table 3). Although the birds from LP remained fatter (P <0.05) at 1500 g liveweight than those from HP (169 vs. 135 g lipid/kg) (Table 6) their rate of gain in lipid during this period was lower on the three highest dietary protein treatments (overall, 11.8 vs. 14.3 g/d). During the period 1500 g to 2000 g liveweight, their lipid gains were also lower (P <0.05), 9.8 vs. 13.8 g/d (Table 7).
Overall, birds that were made fat by giving them a low protein feed had a much-reduced fat content in their subsequent gain, provided that the protein content of the feed was sufficiently high (Experiment 2). In Experiment 1, lipid was 0.08 and 0.18 of the gain in the lean and fat lines that had been made fat initially, but 0.19 and 0.30 of the gain in the two strains kept lean; in Experiment 2, the proportion of lipid in the gain was 0.15 for birds made fat and then fed the highest protein in the period 1000 - 1500 g liveweight versus 0.25 for those birds that were lean at 1000 g liveweight, but the equivalent proportions for birds fed the lowest dietary protein feed were 0.31 and 0.33. Over all the dietary protein treatments, the proportions of lipid in the gain were 0.20 and 0.26 in the first period of Phase 2, and in the second period, 0.16 and 0.23, respectively.
There were differences in the carcass water contents at 1500 g and 2000 g liveweight (Table 6) between broilers on the various dietary treatments and of different initial fatnesses, but the carcass ash and protein contents showed no consistent differences between treatments. However, because of the differences in carcass composition between treatments at the start of Phase 2 of this experiment, gains in carcass water, ash and protein all showed highly significant differences between treatments (Table 7). Carcass water gains differed (P <0.01) between dietary treatments in both periods of Phase 2, with the highest gains being produced by the highest dietary protein contents, and between initial states in the period 1000 g - 1500 g liveweight (39.4 g/d vs. 33.7 g/d), the initially fat birds producing the higher rate of gain; the carcass protein gains generally following the dietary protein content, and being higher for birds that were fat initially (13.0 vs. 10.7 g/d in the period 1000 g -1500 g liveweight).
Discussion
The consumption of food by an animal will dictate the amount of protein and lipid that will be deposited each day, but food intake in turn is dependent, among other factors, on the state of the animal at the time. Emman's (1981) food intake theory suggested that if a broiler is fatter than its inherent fatness, as defined by its genetically determined lipid : protein ratio, then it will attempt to use the extra body lipid as an energy source if and when this is possible. If the food contains a higher protein : energy ratio than that required to maximise protein growth, and the bird contains excess fat, food intake will be reduced to the point at which just sufficient of the first-limiting nutrient is consumed, with the associated energy, and the resultant shortfall in energy will be obtained by mobilising body lipid reserves. This increases the efficiency of utilization of the feed for growth. If there is no excess lipid, then the bird would have to increase food intake, and hence energy, accordingly.
Kyriazakis & Emmans (1992), in considering the growth of animals after a period of nutritional limitation, proposed that any perturbations in body composition at the end of that period will be corrected over time, and that the rates of such corrections are always set by the conditions of rehabilitation. These propositions imply that body lipid content is labile, and the evidence they presented is consistent with that presented here, as are the results of Eits et al. (2003).
A similar model has been described by Broekhuizen et al. (1994), which invokes a measure of well being corresponding closely to the fatness of the animal and is defined as the ratio of the weight of mobilizable reserve tissue to that of structural tissue, the latter being assumed to correlate with skeletal size. In their model, the animal (fish) responds immediately to changes in the reserves : structural tissue ratio (similar to the lipid : protein ratio used here), this ratio depending on feeding history, which provides a simple, implicit memory. Of particular relevance, the key assumption in their model is that the animal has an 'ideal' reserve : structure ratio which it seeks to maintain, almost identical to Emmans' (1981) theory. Small deviations from the ideal are corrected by changes in the proportion of assimilate allocated to the two pools, but major changes are seen as indicating persistent adverse environmental conditions and trigger a protective response.
The results from the experiments reported here throw some light on the problem of growth after a period of under-feeding of protein. There were clear differences between the genotypes used, including sexes. The genotype selected to be lean showed no differences in growth or efficiency, while the genetically fat birds showed some improvement in growth in the males and efficiency in the females. In the commercial females, enhanced growth and efficiency were both shown, provided that a feed with a high enough protein content was used. Differences between strains do occur, although the responses are not always consistent: Cherry et al. (1978) had previously noted that the slower growing broiler strains in their study exhibited more compensatory gain than the faster growing strains, whereas Marks (1978) found that fast growing quail lines were capable of demonstrating greater compensatory growth after a protein restriction than non-selected control quail.
The males and females of the genetically fat strain and the commercial females may be seen as more 'normal' genotypes than the genetically lean strain. In these three normal genotypes, feed efficiency improved when the birds that had been made fat nutritionally were subsequently given a high protein feed. The reason for this, as shown by the body analysis, was straight forward: Birds made fat at the start by giving them a low protein feed had a much-reduced fat content in their subsequent gain, provided that the protein content of this feed was sufficiently high.
From a simulation modelling perspective, it is useful to see the animal as having an inherent lipid : protein ratio, which it attempts to maintain, implying that the animal will be able to consume a feed, or a combination of feeds, that will allow it to retain this ratio, and that at all times it will attempt to correct this ratio if it has previously been made to deviate from it through incorrect feeding in a given environment. This it does by utilizing the excess body lipid as an energy source, provided that the dietary protein content is sufficiently high to allow this. Presumably the rate at which it can make use of this body lipid will depend also on the prevailing environmental temperature and other 'conditions of rehabilitation', as proposed by Kyriazakis & Emmans (1992). The hypothesis that an animal will at all times attempt to retain its inherent lipid : protein ratio cannot be rejected by the evidence presented above.
Conclusions
The evidence presented here suggests that, like growing pigs, broilers will at all times attempt to maintain a genetically determined lipid : protein ratio by utilizing excess body lipid as an energy source when the supply of the limiting nutrient is sufficient to allow this. This means that the optimum protein content of feeds for broilers in each phase of the growing period should not be determined independently of the other feeds being used in the feeding programme.
References
AOAC, 1990. Official Methods of Analysis. (Vol. 1. 15th ed.). Association of Official Analytical Chemists, Virginia, USA. [ Links ]
Broekhuizen, N., Gurney, W.S.C., Jones, A. & Bryant, A.D., 1994.Modelling compensatory growth. Func. Ecol.8, 770-782. [ Links ]
Cherry, J.A., Siegel, P.B. & Beane, W.L., 1978. Genetic nutritional relationships in growth and carcass characteristics of broiler chickens. Poult. Sci. 57, 1482-1487. [ Links ]
Eits, R.M., Kwakkel, R.P., Verstegen, M.W.A. & Emmans, G.C., 2003. Responses of broiler chickens to dietary protein: Effects of early life protein nutrition on later responses. Br. Poult. Sci. 44, 398-409. [ Links ]
Emmans, G.C., 1981. A model of the growth and feed intake of ad libitum fed animals, particularly poultry. In: Computers in Animal Production. pp. 103-110. Occ. Publ. No. 5. Br. Soc. Anim. Prod. [ Links ]
Ferguson, N.S. & Theeruth, B.K., 2002. Protein and lipid deposition rates in growing pigs following a period of excess fattening. S. Afr. J. Anim. Sci. 32, 97-105. [ Links ]
Foot, J.Z., 1972. A note on the effect of body condition on the voluntary intake of dried grass wafers by Scottish Blackface ewes. Anim. Prod. 14, 131-134. [ Links ]
Gous, R.M., Emmans, G.C., Broadbent, L.A. & Fisher, C., 1990. Nutritional effects on the growth and fatness of broilers. Br. Poult. Sci. 31, 495-505. [ Links ]
Jackson, S., Summers, J.D. & Leeson, S., 1982. Effect of dietary protein and energy on broiler carcass composition and efficiency of nutrient utilization. Poult. Sci. 61, 2224-2228. [ Links ]
Jones, G.D.P. & Farrell, D.J., 1989. Reducing body fat in broiler chickens and some physiological consequences. S. Afr. J. Anim. Sci. 19, 179-183. [ Links ]
Kyriazakis, I. & Emmans, G.C., 1992. The growth of mammals following a period of nutritional limitation. J.Theor. Biol. 156, 485-498. [ Links ]
Kyriazakis, I., Stamataris, C., Emmans, G.C. & Whittemore, C.T., 1991. The effects of food protein content on the performance of pigs previously given foods with low or moderate protein contents. Anim. Prod. 52,165-173. [ Links ]
Marks, H.L., 1978. Compensatory growth in Japanese quail following protein restriction. Poult. Sci. 57,1473-1477. [ Links ]
Pesti, G.M. & Fletcher, D.L., 1984. The response of male broiler chickens to diets with various protein contents, during the grower and finisher phases. Br. Poult. Sci. 25, 415-423. Plavnik, I. & Hurwitz, S., 1988. Early feed restriction in chicks: effect of age, duration, and sex. Poult. Sci. 67,384-390. [ Links ] [ Links ]
Plavnik, I. & Hurwitz, S., 1989. Effect of dietary protein, energy, and feed pelleting on the response of chicks to early feed restriction. Poult. Sci. 68, 1118-1125. [ Links ]
Ryan, W.J., Williams, I.H. & Moir, R.J., 1993. Compensatory growth in sheep and cattle. II. Changes in body composition and tissue weights. Aust. J. Agric. Res. 44, 1623-1638. [ Links ]
Sibbald, A.M. & Rhind, S.M., 1997. The effect of previous body condition on appetite and associated insulin profiles in sheep. Anim. Sci. 64, 247-252. [ Links ]
Stamataris, C., Kyriazakis, I. & Emmans, G.C., 1991. The performance and body composition of young pigs following a period of growth retardation by food restriction. Anim. Prod. 53, 373-381. [ Links ]
Whitehead, C.C., 1990. Responses of body composition, growth and feed efficiency to dietary protein in genetically lean and fat broilers up to seven weeks of age. Br. Poult. Sci. 31, 163-172. [ Links ]
Whittemore, C.T., 1998. The Science and Practice of Pig Production, 2nd ed. Blackwell Science Ltd., Oxford. Wilson, P.N. & Osbourn, D.F., 1960. Compensatory growth after undernutrition in mammals and birds. Biol. Rev. 35, 324-363. [ Links ] [ Links ]
Copyright resides with the authors in terms of the Creative Commons Attribution 2.5 South African Licence.
See: http://creativecommons.org/licenses/by/2.5/za Condition of use: The user may copy, distribute, transmit and adapt the work, but must recognise the authors and the South African Journal of Animal Science.
# Corresponding author: gous@ukzn.ac.za














