Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
South African Journal of Animal Science
versão On-line ISSN 2221-4062
versão impressa ISSN 0375-1589
S. Afr. j. anim. sci. vol.41 no.2 Pretoria Jan. 2011
Effects of supplementing humates on rumen fermentation in Holstein steers
C.P. McMurphyI,#; G.C. DuffI; S.R. SandersI; S.P. CuneoI; N.K. ChiraseII
IDepartment of Animal Sciences, The University of Arizona, P.O. Box 210038, Tucson, AZ, USA 85721
IIGTX Technologies, 6500 Canyon Drive, Suite 260, Amarillo, TX, USA 79109
ABSTRACT
Humates have nitrogen-binding qualities that could prove to be beneficial in the retention of ammonia nitrogen in the rumen of Holstein steers fed a high-concentrate finishing diet. Therefore, four ruminally fistulated Holstein steers were used in a 4 x 4 Latin Square design to determine the effects of humates (HA) on serum urea nitrogen, rumen pH, rumen ammonia nitrogen and volatile fatty acid (VFA) production. Steers were randomly assigned one of four treatments; a control (0), 5.0, 10.0 or 15.0 g/kg HA (DM basis). Steers were fed ad libitum for 21 days prior to sample collection with dry matter intake (DMI) determined from days 14 to 21. For DMI, a quadratic response was observed; lowest for 5.0 and 10.0 g/kg HA and highest for 15.0 g/kg HA compared to control. Treatment by time interactions was not observed. No treatment differences were observed for total VFA concentrations (mM), or molar proportions of acetate, propionate, butyrate or valerate, but there was a quadratic response observed for butyrate. It was determined that HA do not alter rumen fermentation of Holstein steers fed high-concentrate diets.
Keywords: Humates, steers, rumen fermentation, serum urea nitrogen, intake
Introduction
Humate substances, or humic acids, are geological deposits in the earth's surface composed mainly of decaying plant and animal matter through the biological activities of microorganisms. Humates, based on solubility in acids and bases and by molecular weight, can be fractionated into three categories: fulvic acid, humic acid and humin (Stevenson, 1982). Humic acids are the major extractable component of soil humates and are dark brown to black in colour. Humates are composed of many functional groups (COOH, OH, C=O) and become more oxidized over periods of time, which decreases solubility due to the decrease in bound oxygen (Stevenson 1982).
Shi et al. (2001) evaluated the use of humic acid to minimize ammonia emissions from beef cattle feedlots and concluded that humates used as soil amendments do minimize ammonia emissions. Brown humate was more effective than black humate in vitro at reducing cumulative ammonia emissions by 67.6% compared to 60.2%, respectively (Shi et al., 2001). These nitrogen-binding qualities could prove to be beneficial in the retention of NH3-N in the rumen. However, there has been minimal research on the utilization of humates in beef cattle diets and their effect on rumen fermentation. Chirase et al. (2000) showed that the addition of humates to feedlot diets did not adversely impact dry matter intake (DMI), average daily gain (ADG) or feed to gain (F:G) ratio. These results were determined during a short 56-day feeding period. These authors suggested that there is no apparent negative effect on palatability or liver function with humate supplementation. Therefore, our objective was to determine the effects of humates on rumen fermentation of Holstein steers fed high-concentrate diets.
Materials and Methods
All procedures were approved by The University of Arizona Institutional Animal Care and Use Committee #05-105.
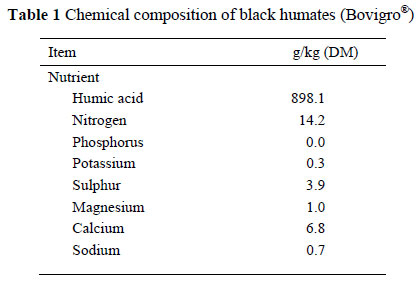
The humate product (Bovigro®, Bio Remedies, San Ysidro, NM, USA) used for this study was black in colour and mined in the northwestern part of New Mexico, United States. The chemical analyses of the humate product were conducted by A&L Western Agricultural Laboratories, Modesto, California and are presented in Table 1.
Six Holstein steers (317 ± 22 kg) (mean body weight ± SD) were brought to the feedlot at the University of Arizona, housed in individual pens, halter broke and fed a common diet until date of surgery. Steers were fistulated at the rumen and monitored for five weeks to ensure there were no post-operative side effects and to make sure they regained their appetite. Four steers were needed to conduct this study, but six steers were fistulated due to possible morbidity from surgery. However, surgery was successful and all steers were deemed healthy.
Four of these steers were used in a 4 x 4 Latin Square design. In each of the four periods, steers were randomly assigned to one of four dietary treatments (DM basis; Table 2): 1) high concentrate finishing diet with no humates (control); 2) control plus 5.0 g HA/kg; 3) control plus 10.0 g HA/kg; 4) control plus 15.0 g HA/kg. Steers were reassigned to diet after each period so that no steer received the same diet twice. Periods lasted 21 days in order to have 14 days of adaptation before diet evaluation. Steers were individually fed once daily at 06:00 to obtain ad libitum intake via slick bunk management. Dry matter intake (DMI) was determined from day 14 until day 21, when rumen and blood samples were collected. Diet samples were collected daily, oven dried at 55 ºC, composited each week and stored for later chemical analyses.
On day 20 at approximately 17:00, the steers were moved through a Silencer squeeze chute (Moly Mfg. Inc., Lorraine, KS, USA), haltered and fitted with jugular catheters. Indwelling polyurethane catheters were inserted via a 14-gauge needle until approximately 13.3 cm of tubing was inside the jugular. Stopcocks were connected to the catheter and secured to the steer with super glue. Catheters were flushed with 1 mL of heparin saline before the steers were returned to their pens. At 06:00 on day 21, 0-hour samples were collected. Blood-samples were collected with a disposable syringe and stored in a BD vacutainer (BD, Franklin Lakes, NJ, USA) where it was allowed to clot at room temperature; serum was harvested via centrifugation at 1,000 x g for 20 minutes and stored (-20 ºC) for later evaluation of serum urea nitrogen (SUN) concentration (mmol/L). Next, 1 mL of heparin saline was flushed through the catheter to prevent coagulation. After blood was collected, 0-hour rumen fluid was collected. The cannula plug was removed by pushing it into the rumen and then pulling it back through the opening, providing access to the rumen fluid. A plastic ladle was used to collect the rumen contents. The contents were strained through four layers of cheesecloth (BBA Nonwovens, Simpsonville, SC, USA) until 100 mL of rumen fluid was obtained in a 150 mL beaker. The pH was immediately determined using a mini Lab professional pH meter (IQ Scientific Instruments, Inc., Carlsbad, CA, USA) and rumen fluid was acidified (1 mL 20% HCl; v:v) and stored in whirl packs (-20 ºC) for subsequent VFA analysis. A small sample was stored in a 115 mL microcentrifuge tube for subsequent ammonia nitrogen analysis. Following 0-hour collection, the steers were haltered and returned to their individual pens. The steers were then individually fed one at a time in 10-minute intervals to allow for ample collection and sample processing time. This ensured that every steer was sampled equally relative to feeding time. The collection process was repeated in individual pens at 1, 2, 4, 8, 12, 16, 20 and 24 h post feeding. At the end of the 24 hour period, feed bunks were swept clean and unconsumed feed was weighed and removed and dry matter (DM) was determined. The steers were fed a steam-flaked maize based diet in a totally mixed ration (TMR); diets were isonitrogenous (Table 1), mixed every three days and stored in Rubbermaid® barrels. Feed intakes were documented, and at the end of the trial, weekly composite feed samples were analyzed for ash (combusted 6-hours in a muffle furnace at 500 ºC), dry matter (DM; oven dried at 110 ºC), crude protein (CP; g/kg N x 6.25; LECO Corporation, St. Joseph, MI, USA), and acid detergent fibre (ADF; Ankom Tech Corp, Fairport, NY, USA). Ort samples were collected at the end of each feeding period and analyzed.
Serum was thawed at room temperature prior to analysis. Serum urea nitrogen was determined using a direct colorimetric determination method according to manufacturer recommendations (TECO Diagnostics, Anaheim, CA, USA). Six prepared standards, two internal controls and 43 samples were tested on a 96 well plate. Samples at 10 µL were added to 1.5 mL SUN Enzyme Reagent, incubated for 5-min at 37 ºC, 1.5 mL Color Developer added and incubated again. Each sample was pipetted at 300 µL across 3 wells and analyzed using a spectrophotometer (Multiskan Ascent, MTX Labsystems Inc., Vienna, VA, USA; filter 595).
Rumen content samples stored in 1 mL microcentrifuge tubes were thawed at room temperature and then analyzed for rumen ammonia nitrogen (RAN) using a phenol-hypochlorite assay adapted from Broderick & Kang (1980) and modified by Galyean (1997), and analyzed using a spectrophotometer (Multiskan Ascent, MTX Labsystems Inc., Vienna, VA, USA; filter 595). Rumen fluid stored in Whirl-paks was thawed overnight in a refrigerator. Rumen fluid was mixed well in the Whirl-pak and poured into 25 mL centrifuge tubes, balanced by weight and centrifuged at 20 000 x g for 10-min. The rumen fluid was then removed from the centrifuge and 5 mL of the supernatant was pipetted into 13 x 100 mL centrifuge tubes. At this time 1 mL of 2-ethyl butyric acid was added as the internal standard. Tubes were vortexed, allowed to stand in ice water for 30 min, and then centrifuged at 10 000 x g for 10 min. Following centrifugation the liquid was filtered through a 0.45 µm filter into 12 x 75 mm storage tubes and placed in the freezer. Rumen fluid was thawed and loaded into gas chromatography (GC) vials at 10 mL. Samples were then shipped to North Dakota State University, Fargo, where subsequent rumen VFA concentrations were determined by gas chromatography (model GC 9A, Shimadzu, Kyoto, Japan) and separated on a packed column (model SP1200; Supelco, Bellefonte, PA, USA) (Goetsch & Galyean, 1983).
Data were analyzed using the PROC MIXED procedures of SAS (SAS Inst. Inc., Cary, NC, USA). For ruminal and serum variables, the model included effects for treatment period, time, and treatment x time. Time was used as a repeated measure and steer (treatment x period) was used as the random effect. The model for intake included treatment and period with steer (treatment x period) as the random effect. Humate treatments were analyzed for linear and quadratic responses. Significance was considered at P <0.05, and LS means along with most conservative SE were reported for each variable.
Results
Effects of supplementing Holstein steers with HA are presented in Tables 3 and 4. For diets containing HA, a quadratic (P = 0.01) response was observed with intake lowest for 5.0 and 10.0 g/kg HA diets and highest for the 15.0 g/kg relative to control steers (0 g/kg HA).
No treatment x time interactions (P >0.05) was observed for RAN concentrations, rumen pH and SUN (Table 2). Therefore, data were analyzed across time. When including HA at 5.0 g/kg in the diet there was a tendency (P = 0.10) for reduced ammonia concentrations.
Ruminal VFA concentrations are presented in Table 4. No treatment x time interaction (P >0.05) was observed for total VFA concentration or for individual molar proportions (P >0.05). Therefore, data were analyzed across time. No differences (P = 0.20) between HA treatments were observed for total VFA concentrations (mM), or for molar proportion of acetate, propionate, butyrate or valerate (P >0.05). Isobutyrate and isovalerate concentrations were higher for control steers than for HA supplemented steers. A quadratic (P = 0.04) response was observed for butyrate with concentrations highest for 5.0 and 10.0 g/kg HA. There was also a higher acetate : propionate ratio for 5.0 g/kg HA supplemented steers.
Discussion
Although not significant, McMurphy et al. (2010) also found that the steers supplemented 15.0 g/kg of HA had the greatest numeric intakes when compared to 5.0 and 10.0 g HA/kg. Chirase et al. (2000) demonstrated a similar decrease in intake during the first 28 days for cattle fed a lower HA concentration (7.8 g/kg) vs. a control and increased concentrations (15.6 and 31.2 g HA/kg). However, no increase in efficiency was observed for the 56 day period. Covington et al. (1997) showed that lambs fed 10.0 and 20.0 g HA/kg during a 63 day feeding period consumed less feed and converted more poorly than lambs fed 0 and 5.0 g/kg. In contrast, Brown et al. (2007) did not show differences in DMI for steers when a humate product was added to a high concentrate diet, nor did Wang et al. (2008) in finishing pigs. Brown et al. (2007) also reported no changes in performance or feed efficiency with the inclusion of humates in the diet. Notably, our study was not a performance trial and inconsistent reports make it difficult to conclude that HA would alter feed intake. Albeit, it does not appear that intake differences can be attributed to palatability.
Fermentation in the rumen produces ammonia nitrogen, and in the unionized state, ammonia (NH3) is readily transferred across the rumen wall and may be excreted in the urine as ammonium salts, used in transamination to form glutamine or converted to urea in the liver and excreted or recycled back to the rumen (Emerick, 1988). The ease at which ammonia traverses the ruminal wall into the blood stream also limits the amounts of dietary non-protein nitrogen that can be fed to ruminants, especially in high roughage diets where ammonia toxicity could result. However, in the ionized state (NH4+), the nitrogen can be utilized by bacteria that tend to produce more propionic acid as opposed to acetic acid (Emerick, 1988), which would be beneficial to steers consuming a high concentrate finishing diet. Rumen ammonia nitrogen production peaked at 1-hour post-feeding, which contradicts the peak time of 2-hours, reported by (Owens & Zinn, 1988). However, this response has been observed in dairy steers (Spires & Clark, 1979) and could possibly be unique to Holstein cattle due to an increase in rumen volume. Humates have been shown in vitro to reduce ammonia emissions when utilized as an amendment to soil, faeces and urine (Shi et al., 2001) or when used as a feed supplement in pig diets (Ji et al., 2006). Similarly, Varadyova et al. (2009) observed a decrease in daily ammonia nitrogen output in a high forage diet and a numerical decrease in a high concentrate diet in vitro with the addition of 10 g/kg of a 610 g/kg of humic acid product. This also coincides with the trend for lower ammonia nitrogen when 5.0 g/kg of HA were included in the diet and those results recorded for beef steers at a different stage of production when 10.0 g/kg of HA was included in the diet (McMurphy et al., 2010). However, the subsequent improvement in propionate was not observed. Acetate concentrations were actually increased in the high concentrate diet with the addition of HA (Varadyova et al., 2009) similar to current findings with the addition of 5 g/kg of HA resulting in a lower acetate : propionate ratio. Nevertheless, these findings have not resulted in improved animal performance in finishing steers consuming high concentrate diets (Brown et al., 2007; McMurphy et al., 2010).
No differences were observed for SUN, also reported by Chirase et al. (2000). McMurphy et al. (2010) also observed similar concentrations of SUN across these same levels of HA. This is potentially due to the lack of ammonia binding in the rumen by HA. Similar SUN concentrations were reported by Gleghorn et al. (2004) when cattle were fed an isonitrogenous, 130.0 g CP/kg, 900.0 g/kg concentrate diet.
No difference in rumen pH was observed. Rumen pH did reach its lowest state at four hours post feeding for diets fed 0 and 5.0 g/kg HA, which is supported by (Owens & Zinn, 1988) and was between 5.5 and 6.3 (Owens & Goetsch, 1988). The pH for all treatments was numerically higher than the control, but not significantly. The lack of significance could be due to higher roughage levels than typical beef cattle finishing diets. Accordingly, this would cause a less acidic rumen pH and therefore these steers may not have benefited from the potential buffering capabilities of HA.
Conclusions
A humate product high in humic acid content did not dramatically impact some aspects of rumen fermentation. However, including dietary humates may slightly alter DMI while reducing rumen ammonia nitrogen when included at 5.0 g/kg. This slight reduction in rumen ammonia nitrogen could benefit nitrogen efficiency in feedlot cattle, reducing nitrogen loss to the environment. However, humate products can be inconsistent and completely different depending on what environment they come from. This could pose a problem with the consistency and validity of research with HA as supplements in animal diets, so further research is needed to validate results.
Acknowledgements
This study was completed at the University of Arizona Feedlot (Tucson, AZ). The authors would like to thank J. English, K. Cannon and M. Townsend for their assistance at the feedlot. This study was made possible through partial financial and product support from Mesa Verde Resources, Placitas, NM, USA.
References
Broderick, G.A. & Kang, J.H., 1980. Automated simultaneous determination of ammonia and total amino acids in ruminal fluid and in vitro media. J. Dairy Sci. 63, 64-75. [ Links ]
Chirase, N.K., Greene, L.W., McCollum, F.T., Auvermann, B.W. & Cole, N.A., 2000. Effect of Bovipro on performance and serum metabolites concentrations of beef steers. Western Section, American Society of Animal Science Proceedings 51, 415-418. [ Links ]
Brown, M.S., Lawrence, T.E., Ponce, C.H., Pulikanti, R., Smith, C.S., Mitchell, D.L, Sumerford, B. & Davenport, J.D., 2007. Effects of a humate product on growth performance, carcass merit, and tissue and serum mineral composition of individually-fed steers. J. Anim. Sci. 85 (Suppl. 1). [ Links ]
Covington, B.R., Ramsey, W.S., Greene, L.W. & Byers, F.M., 1997. Effects of humate on feedlot performance and carcass characteristics in feedlot lambs. J. Anim. Sci. 75 (Suppl. 1). [ Links ]
Emerick, R.J., 1988. Nitrate and Urea Toxicities. In: The Ruminant Animal: Digestive Physiology and Nutrition. Ed. Church, D.C., Prentice-Hall, Englewood Cliffs, NJ. pp. 480-484. [ Links ]
Galyean, M.L., 1997. Laboratory procedures in animal nutrition research. Department of Animal and Food Sciences, Texas Tech University, Lubbock, TX, USA. pp. 98. [ Links ]
Gleghorn, J.F., Elam, N.A., Galyean, M.L., Duff, G.C., Cole, N.A. & Rivera, J.D., 2004. Effects of crude protein concentration and degradability on performance, carcass characteristics, and serum urea nitrogen concentrations in finishing beef steers. J. Anim. Sci. 82, 2705-2717. [ Links ]
Goetsch, A.L. & Galyean. M.L., 1983. Influence of feeding frequency on passage of fluid and particulate markers in steers fed a concentrate diet. Can. J. Anim. Sci. 63,727. [ Links ]
Ji, F., McGlone, J.J. & Kim, S.W., 2006. Effects of dietary humic substances on pig growth performance, carcass characteristics and ammonia emission. J. Anim. Sci. 84, 2482-2490. [ Links ]
McMurphy, C.P., Duff, G.C., Harris, M.A., Sanders, S.R., Chirase, N.K. & Bailey, C. R., 2009. The effects of humic/fulvic acid in beef cattle finishing diets on animal performance, ruminal ammonia and serum urea nitrogen concentration. J. Appl. Anim. Res. 35, 97-100. [ Links ]
NRC, 1996. Nutrient requirements of beef cattle. (7th rev. ed.). National Academy Press, Washington, D.C., USA. [ Links ]
Owens, F.N. & Goetsch, A.L., 1988. Ruminal fermentation. In: The ruminant animal: digestive physiology and nutrition. Ed. Church, D.C., Prentice-Hall, Englewood Cliffs, NJ. pp. 145-171. [ Links ]
Owens, F.N. & Zinn, R.A., 1988. Protein metabolism of ruminant animals. In: The Ruminant Animal: Digestive Physiology and Nutrition. Ed. Church, D.C., Prentice-Hall, Englewood Cliffs, N.J., USA. pp. 227-249. [ Links ]
SAS, 1988. SAS® User's Guide: Statistics. Version 9.1 Edition. SAS Inc., Cary, N.C., USA. [ Links ]
Shi, Y., Parker, D.B., Cole, N.A., Auvermann, B.W. & Mehlhorn, J.E., 2001. Surface amendments to minimize ammonia emissions from beef cattle feedlots. American Society of Agricultural Engineers 44, 677-682. [ Links ]
Stevenson, F.J., 1982. Humus Chemistry: Genesis, Composition, Reactions. 1st edn. Wiley. New York. [ Links ]
Spires, H.R. & Clark, J.H., 1979. Effect of intraruminal urea administration on glucose metabolism in dairy steers. J. Nutr. 109, 1438-1447. [ Links ]
Varadyova, Z., Kisidayova, S. & Jalc, D., 2009. Effect of humic acid on fermentation and ciliate protozoan population in rumen fluid of sheep in vitro. J. Sci. Food Agric. 89, 1936-1941. [ Links ]
Wang, Q., Chen, Y.J., Yoo, J.S., Kim, H.J., Cho, J.H. & Kim, I.H., 2008. Effects of supplemental humic substances on growth performance, blood characteristics and meat quality in finishing pigs. Livest. Sci. 117, 270-274. [ Links ]
Copyright resides with the authors in terms of the Creative Commons Attribution 2.5 South African Licence. See: http://creativecommons.org/licenses/by/2.5/za/
Condition of use: The user may copy, distribute, transmit and adapt the work, but must recognise the authors and the South African Journal of Animal Science
# Corresponding author: casey.mcmurphy@okstate.edu














