Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Animal Science
On-line version ISSN 2221-4062
Print version ISSN 0375-1589
S. Afr. j. anim. sci. vol.40 n.4 Pretoria Jan. 2010
Effect of bromine and iodine in drinking water on production parameters of broilers
J. du Toit; N.H. Casey#
Department of Animal and Wildlife Sciences, University of Pretoria, Pretoria 0002, South Africa
ABSTRACT
Levels of bromine (Br) in groundwater that exceeded guideline concentrations of 0.01 mg/L prompted the investigation of the effect Br and iodine (I) in drinking water on the production parameters of broilers. The research was done over a 42-day growth period, post-hatch, in which the trial design was: Control: 0 mg Br/L and 0 mg I/L; 1 mg Br/L and 0 mg I/L; 3 mg Br/L and 0 mg I/L; 0 mg Br/L and 0.7 mg I/L; 1 mg Br/L and 0.7 mg I/L; 3 mg Br/L and 0.7 mg I/L, by 3 replicates with 30 birds/replicate, using mixed Ross broiler chickens. Br and I were administered as NaBr and KI. Mortalities, water and feed intakes were recorded daily and live weight once a week. Average daily gains and feed conversion rations were calculated. Different treatments of Br, irrespective of I, decreased water and feed intake significantly. The interaction of Br and I had no significant effect on intake of water or feed. I had an effective ameliorating effect on Br. Bromine administered at 1 and 3 mg Br/L or at ingestion rates of 1.59 and 4.44 mg Br/day affected production parameters.
Keywords: Toxicity, halogens, water quality, poultry, livestock
Introduction
The production efficiencies of livestock are determined by the interactions between their genetic potential and the effects of external factors on the physiology of the animals. The efficiency of broiler production relies on the birds currently growing at an average rate of 55.6 g/day with a feed conversion ratios (FCR, g feed/g gain) of 0.87 on Day 7; 1.13 on Day 14; 1.29 on Day 21; 1.44 on Day 28 and 1.60 on Day 36 to attain a target live mass of 2005 g by 36 days of age (Aviagen, 2007). During this period of vigorous growth the birds have a high rate of water intake that in the first week may be equivalent to 73.5% of live weight (Casey et al., 1998). Drinking water is therefore an important route for supplying additional nutrients and pharmaceutical treatments. On the other hand, water may be a conduit for potential hazardous chemicals of concern (PHCC). The PHCC become hazardous under circumstances that may be specific to the livestock type and physiological status of the animal, the production environment, ingestion rates of water quality constituents (WQC) and the agonistic or antagonistic effects of other WQC in the drinking water (Meyer et al., 1997; Coetzee et al., 2000a; b; Meyer & Casey, 2004; Mamabolo et al., 2009).
Various literature and water-source surveys in South Africa analysed by Casey et al. (1998; 2001) indicated that exposure time, production systems, ingestion rates and species tolerance are important factors that need to be taken into account when formulating water quality guidelines for livestock. Verification of the validity of the guidelines in use is required in order to accurately estimate the fitness for use of a water source for livestock production (Meyer et al., 1997). Assessment by Casey & Meyer (2001) of a significant number of water samples over a number of years revealed that high concentrations of bromine (Br) occur naturally within groundwater in South Africa, hence a PHCC. The highest quantities of Br present in the water-sources surveyed were 30 - 132 mg Br/L (Meyer et al., 1997; Coetzee et al., 2000a; b; Meyer & Casey, 2004; Mamabolo et al., 2009), a concentration 30 - 130 times higher than the 1 mg Br/L considered by Kempster et al. (1980) to be acceptable. The relevant safety guideline for Br recommended by Casey & Meyer (2001) is 0.01 mg/L. The Safe Drinking Water Committee (1988) reported chronic exposure levels of Br as 2.3 mg/L. This calculation was based on the Suggested No-Adverse-Response-Level (SNARL) where a 70 kg human consuming 2 L of water per day was included. Russian investigators, El'piner et al. (1972), recommended a maximum bromide concentration of 0.2 mg/L in drinking water.
The toxicity of Br as a PHCC depends upon the susceptibility of the species, the amount and time period over which Br is ingested and the solubility of the form of the Br as found in the water source (Casey et al., 1998; NRC, 2005). Baker et al. (2003) studied the iodine (I) x Br interaction and came to three possible mechanisms by which Br decreases the I concentration. The three likely possibilities are that Br: 1) reduces intestinal absorption of I; 2) enhances urinary excretion of I or; 3) reduces I uptake by the thyroid gland. I concentration did decrease with an increase of Br inclusion levels, but was insignificant within the thyroid gland. Dietary bromide, on the other hand, was reported to reduce I toxicity in chicks (Baker et al., 2003; Baker, 2004; NRC, 2005).
The effect of WQCs on the production parameters of broilers was shown by Coetzee et al. (2000a; b) and Casey et al. (2001). Various manifestations influencing production were reported to occur following the ingestion of Br over different time periods. An elevated dietary intake of bromide by rat dams in lactation was found to cause a very significant decrease in weight gain in the suckling offspring (Pavelka et al., 2002). More trials revealed that only half of these sucklings survived and were in a very poor condition. This was complemented by stagnation in the extent of the consumption of diet and water as well as a drop in milk production rate during the nursing period (Pavelka, 2003). This suggests that bromide ingested by the dam was transported via the milk to the sucklings (Pavelka et al., 2002).
According to Anke et al. (1989) Br may be an essential element. In three long-term experiments with growing, pregnant and lactating goats, poor Br nutrition lead to significantly reduced growth, haemoglobin concentration, haematocrit quantities, conception, milk and fat yield, lower longevity of does and kids and an increased abortion rate. In contrary, Loeber et al. (1983) recorded a suppression of growth hormone release in male rats receiving 19.2 g sodium bromide (NaBr)/kg diet, after four weeks of dietary administration. Velicky et al. (2004) recorded a decrease in body weight in rats fed a semi-synthetic purified diet containing a high concentration of Br. Retarded growth was recorded in dogs and rabbits fed NaBr (Jolles, 1966). Doberenz et al. (1965) found 10 000 mg/kg Br including 1000 mg/kg F fed to chicks resulted in a decrease in body weight.
Most of the work conducted previously on the effect of Br was done on rats and humans, and administrated via the diet where high levels of Br were used. In this trial, treatments closer to the natural Br content of South African ground water sources were used. The objective of this study was to determine the effect of Br and I in drinking water on production parameters of broilers.
Materials and Methods
Procedures for this trial were approved by the University of Pretoria Animal Use and Care Committee (Reference EC080805-032).
The trial design was five treatments and one control x three replicates x 30 Ross broilers of mixed sex per replicate. Treatments administered through the drinking water from Days 1 to 42 were:
| T0 (Control) | = 0 mg Br/L and 0 mg I/L; | |
| T1 | = 1 mg Br/L and 0 mg I/L; | |
| T2 | = 3 mg Br/L and 0 mg I/L; | |
| T3 | = 0 mg Br/L and 0.7 mg I/L; | |
| T4 | = 1 mg Br/L and 0.7 mg I/L; | |
| T5 | = 3 mg Br/L and 0.7 mg I/L. |
The broilers were reared from Day 1 on a litter floor in an environmentally controlled house. Chicks received New Castle disease and bronchitis vaccines at the hatchery and received vaccinations for Gumburo disease on day 12 post-hatching. Vaccines were administered in the form of eye drops and purchased from a reputable animal and veterinary pharmaceutical supplier. Water was delivered from graduated cylinders via bell drinkers for accurate measuring of water intake.
The diets were fed ad libitum throughout the trial and consisted of a starter, finisher and post-finisher ration, formulated for a 42-day commercial broiler programme. Each of the treatment groups received the same diet at a specific stage of the study. The trace element premix contributed 0.001 g/kg iodine to the diets and 0.0 g/kg Br. Table 1 provides the nutrient composition of the starter, finisher and post-finisher diets as provided by the manufacturer.
Water from the Pretoria municipality was used. Chemical analyses of water samples collected throughout the trial were conducted once a week to determine the Br and I levels in the drinking water as well as for quality control purposes. Inductively Coupled Plasma Atomic Emission Spectrometry (ICP-AES) techniques were performed by the ARC-ISCW in Pretoria as well as full quantitative and semi-quantitative procedures (Looke et al., 1998) to measure the mineral element composition of the water. The ARC-ISCW prepared the sampling bottles including a protocol for sampling procedures as described in: "Water analyses methods as described by Looke, Philpott and van Vliet" (Looke et al., 1998). Separate sample bottles were used for macro- and trace elements during these procedures. The final concentrations of the chemical mixture were confirmed by testing the samples at the point of use. The water used in the study contained final concentrations of 1 mg Br/L or 3 mg Br/L as sodium bromide (NaBr), as well as 0.7 mg I/L as potassium iodide (KI).
The reason for selecting 1 and 3 mg Br/L in the investigation was because 1 mg/L is the recommended maximum limit for Br, while Kempster et al. (1980) initially found 3 mg/L to be the maximum permissible level (maximum limit for insignificant risk) and 6 mg/L as crisis limit (maximum limit for low risk), as a general guideline for human consumption. According to Kempster et al. (1980) the recommended limit (maximum limit for no risk) was then set at 1 mg Br/L due to the fact that the presence of 3 mg Br/L could lead to bromate formation. These quality criteria were set for human drinking water and not for livestock, but as stated by McKee & Wolf (1963), water safe for human consumption may be used safely by livestock. The physiology, metabolism and tolerance of animal species and humans differ, which indicates the importance of this trial since no experimental data on Br administered via drinking water is currently available, except for studies done on fish (McKee & Wolf, 1963) and rats. Through the administration of 1 mg Br/L, the recommended level of Br can be verified or it can indicate, by revealing no sub-clinical signs of toxicity, that this recommended limit is too strict.
The inclusion of I is necessary to indicate a possible alleviator effect for these high Br water levels. To determine whether I can possibly alleviate the effect of Br, 0.7 mg I/L was selected because this level falls within the Target Water Quality Range for I which is between 0 and 1 mg/L. Water intake, feed intake, body weight and mortalities were recorded over a 42-day period from Day 1 post-hatching.
The SAS (Statistical Analyses System®) software system was used for the statistical analysis. The GLM procedure was applied to test for differences and interactions between treatments and the level of significance was determined by means of the Fisher's test at a P <0.05.
Results
Water samples were collected and analysed throughout the trial. The municipal water source contained 0.096 mg Br/L and 0.032 mg I/L. This caused that the treatments of 0 mg/L of either Br or I were not truly 0 mg/L, but referred to treatments in which 0 mg/L of either Br or I was added to the water. No corrections were made for the concentrations Br and I in the municipal water when NaBr and KI were added to the treatments, with the result that true ingested concentrations of Br were 0.096, 1.096 and 3.096 mg Br/L and for I 0.032 and 0.732 mg/L. The analyses were done by treatment and not by ingested amounts of Br and I. The amounts of Br and I ingested (mg/bird/day) via the water are shown in Table 2.
The different treatments of Br, irrespective of I, caused a significant decrease in water (P = 0.023) and feed (P = 0.004) intakes. The different levels of I, irrespective of Br, had no significant effect on the intake of water (P = 0.805) or feed (P = 0.936). The interaction of Br and I had no effect on water (P = 0.093) or feed (P = 0.9593) intakes, indicating the ameliorative effect of I on Br (Tables 3 and 5). Tables 3 and 5 indicate that the Br treated groups consumed significantly less water and feed than the control group. The addition of I to the Br treatments resulted in the differences in water and feed consumption being non-significant.
Table 4 illustrates that from Week 3 onwards group T2 consumed significantly less water than the control group. Overall, at Week 6 Br showed a significant influence on cumulative water intake (P = 0.041), since the Br treatment groups (T1 and T2) consumed significantly less water (P = 0.010) than the control group. The overall trend throughout the six weeks was that the control group had the highest water intake and T2 (3 mg Br/L) the lowest. Group T5 had a higher water consumption than T2, but did not differ significantly from the control. T4 had higher water intakes than T1, indicating the ameliorative effect of I on water intake.
Table 6 shows that from Week 3 onwards, T1 consumed significantly less feed than the control group. Iodine alleviated this effect since T3 did not differ significantly from the control group. At Week 6 the overall effect of Br and I on cumulative feed intake was significant (P = 0.005), and both Br (P = 0.004) and I (P = 0.002) showed significant effects. The greater significance at Week 6 than at any other week (Tables 4 and 6) could suggest the 42-day growth period was too short and that the effect of Br would become more prominent with a longer period of exposure.
The five treatments did not have an overall effect on average daily gain (ADG) (P = 0.416). The different levels of I (P = 0.431) and Br (P = 0.306) irrespective of the other element or factor also had no significant influence on ADG, neither did the interaction between Br and I (P = 0.638) influence ADG (Table 7). There was a significant overall effect on ADG(P = 0.0003) only at Week 1, where Br (P = 0.0006) and I (P = 0.0003) individually as well as their interaction (P = 0.0026) had significant influences, but this was not recorded for any of the other weeks. This too is an indication that a longer exposure period is required to quantify the effect of Br on ADG (Table 8).
There were no significant differences within the weeks for FCR, but Br (P = 0.041) irrespective of I and any other factors did have a significant influence on FCR at Week 6, again indicating that a longer exposure period might have indicated an adverse effect of high Br concentrations on the feed conversion of broilers. It is apparent from Table 9 that at Week 1 the control and T3 had the most efficient FCRs and that T2 had the most uneconomical FCR, but they did not differ significantly from each other or any of the other treatments. Then at Week 6 the most efficient FCR was reported as T2 with the most significant uneconomical treatments identified as T4 and T5.
Mortalities during the production period were not linked to Br and liveability ranged from 85.6% to 95.6% (Table 10).
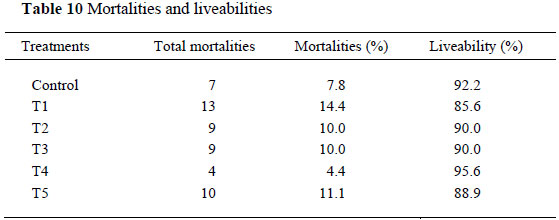
Discussion
The concentration of a WQC in water is a fair indication of the potential exposure to or ingestion of the WQC. A WQC becomes a cause for concern when it exceeds the guideline levels. However, as noted, the question remains as to whether the guidelines may be too stringent or too lax. In addition, elements that occur abundantly in natural groundwater may not appear in the guidelines that may be dated but are still in circulation, for example Ayers & Westcot (1985, reprinted 1994); Casey & Meyer (1996a; b). Verification of the validity of the guidelines in use is required in order to accurately estimate the fitness for use of a water source for livestock production (Meyer et al., 1997).
When water with a potentially toxic constituent such as Br is encountered, ingestion rates need to be calculated. This is important as it is the actual amount of the toxic constituent consumed that is relevant, and not its concentration in the water alone.
The results of the trial conducted on broilers with the Br and I treatments in the drinking water indicated that significantly lower water and feed intakes were induced by the Br treatment groups administered as 1 or 3 mg Br/L in the drinking water or inclusion levels of 4.44 mg Br/L and 1.59 mg Br/L per bird per day. The observation that an alleviator treatment of 0.7 mg I/ L in the drinking water or the intake of 1.1 mg I/L per bird per day, may be used without adverse effects, at least over a 6-week period, may assist in attempting to decrease the risk to communities or production systems that may be exposed to a high Br water source.
Mamabolo et al. (2009) also reported decreased water and feed intakes by broilers when their drinking water was treated with 1 mg Br/L, 0.1 mg As/L and 0.1 mg Pb/L. A decrease in water intake has a very unconstructive influence on broiler production systems as a short term reduction in water intake leads to the reduction in feed intake and a reduction in ADG (Leeson & Summers, 2005); these adverse effects should be prominent in a longer exposure period or with mature birds.
Loeber et al. (1983) recorded a suppression of growth hormone release in male rats receiving sodium bromide (NaBr) enriched diets. On the other hand, Bosshardt et al. (1956) and Huff et al. (1956) demonstrated a nutritional requirement of Br (8 to 15 mg Br/kg administered through the diet for 12 to 31 days) in rats and chicks when an 8 to 10% growth response was evident. Unfortunately these results were obtained where Br was administered through the diet and not through the drinking water. Jolles (1966) and Loeber et al. (1983) recorded a suppression of growth hormone release in male rats receiving sodium bromide (NaBr) enriched diets. On the other hand, retardation in growth of mice and chickens was related to Br deficient diets (Jolles, 1966). The Mamabolo et al. (2009) trial indicated a slight though non-significant improvement in growth with the Br treated group, whereas in this trial neither depression nor induction of growth was observed, thus increasing the necessity for a longer exposure period to Br and its effect on livestock.
Efficiency of feed utilisation was not affected. A longer production period, higher stocking density or rural systems may provide different FCR results. However these results may indicate that the exposure period for this trial was too short, especially in the context of rural communal chicken farming, where chickens are raised under stressful conditions and are provided with poor quality drinking water, feed and environment.
Mortalities were the highest for T1 and lowest for T2 (Table 10), indicating no direct relationship between mortalities and Br administration. It was not unexpected for this trial as 3 mg Br/L was not expected to cause mortality in such a short exposure period, but rather adversely affects production parameters.
Conclusions
The results obtained in this study indicate that the current guidelines (0.01 mg/L) on the Br content of drinking water for broilers were not necessarily too restrictive. This trial confirmed that concentrations of PHCC (Br in this trial) observed in the drinking water of the poultry production unit can adversely influence production parameters regardless of a short exposure period. Significantly lower water and feed intakes were induced by 3 mg Br/ L in the drinking water or inclusion levels of 4.44 mg Br/L per bird per day. The observation that an alleviator treatment of 0.7 mg I/ L in the drinking water or the intake of 1.1 mg I/L per bird per day, may be used without adverse effects, at least over a 6-week period, may assist in attempting to decrease the risk to communities that may be exposed to a high Br water source. Production parameters of broilers that were not significantly influenced by Br over this relatively short term exposure period and low stocking density were, average daily gain or growth, FCR and mortalities. The long term effects of these parameters and exposure of mature animals are yet to be quantified.
Acknowledgements
R.J. Coertze and H.T. Groeneveld for assistance with the statisitical analyses.
References
Anke, M., Groppel, B. & Arnhold, W., 1989. Further evidence for the essentiality of bromine in ruminants. Proc. 6th Int. Trace Element Symposium, University Leipzig-Jena, Germany, 1120. [ Links ]
Aviagen, 2007. Ross 708 Broiler Performance Objectives. Aviagen Limited, Newbridge, Scotland, UK. [ Links ]
Ayers, R.S. & Westcot, D.W., 1985, reprinted 1994. Section 6: Water quality for livestock and poultry. In: Water Quality for Agriculture. Published by the Food and Agriculture Organisation of the United Nations Rome. ISBN 92-5-102263-1. [ Links ]
Baker, D.H., 2004. Iodine toxicity and its amelioration. Mineral view. Exp. Biol. Med. 299, 473-478. [ Links ]
Baker, D.H., Parr, T.M. & Augspurger, N.R., 2003. Oral iodine toxicity in chicks can be reversed by supplemental bromine. J. Nutr. 133, 2309-2312. [ Links ]
Bosshardt, K.D., Huff, J.W. & Barnes, R.H., 1956. Effect of bromine on chick growth. Proc. Soc. Exp. Biol. Med. 92, 219-221. [ Links ]
Casey, N.H. & Meyer, J.A., 1996a. Interim Water Quality Guidelines for Livestock Watering. Published by the Water Research Commission. ISBN 1-86845-225-5. [ Links ]
Casey, N.H. & Meyer, J.A., 1996b. Livestock Watering. In: South African Water Quality Guidelines, Volume 5: Agricultural Use. Published by the South African Department of Water Affairs and Forestry. ISBN 0-7988-5338-7 (set); ISBN 0-7988-5343-3. Vol 5. [ Links ]
Casey, N.H. & Meyer, J.A., 2001. An extension to and further refinement of water quality guideline index system for livestock watering. Volume 1: Rural communal livestock production system and wildlife production system. WRC Report No 857/1/01. [ Links ]
Casey, N.H., Meyer, J.A. & Coetzee, C.B., 1998. An investigation into the quality of water for livestock production with the emphasis on subterranean water and the development of a water quality guideline index system. WRC Reports 644/1/98 and 644/2/98. [ Links ]
Casey, N.H., Meyer, J.A. & Coetzee, C.B., 2001. An extension to further refinement of a water quality guideline index system for livestock watering: Volume 2: Poultry production systems and water quality for ostrich production. WRC Report No 857/2/01. [ Links ]
Coetzee, C.B., Casey, N.H. & Meyer, J.A., 2000a. Quality of groundwater used for poultry production in the Western-Cape. Water SA. 26 (4), 563-568. [ Links ]
Coetzee, C.B., Casey, N.H. & Meyer, J.A., 2000b. The effect of water-borne fluoride on the production of laying hens. Water SA. 26 (1), 115-118. [ Links ]
Doberenz, A.R., Kurnick, A.A., Hulett, B.J. & Reid, B.L., 1965. Bromide and fluoride toxicity in the chick. Poult. Sci. 44, 1500-1504. [ Links ]
El'piner, L.I., Shafirov, Y.B., Khovakh, I.M., Shub, O.A. & Gurvich, I.A., 1972. Hygienic substantiation of permissible content of bromine in drinking water. Gig. Sanit. 37, 13-17. http://67.20.64.230/ss/assets/Tech_Center/Ross_Broiler/Ross_708_Broiler_Performance_Objectives.pdf [ Links ]
Huff, J.W., Bosshardt, D.K., Miller, C.P. & Barnes, R.H., 1956. A nutritional requirement for Bromine. Proc. Soc. Exp. Biol. Med. 92, 216-219. [ Links ]
Jolles, Z.E., 1966. Bromine and Its Compounds. Academic Press, Toronto, Canada. pp. 487-497. [ Links ]
Kempster, P.L., Hattingh, W.H.L. & Van Vliet, H.R., 1980. Summarised water quality criteria. Technical Report No TR 108, Department of Environmental Affairs, Pretoria. 8. [ Links ]
Leeson, S. & Summers, J.D., 2005. Commercial Poultry Nutrition, 3rd ed. Nottingham University Press, Canada. [ Links ]
Loeber, J.G., Franken, M.A.M. & Van Leeuwen, F.X.R., 1983. Effect of sodium bromide on endocrine parameters in the rat as studied by immunocytochemistry and radioimmunoassay. Food Chem. Toxicol. 4, 391-404. [ Links ]
Looke, A., Philpott, M.F. & Van Vliet, H.S., 1998. Water Analyses Methods. Institute for Soil, Climate and Water, Pretoria, South Africa. [ Links ]
Mamabolo, M.C., Casey, N.H. & Meyer, J.A., 2009. Effects of total dissolved solids on the accumulation of Br, As and Pb from drinking water in tissues of selected organs in broilers. S. Afr. J. Anim. Sci. 39, (Suppl. 1), 169-172. [ Links ]
McKee, J.E. & Wolf, H.W., 1963. Water Quality Criteria. 2nd ed. The Resources Agency of California State Water Resources Control Board, California. 112, 148. [ Links ]
Meyer, J.A. & Casey, N.H., 2004. Exposure assessment of potentially toxic trace elements in indigenous goats in the Nothern Region in South African rural communal production systems. 8th Int. Conference on Goats, South Africa, 4-9 July. Book of Abstracts. p. 141. [ Links ]
Meyer, J.A., Casey, N.H. & Coetzee, C.B., 1997. Water quality guidelines for livestock watering in Southern Africa. Water SA. 23 (1), 7-12. [ Links ]
NRC, 2005. Mineral Tolerance of Animals. National Academic Press, Washington D.C., USA. pp. 72-78, pp. 182-198. [ Links ]
Pavelka, S., 2003. The effect of exogenous bromide on the metabolism of iodine. In: Trace Elements in Human: New Perspectives, Part I. Eds Ermidou-Pollet, S. & Pollet, S., University of Athens, Athens. Greece. pp. 615-624. [ Links ]
Pavelka, S., Babicky, A., Vobecky, M. & Lener, J., 2002. Impact of high bromide intake in the rat dam on iodine transfer to the sucklings. Food Chem. Toxicol. 40, 1041-1045. [ Links ]
The Safe Drinking Water Committee, 1988. Drinking Water and Health. Vol. 3. National Academy Sciences, Washington D.C., USA. Chapter 4, 181-187, 215-225. [ Links ]
Velický, J., Titlbach, M., Lojda, Z., Dušková, J., Vobecký, M. & Raška, I., 2004. The effect of bromide on the ultrastructure of rat thyrocytes. Ann. Anat. 186, 209-216. [ Links ]
# Corresponding author. E-mail: norman.casey@up.ac.za














