Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Animal Science
On-line version ISSN 2221-4062
Print version ISSN 0375-1589
S. Afr. j. anim. sci. vol.39 n.5 Pretoria Jan. 2009
The nutritive value of South African Medicago sativa L. hay
G.D.J. ScholtzI,#; H.J. van der MerweI; T.P. TylutkiII
IDepartment of Animal-, Wildlife- and Grassland Sciences, P.O. Box 399, University of the Free State, Bloemfontein 9300, South Africa
IIAgricultural Modelling and Training Systems, 418 Davis Rd, Cortland, NY 13045, USA
ABSTRACT
A study was conducted to evaluate the variation and expand the existing and limiting nutritive value database of Medicago sativa L. hay (168 near-infrared reflectance spectroscopy spectrally selected samples) in South Africa. The highest moisture content recorded (140 g/kg) was safely below the critical moisture level of 160 g/kg for effective storage. Coefficient of variation (CV) ranged from 1.2% for dry matter (DM) up to 66.2% for acid detergent fibre-crude protein (ADF-CP). The average ash content was 130 g/kg (73 to 295 g/kg), indicating soil contamination. Fibre fractions varied as follows: acid detergent fibre (ADF) (213 to 473 g/kg), neutral detergent fibre (NDF) (289 to 659 g/kg), lignin (43 to 163 g/kg), cellulose (163 to 364 g/kg) and hemicellulose (53 to 199 g/kg). The mean IVOMD for both 24 and 48 h (693 and 732 g/kg DM, respectively), was representative (CV = ± 8%) of the Medicago sativa L. hay population. Crude protein (CP) (average = 207 g/kg DM) consists of 76.9% true protein. According to ADF-CP, 6% of the samples were heat damaged. High mean Ca (13.5 g/kg), P (25.3 g/kg) and Fe (874 mg/kg) values were recorded.
Keywords: Lucerne, quality, chemical analysis
Introduction
Lucerne hay quality (nutritive value) could be influenced by factors such as harvesting at specific physiological stages, climatic factors, edaphic factors such as soil conditions, leaf losses during haymaking, storage and feeding practice, diseases and insects, weeds, lucerne cultivar, moisture content during storage, water supply and fertilisation (Putnam et al., 1997). Given that dairy rations could contain up to 40% lucerne hay, milk production variability can be partially related to lucerne hay. Accordingly, variation in quality of South African lucerne hay could hamper the accurate formulation of ruminant diets especially for dairy cattle.
Van Wyk et al. (1955) published the first nutritive values for lucerne hay in South Africa, based on only 38 samples. This was followed by Van der Merwe (1970) who classified the quality of lucerne according to stage of harvesting (bloom stage). Van der Merwe (1970) emphasised that the nutritive value of roughages can vary considerably. Therefore their values (CP, crude fibre, fat) can serve at best as a general guideline in diet formulations and should be verified by actual analysis. These values also do not include later developments in nutrition like analysis for acid detergent fibre (ADF), neutral detergent fibre (NDF) and the extent of protein and fibre degradation. Mertens (1992), McDonald et al. (1995) and NRC (2001) reported values in this regard. The only South African data available are the average values of Erasmus et al. (1990) based upon three samples from various locations. Information is also lacking on nutritional value of South African lucerne hay with regards to nutrient fractions required for modern diet formulation programmes including National Research Council (NRC) (2001) and the Cornell Net Carbohydrate and Protein System (CNCPS) (Tylutki, 2007). Information is required on protein and carbohydrate fractions, detailed nutrient composition, predicted energy values and rumen degradability. This clearly illustrates the urgent need for more reliable data on the nutritive value of South African lucerne hay.
The aim of this study was to evaluate the variation- and expand the existing and limiting nutritive value database of lucerne hay in South Africa.
Materials and Methods
During 2004 and 2005, samples of lucerne hay (n = 600) (Medicago sativa L.) were collected from 600 cuttings at different times in the season (September 2004 to April 2005) from 300 (state number for each area) commercial irrigation farms in the main lucerne producing areas (sites) in South Africa, viz. Cradock (60), Douglas (60), Groblershoop (60), Hartswater (60) and Vaalharts (60) irrigation scheme, and Hopetown and Upington. These sites represent a wide range of soil characteristics (texture, organic matter, N and P content, pH) and farm management practices. The objective was to obtain an as wide as possible range of samples that would vary more in chemical and digestible properties than would lucerne sampled from a distinct area, thus representing the South African lucerne hay population.
Each lucerne hay sample was milled under controlled conditions through a 1-mm screen using a LM 3100 mill (Perten Instruments AB, Huddinge, Sweden). The samples were sealed in airtight containers and stored in a refrigerator at 3 ºC for subsequent near-infrared reflectance spectroscopy (NIRS) scanning and chemical analysis.
The required number of samples to represent the South African population was selected by scanning the 600 milled lucerne hay samples with a NIR-System 5000 monochromater (NIR Systems, Silver Spring, MD, USA) resulting in 168 samples representing a wide spectral range. These 168 samples were analysed by Cumberland Valley Analytical Services, Inc., (Maugansville, Maryland, USA) via wet chemistry. Crude protein, ash and ADF content were determined by means of the procedures described by AOAC (2000). Dry matter was determined by a two-step process: First step: Partial dry matter adapted from Goering & Van Soest (1970), second step: Modified to 105 ºC for 3 h as described by the National Forage Testing Association recommendations, 2002 (Ward, R.T., 2005, Pers. Comm., Cumberland Valley Analytical Services, Inc., 14515 Industry Drive, Hagerstown, MD 21742, USA). Neutral detergent fibre and acid detergent lignin (ADL) were determined according to the procedures of Goering & Van Soest (1970) with a modification to the NDF procedure, which excludes sodium sulphite (neutral detergent solution) and includes 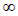 -amylase to hydrolyse the starch component (Dowman & Collins, 1982). Soluble protein (SP) was determined by the borate-phosphate procedure as detailed by Krishnamoorthy et al. (1982). Acid detergent insoluble crude protein (ADICP) and neutral detergent insoluble crude protein (NDICP) were determined by measuring the CP content of the ADF and NDF residue respectively using a Leco FP-528 Nitrogen Combustion Analyzer (Leco. 3000 Lakeview Avenue, St. Joseph, MI 49085). Fat (EE) was analysed according to the method described by the AOAC (1990). Starch was determined by a modification of the method of Holm et al. (1986) as cited by Hall (2000) and sugar by Dubois et al. (1957), as cited by Hall (2000). Mineral elements (Fe, Mn, Zn, Cu, Ca, P, Mg, K, Na, S, Cl) were analysed by means of the procedures described by AOAC (2000).
-amylase to hydrolyse the starch component (Dowman & Collins, 1982). Soluble protein (SP) was determined by the borate-phosphate procedure as detailed by Krishnamoorthy et al. (1982). Acid detergent insoluble crude protein (ADICP) and neutral detergent insoluble crude protein (NDICP) were determined by measuring the CP content of the ADF and NDF residue respectively using a Leco FP-528 Nitrogen Combustion Analyzer (Leco. 3000 Lakeview Avenue, St. Joseph, MI 49085). Fat (EE) was analysed according to the method described by the AOAC (1990). Starch was determined by a modification of the method of Holm et al. (1986) as cited by Hall (2000) and sugar by Dubois et al. (1957), as cited by Hall (2000). Mineral elements (Fe, Mn, Zn, Cu, Ca, P, Mg, K, Na, S, Cl) were analysed by means of the procedures described by AOAC (2000).
Cellulose and hemi-cellulose concentrations were estimated as the difference between ADF and ADL concentrations and NDF and ADF concentrations, respectively.
In vitro organic matter digestibility at 48 h (IVOMD48) was estimated using the two-stage rumen fluid-pepsin technique described by Tilley & Terry (1963). For the estimation of in vitro organic matter digestibility at 24 h (IVOMD24) the technique was, however, modified to 24 h without sulphite (neutral detergent solution) or pepsin/hydrochloric acid (HCL) treatment, but with the inclusion of amylase (Ward, 2005, Pers. Comm.). Rumen fluid was collected, and mixed, from six early to mid lactation cows consuming a high production total mix ration (TMR) of lucerne haylage, maize silage and high moisture shelled maize (HMSC) (Ward, 2005, Pers. Comm.). The 24 h and 48 h in vitro digestibilities of NDF (NDFD24 and NDFD48, respectively) were determined using the modified Tilley and Terry procedures as described above.
Statistical analyses were performed using SAS 9.1.3 Service Pack 4 (2002-2003). Descriptive statistics namely the mean, standard deviation (SD), coefficient of variation (CV), minimum and maximum values were calculated for the quantitative variables.
Results and Discussion
The chemical composition of the samples representing the South African lucerne hay population is set out in Table 1. A high DM content in lucerne hay was recorded. Accordingly, the moisture content was within the desired range (lower then 160 g/kg) for effective storage and prevention of heat damage and mould formation (Bath & Marble, 1989). On the other hand, the relatively high DM content observed for the lucerne hay in the current study could be explained by moisture losses during sample preparation (grinding) and storage. A relatively low moisture content in the lucerne hay was supported by a low average ADF-CP content, which is usually associated with heat damage caused by a high moisture content (Van Soest, 1994). Linn & Martin (1999) recommended an adjustment for CP availability when the proportion of CP in the ADF fraction increases above 120 g/kg of total CP. According to this figure heat damage only occurred in a small percentage (6%) of the samples. Accordingly, the low ADF-CP content could be an indication that lucerne hay moisture content was mostly within the desired range for effective storage.
The wide range observed for ash in this study may be explained by soil contamination (exogenous ash) due to harvest practices (Abreu & Bruno-Soares, 1996). This is confirmed by Bath & Marble (1989), which noted that values above 140 g/kg DM for lucerne hay are usually an indication of soil contamination. Thus, it is evident from the data set that 24% of the samples in the present study were prone to soil contamination. Accordingly, the high mean ash value (130 g/kg of lucerne hay in the current study could have had a detrimental effect on the energy value due to the zero energy contribution of ash. It is also important to note the high level of Fe in South African lucerne hay. This might interfere with the absorption of Cu and Zn in the diet which could lead to depletion of these minerals in high producing dairy cattle.
Similar to ash, lignin has also theoretically, no digestible energy. Therefore, the variation in lignin, and its role as a nutritional entity especially in forages, should be acknowledged in the quest for developing an accurate quality model for assessing animal performance. Variability in lignin was found to be the second highest (CV = 23%) of the fibre fractions. This was probably due to the lower and higher lignin and NDF content, respectively, found in grasses compared to lucerne hay. The probable overall effect of grass contamination of lucerne hay in the current study seems to be manifested in the majority of the energy related parameters (ADF, NDF, lignin, NDFD, etc.). Furthermore, the relatively low lignin/NDF relation (16.2% of NDF) of the samples in the present study was enough evidence to claim that the majority of lucerne hay grown in South Africa is not from pure strands but is a mixture of lucerne and grass. This phenomenon produced a dataset for South African lucerne hay with generally a larger variation than normally found in the available literature for this species.
The high NDF content of several lucerne hay samples in the current study proposed possible intake restriction thereof in high yielding dairy diets. Zinn et al. (2004) suggested that the lucerne hay in high producing dairy diets should contain a NDF content of 400 g/kg or less. Accordingly, only 33% of the South African population would have fitted this criterion. On the other hand, 8% of these samples contain less than 35 g NDF/kg; therefore, nutritionally disqualifying it as a roughage (Zinn et al., 2004).
The mean IVOMD at 24 h (IVOMD24) (69%) and 48 h (IVOMD48) (73%) values in the current study seem to be relatively similar, irrespective of incubation time, and are accordingly representative of the South African lucerne hay population. From these results it is evident that in vitro incubation time had a minor effect on digestibility of lucerne hay organic matter (OM) due to its low mean fibre content (Table 1), brittle texture and thus high degradability.
According to the results of the present study the mean crude protein (CP) (200.7 g/kg DM) and soluble protein (SP) (62 g CP/kg) content of lucerne hay was higher and lower, respectively, than those found in the literature. Therefore, it seems that the South African lucerne hay population contains a lower concentration of rapidly degradable protein as reported by the NRC (2001).
Conclusions
From the results of the present study the variation in energy and protein composition as well as utilisation of nutrients in lucerne hay is evident. It seems that this variation was exaggerated by soil and grass contamination.
References
Abreu, J.M.F. & Bruno-Soares, A.M., 1996. Soil intake by sheep - Effects in digestibility of roughage material. An. Inst. Sup. Agron. 45, 523-532. [ Links ]
AOAC, 1990. Official methods of analysis. 15th ed. Association of Official Analytical Chemists, Arlington, VA, USA. [ Links ]
AOAC, 2000. Official Methods of Analysis of AOAC International. Volume 1. 17th Edition. Ed. Horwitz, W., AOAC International® [ Links ].
Bath, D.L. & Marble V.L., 1989. Testing Alfalfa for its Feeding Value. Leaflet 21457. WREP 109. University of California Cooperative Extension, Agriculture and Natural Resources. 6701 San Pablo Ave., Oakland, CA 94608. [ Links ]
Dowman, M.G. & Collins, F.C., 1982. The use of enzymes to predict the digestibility of animal feeds. J. Sci. Food Agric. 33, 689-696. [ Links ]
Erasmus, L.J., Prinsloo, J., Botha, P.M. & Meissner, H.H., 1990. Establishment of a ruminal protein degradation database for dairy cattle using the in situ polyester bag technique. 3. Roughages. S. Afr. J. Anim. Sci. 20, 124-134. [ Links ]
Goering, H.K. & Van Soest, P.J., 1970. Forage fibre analyses (Apparatus, reagents, procedures and some applications). Agricultural Handbook No. 379. A.R.S., U.S. Dept. of Agric. [ Links ]
Hall, M.B., 2000. Neutral detergent soluble carbohydrates - Nutritional Relevance and Analysis. Univ. of Florida, Bulletin number 339. [ Links ]
Linn, J.G. & Martin, N.P., 1999. Forage quality tests and interpretations. College of Agricultural, Food and Environmental Sciences. University of Minnesota, USA. [ Links ]
McDonald, P., Edwards, R.A., Greenhalgh, J.F.D. & Morgan, C.A., 1995. Animal Nutrition, 5th ed., Longman. Singapore. [ Links ]
Mertens, D.R., 1992. Nonstructural and Structural Carbohydrates. In: Large Dairy Herd Management. Eds. Van Horn, H.H. & Wilcox, C.J., American Dairy Science Association, Champaign, IL. pp. 219-235. [ Links ]
NRC, 2001. Nutrient requirements of dairy cattle. 7th rev. ed. National Academy Press, Washington, DC. [ Links ] Putnam, D.H., Lamb, C., Peterson, G., Orloff, S. & Kirby, D., 1997. 1995 Alfalfa cultivar forage quality trial result. Agronomy progress report No. 256. University of California, Davis, USA. [ Links ]
SAS, 2003. SAS 9.1.3 Service Pack 4, SAS Institute Inc, Cary N.C., USA. [ Links ]
Tilley, J.M.A. & Terry, R.A. 1963. A two-stage technique for the in vitro digestion of forage crops. J. Br. Grassl. Soc. 18, 104-111. [ Links ]
Tylutki, T.P., 2007. Study guide, AMTS. Cattle version 1.1.0.1, AMTS, LLC, 418 Davis RD, Cortland, NY. 13045, USA. [ Links ]
Van der Merwe, F.J., 1970. Dierevoeding. Kosmo-uitgewery Edms Bpk., Stellenbosch, South Africa. [ Links ]
Van Soest, P.J., 1994. Nutritional Ecology of the Ruminant, 2nd Ed. Cornell Univ. Press, Ithaca, N.Y. [ Links ]
Van Wyk, H.P.D., Oosthuizen, S.A. & Meyer, E.E., 1955. Nutritive value of South African feeds. III. Hay and pasture crops, silage, cereals, tubers and pods. Science Bull. 354, 19-23. [ Links ]
Zinn, R.A., Corona, L. & Ware, R.A. 2004. Forage Quality: Impacts on cattle performance and economics. In: Proc. National Alfalfa Symposium, 13-15 Dec., San Diego, C.A. USA. [ Links ]
# Corresponding author. E-mail: Scholtzgd@ufs.ac.za














