Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Animal Science
On-line version ISSN 2221-4062
Print version ISSN 0375-1589
S. Afr. j. anim. sci. vol.38 n.2 Pretoria Feb. 2008
Threonine and lysine requirements for maintenance in chickens
M.K. Nonis; R.M. Gous
Animal and Poultry Science, School of Agricultural Sciences and Agribusiness, University of KwaZulu-Natal, Private Bag X01, Scottsville 3209, South Africa
ABSTRACT
In order to describe the response of broiler breeder hens to dietary amino acids and to develop an effective model for the precise feeding of these birds after sexual maturity, accurate estimates of the amounts of each amino acid required for maintenance and egg production are needed. The maintenance requirement for threonine and lysine were estimated in two different experiments by measuring the nitrogen balance of adult male cockerels. Measured amounts of a diet first-limiting in threonine or lysine were fed by intubation each day for 4 d to give a range of intakes (unbalanced series) of from 0 to 239 mg threonine/kg or 0 to 40 mg lysine/kg body weight. To confirm that threonine or lysine was first-limiting and that the response obtained was to threonine or lysine and not to protein, a second series of diets was used (balanced series) in which synthetic threonine or lysine was added to each diet in the unbalanced series. A nitrogen-free diet containing energy, vitamins and minerals was made available ad libitum during the balance period, to ensure that the birds remained in positive energy balance. The balance period was three days and excreta were collected in colostomy bags (threonine) or in trays (lysine) during that time. The nitrogen content of the excreta was determined on dried homogenised samples. The resultant linear regression equations describing the effect of threonine or lysine intakes on nitrogen retention were: N retention = -230.4 (± 27.6) + 4.134 (± 0.274) I (R2 = 91.9) and N retention = -256.8 (± 37.7) + 6.89 (± 1.37) I (R2 = 70.8), respectively, where I is the intake of threonine or lysine in mg/kg body weight day. The threonine and lysine required to maintain the body at zero nitrogen retention was therefore estimated to be 56 and 37 mg/kg body weight day, respectively.
Keywords: Maintenance requirements, amino acids, cockerels
Introduction
In order to develop an effective model for the precise feeding of broilers during growth and of broiler breeder hens after sexual maturity, the maintenance requirement for each essential amino acid must be known, particularly as the maintenance requirement becomes the major proportion of the total requirement as birds age. One method of determining these requirements is to use a factorial approach, such as is applied in the Reading Model (Fisher et al., 1973). Where this has been used, the coefficients of response of broiler breeders for maintenance were calculated by Bowmaker & Gous (1991) to be 11.2 mg/W.d and 1.52 mg/W.d for lysine and methionine respectively, where W is body weight, kg, these values being considerably lower than when derived from laying hens (73W and 31W for lysine and methionine, respectively) (McDonald & Morris, 1985). Such differences are unacceptably high, so a more satisfactory method of determining these requirements is needed.
A method of measuring maintenance requirements using adult male cockerels was suggested by Gous et al. (1984) and was applied successfully by Burnham & Gous (1992) for measuring the isoleucine required for maintenance of broilers. Maintenance amino acid requirements are normally defined at nitrogen (N) equilibrium, the state in which N intake exactly equals the sum of N losses so that the N content of the body remains constant (Sakomura & Coon, 2003). Ideally such studies should be conducted on adult male fowl, as these birds are most likely to be in a steady state thereby avoiding the confounding effect, when interpreting the results of trials conducted on growing birds, of partitioning the consumed amino acids between growth and maintenance. Nevertheless, most authors have approached the problem of estimating the amounts of an amino acid required for maintenance using response trials with populations of laying hens or growing birds. Fisher et al. (1973) developed the so-called Reading Model to estimate the coefficients of response (mg amino acid required per g egg output and per kg body weight.d) to amino acid intake in laying hens, and this technique was subsequently used relatively successfully with growing birds (Clark et al., 1982; Boorman & Burgess, 1986) even though the Reading Model applies only to populations that are in a steady state, which is not the case with growing animals. No attempt was made in these latter trials to reduce growth rate to zero when attempting to determine the maintenance requirements of the birds. Instead, the growth rate was extrapolated to zero and the intercept on the X-axis (amino acid intake/bird or /kg) produced the required coefficient. The technique used by Edwards et al. (1997; 1999) and Sakomura & Coon (2003), who used growing broilers and broiler breeder pullets respectively, differed considerably from the above technique. They used feeds that contained increasing amounts of the test amino acid as the only protein source and measured accretion of that amino acid by whole body analysis. This begs the question of whether growing birds would use a single amino acid for maintenance in the absence of any others, and whether this technique adequately addresses the problem of some birds losing weight whilst others gain weight during the balance period of three weeks.
An alternative approach to estimating these coefficients is to measure one of the coefficients independently of the other, i.e. measure maintenance in the absence of growth through the use of adult roosters (Leveille & Fisher, 1958; Gous et al., 1984; Gous, 1986). The maintenance requirement for a number of amino acids including threonine and lysine by adult roosters has been determined by Leveille & Fisher (1959; 1960) in which the N source used was a whole egg diet and a free amino acid mixture containing 13 amino acids, supplied at the same level as found in whole egg protein. The resultant curvilinear response for isoleucine in that trial suggested that an amino acid other than the one under test was probably first-limiting, and the observed food refusal of birds eating pelleted food first-limiting in threonine cast doubts as to the validity of the results obtained.
The present two studies were designed so as to eliminate the problems that have been indentified in earlier attempts to measure the maintenance requirements for threonine and lysine in poultry, by measuring the N balance of mature cockerels subjected to a range of intakes of a feed known to be first limiting in threonine or lysine. A technique similar to that suggested by Gous et al. (1984) was applied in these trials to measure the maintenance requirements for threonine and lysine.
Materials and Methods
Twenty four adult cockerels of a laying strain were housed in individual cages in both studies. Each cage was supplied with one nipple drinker and drip cup, and one feeder. The house was cross-ventilated using up to six fans. The lighting program used throughout the experimental period was 16L : 8D (04:00 -20:00).
For the threonine study only, a colostomy bag was used for collecting excreta (Burnham & Gous, 1992). Feathers around the cloaca were removed the day before the trial began, to allow better adhesion of the ring to that area and also for sanitary reasons. In the lysine study, feathers around the cloaca were also removed but no colostomy bag was used; instead, a tray covered with foil was suspended beneath each cage to catch the excreta.
In both studies, a protein-free basal diet was made available to the birds ad libitum to allow each bird to meet its daily energy, vitamin and mineral needs. A summit diet was formulated to contain all the essential amino acids in the balance assumed to meet the maintenance requirement of a bird, but with threonine or lysine at a lower concentration than required, thereby making this the limiting amino acid in the feed. The composition of the protein-free and summit diets used in the threonine (PFDT and SDT) and lysine (PFDL and SDl) studies are presented in Table 1. Quantities of the summit diet were weighed out (0, 10, 20, 30, 40 and 50 g, for the threonine study and 0, 2, 4, 6, 8 and 10 g for the lysine study) giving a series of intakes of threonine and lysine (unbalanced series) ranging from 0 to 239 mg/kg and 0 to 40 mg/kg body weight day, respectively. The range of intake was chosen to ensure that the intake resulting in zero N balance would be lying somewhere between the two extremes. The N-free diet was then added to the summit diet to make the amount of food allocated to the bird each day by intubation up to 50 g. To confirm that threonine or lysine was the limiting amino acid in the summit feed, and that the response obtained was to threonine or lysine and not to protein, a second series (balanced series) was prepared by adding to the summit feed 2 g/kg or 4 g/kg of synthetic threonine and lysine, respectively. The same amounts of the feeds in the balanced series were weighed out and N-free diet was also added to these feeds to make the daily allocation by intubation up to 50 g per day. Each feeding treatment was replicated twice. The threonine trial ran for three periods of 6 d, with the treatments being randomly allocated to the roosters in each period, i.e. each feeding treatment was replicated six times. The lysine trial ran for 6 d.
All the food was removed from the birds the day before starting the 6 d experimental period in order to clear the digestive tract of protein-containing material and this was replaced, on the first day of the 6 d trial period (24-hour after the start of the fasting period), with the N-free diet, which was made available ad libitum. On the second day (48-hour after the start of the fasting period), the test feeds were given by intubation, and this was repeated at the same time each day for 4 d. The procedure for fasting the birds prior to intubation, and the intubation procedure itself, is a recognised method for measuring digestibility (Fisher & McNab, 1987). On the morning of the third day (first day of the 3 d balance period) the body weight was measured, and after being force fed, for the threonine study only, each bird was fitted with a colostomy bag, which was attached over its cloaca, after which the bird was returned to its cage. On each subsequent day the bag or the tray from the previous day was removed before the bird was fed, and another bag was attached and tray (covered with a new piece of foil) replaced before the bird was returned to the cage. Ethical approval for these procedures was obtained from the University of KwaZulu-Natal animal ethics committee.
Three bags or three pieces of foil of excreta, each representing the daily output for the previous day, were thus collected for each bird. Each bag was cleaned with water, and the excreta were transferred into pre-weighed aluminium dishes and placed in an oven kept at 60 °C to dry for 48 h. Each piece of foil was directly placed in the oven. Subsequently, each aluminium dish or piece of foil plus contents was weighed to determine the weight of dry excreta by difference, and the dry faeces were milled. The N content of the dry sample was then determined using a Leco N analyser and thus the N output of each bird was calculated.
A linear regression of N retention on threonine and lysine intakes was fitted to the data from each replication, and a test of significance (linear regression with groups) was performed on the regression equations to establish whether they differed significantly from one another, using Genstat (1997).
Results
Four of the 24 birds in the threonine trial lost their colostomy bag during the collection period and were consequently removed from the trial, and four other birds died on the first day of the trial period. The remaining 16 birds were used in the first 6 d trial period. Three birds during the second 6 d trial period and one bird during the third period lost their colostomy bags, resulting in 13 birds and 12 birds being used during the second and the third trial periods, respectively.
It became clear from the threonine study results that the N balance of birds receiving more than 180 mg/kg body weight varied considerably thereby increasing the standard error of the regression equation. Given that the requirement of threonine for maintenance was in the region of 60 mg/kg, it was decided to use in the analysis only data of birds fed up to 180 mg threonine/kg body weight day.
The body weights of the cockerels at the start of the balance periods, the intakes of threonine or lysine and the N retention for each of the remaining replications are presented in Tables 2 and 3.
The resultant regression equations for the balanced and the unbalanced series are given in Table 4 for the threonine sudy and Table 5 for the lysine study.
The slopes and the intercepts of the two series did not differ significantly one from the other in both studies. Consequently, data from both two series were pooled to increase the number of replications of each feeding treatment. The resultant regression equation for the threonine and lysine studies were: N retention = -230.4 (± 27.6) + 4.134 (± 0.274) I (R2 = 91.9) and N retention = -256.8 (± 37.7) + 6.89 (± 1.37) I (R2 = 70.8), respectively, where I is the intake of threonine or lysine in mg/kg body weight day (Figures 1 and 2).
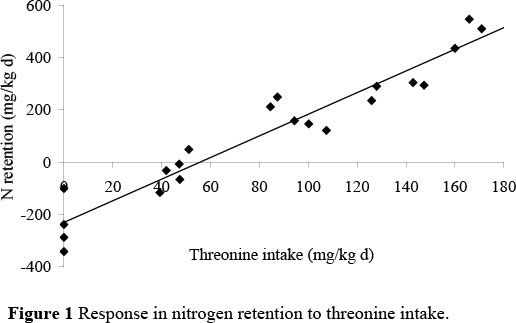
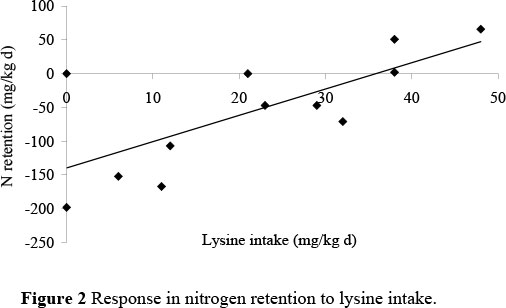
The maintenance requirement for threonine and lysine, calculated as the threonine and lysine intake required to maintain the body of an adult cockerel at zero N retention, were estimated to be 56 mg threonine and 37 mg lysine per kg body weight day.
Discussion
When modelling amino acid requirements for maintenance of broiler breeders after sexual maturity, an important concern is to determine the amount of each amino acid required to maintain a hen in a functioning state at zero egg production. The maintenance requirements for threonine have been estimated for cockerels by Leveille & Fisher (1960) as 74 and 55 mg/kg for what they termed the maintenance and the minimum maintenance requirements, respectively. The maintenance requirement was defined as the lowest amount of dietary amino acid which maintained the same N balance as observed on a whole egg protein diet or an amino acid mixture containing all the essential amino acids in the same composition as whole egg protein.
The minimum maintenance requirement was defined as the lowest dietary inclusion rate of an amino acid which maintained zero N balance and was equivalent to the definition used in the present study. The requirement for lysine was found not to exceed 29 mg/kg d and no minimal maintenance level could be demonstrated (Leveille & Fisher 1959). Bowmaker & Gous (1991) found that broiler breeders have a lysine requirement for maintenance of 11.2 mg/kg body weight, these values being considerably lower than 73 mg/kg when derived from laying hens (McDonald & Morris, 1985). The result obtained for the threonine requirement is very close to Leveille's values, which shows the confidence of using the method employed in this trial to estimate the amount of amino acid required for maintenance. Indeed, as suggested by Burnham & Gous (1992) for isoleucine, the technique used here ensures that the correct daily amount of the first-limiting amino acid is consumed. The collection of excreta directly into colostomy bags prevents contamination of faeces by feathers, skin, feed and other debris, rich in N, that will increase the apparent N output, resulting in a lower estimate of the maintenance requirement for that amino acid. Free access to a N-free diet allows the bird to meet its energy demand without having to mobilise body protein reserves and supplies essential trace nutrients. On the other hand, the lysine requirement for maintenance obtained in the study is higher than the one found previously in broiler breeders, showing that collecting excreta directly into trays exposed faeces to contamination by debris rich in N. We believe that the experimental procedure employed in the threonine study is the most sensible approach for determining the daily requirement of each amino acid for maintenance of poultry: the results between the three trial periods were uniform, the slopes of the responses to the unbalanced and balanced series of diets were the same, and so the response obtained was to threonine and not to protein. There appeared to be no specific reason for the mortality that occurred on the first day of the first trial. The same technique was used previously (Burnham & Gous, 1992) with no such adverse effects, nor has this problem occurred subsequently during further studies in our facilities.
A question regarding the amino acid nutrition of hens that requires closer scrutiny is the extent to which the maintenance requirements are related to body weight as opposed to body protein content. Because adult birds generally have considerable lipid reserves (the lipid content of carcasses of broiler breeders can vary from 15.1 to 17.3% from 30 to 60 weeks of age, respectively) (Unpublished data, University of KwaZulu-Natal, 2005) and because it can be argued that there is no energy cost in maintaining such lipid reserves (Emmans & Oldham, 1988), it is unlikely that the amino acids required for maintenance (as defined by the b value of the Reading Model) would be related directly to the body weight of the bird. A more accurate estimate of the amount required for maintenance, when comparing adults of different size and body composition, would be that related to the protein content of the body. To this end, the maintenance requirement was scaled according to the feather-free body protein content and its degree of maturity by Emmans & Fisher (1986) in their adaptation of Taylor's (1980) size scaling rule. The equation suggested by Emmans and Fisher (1986) is: MP = mpPm0.73u, where MP = maintenance protein requirement (g/day), mp = 0.008 kg ideal protein/unit day, Pm = mature protein weight (kg) and u = degree of protein maturity (P/Pm), equal to 1 in this study. Assuming a mean feather-free body protein content of 160 g/kg body weight and a body weight of 2.5 kg for the birds used in this trial, the body protein weight would be 0.40 kg. From the result of both studies, the maintenance requirement can be expressed as (56*2.5) = 140 mg threonine and (37*2.5) = 93 mg lysine /bird day and because the birds were mature, as (140/0.40073) = 273 mg threonine and (93/0.40073) = 182 mg lysine /unit Pm073 day. Using the above equation of Emmans & Fisher (1986), the threonine and lysine body contents are thus 273/8 = 34 g/kg and 182/8 = 23 g/kg protein, which is lower than the coefficients of 42 g/kg and 75 g/kg protein suggested by Emmans (1989). To obtain a coefficient of 44 g threonine/kg protein, which is closer to that suggested by Emmans (1989), the maintenance requirement should be related to body protein and not body protein^0.73.
Conclusion
The maintenance requirement for amino acids is an area of nutrition presenting many problems because the concept is not well defined. However, the improved method suggested by Gous et al. (1984) has proved to be of value in determining the maintenance requirements for amino acids in mature birds (Burnham & Gous, 1992; the present study) and thus further experiments of this nature are warranted, in which the requirements of all of the essential amino acids for maintenance are measured, as these values will improve the accuracy of models of amino acid requirements for broiler breeders.
Acknowledgements
The authors would like to express their sincere appreciation to Oscar Blanco (Animal and Poultry Science, University of KwaZulu-Natal, Pietermaritburg) and Natalie Armour (Allerton Provincial Veterinary Laboratory, Pietermaritzburg) for their veterinary assistance, and to the staff at the poultry section of the University Research Farm, D. Davies, M. Hundley and S. Opperman for their technical assistance.
References
Boorman, K.N. & Burgess, A.D., 1986. Responses to amino acids. In: Nutrient Requirements of Poultry and Nutritional Research. Eds. Fisher, C. & Boorman, K.N., Oxford, Butterworths. pp. 99-123. [ Links ]
Burnham, D. & Gous, R.M., 1992. Isoleucine requirements of the chicken: requirement for maintenance. Br. Poult. Sci. 33, 59-69. [ Links ]
Clark, F.A., Gous, R.M. & Morris, T.R., 1982. Response of broilers to well-balanced protein mixtures. Br. Poult. Sci. 23, 433-446. [ Links ]
Edwards III, H.M., Baker, D.H., Fernandez, S.R. & Parsons, C.M., 1997. Maintenance threonine requirement and efficiency of its use for accretion of whole-body threonine and protein in young chicks. Br. J. Nutr. 78, 111-119. [ Links ]
Edwards III, H.M., Fernandez S.R. & Baker, D.H., 1999. Maintenance lysine requirement and efficiency of using lysine for accretion of whole-body lysine and protein in young chicks. Poult. Sci. 78, 1412-1417. [ Links ]
Emmans, G.C., 1989. The growth of turkeys. In: Recent Advances in Turkey Science. Eds. Nixey, C. & Grey, T.C., London, Butterworths. pp. 135-166. [ Links ]
Emmans, G.C. & Fisher, C., 1986. Problems in nutritional theory. In: Nutrient Requirements of Poultry and Nutritional Research. Eds. Fisher, C. & Boorman, K.N., London, Butterworths. pp. 9-40. [ Links ]
Emmans, G.C. & Oldham, J.D., 1988. Modelling of growth and nutrition in different species. In: Modelling of Livestock Production Systems. Eds. Karver, S. & Van Arendonk, J.A.M., Dordrecht, Kluwer Academic Publishers. pp. 13-21. [ Links ]
Fisher, C. & McNab, J.M., 1987. Techniques for determining the metabolizable energy (ME) content of poultry feeds. In: Recent Advances in Animal Nutrition. Eds. Haresign, W., Cole, D.J.A., London, Butterworths. pp. 3-18. [ Links ]
Fisher, C., Morris, T.R. & Jennings, R.C., 1973. A model for the description and prediction of the response of laying hens to amino acid intake. Br. Poult. Sci. 14, 469-484. [ Links ]
Gous, R.M., 1986. Measurements of response in nutritional experiments In: Nutrient Requirements of Poultry and Nutritional Research. Eds. Fisher, C. & Boorman, K.N., Oxford, Butterworths. pp. 41-57. [ Links ]
Gous, R.M. Fisher, C. & Broadbent, L.A., 1984. Measurements of the amino acid requirement for maintenance of adult cockerels. Proc. XVII World's Poult. Congress, Helsinski, Finland. [ Links ]
Leveille, G.A. & Fisher, H., 1958. The amino acid requirements for maintenance in the adult rooster. I. Nitrogen and energy requirements in normal and protein-depleted animals receiving whole egg protein and amino acid diets. J. Nutr. 66, 441-453. [ Links ]
Leveille, G.A. & Fisher, H., 1959. Amino acid requirements for maintenance in the adult rooster. II. The requirements for glutamic acid, histidine, lysine and arginine. J. Nutr. 69, 289-294. [ Links ]
Leveille, G.A. & Fisher, H., 1960. Amino acid requirements for maintenance in the adult rooster. III. The requirements for leucine, isoleucine, valine and threonine, with reference also to the utilisation of the D-isomers of valine, threonine and isoleucine. J. Nutr. 70, 135-140. [ Links ]
McDonald, M.W. & Morris, T.R., 1975. Quantitative review of optimum amino acid intakes for young laying pullets. Br. Poult. Sci. 26, 253-264. [ Links ]
Sakomura, N.K. & Coon, N.C., 2003. Amino acid requirements for maintenance of broiler breeder pullets. Proc. 14th European Symp. Poult. Nutr., Lillehammer, Norway. [ Links ]
Taylor, St. C.S., 1980. Genetic scaling rules in animal growth. Anim. Prod. 30, 161-165. [ Links ]
 Correspondence:
Correspondence:
E-mail: nonis_fr@yahoo.fr














