Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
SAMJ: South African Medical Journal
versão On-line ISSN 2078-5135
versão impressa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.114 no.4 Pretoria Abr. 2024
http://dx.doi.org/10.7196/SAMJ.2024.v114i4.1296
RESEARCH
Policy brief: Optimising antimicrobial usage in paediatric inpatient hospital settings
J CloeteI; M KarsasII; T ChettyIII; A PillayIV; M ArcharyV; D MooreVI; T ReddyVII; Y BalakrishnaVIII; F NakwaIX; R LancasterX; A GogaXI; P JeenaXII
IMB ChB, MMed (Paeds); Department of Paediatrics and Child Health, Steve Biko Academic Hospital, University of Pretoria, South Africa; and Maternal and Infant Health Care Strategies Research Unit Centre, University of Pretoria, South Africa
IIMB ChB, FC Paeds (SA); Department of Paediatrics and Child Health, Steve Biko Academic Hospital, University of Pretoria, South Africa; and Maternal and Infant Health Care Strategies Research Unit Centre, University of Pretoria, South Africa
IIIMB ChB, PhD; HIV and Other Infectious Diseases Research Unit, South African Medical Research Council, Durban, South Africa; and Discipline of Public Health Medicine, University of KwaZulu-Natal, Durban, South Africa
IVFC Paed (SA),Cert ID Paed (SA); Department of Paediatrics and Child Health, Nelson R Mandela School of Medicine University of KwaZulu-Natal, Durban, South Africa
VMB ChB, PhD; Department of Paediatrics and Child Health, Nelson R Mandela School of Medicine University of KwaZulu-Natal, Durban, South Africa
VIFC Paed (SA), PhD; Department of Paediatrics and Child Health, Chris Hani Baragwanath Academic Hospital, School of Clinical Medicine, Faculty of Health Sciences, University of the Witwatersrand, Johannesburg, South Africa; and South African Medical Research Council Vaccine and Infectious Diseases Analytics (VIDA) Research Unit, University of the Witwatersrand, Johannesburg, South Africa
VIIMSc, PhD; Biostatistics Research Unit, South African Medical Research Council, Durban, South Africa; and School of Mathematics, Statistics and Computer Science, University of KwaZulu-Natal, Durban, South Africa
VIIIPhD (Statistics); Biostatistics Research Unit, South African Medical Research Council, Durban, South Africa; and School of Mathematics, Statistics and Computer Science, University of KwaZulu-Natal, Durban, South Africa
IXMB BCh, MMed Paeds; Department of Paediatrics and Child Health, Chris Hani Baragwanath Academic Hospital, School of Clinical Medicine, Faculty of Health Sciences, University of the Witwatersrand, Johannesburg, South Africa; and South African Medical Research Council Vaccine and Infectious Diseases Analytics (VIDA) Research Unit, University of the Witwatersrand, Johannesburg, South Africa
XB Pharm, Pharm D (Rhodes); Affordable Medicines Directorate, National Department of Health, Pretoria, South Africa
XIMB ChB, PhD; HIV and Other Infectious Diseases Research Unit, South African Medical Research Council, Durban, South Africa; and Discipline of Public Health Medicine, University of KwaZulu-Natal, Durban, South Africa
XIICert Paeds Pulm (SA), PhD; Department of Paediatrics and Child Health, Nelson R Mandela School of Medicine University of KwaZulu-Natal, Durban, South Africa
ABSTRACT
This policy brief document utilised an original published study from three academic hospitals in South Africa (SA) and all relevant published literature to develop recommendations on several strategies, summarised by the acronym 'PRACTICE', to optimise antimicrobial prescribing practices. These include implementing standardised policies for empirical antimicrobial use, routine review of antimicrobial therapy, age-specific antimicrobial stewardship programmes and continued collaborative efforts and research. Individualised treatment plans, improved infection prevention and control measures, ongoing surveillance and exploring electronic technology for antimicrobial stewardship are also crucial in combating the growing threat of antimicrobial resistance and improving patient outcomes in low- and middle-income countries (LMICs) such as SA.
Antimicrobial resistance (AMR) poses a global threat to health, partly fueled by antimicrobial overuse. Paediatric inpatients are particularly vulnerable to infections, leading to high antimicrobial consumption. In low- and middle-income countries (LMICs) such as South Africa (SA), research on antimicrobial usage for neonatal and paediatric healthcare-associated infections (HAI) is limited.
Antimicrobial overuse is an important contributor to the development of AMR globally. A high use of antimicrobials has been reported among paediatric inpatients due to infectious pathologies, nonspecific disease presentations of sepsis,[1] and difficulties in excluding various infections. The utilisation of antimicrobials among paediatric inpatients ranges widely from 33% to 93% in Europe and India.[2,3] In SA, a LMIC burdened by multiple healthcare challenges such as HIV, tuberculosis (TB), malnutrition, COVID-19 and prematurity, there remains a dearth of research on the usage of antimicrobials for neonatal and paediatric HAIs and community-acquired infections (CAI). [4]
HAIs have been linked to increased antimicrobial usage, which further contributes to the development of AMR.[5] Noteworthy variations in antimicrobial use for HAIs have been observed ranging from 14.4% among African countries in 2012 to 29% in a SA academic hospital in 2018.[3,5] Limited SA literature on hospitalised neonates and children indicates that HAI primarily consists of bloodstream infections, urinary tract infections and hospital- acquired pneumonia.[6-8] Several risk factors have been identified for HAI, including prematurity, malnutrition, intensive care admission and having indwelling devices.[7,9-11] Additionally, HIV exposure and infection have been identified as a significant risk for HAI in SA children.[7,12] Given the urgency of the need for treatment of neonatal infections and sepsis, early use of antimicrobial agents, particularly antibiotics, is justified.[13,14] In a global cross-sectional survey in neonatal intensive care units in LMICs, there was considerable variability in antimicrobial usage, from 17% to 48%. Antimicrobial therapy for HAIs followed an empirical approach, accounting for 55% (293/531) of cases, with 38% targeted towards specific infections.[11]
Sparse data exist on whether HAI drives antimicrobial use in LMIC settings. In a cross-sectional study that evaluated antimicrobial usage in three academic public sector hospitals in SA to improve appropriateness, 22.9% of hospitalised children received at least one prescribed antimicrobial, with neonates, infants and adolescents having higher prescription rates for HAIs.[4,15] Common antimicrobials prescribed included beta-lactamase-sensitive penicillin, aminoglycosides and carbapenems. Antimicrobial selection aligned with the World Health Organization (WHO) Access Watch and Reserve (AWaRe) classification system. HIV infection did not emerge as a risk factor for HAIs or excessive antimicrobial usage. This policy brief presents findings and recommendations from a from this study.[15]
Policy objective
To improve the appropriate utilisation of antimicrobials in paediatric hospitals in LMICs.
Study design
A cross-sectional study was conducted between 22 September 2021 and 5 January 2022, using anonymised data from inpatients at three hospitals. The study included all newborns and children (aged 0 - 15 years) who were admitted to three hospitals: Inkosi Albert Luthuli Central Hospital in KwaZulu-Natal Province, and Steve Biko Academic Hospital and Chris Hani Baragwanath Academic Hospital in Gauteng Province, all in SA. The study methodology adhered to the guidelines provided by the WHO methodology for conducting antimicrobial point prevalence surveys (PPS) in hospitalised children.[15]
Ethical approval
Ethical approval for the study was received from all the institutions ethics boards of the University of Kwazulu-Natal (ref. no. BREC 000002607/2021)/, the University of Witwatersrand (ref. no. R14/49), the University of Pretoria (ref. no. 515/2020), and the South African Medical Research Council (ref. no. EC023-5/2021), and site-specific institutional andprovincial approvals were also obtained. No personal identifying data other than date of birth was collected, and therefore informed consent was waived. Staff privacy was ensured, and a no-blame approach to antimicrobial prescribing was adopted.
Participants
Inclusion criteria: All children from 1 day of life to 15 years old who were hospitalised and had an antimicrobial prescription at the respective neonatal and paediatric wards (including neonatal and paediatric intensive care units (ICUs)) at 08h00 on each day of the survey were included in the study.
Exclusion criteria: Children previously recruited into the study during the study period; undergoing treatment as inpatients and discharged on the same day; or hospitalised children receiving only topical or ophthalmologic antibiotics were excluded.
Patient sampling
Antimicrobial prevalence among hospitalised patients was assumed to be 40% with a precision of 4% standard deviation. To achieve a margin of error of 6% and account for the design effect of 1.5 per site, a minimum sample of 384 children per hospital was required to estimate prevalence rates ranging from 30% to 40%. Each hospital aimed to recruit 400 patients to accommodate potential data quality issues. Chris Hani Baragwanath Hospital had almost twice as many paediatric and neonatal inpatient beds as the other hospitals, and therefore every second patient meeting the eligibility criteria from an alphabetised list created on the survey day was sampled, as per the WHO PPS methodology. All patients meeting the eligibility criteria were sampled from the other two hospitals.
Data analysis
Descriptive statistical methods were used to present results as frequencies, percentages and cross-tabulations. Overall antimicrobial prescribing prevalence, and prescribing prevalence stratified by ward and site, were calculated. The antimicrobials considered for analysis included antibiotics, antimycobacterial agents and antifungals. Data were analysed across all sites using Stata version 16 (STATACorp, USA).
Key indicators assessed in the study were as follows: antimicrobial prescription prevalence rate; indication for an antimicrobial prescription (CAI, HAI, or prophylaxis); antimicrobial spectrum prescribed (antibacterial, antimycobacterial, antifungal); the proportional contribution to overall antimicrobial usage and classification of prescribed antibiotics based on the WHO AWaRe classification system.
Furthermore, multivariable Poisson regression analyses were conducted to examine associations with HAI (p<0.1). These analyses aimed to explore potential factors influencing the occurrence of HAIs and their relationship with antimicrobial usage.
Results
A total of 1 946 antimicrobials were prescribed to 1 191 children. Of the 5 200 hospitalised children, 22.9% (95% CI 15.5 - 32.5%) received at least one prescribed antimicrobial. Among the 1 191 children who had prescriptions for antimicrobials, those <1 year of age accounted for 66.7% of the population on antibiotics and antifungals. The prescriptions for neonates was 24.2% (288), infants 29.1% (347), while children aged 1 - 5 years and 6 - 12 years constituted 27.2% (324) and 19.5% (215), respectively.
The commonly prescribed antibiotics in children <1 year of age were beta-lactamase-sensitive penicillin (benzylpenicillin, combination of benzylpenicillin, and procaine-benzylpenicillin), aminoglycosides (amikacin and gentamicin) and carbapenems (ertapenem, imipenem and meropenem). Older children (1 - 15 years) were frequently prescribed a combination of penicillin (amoxicillin-clavulanate) and carbapenems (ertapenem, imipenem and meropenem). The selection of antimicrobials at the three academic hospitals aligned appropriately with the AWaRe classification (Fig. 1).[15]
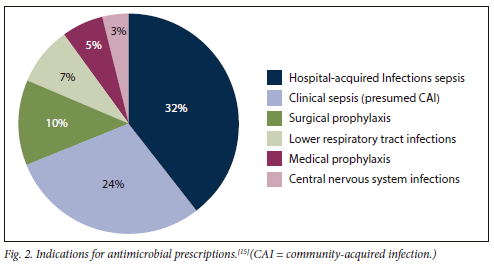
The top primary reasons for which antimicrobials were prescribed were HAI sepsis (32%, 382), clinical sepsis (24%, 287), surgical prophylaxis (10%, 123), lower respiratory tract infections (7%, 92), medical prophylaxis (5%, 66) and central nervous system infections (3%, 42) (Fig. 2).
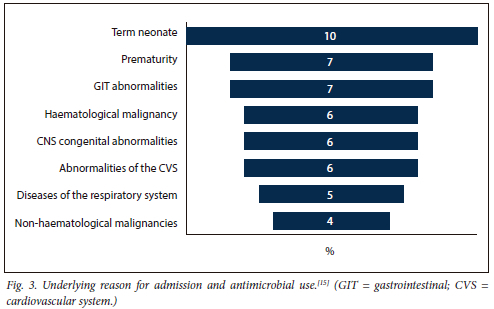
The predominant reason for admission for the patients was mainly neonatal conditions (17%) followed by other system abnormalities (Fig. 3).
In the case of SARS-CoV-2-infected participants, the high rate of antimicrobial usage, especially antibiotic utilisation (48.2%, 54) was attributed to suspected or confirmed co-infections. However, due to the limited sample size, further data are required to evaluate the appropriateness of antibiotic use in children with SARS-CoV-2 infection.
In this study, a small proportion of children were living with HIV (2.8%, 33), most of whom (84.8%, 28) were receiving antiretroviral therapy, indicative of an effective prevention of mother-to-child transmission programme. Notably, only 70 microbial prescriptions were written for children living with HIV, and did not emerge as a risk factor for HAI or excessive antimicrobial usage.
In multivariable analysis, various age groups (neonates (0 - 28 days); infants (29 days - 364 days); children (1 - 5 years); children (6 - 12 years) and adolescents (13 - 15 years)) showed various levels of risk for antimicrobial prescriptions related to HAI. Using 6 - 12 years as the reference group, neonates had a 1.64 times higher risk of antimicrobial use for HAI (adjusted relative risk (aRR) 1.64; 95% CI 1.06 - 2.53). Infants (29 - 364 days old) had 1.57 times higher risk (95% CI 1.12 - 2.21), and adolescents (13 - 15 year old) had the highest risk with a 2.18 - fold increase of antimicrobials for HAI (95% CI 1.45 - 3.29). Furthermore, preterm birth (aRR 1.33; 95% CI 1.04 - 1.70) and underweight (aRR 1.25; 95% CI: 1.01 - 1.54) were independently associated with antimicrobial prescribing to treat confirmed and suspected HIA.[15]
Recommendations: Addressing antimicrobial usage for HAIs
The recommendations are based on the study described above.[15] The study revealed extremely high antimicrobial usage for HAIs, variations in prescribing practices across hospitals, age-related disparities in antimicrobial prescription rates for HAIs, as well as the association of preterm birth and underweight status with higher prescription rates. We recommend the following to guide clinicians in optimising antimicrobial prescribing practices, and by implementing these recommendations, clinicians can contribute to reducing antimicrobial prescribing disparities in HAIs, optimise patient care, mitigate the risk of antimicrobial resistance and safeguard the health and wellbeing of paediatric patients. We created an acronym 'PRACTICE' (Fig. 4) to aid clinicians in remembering steps to improve antimicrobial practices.
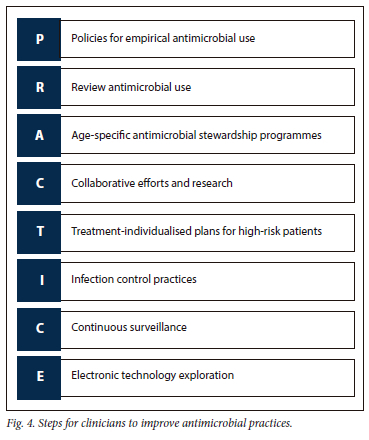
Policies for empiric antimicrobial use: Based on the variations observed in overall antimicrobial prescribing prevalence across different sites (29.1% at Hospital A, 40.8% at Hospital B and 14.1% at Hospital C), healthcare facilities must implement standardised policies regarding empiric antimicrobial use.[15]
Review antimicrobial use: One effective approach is to routinely de-escalate or discontinue all empiric antimicrobials at 72 hours unless there is a justified need for their continuation. This ensures that antimicrobial therapy is appropriate, tailored to the patient's clinical condition, and minimises unnecessary use, thereby combating antimicrobial resistance. For patients with HAIs, changing and adjusting antimicrobials according to the microbial cultures and sensitivity is of vital importance.
Age-specific antimicrobial stewardship programmes: Implement age-specific antimicrobial stewardship programmes to address the observed variations in antimicrobial prescription rates across different age groups. Tailored interventions should be designed to promote judicious use of antimicrobials, especially in neonates, infants and adolescents, who face significantly higher prescription risks for HAIs.
These programmes should encompass educational initiatives, clinical guidelines and regular audits to monitor prescribing practices.
Continued collaborative efforts and research: Foster multidisciplinary collaboration among clinicians, infectious disease specialists, microbiologists, pharmacists and infection control teams to establish a comprehensive approach to antimicrobial stewardship.
Regular communication and sharing of best practices can help to identify areas for improvement, implement evidence-based strategies and monitor the impact of interventions. We must sustain efforts to monitor and evaluate antimicrobial prescribing practices, particularly in the context of HAIs, to assess the effectiveness of interventions and identify areas requiring further improvement.
Individualised treatment plans: Develop individualised treatment plans for patients at higher risk of infection, considering their age, underlying medical conditions and other risk factors. Ensure that antimicrobial therapy is prescribed judiciously and tailored to the specific microbial pathogens involved. This includes assessing the duration, dosage and selection of antimicrobials according to evidence-based guidelines and patient-specific factors.
Improved infection prevention and control: Enhance infection prevention and control measures, with a focus on reducing the incidence of HAIs in vulnerable populations, such as preterm infants and underweight children. Emphasise strict adherence to hand hygiene protocols, appropriate disinfection practices and targeted interventions for patients requiring invasive procedures or advanced monitoring. Robust infection prevention practices can help minimise the need for antimicrobial treatment in these high-risk populations.
Continued surveillance: Ongoing surveillance can provide valuable insights into trends, resistance patterns and emerging infectious risks, enabling clinicians to adapt strategies and guidelines accordingly. Utilising current National Institute for Communicable Diseases surveillance programmes or local laboratory surveillance is a vitally important collaboration.
Explore the use of electronic technology for antimicrobial stewardship: In healthcare settings where there is a shortage of medical personnel, leveraging electronic technology to link microbiological results to anitimcrobial prescribing may be highly beneficial. This approach would enable timely access to microbiology data, facilitating more targeted and informed decision-making in antimicrobial therapy. By integrating microbiological information into the prescribing process, healthcare providers can make evidencebased decisions, optimise antimicrobial treatment and contribute to a reduction of antimicrobial resistance.
By implementing these PRACTICE recommendations, clinicians can contribute to reducing antimicrobial prescribing disparities in HAIs, optimising patient care, mitigating the risk of antimicrobial resistance and safeguarding the health and wellbeing of paediatric patients.
Conclusions
While additional research is warranted to gather more comprehensive insights into antimicrobial utilisation and associated outcomes in diverse patient populations, the recommendations should be implemented immediately to avoid a major catastrophe of having no antimicrobial to treat extreme and multidrug resistance pathogens.
Declaration. None.
Acknowledgements. The authors acknowledge the contributions of the following clinical staff members who assisted in facilitating the set-up of this study at the Chris Hani Baragwanath Academic Hospital site: Prof. S Velaphi, Department of Pediatrics and Child Health; Prof. G Naidu, Department of Pediatric Hematology-Oncology; Prof. J Loveland, Department of Pediatric Surgery; Prof. M T Ramokgopa, Department of Orthopedic Surgery; Prof. R Ouma and Dr D Poati, Department of Neurosurgery; Dr M R Ahmed, Department of Otorhinolaryngology; Prof. R Mathivha and Dr K Naidoo, Pediatric Critical Care; Dr J Wadula, Clinical Microbiology, National Health Laboratory Service; Matron M Ntikana, Paediatric Nursing. We further acknowledge the following staff members who assisted with data capture at the Chris Hani Baragwanath Academic Hospital site: Mr J Serumula and Mr T Mentoor. The team at IALCH would like to acknowledge Dr SA Thula, Paediatric Critical Care; Dr L Naidoo, Neonatal Intensive Care; Dr L Mubaiwa, Department of Pediatric Neurology; Prof. R Bhimma, Department of Pediatric Nephrology; Dr E Hoosen, Department of Pediatric Cardiology; Dr S H Sheik-Gafoor, Department of Pediatric Surgery; Dr W Kuhn, Department of Otorhinolaryngology; Dr B Enicker, Department of Neurosurgery; Prof. M Madaree, Department of Plastic & Reconstructive Surgery; Dr B Neethling, Department of Pediatric Hematology/Oncology; Prof. T Hardcastle, Department of Trauma & Burns; Dr R Madansein, Department of Cardiothoracic and Dr Y Mahabeer, Clinical Microbiology, National Health Laboratory Services. The team at SBAH would like to acknowledge the following clinical staff members who assisted in facilitating the set-up of thisstudy: Prof. R Green, Department of Pediatrics and Child Health, Dr E Müller, Department of Paediatric Surgery; Prof. R Goller, Department of Pediatric Orthopedic Surgery; Prof. M Tshifularo, Department of Otorhinolaryngology; Dr N Lourens, Department of Paediatric Urology. We further acknowledge the following staff members who assisted with data capture at the Steve Biko Academic Hospital: Dr T Gokar, Dr S Ndlovu and Ms C Shabalala. We would like to acknowledge and thank the data management team: Ms. Natasha Titus, Mr. Skhumbuzo Mzimela and Ms. Khanya Mohlabi for the development of the database and data cleaning and curation.
Author contributions. JC: conceptualisation, methodology, writing original draft with reviewing and editing, visualisation; MK, TC, AP, MA, DM, AG, PJ: conceptualisation, methodology, writing reviewing and editing, visualisation; TR, yb: conceptualisation, methodology, data curation, formal analysis, writing reviewing and editing; FN, RL: writing, reviewing and editing
Funding. This study was funded by UNICEF (grant number ZAR/PCA2021212/PD2021219-1) with contributions from the South African Medical Research Council. DM is partly supported by a grant awarded by the Carnegie Corporation of New York.
Conflicts of interest. None.
References
1. Hotchkiss RS, Moldawer LL, Opal SM, Reinhart K, Turnbull IR, Vincent JL. Sepsis and septic shock. Nat Rev 20163(1):1-21. https://doi.org/10.1038/nrdp.2016.45 [ Links ]
2. Gandra S, Singh SK, Jinka DR, et al. Point prevalence surveys of antimicrobial use among hospitalised children in six hospitals in India in 2016. Antibiotics 2017;13;6(3):19. https://doi.org/10.3390/antibiotics6030019 [ Links ]
3. Versporten A, Bielicki J, Drapier N, et al. The Worldwide Antibiotic Resistance and Prescribing in European Children (ARPEC) point prevalence survey: Developing hospital-quality indicators of antibiotic prescribing for children. J Antimicrob Chemother 2016;71(4):1106-1117. https://doi.org/10.1093/JAC/DKV418 [ Links ]
4. Godman B, Egwuenu A, Haque M, et al. Strategies to improve antimicrobial utilisation with a special focus on developing countries. Life 2021;11(6):528. https://doi.org/10.3390/LIFE11060528 [ Links ]
5. Koopmans LR, Finlayson H, Whitelaw A, Decloedt EH, Dramowski A. Paediatric antimicrobial use at a South African hospital. Int J Infect Dis 2018;74:16-23. https://doi.org/10.1016/jijid.2018.05.020 [ Links ]
6. Dusé AG. Infection control in developing countries with particular emphasis on South Africa. S Afr J Epidemiol Infect 201530(2):37-41. https://doi.org/10.1080/10158782.2005.11441230 [ Links ]
7. Dramowski A, Whitelaw A, Cotton M. Burden, spectrum, and impact of healthcare-associated infection at a South African children's hospital. J Hosp Infect 2016;94(4):364-372. https://doi.org/10.1016/j.jhin.2016.08.022 [ Links ]
8. Cotton M, Berkowitz FK, Berkowitz Z, Becker P, Heney C. Nosocomial infections in black South African children. Pediatr Infect Dis J 1989;8(10):676-682. https://journals.lww.com/pidj/Abstract/1989/10000/Nosocomial_infections_in_Black_South_African.3.aspx (accessed 1 July 2022). [ Links ]
9. Murni IK, Duke T, Kinney S, Daley AJ, Wirawan MT, Soenarto Y. Risk factors for healthcare-associated infection among children in a low-and middle-income country. BMC Infect Dis 2022;22(1):1-9. https://doi.org/10.1186/S12879-022-07387-2 [ Links ]
10. Zingg W, Hopkins S, Gayet-Ageron A, et al. Health-care-associated infections in neonates, children, and adolescents: An analysis of paediatric data from the European Centre for Disease Prevention and Control point-prevalence survey. Lancet Infect Dis 2017;17(4):381-389. https://doi.org/10.1016/S1473-3099(16)30517-5 [ Links ]
11. Prusakov P, Goff DA, Wozniak PS, et al. A global point prevalence survey of antimicrobial use in neonatal intensive care units: The no-more-antibiotics and resistance (NO-MAS-R) study. E Clin Med 2021;32. https://doi.org/10.1016/j.eclinm.2021.100727 [ Links ]
12. Archary M, Adler H, La Russa P, Mahabeer P, Bobat RA. Bacterial infections in HIV-infected children admitted with severe acute malnutrition in Durban, South Africa. Paediatr Int Child Heal 2017;37(1):6-13. https://doi.org/10.1080/20469047.2016.1198561 [ Links ]
13. Viswanad V, Abraham S, Abraham A, Anupama P, Muralidharan A, Arya S. Confrontational use of antibiotics in pediatric prescriptions. Deccan J Pharm Cosmetol 2010;1(2):52-56. [ Links ]
14. Ashraf H, Handa S, Khan N. Prescribing pattern of drugs in outpatient department of child care centre in Moradabad city. Int J Pharm Sci Rev Res 2010;3(2):1-5. [ Links ]
15. Chetty T, Pillay A, Balakrishna Y et al. Healthcare-associated infections drive antimicrobial prescribing in pediatric departments at three academic hospitals in South Africa. Pediatr Infect Dis J 2023;42:e282- 289. https://doi.org/10.1097/inf.0000000000003954 [ Links ]
 Correspondence:
Correspondence:
J Cloete
jeane.cloete@up.ac.za
Accepted 25 January 2024














