Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
SAMJ: South African Medical Journal
versão On-line ISSN 2078-5135
versão impressa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.113 no.2 Pretoria Fev. 2023
http://dx.doi.org/10.7196/SAMJ.2022.v113i2.16758
IN PRACTICE
Transcatheter vascular plugs for the treatment of massive haemoptysis from Rasmussen aneurysms
J A ShawI; T J JohnII; C F N KoegelenbergIII; M da SilvaIV; B W AllwoodV; H WeichVI
IMB ChB, MPhil (Pulm); DSI-NRF Centre of Excellence for Biomedical Tuberculosis Research; South African Medical Research Council Centre for Tuberculosis Research; Biomedical Research Institute, Division of Molecular Biology and Human Genetics, and Division of Pulmonology, Department of Medicine, Stellenbosch University and Tygerberg Hospital, Cape Town, South Africa
IIMB ChB, MPhil (Cardio); Division of Cardiology, Department of Medicine, Stellenbosch University and Tygerberg Hospital, Cape Town, South Africa
IIIMB ChB, PhD; Division of Pulmonology, Department of Medicine, Stellenbosch University and Tygerberg Hospital, Cape Town, South Africa
IVMB BCh, FC Rad Diag (SA); Division of Radiodiagnosis, Department of Medical Imaging and Clinical Oncology, Stellenbosch University and Tygerberg Hospital, Cape Town, South Africa
VMB BCh, PhD; Division of Pulmonology, Department of Medicine, Stellenbosch University and Tygerberg Hospital, Cape Town, South Africa
VIMB ChB, Cert Cardiol (SA); Division of Cardiology, Department of Medicine, Stellenbosch University and Tygerberg Hospital, Cape Town, South Africa
ABSTRACT
Rasmussen aneurysms are abnormalities of the pulmonary arterial system caused by tuberculosis (TB). They are associated with a high mortality rate when they cause life-threatening haemoptysis. High TB-prevalence regions have a large burden of TB-related haemoptysis but often limited resources. This series of 25 patients who presented with life-threatening haemoptysis from current and/or previous TB were found to have abnormal pulmonary arteries on computed tomography pulmonary angiogram (CTPA), which were judged to be likely contributors to their bleeding, either in isolation or with concomitant abnormal bronchial or systemic vasculature. These patients underwent transcatheter placement of Amplatzer vascular plugs in the feeder pulmonary artery. Bronchial and systemic lesions were addressed separately as needed. Immediate technical success was achieved in all patients, but four of them experienced intraoperative haemoptysis related to dislodgement of the occluding platelet plug by the high-pressure automatic injector and wire. At 48 hours after the procedure, 18 (72%) remained haemoptysis-free. Six of these experienced recurrence within 1 year of their procedure. Pulmonary artery placement of an Amplatzer vascular plug is a feasible option for treating bleeding Rasmussen aneurysms, but should be part of a combined approach to addressing suspected culprit vascular lesions in all intrathoracic vascular systems.
Haemoptysis caused by bleeding from the pulmonary artery (PA) is a rare but potentially life-threatening complication of lung infection.[1] While virulent organisms such as Staphylococcus usually cause true aneurysms (focal dilatation of all three layers of the artery wall), tuberculosis (TB) is the most common cause of PA pseudoaneurysm (bleeding from the artery that is not contained by the artery wall), named Rasmussen aneurysms.[1] This term has evolved to become a catch-all for any PA bleeding caused by TB, as distinguishing between true aneurysm and pseudoaneurysm on imaging is often not possible, and extravasation from the PA may even be seen without aneurysmal changes.[2]
South Africa (SA) has an extremely high prevalence of pulmonary TB, in excess of 1 000/100 000 in some subpopulations, and consequently a high burden of TB-related haemoptysis.[3,4] Haemoptysis can occur during active TB infection, or as a sequel of previous TB infection, and may present as either trivial or life threatening. PA involvement is a risk factor for death in haemoptysis, and it is thought that between 5% and 10% of cases of life-threatening haemoptysis arise from the pulmonary arterial system.[5,6] It is, however, very difficult to locate the source of bleeding with certainty in a large number of cases, and the true incidence of PA involvement may be different.
The management of life-threatening haemoptysis from a Rasmussen aneurysm is complex, and generally requires a high level of institutional expertise. Emergency surgical resection of the affected portion of the lung is often necessary, but carries a high mortality and morbidity.[1] Moreover, in under-resourced, overburdened centres there may be a considerable delay between presentation and surgery, and patients with severe post-TB lung disease or other comorbidities may not be candidates for surgical intervention.[7] Several endovascular techniques have been reported for the treatment of PA aneurysms other than Rasmussen's, among these, the Amplatzer vascular plug (AVP; Abbott Laboratories, USA), shown in Fig. 1. Here we report the first series of 25 patients with massive haemoptysis due to Rasmussen aneurysms who were treated with emergency transcatheter placement of the AVP.
Case series
The patients in this report were treated at Tygerberg Hospital, a tertiary referral centre in Cape Town, SA, in which 9.8% of acute respiratory admissions are for haemoptysis.[4] Between November 2018 and June 2021, patients referred for the management of life-threatening haemoptysis (defined as haemoptysis causing haemodynamic instability, or a drop in measured haemoglobin, the need for blood transfusion, respiratory support for haemorrhage-related hypoxaemia, or witnessed expectoration of >500 mL in 24 hours) underwent chest computed tomography pulmonary angiogram (CTPA) once clinically stabilised. A CTPA was performed with a Somatom Definition Edge TM 128 MDCT (Siemens, Germany). The protocol for a patient presenting with haemoptysis at our institution consists of a single-phase, single-acquisition CT scan with intravenous administration of 80 mL OmnipaqueTM 350 at a rate of 4 mL/sec using an injector pump and bolus tracking. The presence of PA abnormalities such as true aneurysms, pseudoaneurysms and extravasation from a PA vessel, as well as abnormalities of the bronchial and systemic vasculature and underlying pulmonary pathology, were documented by a specialist radiologist. Those with only bronchial or systemic arterial abnormalities underwent bronchial artery embolisation (BAE) as recommended.[8] Patients with PA abnormalities judged to be a likely contributor to bleeding (Fig. 2) were offered transcatheter placement of an AVP. In cases with both bronchial and PA abnormalities, a combined approach was used with the vascular system judged most likely to be the culprit targeted first. In patients who consented to AVP placement, procedures were performed in a cardiac catheterisation laboratory under local anaesthesia with cardiac monitoring. Targeted pulmonary angiography was performed through femoral vein vascular access (Fig. 3). The most likely culprit was identified using both angiography and CTPA imaging. The feeder artery was selectively engaged with an appropriately shaped guide catheter and guidewire, usually a Terumo Radifocus Guide Wire M (Terumo Medical, Japan). In cases with difficult access to the feeder vessel, two catheters and a mother-daughter technique were used. Cautious angiography of the culprit vessel was performed, allowing operators to size the feeder vessel. A plug was chosen that was oversized by 20%. Two types of plugs (AVP 2 or 4) were chosen dependent on the length of the landing zone, diameter of catheter required and availability of plugs. The position of the plug was evaluated by contrast injection and if deemed adequate, the plug was released. A final angiogram was done to ensure that the feeder artery was covered. Patients were observed for 48 hours after the procedure. If haemoptysis recurred during this time, further interventions were decided by the multidisciplinary team on an individual patient basis.
During the study period, 25 patients (17 (68%) males, median age 40 (range 19 - 66) years) underwent the procedure. The characteristics of the patients and the procedures are shown in Table 1. Most notably, 18 (72%) had current active pulmonary TB, and 21 (84%) had previous TB, of whom 7 (28%) had >1 previous episode. Six patients had intracavitary mycetomas (aspergillomas). Six (24%) were HIV-positive (median CD4 count 102 cells/mL, range 5 - 530). PA abnormalities were identified in the right lung in 13 patients (upper lobe n=5; lower lobe n=8) and on the left in 14 patients (upper lobe n=11, lower lobe n=3). Three patients had multiple PA lesions. No patients had bilateral PA lesions. Seven (28%) patients also had associated abnormal bronchial and nonbronchial systemic collaterals.
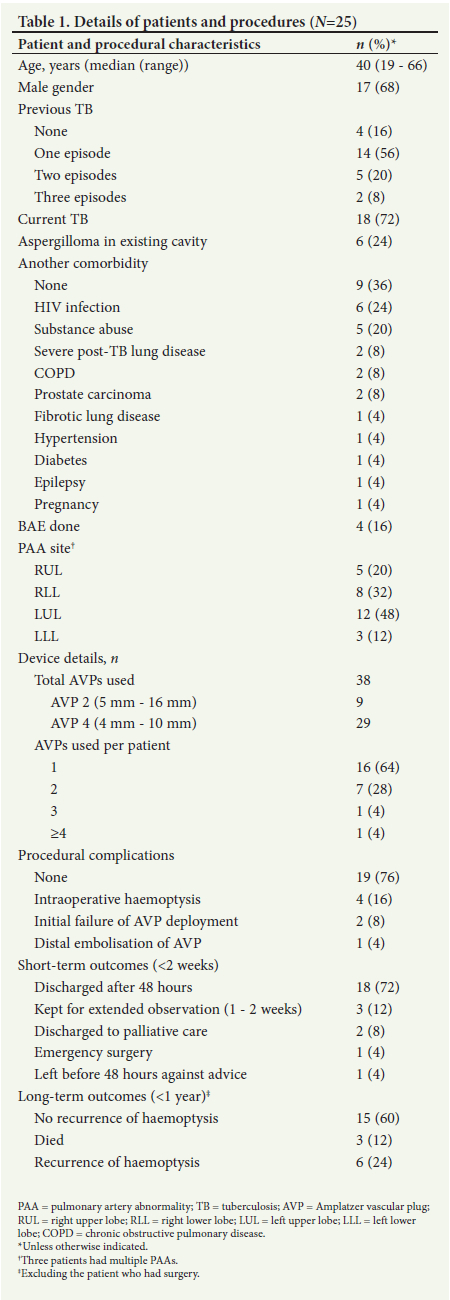
Four patients experienced significant intraoperative haemoptysis, likely from the high-pressure automatic injector and wire-related dislodgement of the occluding platelet plug. In two procedures there was an initial failure of deployment of the AVP, requiring resheathing, retraction and redeployment. In one procedure, the AVP embolised distally to lodge in a non-culprit vessel and a further AVP had to be deployed in the feeder vessel.
Immediate technical success was achieved in all patients. Most of the patients (18; 72%) remained free of haemoptysis for 48 hours after the procedure and were discharged home with a follow-up plan. One patient left hospital against medical advice before 48 hours but had no later recurrence. One patient, known to have an aspergilloma, needed an emergency left upper lobectomy during the same admission for persistent life-threatening haemoptysis. Three patients were kept for extended observation for persistent minor haemoptysis, and were discharged home after 1 - 2 weeks. Two patients were discharged to palliative care for severe inoperable post-TB lung disease (one had two attempted BAEs as well as the AVP procedure, the second also suffered from advanced HIV disease) and died within a month of the procedure. Six patients (excluding the two who died) had recurrent haemoptysis in the following 12 months: one defaulted initial TB treatment, one presented with presumed reinfection after treatment completion, one suffered rifampicin-induced thrombocytopenia, two had concomitant aspergillomas and one who was pregnant at the time of her procedure elected to delay definitive intervention. One patient presented with recurrent pneumonia (without haemoptysis) at 4 months and 6 months after the procedure and died from the second episode.
Discussion
Our series of 25 patients with massive haemoptysis from Rasmussen aneurysms treated with transcatheter placement of an AVP shows that this procedure is feasible, and potentially life-saving. The Amplatzer vascular plug is a braided nitinol mesh disc, attached to a delivery wire and micro-screw which, when deployed in a vessel, acts as an embolic agent by promoting clot formation as blood flows over and through the mesh.[10] This device has been successfully used in the pulmonary arteries in Behcet's disease, Hughes-Stovin syndrome, iatrogenic PA aneurysms, idiopathic aneurysms and pulmonary arteriovenous malformations (AVMs), among others.[11-13] However, to our knowledge this is the only series of Rasmussen aneurysms managed with the AVP to date.
Despite recent advances in both diagnosis and treatment, TB remains the most common cause of massive haemoptysis in the world[9]
TB-related bleeding from the pulmonary vasculature is often fatal, yet few institutions where this problem is common have the resources to offer urgent surgical intervention to all cases, many patients with the problem are unfit for surgery and it is not known if endovascular treatment is sufficient. In a recent review of the literature, Gupta et al.[l] identified 248 cases of pulmonary artery aneurysms/pseudoaneurysms, 32 of them caused by infection, only a third of which were due to TB. Management strategies in this review varied widely, including conservative or medical treatment, endovascular intervention with embolised coils or vascular plugs (not specifically AVP) and surgery. The most common endovascular treatment for infectious PA aneurysms/pseudoaneurysms in the literature is coil embolisation, the first descriptions of which were from Remy et al.[5,14] almost 40 years ago. In one of the recent larger series, Shin et al.[15] report on 24 cases of infectious peripheral PA pseudoaneurysms, of which 16 were Rasmussen's and 5 associated with aspergillomas. In this series, the feeder arteries were embolised with microcoils or N-butyl cyanoacrylate (glue), with varying success depending on the presence of systemic shunts. Most recently, Lal et al.[16] described direct percutaneous embolisation of 29 PA pseudoaneurysms with glue or reconstituted fibrin, of which 23 (85%) were Rasmussen aneurysms. The AVP offers several advantages over traditional coil or glue embolisation: the delivery mechanism can achieve exact placement and allows for repositioning; it has minimal risk of migration, even in high flow vessels and short landing zones; and it is possible to occlude a large vessel with a single device (as opposed to multiple coils).[10]
In our series the AVP was unsuccessful in stopping bleeding within 48 hours in six (24%) patients, and a further six (24%) of those with early success had recurrence of bleeding within a year. Ideally patients should have repeat CTPA after an interval to confirm continued vessel obstruction and healing of the surrounding destructive infectious process, which we were unable to do. However, the literature on the use of AVP for pulmonary AVMs reports rates of recanalisation of the vessel of only 1% -7%, suggesting that the recurrent bleeding in our series may have been from other vascular sites.[10] In fact, in the cases where the PA bleeding occurred during active infection, we suspect that the development of fibrous scar tissue around the Rasmussen aneurysm during the healing process could prevent recurrent bleeding even in the case of vessel recanalisation. Those patients who experienced haemoptysis from Rasmussens that were the result of a previous infection, or who have associated aspergillomas, may still need a more definitive intervention, and in these patients the PA plug is a good bridging technique. In those who are unfit for surgery, the PA intervention may be their only chance for survival.
Evidence is mounting that the traditional approach of first performing BAE is insufficient in a significant proportion of patients presenting with massive haemoptysis related to TB.[15] Although peripheral PA aneurysms/pseudoaneurysms may be simultaneously perfused by both pulmonary and systemic systems, particularly in patients with cavitary or destructive parenchymal infections, our series and others[15,17] suggest that occlusion of the PA feeder alone is successful in most cases where there are minimal bronchial and non-bronchial systemic collaterals identified on CT angiogram. We recommend that patients who have evidence of a PA and bronchial or systemic source of bleeding on their initial CT angiogram should have both lesions addressed in parallel.
Conclusion
In conclusion, we believe the AVP offers a feasible alternative treatment option for Rasmussen aneurysms.
Declaration. None.
Acknowledgements. None.
Author contributions. HW, JAS and TJJ conceived of and designed the study; all authors were responsible for data acquisition, analysis and interpretation; JAS drafted the manuscript, which was critically revised by all authors; all authors approved the final version.
Funding. JAS is supported by funding from the SA Medical Research Council (SAMRC) through its Division of Research Capacity Development under the Bongani Mayosi National Health Scholars Programme, from funding received from the Public Health Enhancement Fund/SA National Department of Health. The content hereof is the sole responsibility of the authors and does not necessarily represent the official views of the SAMRC.
Conflicts of interest. None.
References
1. Gupta M, Agrawal A, Iakovou A, et al. Pulmonary artery aneurysm: A review. Pulm Circ 2020;10(1):2045894020908780. https://doi.org/10.1177/2045894020908780 [ Links ]
2. Gothi R. Rasmussen aneurysm: A pseudoaneurysm of the pulmonary, not bronchial, artery. Radiology 2021;302(3):211647. https://doi.org/10.1148/radiol.211647 [ Links ]
3. Van der Walt M, Moyo S. The First National TB Prevalence Survey, South Africa 2018: Short report. Cape Town: South African Medical Research Council Human Sciences Research Council, 2021. https://www.knowledgehub.org.za/system/files/elibdownloads/2021-02/A4_SA_TPS%20Short%20Report_10June20_Final_highres.pdf (accessed 6 August 2021) [ Links ]
4. Ngah V, Maud P, Baines N, et al. Respiratory presentations to acute services at a tertiary hospital in South Africa. S Afr Med J 2021:111(11):1104-1109 https://doi.org/10.7196/SAMJ.2021.v111i11.15711 [ Links ]
5. Remy J, Lemaitre L, Lafitte JJ, et al. Massive hemoptysis of pulmonary arterial origin: Diagnosis and treatment. AJR Am J Roentgenol 1984;143(5):963-969. https://doi.org/10.2214/ajr.143.5.963 [ Links ]
6. Fartoukh M, Khoshnood B, Parrot A, et al. Early prediction of inhospital mortality of patients with hemoptysis: An approach to defining severe hemoptysis. Respiration 2012;83(2):106-114. https://doi.org/10.1159/000331501 [ Links ]
7. Masoud SR, Irusen EM, Koegelenberg CFN, et al. Outcomes of resectable pulmonary aspergilloma and the performance gap in a high tuberculosis prevalence setting: A retrospective study. Afr J Thorac Crit Care Med 2017;23(1):8-13. https://doi.org/10.7196/SARJ.2017.v23i1.154 [ Links ]
8. Davidson K, Shojaee S. Managing massive hemoptysis. Chest 2020;157(1):77-88. https://doi.org/10.1016/j.chest.2019.07.012 [ Links ]
9. Singh SK, Tiwari KK. Etiology of hemoptysis: A retrospective study from a tertiary care hospital from northern Madhya Pradesh, India. Indian J Tuberc 2016;63(1):44-47. https://doi.org/10.1016/j.ijtb.2016.02.007 [ Links ]
10. Lopera JE. The Amplatzer vascular plug: Review of evolution and current applications. Semin Intervent Radiol 2015;32(4):356-369. https://doi.org/10.1055/s-0035-1564810 [ Links ]
11. Ianniello A, Carrafiello G, Nicotera P, et al. Endovascular treatment of a ruptured pulmonary artery aneurysm in a patient with Behcet's disease using the Amplatzer Vascular Plug 4. Korean J Radiol 2013 Mar-Apr;14(2):283-286. https://doi.org/10.3348/kjr.2013.14.2.283 [ Links ]
12. Landi A, Nai Fovino L, Fabris T, et al. Pulmonary artery rupture during right heart catheterisation: Successful endovascular treatment with Amplatzer vascular plug. Eur Heart J 2018;39(44):3982. https://doi.org/10.1093/eurheartj/ehy400 [ Links ]
13. Tamura T, Yabuki T, Kawajiri T, et al. Transcatheter embolisation for idiopathic peripheral pulmonary arterial aneurysm: A case report. Clin Case Rep 2019;7(5):1021-1024. https://doi.org/10.1002/ccr3.2154 [ Links ]
14. Remy J, Smith M, Lemaitre L, et al. Treatment of massive hemoptysis by occlusion of a Rasmussen aneurysm. Am J Roentgenol 1980;135(3):605-606. https://doi.org/10.2214/ajr.135.3.605 [ Links ]
15. Shin S, Shin T, Choi H, et al. Peripheral pulmonary arterial pseudoaneurysms: Therapeutic implications of endovascular treatment and angiographic classifications. Radiology 2010;256(2). https://doi.org/10.1148/radiol.10091416 [ Links ]
16. Lal A, Bansal A, Chaluvashetty SB, et al. Percutaneous transthoracic embolisation for massive haemoptysis secondary to peripheral pulmonary artery pseudoaneurysms. Eur Radiol 2021;31(4):2183-2190. https://doi.org/10.1007/s00330-020-07348-w [ Links ]
17. Khalil A, Parrot A, Nedelcu C, et al. Severe hemoptysis of pulmonary arterial origin: Signs and role of multidetector row CT angiography. Chest 2008;133(1):212-219. https://doi.org/10.1378/chest.07-1159 [ Links ]
 Correspondence:
Correspondence:
J A Shaw
janeshaw@sun.ac.za
Accepted 5 September 2022














