Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
SAMJ: South African Medical Journal
versão On-line ISSN 2078-5135
versão impressa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.112 no.10 Pretoria Out. 2022
http://dx.doi.org/10.7196/SAMJ.2022.v112i10.16397
RESEARCH
Improving primary care antimicrobial stewardship by implementing a peer audit and feedback intervention in Cape Town community healthcare centres
E de VriesI; Y JohnsonII; B WillemsIII; W BedekerIV; T RasV; R CoetzeeVI; Y TemboVII; A BrinkVIII, IX
IMB ChB, MFamMed; School of Public Health and Family Medicine, Faculty of Health Sciences, University of Cape Town, South Africa
IIB Pharm, MPH; Pharmacy Services, Western Cape Government Health, Cape Town, South Africa
IIIMB ChB, MMed (Public Health); Nga Tai Ora - Public Health Northland, New Zealand
IVMB ChB, MMed (Fam Med); Family physician, Campbell River, Canada
VMB ChB, MMed (Fam Med); School of Public Health and Family Medicine, Faculty of Health Sciences, University of Cape Town, South Africa
VIB Pharm, Pharm D; School of Pharmacy, Faculty of Natural SciencesUniversity of the Western Cape, Cape Town, South Africa
VIIBSc, MB ChB; School of Public Health and Family Medicine, Faculty of Health Sciences, University of Cape Town, South Africa
VIIIMB ChB, MMed (Med Micro); Division of Medical Microbiology, Faculty of Health Sciences, University of Cape Town, and National Health Laboratory Services, Groote Schuur Hospital, Cape Town, South Africa
IXMB ChB, MMed (Med Micro); Institute of Infectious Disease and Molecular Medicine, Faculty of Health Sciences, University of Cape Town, South Africa
ABSTRACT
BACKGROUND: The increasing prevalence of antibiotic resistance is a major threat to public health. Primary care, where 80% of antibiotics are consumed, is a pivotal setting to direct antimicrobial stewardship (AMS) efforts. However, the ideal model to improve antibiotic prescribing in primary care in low-resource settings is not known
OBJECTIVE: To implement a multidisciplinary audit and feedback AMS intervention with the aim to improve appropriate antibiotic prescribing at primary care level
METHODS: The intervention was implemented and monitored in 10 primary care centres of the Cape Town metropole between July 2017 and June 2019. The primary and secondary outcome measures were monthly adherence to a bundle of antibiotic quality process measures and monthly antibiotic consumption, respectively. Multidisciplinary audit and feedback meetings were initiated and integrated into facility clinical meetings. Two Excel tools were utilised to automatically calculate facility audit scores and consumption. Once a month, 10 antibiotic prescriptions were randomly selected for a peer review audit by the team. The prescriptions were audited for adherence to a bundle of seven antibiotic process measures using the standard treatment guidelines (STG) and Essential Medicines List (EML) as standard. Concurrently, primary care pharmacists monitored monthly antibiotic consumption by calculating defined daily doses (DDDs) per 100 prescriptions dispensed. Adherence and consumption feedback were regularly provided to the facilities. Learning collaboratives involving representative multidisciplinary teams were held twice-yearly. Pre-, baseline and post-intervention periods were defined as 6 months before, first 6 months and last 6 months of the study, respectively
RESULTS: The mean overall adherence increased from 19% (baseline) to 47% (post intervention) (p<0.001). Of the 2 077 prescriptions analysed, 33.7% had an antibiotic prescribed inappropriately. No diagnosis had been captured in patient notes, and the antibiotic chosen was not according to the STG and EML in 30.1% and 31.7% of cases, respectively. Seasonal variation was observed in prescribing adherence, with significantly lower adherence in winter and spring months (adjusted odds ratio 0.60). A reduction of 12.9 DDDs between the pre- and post-intervention periods (p=0.0084) was documented, which represented a 19.3% decrease in antibiotic consumption
CONCLUSION: The study demonstrated that peer reviewed audit and feedback is an effective AMS intervention to improve antibiotic prescribing in primary care in a low-resource setting. The intervention, utilising existing resources and involving multidisciplinary engagement, may be incorporated into existing quality improvement processes at facility level, to ensure sustainable change
In response to increases in antibiotic consumption[1] and to the rising prevalence of multidrug and extensive drug resistance among common bacterial infections in South Africa (SA), a One Health-based national strategic framework for antimicrobial resistance,[2] an implementation plan[3] and a mechanism for the governance of antimicrobial stewardship (AMS) at national, provincial, district and hospital levels[4] was developed by the National Department of Health. While the importance of improving outpatient antibiotic use is acknowledged, no comprehensive recommendations to curb the use in the primary healthcare setting have been made, owing to the lack of sufficient data to guide such efforts.[5] A targeted application of AMS principles to the ambulatory setting has the potential to affect the most common indications for systemic antibiotic use, in that the majority (80%) of antibiotic use occurs in primary care, with acute respiratory-tract infections being the most common indication.[5,6]
AMS is defined as the systematic approach to optimising the appropriate use of antimicrobials to improve patient outcomes and limit emergence of resistant pathogens while ensuring patient safety.[4] However, the reasons driving the excessive prescription of antibiotics in the community are complex, and include constraints on consultation time, lack of appreciation of the impact on resistance, considerable
diagnostic uncertainty and perhaps equally importantly, perceived patient and parental/guardian pressures.[5,7,8] Therefore, multifaceted, multicomponent interventions with effect sizes of sufficient magnitude to potentially reduce the incidence of antibiotic-resistant bacteria are generally recommended for primary care.[5] Whereas educational interventions may be implemented on many levels after addressing local barriers to change, expecting adherence to antibiotic guidelines without concurrent interventions is of little value.
In fact, a recent survey of antibiotic prescribing practices in the Cape Town metropole confirmed 55% non-adherence to the standard treatment guidelines (STG) and Essential Medicines List (EML) for SA.[9] Undocumented antibiotic indication (30.5%) and inappropriate antibiotic dose (12.9%), choice (11.5%) and duration (9.5%) were the main reasons for non-adherence.[9] In addition, the problem in SA is confounded by lack of undergraduate AMS training, as final-year medical undergraduates have been found to be inadequately prepared for practice.[10] Crucial gaps in knowledge and poor understanding of AMS principles were recently demonstrated in a multisite survey in SA.[11]
Many different interventions have been implemented to improve antibiotic prescribing in primary care settings.[12] A Cochrane review concluded that C-reactive protein testing, shared decision-making between prescriber and client and procalcitonin-guided management reduce antibiotic prescribing for acute respiratory infections in general practice.[12] In one study, personalised prescription feedback did not have a significant effect in changing the antibiotic consumption between the intervention and control group,[13] but in primary care practices, accountable justification and peer comparison as behavioural interventions resulted in lower rates of inappropriate prescribing.[14]
A systematic review concluded that quality improvement (QI) methods are effective at reducing antibiotic use in ambulatory settings, but suggested further improving outcomes by active clinician education and targeted management of all acute infections rather than focusing on single conditions or single age groups of clients.[15] In the UK, stakeholder consultation, APEASE (i.e. affordability, practicability, effectiveness, acceptability, safety and equity) criteria were used to identify the highest-rated interventions to improve antibiotic prescribing. The top four were quality improvement, multidisciplinary peer learning, appointing AMS leads and auditing individual level prescribing.[16]
In the SA public sector, there is no standardised way of integrating AMS governance processes into primary care facility operations. At hospital level in the Western Cape Province, AMS committee activities have included antimicrobial prescription chart reviews and audits, AMS ward rounds, antimicrobial restriction policies and training.[17,18]
The ideal model to improve antibiotic prescribing in primary care in low-resource settings is not known. Utilising existing staff, we therefore designed and implemented a multidisciplinary AMS intervention involving an audit and feedback strategy, with the objective of improving appropriate prescribing of antibiotics at primary care level as defined in the STG and EML.'191 A peer review approach was chosen rather than a formal audit by outsiders, to increase ownership of the process by facilities and embed it in local quality improvement processes. It is hypothesised that such an intervention will result in improved prescribing patterns and adherence to evidence-based guidelines.
Methods
The multicentre AMS intervention was implemented in community health centres (CHCs) and community day centres (CDCs) of the Cape Town metropole between July 2017 and June 2019.
Ethical approval
Approval to conduct the study was granted by the Human Research Ethics Committee at the University of Cape Town, (ref. no. 290/2016), as well as Western Cape Government Health. For the purpose of the folder review, informed consent from patients was not required by the approval committees, as folder review was retrospective, data were anonymised before analysis and there was no direct patient contact.
Participants
Thirteen primary care facilities in the Cape Town metropole were initially invited to participate in the quality improvement implementation study. Ten facilities provided adherence and consumption data, and were included in the 24-month study period (July 2017 - June 2019). Characteristics of the facilities are shown in Table 1.
Interventions
Multidisciplinary audit and feedback meetings (consisting of pharmacists, doctors and nurse prescribers) were initiated and integrated into the facility clinical meetings. Once a month, at each of these facilities, 10 antibiotic prescriptions were randomly selected for a peer review audit by the team. These prescriptions were then assessed and scored for adherence to a bundle of seven measures, including antibiotic choice, dose, duration and frequency with the STG and EML[19] as standard, as shown in Table 2. All measures had to be met for the prescription to be considered correct, i.e. the all-or-nothing concept.

Concurrently, primary care pharmacists monitored monthly antibiotic consumption for the six oral antibiotics prescribed most frequently, i.e. amoxicillin, amoxicillin/clavulanic acid, azithromycin, ciprofloxacin, flucloxacillin and Penicillin VK.[9] Intravenous antibiotics were excluded. Monthly consumption data and the number of prescriptions dispensed were obtained from the electronic pharmacy information system. Consumption was calculated as defined daily doses (DDDs) per 100 prescriptions dispensed.[20]
Two tools (in Excel; Microsoft, USA), one for audit and one for measuring consumption, were developed. The audit tool was piloted by a family physician at a CDC during February - April 2017, prior to implementation in the rest of the participating CHCs.
Monthly, each facility provided feedback on the monthly folder review audit and consumption data to two project managers (one for adherence and one for consumption), using the two Excel tools that automatically calculated facility audit scores and DDDs per 100 prescriptions, respectively. The project managers collated the data from the various facilities 6-monthly, and provided comparative feedback to the pharmacists, who in turn provided verbal feedback in monthly clinical meetings.
Learning collaborations involving pharmacists and prescribers from participating facilities were held twice yearly, with the following aims: to facilitate collaborative learning among CHCs; to clarify requirements of AMS implementation; to brainstorm ideas to overcome obstacles to implementation; to share successes; to evaluate accuracy and consistency of data; and to provide comparative feedback on progress and improvements or otherwise. The feedback from the participating teams regarding successful components to achieve improvements in adherence to the antibiotic audit measures was collated.
Study outcomes
To enable measurement of process measures and sustainable improvement or otherwise, as well as consumption, pre-, baseline and post-intervention periods were defined as 6 months before the intervention, during the first 6 months of the intervention and the last 6 months of the intervention, respectively.
Primary
The primary study outcome was monthly adherence to the bundle of antibiotic prescription quality process measures (Table 2). The percentage of correct prescriptions (out of 10) was reported as the performance for the month per facility, and compared over the study period. Facility percentages were combined to form a 'system' outcome measure and reported on an annotated run chart.
Secondary
The secondary outcome was monthly antibiotic consumption calculated in DDDs per 100 prescriptions (100 Rx) per month, as a pragmatic metric to assess trends in consumption over the study period.
Statistical analysis
Regression was carried out using R version 3.6.2 (The R Foundation, USA), to compare the adherence to the bundle of antibiotic prescription quality processes, comparing the first 6 months of the intervention to subsequent time periods in 6-month intervals for the duration of the study. Regression analysis was also carried out to explore the factors affecting the adherence to the bundle of antibiotic prescription quality process measures per facility. Unadjusted and adjusted analysis is presented for prescriber type, age of patient, season, physiological system and antimicrobial prescribed.
Stata/SE 14.0 (Stata Corp., USA) was used to generate paired f-tests and two-way scatter graphs to compare antibiotic consumption 6 months pre-intervention, with the last 6 months (post-intervention period) at facility level, over the same seasonal periods.
Results
Adherence to antibiotic process audit bundle
The response rate of facilities submitting monthly audits was 208/240 (86.6%), with 20% of facilities managing 100%. After 24 months of conducting the audit and feedback intervention with constant small adaptations, the mean overall level of adherence increased from 11% in July 2017 to 53% in June 2019 (Fig. 1), with a mean of 19% correct prescriptions in the first 6 months to a mean of 47% correct prescriptions in the last 6 months (p<0.001). A positive linear trend over time was documented, and the adherence improved by 2% each month, on average. When compared with the first 6 months, the odds of adherence to process measures for the last 6 months of the study period were 3.72 (95% confidence interval (CI) 2.85 - 4.88; p<0.001 (appendix https://www.samedical.org/file/1883).

Of the 2 077 prescriptions analysed, 33.7% (n=700) had an antibiotic that was prescribed incorrectly. Of these incorrect prescriptions, no diagnosis had been captured in patient notes in 30.1% (211/700), the antibiotic prescribed was different from what was recommended in the EML guidelines[19] in 31.7% (222/700), 6.1% (43/700) of prescriptions were incorrect as a result of differences between the EML[19] and Integrated Management of Childhood Illness 2014 guidelines[21] and only 0.3% (2/700) were incorrect owing to stockout of the EML-recommended drugs.
Univariable and multivariable logistic regression for adherence to the audit measures are shown in Table 3. Adherence to the audit measures differed between the facilities, and seasonal variation was observed in prescribing adherence, with significantly lower adherence in winter and spring months (adjusted odds ratio (aOR) 0.60; p<0.01) compared with summer and autumn months. Adherence to audit measures also differed by physiological system: prescriptions for sexually transmitted infections (aOR 1.83; p=0.05 ) and urology (aOR 1.49; p=0.20) were more likely to be adherent to audit measures. Antibiotic prescriptions for gastrointestinal tract infections (aOR 0.65; p=0.32) were less likely to adhere, although this was not found to be statistically significant. Comparing the top six antimicrobials prescribed, prescriptions involving flucloxacillin (aOR 3.10; p<0.01) and penicillin (aOR 2.62; p<0.01) were more likely to adhere to audit measures than prescriptions for other antimicrobials.
Consumption
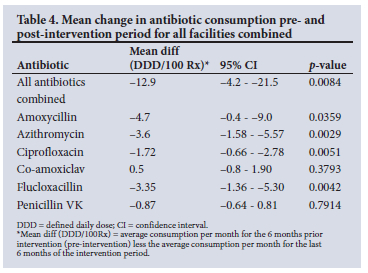
Facility response rate for submission of monthly consumption data was 100%. The mean differences in antibiotic consumption between the pre- and post-intervention periods are shown in Table 4. A significant reduction of 12.9 DDD/100 Rx between the pre - and post-intervention period was documented, representing a 19.3% (95% CI: 6.3% -32.3%) decrease in consumption. Except for amoxicillin/clavulanic acid and Penicillin VK, all other antibiotics showed a significant decrease in consumption. The greatest decreases in consumption were observed for amoxicillin, azithromycin and flucloxacillin. Overall, antibiotic consumption (DDDs/100 Rx) was higher during the winter months (June and July) than summer months (December and January). The overall antibiotic consumption per facility is depicted in appendix 2 (https://www.samedical.org/file/1884). A decrease in DDD/100 Rx was observed in the majority of facilities (80%). Only one facility showed an increase in overall consumption (13.19), with increases recorded for all antibiotics except ciprofloxacin.
The feedback from the participating teams during collaborative learning sessions regarding successful components to achieve improvements in adherence to the antibiotic audit measures are summarised in Table 5.
Discussion
Adherence to guidelines
In SA, AMS interventions at primary care have not been extensively reported, with more focus on hospital-based interventions at a tertiary hospitals,[22] private hospitals[23] and district hospitals.[18,24] Our study demonstrated that audit and feedback are an effective AMS intervention for primary care in a low-resource setting. While it is difficult for low- and middle-income countries to replicate resource-abundant stewardship models, utilisation of existing resources such as pharmacists and nurses is critical in implementing AMS.[25]
This pilot study illustrated that a quality improvement approach that involved peer review of prescribing practices, feedback and learning collaboratives resulted in a fivefold improvement in guideline adherence. The results revealed sustainability, substantiated by continuous improvement over the 2-year period. Spikes of improvements after the learning collaborative meetings were observed, reflecting the importance of collaborative feedback.
Furthermore, there was no significant difference in prescribing adherence between nurses and doctors, or for prescribing for adults compared with children. However, prescribing adherence among facilities differed significantly, indicating that specific facilities could be targeted and provided with additional AMS support and guidance. Challenges in implementing AMS involved high staff turnover, resource constraints and competing priorities. Strong AMS leadership, understanding the value of AMS, regular feedback and support from mentors promoted implementation at facilities. Linking AMS to patient safety, i.e. recording allergies as a pivotal process measure, was another enabler for change.
Similar to the Gasson et al.[9] study, differences were found comparing physiological systems, with prescribing most adherent in sexually transmitted infections, dermatology and ear, nose and throat infections. Poor adherence was seen in respect of the respiratory tract infections (RTIs) and gastrointestinal infections. For urinary tract infections (UTIs), conflicting results were found between the Gasson et at.[9] study, which was the site of infection where prescribing was least adherent to the guidelines, and our study.
One plausible explanation may relate to a greater focus on UTI treatment, as a guideline change due to a medicine safety warning for fluoroquinolones occurred during the study period. Notably, this suggests that guidelines for specific infections could be targeted for improvement.
Prescribing adherence was significantly lower in winter and spring, concurrent with higher antibiotic consumption, which may reflect inappropriate antibiotics for increased viral RTI months. Seasonal variation in antibiotic prescribing is a global phenomenon.[26,27] In our study, increased inappropriate prescribing, especially of amoxicillin, contributed to the increased consumption.
Consumption
Overall, there was a significant 19.3% decrease in antibiotic consumption over the intervention period. Notably, the four facilities with the highest adherence also had the greatest decreases in antibiotic consumption. Consumption variation between facilities may be related to different factors, including patient populations. A previous survey of non-communicable disease profiles at most of the primary healthcare facilities recruited in our study documented major variations in the patient demographics and proportions of acute and chronic conditions.[28]
Role of learning collaboratives
During learning collaboratives, facilities identified key components of a successful implementation strategy that included formalising an AMS team that comprised of at least health facility pharmacists and family physicians to ensure that monthly audits take place. The folder peer review audit took 1 hour per month to perform, and facility staff reported that it was a useful process, and that prescribers learned by doing the audit and reviews themselves. In addition, comparing the antibiotic process measures against the EML and STG was found to be a good way to learn. A common challenge in implementing AMS processes in our study was the high turnover of prescribers, especially interns and community service doctors: therefore strategies that promote consistency, oversight or mentorship across facilities may be of value. In this regard, AMS activities should be integrated into the induction or orientation of all new prescribers who join a facility.
Other multimodal components for successful implementation that were identified during the collaborative learning meetings to improve adherence included staff surveys to identify challenges and sharing resolutions, a '7-yes' criteria sticker on prescriptions to indicate adherence to all measures and immediate feedback to the prescriber for correction. Furthermore, posters on awareness of antimicrobial resistance, as well as medicine use evaluations to identify problem areas for specific antibiotics, etc., were other enablers for sustained improvement over the study period.
Limitations
The following limitations apply to our study. It was not compared with other primary care facilities in the Cape Town metropolitan area, where antibiotic audit and feedback are not standard of practice. Furthermore, we did not include outcome measures such as adverse events or repeat visits for the same infection (bacteriological or clinical failure), and neither did we follow up patients to calculate infection-related hospital admission rates. While appropriate prescribing should improve patient outcomes, measuring patient-level outcomes of any AMS intervention is pivotal in future primacy care interventions.
In addition, we did not have the resources to provide individual monthly antibiotic adherence rates to every prescriber. Nevertheless, we believe the peer and institutional feedback led to continuous improvement in adherence over the study period. A systematic review[28] that included 140 randomised controlled trials showed that audit and feedback have small to moderate but important effects on health professionals' behaviour. Moreover, in this Cochrane review, the evidence informing its effectiveness for changing antibiotic prescribing behaviour within the primary care setting was sparse, with only four of the trials studying the effect of feedback on prescribing within primary care. Therefore the reasons for the decrease in consumption in our study are not entirely clear, and future studies in this setting should incorporate qualitative research approaches, including comparison of the effectiveness of different ways of providing feedback. Finally, the effect size of randomly selecting more folders monthly on the impact of this intervention is unknown.
Conclusion
The strength of this study is that it demonstrated that peer audit and feedback may be an effective AMS intervention in primary care in a low-resource setting. Prescriber adherence to guidelines improved during the study period, and antibiotic consumption decreased. It represents a feasible intervention that can seamlessly be incorporated into existing quality improvement processes at facility level, utilising existing staff. Improvement methodology supports this type of intervention, as it facilitates regular measurements, and peer-reviewed performance assessments with ongoing feedback. Support of the facility management and a multidisciplinary team approach is critical, with the pharmacist playing a key co-ordinating and interdisciplinary role.[29] A qualitative approach and analysis of barriers and enablers for further improvement are required prior to provincial-wide implementation.
Declaration. None.
Acknowledgements. The authors would like to acknowledge the family physicians, pharmacists and their teams at the 10 facilities who ensured that the monthly audit and feedback took place, as well as Renfred Joshua who assisted with the development of the electronic AMS review and DDD tools.
Author contributions. EDV, YJ, WB, TR, BW, RC and AB conceptualised the research project. EDV, YJ, BW, TR, RC and AB participated in the learning collaboratives. BW, YJ and YT did the initial analysis. All authors contributed to writing and editing the manuscript and approved the final manuscript.
Funding. None.
Conflicts of interest. None.
References
1. Van Boeckel TP, Gandra S, Ashok A, et al. Global antibiotic consumption 2000 to 2010: An analysis of national pharmaceutical sales data. Lancet Infect Dis 2014;14(8):742-750. https://doi.org/10.1016/s1473-3099(14)70780-7 [ Links ]
2. National Department of Health, South Africa. South African Antimicrobial Resistance National Strategy Framework 2017 - 2024. Pretoria: NDoH, 2017. https://www.knowledgehub.org.za/elibrary/south-african-antimicrobial-resistance-national-strategy-framework-one-health-approach (accessed 28 September 2021). [ Links ]
3. National Department of Health, South Africa. Implementation Plan for the Antimicrobial Resistance Strategy Framework in South Africa: 2014 - 2019. Pretoria: NDoH, 2015. https://www.knowledgehub.org.za/elibrary/implementation-plan-antimicrobial-resistance-strategy-framework-south-africa-2014-2019 (accessed 28 September 2021). [ Links ]
4. National Department of Health, South Africa. Guidelines on Implementation of the Antimicrobial Strategy in South Africa: One Health Approach and Governance. Pretoria: NDoH, 2017. https://www.knowledgehub.org.za/elibrary/guidelines-implementation-antimicrobial-strategy-south-africa-one-health-approach (accessed 28 September 2021). [ Links ]
5. Brink AJ. Antimicrobial stewardship (AMS) in the community. Clin Pulm Med 2016;23(1):1-10. https://doi.org/10.1097/CPM.0000000000000107 [ Links ]
6. Brink AJ, van Wyk J, Moodley VM, et al. The role of appropriate diagnostic testing in acute respiratory tract infections: An antibiotic stewardship strategy to minimise diagnostic uncertainty in primary care. S Afr Med J 2016;106(6):554-561. https://doi.org/10.7196/samj.2016.v106i6.10857 [ Links ]
7. Farley E, van den Bergh D, Coetzee R, Stewart A, Boyles T. Knowledge, attitudes and perceptions of antibiotic use and resistance among patients in South Africa: A cross-sectional study. S Afr J Infect Dis 2019;34(1):1-9. https://doi.org/10.4102/sajid.v34i1.118 [ Links ]
8. Farley E, Stewart A, Davies MA, Govind M, van den Bergh D, Boyles TH. Antibiotic use and resistance: Knowledge, attitudes and perceptions among primary care prescribers in South Africa. S Afr Med J 2018;108(9):763-771. https://doi.org/10.7196/samj.2018.v108i9.12933 [ Links ]
9. Gasson J, Blockman M, Willems B. Antibiotic prescribing practice and adherence to guidelines in primary care in the Cape Town Metro district, South Africa. S Afr Med J 2018;108(4):304-310. https://doi.org/10.7196/samj.2017.v108i4.12564 [ Links ]
10. Brink A, Schoeman J, Muntingh G. Undergraduate antibiotic stewardship training: Are we leaving our future prescribers 'flapping in the wind'? S Afr Med J 2017;107(5):357-358. https://doi.org/10.7196/samj.2017.v107i5.12496 [ Links ]
11. Wasserman S, Potgieter S, Shoul E, et al South African medical students' perceptions and knowledge about antibiotic resistance and appropriate prescribing: Are we providing adequate training to future prescribers? S Afr Med J 2017;107(5):405-410. https://doi.org/10.7196/samj.2017.v107i5.12370 [ Links ]
12. Tonkin-Crine S, Tan P, van Hecke O, et al. Clinician-targeted interventions to influence antibiotic prescribing behaviour for acute respiratory infections in primary care: An overview of systematic reviews. Cochr Datab Syst Rev 2017;(9)CD012252. https://doi.org/10.1002/14651858.CD012252.pub2 [ Links ]
13. Hemkens LG, Saccilotto R, Reyes SL, et al. Personalised prescription feedback using routinely collected data to reduce antibiotic use in primary care: A randomised clinical trial. JAMA Intern Med 2017;177(2):176-183. https://doi.org/10.1001/jamainternmed.2016.8040 [ Links ]
14. Meeker D, Linder JA, Fox CR, et al. Effect of behavioral interventions on inappropriate antibiotic prescribing among primary care practices: A randomised clinical trial. JAMA 2016;315(6):562-570. https://doi.org/10.1001/jama.2016.0275 [ Links ]
15. Ranji SR, Steinman MA, Shojania KG, Gonzales R Interventions to reduce unnecessary antibiotic prescribing: A systematic review and quantitative analysis. Med Care 2008;46(8):847-862. https://doi.org/10.1097/mlr.0b013e318178eabd [ Links ]
16. Borek AJ, Wanat M, Sallis A, et al How can national antimicrobial stewardship interventions in primary care be improved? A stakeholder consultation. Antibiotics 2019;8(4):207. https://doi.org/10.3390%2Fantibiotics8040207 [ Links ]
17. Peters SM, Sheik S, Werner JL, Davies MA, Willems B. Antimicrobial stewardship in the Western Cape: A situational analysis of existing facility-level initiatives. S Afr Med J 2021;111(5):421-425. https://doi.org/10.7196/samj.2021.v111i5.14645 [ Links ]
18. Von Pressentin KB, Swanepoel H, Opie JJS, Jenkins LS. Antimicrobial stewardship in rural districts of South Africa: Growing a positive culture. S Afr Fam Pract 2019;61(6):276-281. https://doi.org/10.1080/20786190.2019.1680009 [ Links ]
19. National Department of Health, South Africa. Standard Treatment Guidelines and Essential Medicines List for South Africa: Primary Healthcare Level 2020 Edition. Pretoria: NDoH, 2020. https://www.idealhealthfacility.org.za/ (accessed 18 October 2021). [ Links ]
20. World Health Organization (WHO). Defined daily dose (DDD). https://www.who.int/toolkits/atc-ddd-toolkit/about-ddd (accessed 28 September 2021). [ Links ]
21. National Department of Health, South Africa. Integrated Management of Childhood Illness. Pretoria: NDoH, 2014. https://www.knowledgehub.org.za/system/files/elibdownloads/2019-07/IMCICB-2014.pdf (accessed 12 December 2021). [ Links ]
22. Boyles TH, Naicker V, Rawoot N, Raubenheimer PJ, Eick B, Mendelson M. Sustained reduction in antibiotic consumption in a South African public sector hospital: Four-year outcomes from the Groote Schuur Hospital antibiotic stewardship programme. S Afr Med J 2017;107(2):115-118. https://doi.org/10.7196/samj.2017.v107i2.12067 [ Links ]
23. Brink AJ, Messina AP, Feldman C, et al Antimicrobial stewardship across 47 South African hospitals: An implementation study. Lancet Infect Dis 2016;16(9):1017-1025. https://doi.org/10.1016/s1473-3099(16)30012-3 [ Links ]
24. Peters SM, Sheik S, Werner JL, Davies MA, Willems B. Antimicrobial stewardship in the Western Cape: A situational analysis of existing facility-level initiatives. S Afr Med J 2021;111(5):421-425. https://doi.org/10.7196/samj.2021.v111i5.14645 [ Links ]
25. Pierce J, Apisarnthanarak A, Schellack N, et al. Global antimicrobial stewardship with a focus on low- and middle-income countries. Int J Infect Dis 2020;96:621-629. https://doi.org/10.1016/j.ijid.2020.05.126 [ Links ]
26. Durkin MJ, Jafarzadeh SR, Hsueh K, et al Outpatient antibiotic prescription trends in the United States: A national cohort study. Infect Control Hosp Epidemiol 2018;39(5):584-589. https://doi.org/10.1017%2Fice.2018.26 [ Links ]
27. Safaeian L, Mahdanian AR, Salami S, Pakmehr F, Mansourian M. Seasonality and physician-related factors associated with antibiotic prescribing: A cross-sectional study in Isfahan, Iran. Int J Prevent Med 2015;6(1):1. https://doi.org/10.4103/2008-7802.151431 [ Links ]
28. Isaacs AA, Manga N, le Grange C, Hellenberg DA, Titus V, Sayed R. A snapshot of noncommunicable disease profiles and their prescription costs at ten primary healthcare facilities in the in the western half of the Cape Town Metropole. S Afr Fam Pract 2014;56(1):43-49. https://doi.org/10.1080/20786204.2014.10844582 [ Links ]
29. Ivers N, Jamtfedt G, Flottorp S, et al Audit and feedback: Effects on professional practice and healthcare outcomes. Cochrane Database Syst Rev 2012;(6):CD000259. https://doi.org/10.1002/14651858.CD000259.pub3/full [ Links ]
30. Brink AJ, van den Bergh D, Mendelson M, Richards GA. Passing the baton to pharmacists and nurses: New models of antibiotic stewardship for South Africa? S Afr Med J 2016;106(10):947-948. https://doi.org/10.7196/samj.2016.v106i10.11448 [ Links ]
 Correspondence:
Correspondence:
E de Vries
elma.devries@uct.ac.za
Accepted 21 June 2022














