Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
SAMJ: South African Medical Journal
versão On-line ISSN 2078-5135
versão impressa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.111 no.9 Pretoria Set. 2021
http://dx.doi.org/10.7196/SAMJ.2021.v111i9.15819
CORRESPONDENCE
Re-evaluating blood culture incubation times during SARS-CoV-2: Can we shorten incubation times without compromising results?
To the Editor: The first COVID-19 case in South Africa was reported on 5 March 2020, with a lockdown period implemented within the same month. The second wave of COVID-19 started on 12 November in Western Cape Province. Currently the country is in the third wave. The increase in febrile patients during the spread of SARS-CoV-2 resulted in an increase in blood cultures (BCs) submitted to our diagnostic laboratory (National Health Laboratory Service, Groote Schuur Hospital) for processing. The value of a BC to direct therapy and identify a disease-causing pathogen is widely appreciated. However, little is known of its usefulness in COVID-19 confirmed cases, and the evidence suggests a low incidence of co-infections with bacteraemia.[1-3] These surges in BC requests during COVID-19 peaks have the potential to overwhelm resource-limited laboratories that have limited space in their automated BC incubation instruments. Delaying the time to incubation of newly received bottles may delay the identification of pathogens as well as decrease the diagnostic yield. These factors can impact negatively on clinician antimicrobial treatment decisions, and from an antimicrobial stewardship stance may lead to missed opportunities to de-escalate or stop antimicrobials.[4] To mitigate this problem in future, it is worth exploring whether incubation times can be shortened, thereby freeing up incubator space more quickly.
The objective of this study was to determine the proportion of positive BCs that were processed over a 6-month period prior to and during the first wave of the COVID-19 pandemic; the incubation time to positivity; and the organisms identified from those BCs that only flagged positive on day 5 of incubation. We retrospectively analysed BCs submitted from various healthcare institutions to the National Health Laboratory Service at Groote Schuur Hospital from 1 January 2020 to 30 June 2020. Ethical and institutional approval for the study was obtained (ref. no. HREC 714/2020).
Of 22 156 BCs collected over 6 months, 3 228 (14.6%) had a micro-organism cultured. Of these positive cultures, 1 109 (34.4%) were probable contaminants (coagulase-negative staphylococci, Cutibacterium spp., Micrococcus spp., Bacillus spp., Corynebacterium spp., Aerococcus spp. and Viridans streptococci). Most of the pathogenic bacteria cultured belonged to the Enterobacterales order (Fig. 1A). The mean number of BCs sent to the laboratory during May and June increased by 14% compared with the previous 2 months and exceeded our automated instrument testing capacity. Most BCs (91.8%) signalled positive within the first 48 hours of incubation between 1 January 2020 and 30 June 2020 (Fig. 1B). Only 40 (1.2%) of the positive BCs submitted cultured a microorganism on day 5 (Fig. 1C), mainly with bacteria deemed contaminants (70.0%). The low yield of positive BCs during day 5 may justify a shorter period of incubation during peak times.[5] The exceptions to a shortened incubation duration are a suspicion of or proven fungaemia; specific clinical scenarios, e.g. infective endocarditis, that are known to be caused by fastidious organisms; and known Staphylococcus aureus bacteraemia, because missing this can have serious health consequences.[5]
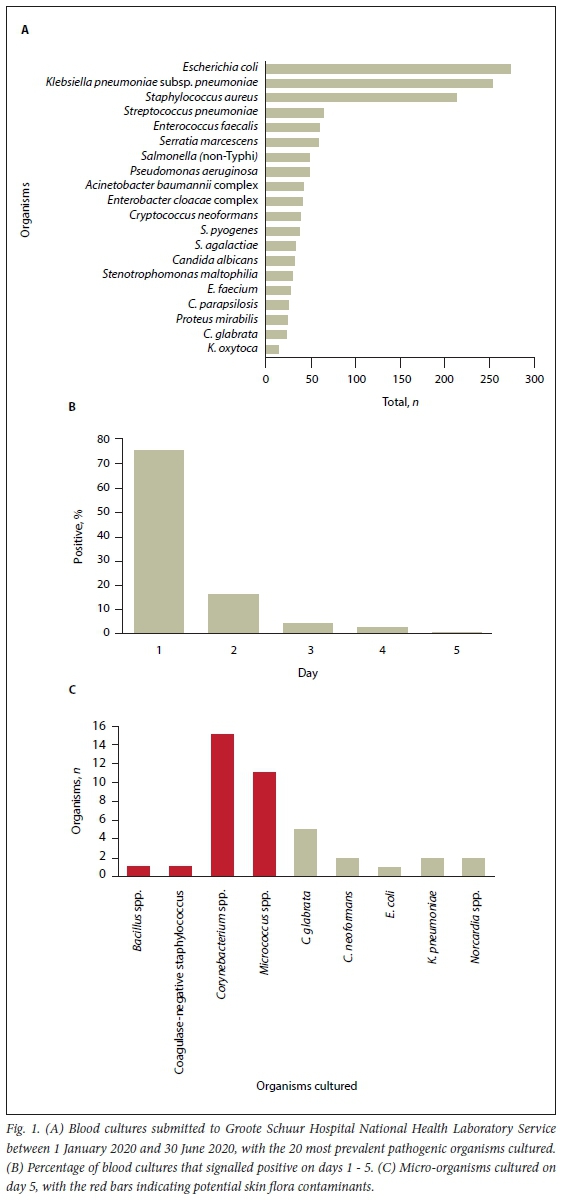
It has been recommended that empirical antibiotics are withheld in the absence of compelling evidence ofbacterial co-infection in a patient with COVID-19.[6] Reducing BC requests coupled with decreasing incubation times of BCs may preserve operational capacities during subsequent COVID-19 peaks. The rationale for increased BCs during the COVID-19 pandemic was not determined, but it may be that clinicians overestimated the frequency of co-infection or lacked the confidence to exclude co-infection on clinical presentation alone. In conclusion, the yield of positive BCs on day 5 remained low, with only 1.2% of cultures flagging positive; 70.0% (n=28/40) of these are likely to have been contaminants. It is therefore reasonable to shorten standard incubation periods when testing demand increases. Clinicians should provide clinical information on the laboratory request forms so that it can be determined whether a patient falls into an 'exception category' that justifies a standard incubation duration over a shortened one. BCs with no growth after day 3 are unlikely to become positive, and clinicians should consider this when consulted on patient management.
C J Opperman, N Samodien
Division of Medical Microbiology, National Health Laboratory Service, University of Cape Town and Groote Schuur Hospital, Cape Town, South Africa stefanoppermanl@gmail.com
References
1. Engsbro AL, Israelsen SB, Pedersen M, et al. Predominance of hospital-acquired bloodstream infection in patients with Covid-19 pneumonia. J Infect 2020;52(12):919-922. https://doi.org/10.1080/23744235.2020.1802062 [ Links ]
2. Langford BJ, So M, Raybardhan S, et al. Bacterial co-infection and secondary infection in patients with COVID-19: A living rapid review and meta-analysis. Clin Microbiol Infect 2020; 26(12):1622-1629. https://doi.org/10.1016/j.cmi.2020.07.016 [ Links ]
3. Lansbury L, Lim B, Baskaran V, et al. Co-infections in people with COVID-19: A systematic review and meta-analysis. J Infect 2020;81(2):266-275. https://doi.org/10.2139/ssrn.3594598 [ Links ]
4. Ling CL, Roberts T, Soeng S, et al. Impact of delays to incubation and storage temperature on blood culture results: A multi-centre study. BMC Infect Dis 2021;21:173 https://doi.org/10.1186/s12879-021-05872-8 [ Links ]
5. Bourbeau PP, Foltzer M. Routine incubation of BacT/ALERT FA and FN blood culture bottles for more than 3 days may not be necessary. J Clin Microbiol 2005;43(5):2506-2509. https://doi.org/10.1128/JCM.43.5.2506-2509.2005 [ Links ]
6. Sepulveda J, Westblade LF, Whittier S, et al. Bacteremia and blood culture utilization during COVID-19 surge in New York City. J Clin Microbiol. 2020;58(8):e00875-20. https://doi.org/10.1128/JCM.00875-20 [ Links ]














