Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
SAMJ: South African Medical Journal
versão On-line ISSN 2078-5135
versão impressa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.111 no.3 Pretoria Mar. 2021
http://dx.doi.org/10.7196/samj.2021.v111i3.14987
RESEARCH
The next generation: Pregnancy in adolescents and women living with perinatally acquired HIV in South Africa
K AndersonI; T MutemaringaII, III; K-G TechnauIV; L F JohnsonV; K BraithwaiteVI; E MokotoaneVII; A BoulleVIII, IX; M-A DaviesX, XI
IMB ChB, Dip HIV Man (SA), MPH (Epidemiology); Centre for Infectious Disease Epidemiology and Research, School of Public Health and Family Medicine, Faculty of Health Sciences., University of Cape Town, South Africa
IIMPhil; Centre for Infectious Disease Epidemiology and Research, School of Public Health and Family Medicine, Faculty of Health Sciences., University of Cape Town, South Africa
IIIMPhil; Directorate of Health Impact Assessment, Western Cape Department of Health, Cape Town, South Africa
IVMB ChB, DCH (SA), Dip HIV Man (SA); Empilweni Services and Research Unit, Department of Paediatrics and Child Health, Rahima Moosa Mother and Child Hospital Faculty of Health Sciences, University of the Witwatersrand, Johannesburg, South Africa
VBBusSc, AIA, PhD; Centre for Infectious Disease Epidemiology and Research, School of Public Health and Family Medicine, Faculty of Health Sciences., University of Cape Town, South Africa
VIMB ChB; Empilweni Services and Research Unit, Department of Paediatrics and Child Health, Rahima Moosa Mother and Child Hospital Faculty of Health Sciences, University of the Witwatersrand, Johannesburg, South Africa
VIIBSc; Centre for Infectious Disease Epidemiology and Research, School of Public Health and Family Medicine, Faculty of Health Sciences., University of Cape Town, South Africa
VIIIMB ChB, MSc, FCPHM (SA), PhD; Centre for Infectious Disease Epidemiology and Research, School of Public Health and Family Medicine, Faculty of Health Sciences., University of Cape Town, South Africa
IXMB ChB, MSc, FCPHM (SA), PhD; Directorate of Health Impact Assessment, Western Cape Department of Health, Cape Town, South Africa
XMB ChB, MMed, PhD, EC; on behalf of IeDEA-SA Pediatrics; Centre for Infectious Disease Epidemiology and Research, School of Public Health and Family Medicine, Faculty of Health Sciences., University of Cape Town, South Africa
XIMB ChB, MMed, PhD, EC; on behalf of IeDEA-SA Pediatrics; Directorate of Health Impact Assessment, Western Cape Department of Health, Cape Town, South Africa
ABSTRACT
BACKGROUND: An increasing number of girls living with perinatally acquired HIV (PHIV) are reaching adolescence and adulthood and becoming pregnant. Youth living with PHIV (YLPHIV) may have HIV-associated infections/complications, long-term exposure to antiretroviral treatment (ART), drug resistance and increased psychosocial challenges, which may adversely affect pregnancy outcomes. There is a lack of published studies on pregnancy in YLPHIV in sub-Saharan Africa
OBJECTIVES: To describe characteristics of pregnant South African (SA) YLPHIV and their pregnancy outcomes
METHODS: We retrospectively identified pregnancies in YLPHIV, who were diagnosed with HIV when they were <12 years old and before their first pregnancy (as a proxy for perinatal route of infection), from routinely collected data in Western Cape Province, SA (2007 - 2018). We combined these with pregnancies from a Johannesburg cohort of YLPHIV
RESULTS: We identified 258 pregnancies among 232 females living with likely PHIV; 38.8% of pregnancies occurred in YLPHIV <16 years old, 39.1% at age 17-19 years and 22.1% at age >20 years. In recent years, a steady increase in the number of pregnancies in YLPHIV was noted; more than two-thirds occurred during 2016 - 2018. ART was commenced prior to pregnancy in 84.9% of YLPHIV, during pregnancy in 6.6% and was not commenced by pregnancy end date in 8.5%. Of the pregnancies in young women with documented outcomes (88.8%; n=229), 80.3% were live births, 14.4% terminations, 3.1% miscarriages and 2.2% stillbirths. Mother-to-child transmission of HIV occurred in 2.2% of infants, 75.3% were uninfected when last tested and 22.6% had unknown HIV status. Among YLPHIV with CD4 counts available within 12 months of pregnancy end date (n=202), 20.3% had a CD4 count <200 cells/μL, 43.1% CD4 count 200 - 499 cells/uL and 36.6% CD4 count >500 cells/uL. Among those with a viral load (VL) available within 12 months of pregnancy end date (n=219), 66.7% had a VL <400 copies/mL, 5.0% VL 400 - 999 copies/mL and 28.3% VL >1 000 copies/mL. Of 186 neonates, 20.4% were preterm deliveries (<37 weeks' gestation). Among neonates with known birthweight (n=176), the mean birthweight was 2 900 g (95% confidence interval (CI) 2 747 - 2 935 g) and 20.5% had a low birthweight (<2 500 g). One congenital malformation (musculoskeletal) and 2 neonatal deaths were recorded
CONCLUSIONS: In recent years, the number of pregnancies in YLPHIV has increased. A considerable proportion of pregnancies occurred in YLPHIV <16 years old. A high proportion of pregnancies was electively terminated. The prevalence of elevated VL and poor immunological status among pregnant YLPHIV is concerning
Paediatric antiretroviral treatment (ART) has improved survival in children living with perinatally acquired HIV (PHIV).[1] Consequently, an increasing number of girls, who were vertically infected by their mothers, are entering adolescence and adulthood, and are becoming pregnant. These young women have grown up with the effects of chronic HIV infection and long-term exposure to ART. They may also have increased psychosocial challenges, ART adherence difficulties and resistance mutations,[2-4] which may adversely affect their pregnancy outcomes.
There are several published studies on the outcomes of pregnancies in youth living with PHIV (YLPHIV), but they are mostly from resource-rich settings: the USA,[5-16] the UK and Ireland[17-19] and other European countries.[20-22] Only a few studies are from resource-limited settings, including Brazil,[23-25] Puerto Rico[26] and India.[27] Most studies are small: n<50 pregnant YLPHIV per study, except for 2 studies[5,6] that included 232 and 235 females, respectively Some findings suggest that, in comparison with youth living with non-perinatally acquired HIV, those living with PHIV have higher rates of elective termination;[7,17] poorer immunological status during pregnancy;[5-7,10,11,14,15,17] poorer virological outcomes during pregnancy[5,6,8,14,15,17] and after pregnancy;[14] and are more likely to have drug resistance.[8,12] In some studies, infants born to mothers with PHIV had high rates of preterm birth[16] and, compared with infants of mothers with non-perinatally acquired HIV, higher mother-to-child transmission (MTCT) of HIV,[5] lower birthweight[14,15] and birthweight-for-age,[11] and shorter stature at 12 months of age.[10]
There is a lack of published data specifically addressing pregnancy outcomes in YLPHIV in sub-Saharan Africa, where >90% of the estimated 1.8 million children < 15 years of age living with HIV reside. [28] Over time, these children will enter childbearing age; therefore the number of pregnancies emerging from this group is expected to increase. The lack of studies in sub-Saharan Africa may be due to several reasons, including limited capacity/resources for routine data collection. Less access to ART during earlier years of the epidemic may also have led to sub-Saharan African countries lagging behind many other countries, with few children surviving to reproductive age until relatively recently. Pregnant YLPHIV may be managed by services/ facilities (e.g. antenatal clinics) different to those where they receive their HIV care, resulting in pregnancy information not being captured with routine HIV program data. Furthermore, children, adolescents and adults are often seen together in integrated HIV clinics, which may make it less easy to monitor YLPHIV as a distinct group.
Our study is descriptive and aims to report the characteristics of pregnant YLPHIV in South Africa (SA) in terms of age; immunological and virological status; and ART in relation to pregnancy. We describe pregnancy outcomes in terms of live births, stillbirths, miscarriages and termination of births; and neonatal outcomes in terms of MTCT congenital abnormalities, deaths, preterm deliveries and birthweight.
Methods
The International epidemiologic Databases to Evaluate AIDS Southern Africa collaboration (IeDEA-SA) undertakes research to improve the long-term delivery of HIV care and ART services. The combined database includes individual-level data from several large collaborating cohorts in Malawi, Zambia, SA, Mozambique, Lesotho and Zimbabwe. All IeDEA-SA sites were invited to contribute data to this analysis. However, most sites, except those in Western Cape Province of SA and one in Johannesburg, did not have routinely collected data on pregnancies.
The Western Cape Department of Health has a variety of electronic record systems for routine delivery of healthcare, including the management of hospital and primary care administration, drug dispensing and laboratory records. Patient records are linked across systems through a unique patient identifier. The integration of this data is managed by the Provincial Health Data Centre (PHDC), which provided the Western Cape data for this analysis.'291 We used routinely collected data from the Western Cape PHDC between 2007 and 2018. The data were received late in 2018; therefore, the 2018 data were not complete.
Pregnancy in women is provisionally identified by the PHDC if there is a rhesus antibody test, midwife obstetric unit visit, International Classification of Diseases 10th Revision (ICD-10) code indicating a pregnancy or pregnancy outcome, drugs dispensed for the termination of pregnancy, or a birth recorded on the birth register Based on these integrated data, a pregnancy outcome and outcome date are inferred for each woman. If additional data are available, the outcome date is inferred by using the last menstrual period date, delivery date or gestational age. If no outcome is recorded, the pregnancy end date is predicted to be 41 weeks after the first evidence of pregnancy. Pregnancy confidence scores are calculated by the PHDC to measure the degree of confidence that the data sources used for inferring pregnancy truly indicate a pregnancy. We restricted our analysis to women with a pregnancy confidence score > 0.7 ('probable pregnancies'), with outcome dates between 1 January 2007 and 31 December 2018. As the mode of HIV transmission is not routinely collected, we identified pregnancies in those who received HIV care before the age of 12 years and before the first pregnancy, as a proxy for perinatal route of infection.
We combined pregnancy data from the Western Cape with those from a smaller cohort of adolescents with PHIV from Rahima Moosa Hospital in Johannesburg, SA, from the same time period (2007 -2018). Rahima Moosa Hospital has a paediatric and adolescent HIV clinic that currently includes ~1 600 patients, of whom ~1 000 are adolescents. Information on mode of HIV transmission, pregnancy and pregnancy outcomes is collected routinely in the clinic and recorded in patient files and on the electronic database. Proxies and confidence scores were not applied, as the Johannesburg data were recorded directly from patient files; hence, there is 100% certainty about those pregnancies.
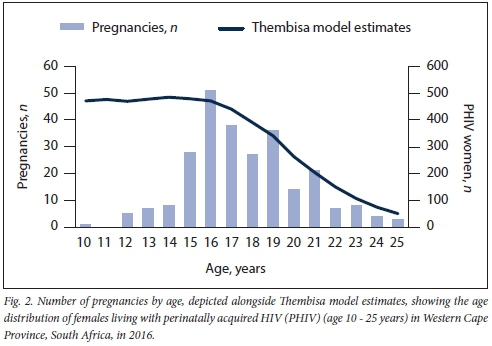
The observed age distribution of pregnant YLPHIV was compared with that of female YLPHIV in the Western Cape in 2016, as estimated by the Thembisa model (version 4.2).[30] Thembisa is a combined demographic and HIV model that has been developed for SA and applied to each province, and is the main source of Joint United Nations Programme on HIV and AIDS (UNAIDS) estimates for SA. The model estimate of YLPHIV was obtained by taking estimated numbers of people living with HIV at each age and subtracting the numbers that would have been predicted by the model if there was no MTCT.
After combining the Western Cape and Johannesburg data, CD4 counts of the young women were recorded as the result closest to pregnancy end date, within 12 months before or after pregnancy end date. Viral load (VL) was also recorded as the result closest to pregnancy end date, within 12 months before or after pregnancy end date. Furthermore, among pregnancies with an outcome of live birth, the VL measurement during pregnancy and nearest to delivery date was recorded (within 41 weeks before delivery or 7 days after delivery) and compared with the first VL available postpartum (measured >7 days post-delivery date and within 12 months of delivery date). Preterm births were defined as those occurring at <37 weeks' gestation. Low birthweight was defined as <2 500 g.
Ethical approval
Analysis of the Western Cape data was undertaken as part of a broader evaluation of the impact of expanded access to ART in pregnancy, approved by the Human Research Ethics Committee at the University of Cape Town (ref. no. UCT HREC 541/2015). The contributions made by the Johannesburg site were approved by the University of the Witwatersrand Human Research Ethics Committee (ref. no. M170788), which includes approval to share anonymised data with the IeDEA Data Centre at UCT for analysis. The IeDEA Data Centre has approval for the analysis of anonymous data from any IeDEA-SA collaboration site (ref. no. HREC 084/2006). Data were collected through routine health services and were anonymised before data sharing with the researchers.
Results
Of 804 495 pregnancies recorded from Western Cape health facilities between 2007 and 2018, 163 351 (20.3%) had evidence of maternal HIV. Of these, 228 had evidence of maternal HIV before age 12 years and before the estimated start date of the first pregnancy. Data were also provided from Rahima Moosa Hospital on 30 pregnancies among adolescents known with PHIV. In total, 258 pregnancies among 232 adolescents and women living with likely PHIV were identified.
Outcomes were documented in 88.8% of pregnancies (n/N=229/258). Among these, 80.3% (n=184) resulted in live births (including 2 sets of twins), 14.4% (n=33) elective terminations, 3.1% (n=7) miscarriages and 2.2% (n=5) stillbirths.
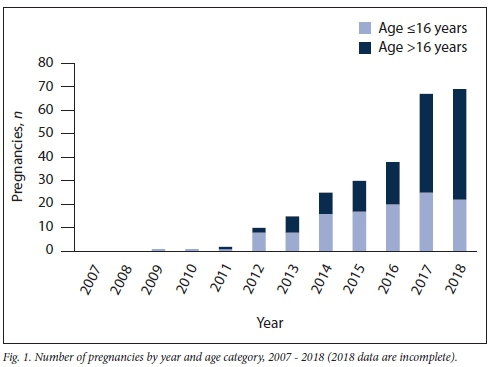
The number of YLPHIV with pregnancies increased annually (Fig. 1), with 67.4% during 2016 - 2018. The proportion of pregnancies in YLPHIV >16 years of age increased annually. Age at the start of pregnancy ranged from 10 to 24 years (Fig. 2); 46.1% of pregnancies occurred in those aged <16 years, 32.6% at 17 - 19 years of age, while 21.3% were in adults >20 years old. Moreover, the changing age distribution of YLPHIV is demonstrated in Fig. 2, which shows estimates of the age distribution of females (aged 10 - 25 years) living with PHIV in the the Western Cape in 2016. For 232 women, we recorded a single pregnancy, for 21 women, 2 pregnancies and for 5 women, 3 pregnancies. ART was commenced before pregnancy in 84.9% of cases, during pregnancy in 6.6% and not commenced by pregnancy end date in 8.5%.
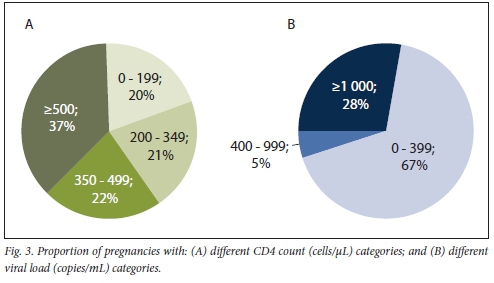
In pregnant women with CD4 counts available within 12 months of pregnancy end date (n/N=202/258; 78.3%), 20.3% had severe immunosuppression, with a CD4 count <200 cells/uL, 21.3% CD4 count 200 - 349 cells/μL, 21.8% CD4 count 350 - 499 cells/uL and 36.6% had optimal immunological status (>500 cells/μL) (Fig. 3A). Median time between pregnancy end date and CD4 count was 104 (interquartile range (IQR) 51 - 173) days. Among those with VL available within 12 months of pregnancy end date (n/N=219/258; 84.9%), 66.7% had a VL <400 copies/mL, 5.0% VL 400 - 999 copies/mL and 28.3% VL >1 000 copies/mL (Fig. 3B). Median time between pregnancy end date and VL was 36 (IQR 11-84) days.
Among the subset of pregnancies in women who had live birth outcomes, VL during pregnancy was compared with VL postpartum. Of those with VL available during pregnancy (n/N=143/184; 77.7%), 66.4% had a VL <400 copies/mL, 5.6% VL 400 - 999 copies/mL and 28.0% VL >1 000 copies/mL. Among those with VL available postpartum (n/N=90/184; 48.9%), 60.0% had a VL <400 copies/mL, 10.0% VL 400 - 999 copies/mL and 30.0% VL >1 000 copies/mL. For women with pregnancies that ended in 2018, the data were incomplete for the postpartum period, resulting in many unknown postpartum VL values. After excluding the pregnancies from 2018, among the subset of pregnant women who had live birth outcomes and VL available during pregnancy (n/N=101/139; 72.7%), 68.3% had a VL <400 copies/mL, 5.9% VL 400 - 999 copies/mL and 25.7 % VL>1 000 copies/mL. Among those with VL available postpartum (n/N=86/139; 61.9%), 61.6% had a VL <400 copies/mL, 10.5% VL 400 - 999 copies/mL and 27.9% VL >1 000 copies/mL. McNemar's X5 test showed that there was a statistically significant decrease in the proportion with VL measurements available postpartum v. during pregnancy (38.1% v. 27.3%; p=0.03).
Among the 186 live births, MTCT of HIV occurred in 2.2% of infants (n=4), 75.3% were uninfected when last tested and in 22.6% HIV status was unknown. Mothers who transmitted HIV to their infants were young (age 13, 15, 16 and 16 years at conception). One of the mothers had an undetectable VL before delivery (measured 2 months prior to delivery). Two of the other mothers had a raised VL at delivery (>1 000 copies/mL), whereas the remaining mother did not have VL monitoring during pregnancy (although VL prior to pregnancy was >l 000 copies/mL). There were 2 neonatal deaths and 1 congenital abnormality, which was described as a proximal focal femoral deficiency. Of the pregnancies that resulted in live births, 20.4% were preterm deliveries. Among neonates with known birthweights (n/N= 176/186), the mean birthweight was 2 900 g (95% confidence interval (CI) 2 747 -2 935 g) and 20.5% of neonates had a low birthweight (<2 500 g).
Discussion
In recent years, there has been a striking increase in pregnancies among YLPHIV; over two-thirds occurred during the past 3 years, with a large proportion in adolescents aged <16 years. Concerningly in one-third of pregnancies, YLPHIV had elevated VLs (>400 copies/mL) and in one-fifth of pregnancies, YLPHIV had severe immuno-suppression (CD4 count <200 cells/uL). The age distribution of these pregnant young women, who included many adolescents aged <16 years, reflects the current context of YLPHIV in the Western Cape, where most of the YLPHIV population are in this age category, with relatively few in the older age categories. Fig. 2 demonstrates how the age distribution of pregnant YLPHIV mirrors the estimates of the age distribution of females living with PHIV in the Western Cape in 2016, whereas Fig. 1 shows that the proportion of pregnancies among YLPHIV >16 years of age has increased annually, as the YLPHIV cohort has grown older.
A high proportion of YLPHIV electively terminated their pregnancies (14.4%), suggesting reasonable access to this service, although this also implies that rates of unintended pregnancy are high and suggests poor access to or poor uptake of contraception. Because routine data from health services were used to assess whether pregnancies were terminated, it is likely to be fairly representative of terminations in the public health service as opposed to self-reported outcomes, which are more prone to bias. The majority of pregnancies occurred in adolescents. Although this most likely reflects the current age distribution of SAs YLPHIV population, it is evident that many YLPHIV become sexually active while still under the care of paediatric HIV services, where provision of integrated family planning services may not be well provided. There may be a need to provide more proactive sexual and reproductive health services and counselling to these youth. Earlier referral of young people to adolescent/youth services or differential care models to increase access to contraceptives may be preferable to keeping adolescents/ youth in paediatric care models where this may be poorly implemented.
The high proportion of pregnant YLPHIV who had elevated VLs (>400 copies/mL) or severe immunosuppression (CD4 count <200 cells/uL) is a concern, as it can be anticipated that in the years to come, an increasing number of pregnancies will occur among women living with PHIV, who may have similar poor virological and/or immunological status. Elevated maternal VL places infants at higher risk of MTCT, whereas severe maternal immunosuppression places mothers at higher risk of opportunistic infections and/or death, which in turn is a risk for poor infant outcomes.
A small study of long-term retention in care and viral suppression in postpartum women with PHIV,[9] as well as a systematic review and meta-analysis to estimate ART adherence rates during pregnancy and postpartum (not specifically PHIV women),[31] has demonstrated significant decreases in postpartum ART adherence. In our cohort, among pregnancies that resulted in live births, a decrease was observed in the proportion of YLPHIV known to be virally suppressed postpartum (61.6% v. 68.3% during pregnancy). However, there was a significantly higher proportion of YLPHIV with unknown postpartum VL (38.1% v. 27.3% during pregnancy), which makes interpretation of these findings difficult. Programmatically, it can be anticipated that women receiving ART should have annual VL monitoring; therefore, it may be that many of those without postpartum VL dropped out of care and were not virally suppressed. It may also be that less attention is paid to VL monitoring in the postpartum period. Poor monitoring of VL postpartum is concerning, especially as breastfeeding is generally promoted. Adolescents are typically transitioned to adult care when pregnant, and if the transition is to a less supportive environment, it may influence adherence.
MTCT of HIV is known to have occurred in 2.2% of neonates, comparable to a reported national early MTCT rate of 2.4%, [32]although 22.6% of the exposed infants in this cohort had not been tested for HIV, and additional transmissions from breastfeeding may have subsequently occurred among the infants who initially tested negative.
Study limitations
Limitations for this study include that the Western Cape data were received late in 2018; therefore the 2018 data were incomplete and the total number of pregnancies for 2018 is likely to be higher than the number reported. Pregnancies prior to 2007 were not included therefore, it is likely that the total YLPHIV pregnancies are under-estimated. Pregnancy and outcome data were not collected directly from young women in the Western Cape and were inferred in some cases; consequently there may be missing/incomplete/inaccurate data. Moreover, we used strict criteria to identify pregnancy in the Western Cape dataset (confidence score >0.7), resulting in many 'possible pregnancies' being excluded. A further limitation was that we did not have a comparison group of women with non-perinatally acquired HIV; therefore, the study is purely descriptive. Survival bias is present for those who are well enough to age into adolescence, with child-bearing potential.
The prevalence of preterm birth and low birthweight in women living with HIV in SA is not known. A large prospective study of a Cape Town cohort from a community-based primary care clinic (not disaggregated by mode of infection) found that, among women with HIV, preterm deliveries occurred in 22% of cases and low birthweight among 14% of neonates.[33] However, a large cohort study from rural KwaZulu-Natal found that, among neonates born to women with HIV, 10.4% were born prematurely and 9.6% had a low birthweight.[34] In our YLPHIV cohort 20.4% of births were preterm deliveries and 20.5% of neonates had a low birthweight. In addition to the effects of chronic HIV infection and long-term exposure to ART, because they are mostly still adolescents and young adults, YLPHIV may typically be at higher risk of pregnancy complications such as preterm delivery and low birthweight. Studies comparing pregnancies in women with PHIV and women with non-perinatally acquired HIV in SA are needed to assess whether these outcomes differ.
Conclusions
Albeit a largely descriptive study, this is one of the first studies on pregnancy outcomes in YLPHIV in sub-Saharan Africa. The next step should be a comparison with pregnancies in women in resource-limited settings who have non-perinatally acquired HIV to assess whether the perinatally acquired group is at risk of worse outcomes.
Declaration. None.
Acknowledgements. The YLPHIV and the staff at the Western Cape sites and Rahima Moosa Hospital are acknowledged for their data and clinical service provision, respectively. The Western Cape Provincial Health Data Centre is credited for providing data.
Author contributions. M-AD and KA contributed to the study conception and design; AB and K-GT contributed data; LFJ, TM and KA performed data analysis; EM contributed to data management; KA prepared the manuscript; M-AD, AB, LFJ, KB and K-GT revised the manuscript and appraised several drafts before approving the final version submitted for publication.
Funding. The project was supported by grant number U01AI069924 from the National Institutes of Health (NIAID, NICHD, NCI, NIDA, NIMH) -PI: M Egger and M-A Davies; and by grant number R01HD080465 from NIH (NICHD) - PI: A Boulle. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NIH.
Conflicts of interest. None.
References
1. Davies M-A, Gibb D, Turkova A. Survival of HIV-1 vertically infected children. Curr Opin HIV AIDS 2016;11(5):455-464. https://doi.org/10.1097/COH.0000000000000303 [ Links ]
2. Nachega JB, Hislop M, Nguyen H, et al. Antiretroviral therapy adherence, Virologie and immunologic outcomes in adolescents compared with adults in southern Africa. J Acquir Immune Defic Syndi 2009;51(1):65-71. https://doi.org/10.1097/QAI.0b013e318199072e [ Links ]
3. Nglazi MD, Kranzer K, Holele P, et al. Treatment outcomes in HIV-infected adolescents attending a community-based antiretroviral therapy clinic in South Africa. BMC Infect Dis 2012;12:21. https://doi.org/10.1186/1471-2334-12-21 [ Links ]
4. Sohn AH, Hazra R. The changing epidemiology of the global paediatric HIV epidemic. Keeping track of perinatally HIV-infected adolescents. J Int AIDS Soc 2013;16(1):18555. https://doi.org/10.7448/IAS.16.1.18555 [ Links ]
5. Goodenough CJ, Patel K, van Dyke RB. Is there a higher risk of mother-to-child transmission of HIV among pregnant women with perinatal HIV infection? Pediatr Infect Dis J 2018;37(12):1267-1270. https://doi.org/10.1097/INR0000000000002084 [ Links ]
6. Jao J, Kacanek D, Williams PL, et al. Birth weight and preterm delivery outcomes of perinatally vs nonperinataliy human immunodeficiency virus-infected pregnant women in the United States. Results from the PHACS SMARTT study and IMPAACT P1025 protocol. Clin Infect Dis 2017;65(6):982-989. https://doi.org/10.1093/cid/cix488 [ Links ]
7. Agwu AL, Jang SS, Korthuis PT, Araneta MRG, Gebo KA. Pregnancy incidence and outcomes in vertically and behaviorally HIV-infected youth. JAMA 2011;30579(510):468-470. https://doi.org/10.100l/jama.2011.79 [ Links ]
8. Badell ML, Kachikis A, Haddad LB, Ly Nguyen M, Lindsay M. Comparison of pregnancies between perinatally and sexually HIV-infected women. An observational study at an urban hospital. Infect Dis Obstet Gynecol 2013;2013. https://doi.org/10.1155/2013/301763 [ Links ]
9. Brogiy SB, Watts DH, Ylitalo N, et al. Reproductive health of adolescent girls perinatally infected with HIV Am J Public Health 2007;97(6):1047-1052. https://doi.org/10.2105/AJPH.2005.071910 [ Links ]
10. Jao J, Agwu A, Mhango G, et al. Growth patterns in the first year of life differ in infants born to perinatally vs. nonperinataliy HIV-infected women. AIDS 2015;29(1):111-116. https://doi.org/10.1097/QAD.0000000000000501 [ Links ]
11. Jao J, Sigel KM, Chen KT, et al. Small for gestational age birth outcomes in pregnant women with perinatally acquired HIV AIDS 2012;26(7):855-859. https://doi.org/10.1097/QAD.0b013e328351f6ef [ Links ]
12. Lazenby GB, Mmeje O, Fisher BM, et al. Antiretroviral resistance and pregnancy characteristics of women with perinatal and nonperinatal HIV infection. Infect Dis Obstet Gynecol 2016;2016:4897501. https://doi.org/10.1155/2016/4897501 [ Links ]
13. Meade CM, Hussen SA, Momplaisir F, Badell M, Hackett S, Sheth AN. Long term engagement in HIV care among postpartum women with perinatal HIV infection in the United States. AIDS Care 2018;30(4):488-492. https://doi.org/10.1080/09540121.2017.1417531 [ Links ]
14. Munjal I, Dobroszycki J, Fakioglu E, et al. Impact of HIV-1 infection and pregnancy on maternal health. Comparison between perinatally and behaviorally infected young women. Adolesc Health Med Ther 2013;4:51-58. https://doi.org/10.2147/AHMT.S39885 [ Links ]
15. Phillips UK, Rosenberg MG, Dobroszycki J, et al Pregnancy in women with perinatally acquired HIV-infection. Outcomes and challenges. AIDS Care 2011;23(9):1076-1082. https://doi.org/10.1080/09540121.2011.554643 [ Links ]
16. Williams SF, Keane-Tarchichi MH, Bettica L, Dieudonne A, Bardeguez AD. Pregnancy outcomes in young women with perinatally acquired human immunodeficiency virus-1. Am J Obstet Gynecol 2009;200(2):149.el-149.e5. https://doi.org/10.1016/j.ajog.2008.08.020 [ Links ]
17. Byrne L, Sconza R, Foster C, Tookey PA, Cortina-Borja M, Thorne C. Pregnancy incidence and outcomes in women with perinatal HIV infection. AIDS 2017;31(12):1745-1754. https://doi.org/10.1097/QAD.0000000000001552 [ Links ]
18. Croucher AP, Jose S, McDonald S, Foster C, Fidler S. Sexual and reproductive health in a UK cohort of young adults perinatally infected with HIV. Sex Transm Infect 2013;89:392-394. https://doi.org/10.1136/sextrans-2012-050831 [ Links ]
19. Kenny J, Williams B, Prime K, Tookey P, Foster C. Pregnancy outcomes in adolescents in the UK and Ireland growing up with HIV HIV Med 2012;13(5):304-308.w https://doi.org/10.1111/j.l468-1293.2011.00967.x [ Links ]
20. Calitri C, Gabiano C, Galli L, et al The second generation of HIV-1 vertically exposed infants. A case series from the Italian register for paediatric HIV infection. BMC Infect Dis 2014;14(1):277. https://doi.org/10.1186/1471-2334-14-277 [ Links ]
21. Prieto LM, McPhee CF, Rojas P, et al. Pregnancy outcomes in perinatally HIV-infected young women in Madrid, Spain-. 2000 - 2015. PLoS ONE 2017;12(8). https://doi.org/10.1371/journal.pone.0183558 [ Links ]
22. Thorne C, Townsend CL, Peckham CS, Newell M-L, Tookey PA. Pregnancies in young women with vertically acquired HIV infection in Europe. AIDS 2007;21(18):2552-2556. https://doi.org/10.1097/QAD.0b013e3282f08b5f [ Links ]
23. Cruz ML, Cardoso CA, João EC, et al Pregnancy in HIV vertically infected adolescents and young women. A new generation of HIV-exposed infants. AIDS 2010;24(17):2727-2731. https://doi.org/10.1097/QAD.0b013e32833e50d4 [ Links ]
24. Cruz ML, Santos E, Teixeira M de LB, et al. Viral suppression and resistance in a cohort of perinatally-HIV infected (PHIV+) pregnant women. Int J Environ Res Public Health 2016;13(6):568. https://doi.org/10.3390/ijerphl3060568 [ Links ]
25. Lundberg P, Andersson R, Machado ES, da Costa TP, Hofer CB. Pregnancy outcomes in young mothers with perinatally and behaviorally acquired HIV infections in Rio de Janeiro. Brazil J Infect Dis 2018;22(5):412-417. https://doi.org/10.1016/j.bjid.2018.08.005 [ Links ]
26. Zorrilla C, Febo I, Ortiz I, et al Pregnancy in perinatally HIV-infected adolescents and young adults -Puerto Rico, 2002. MMWR 2003;52(08):149-151. [ Links ]
27. Chibber R, Khurranna A. Birth outcomes in perinatally HIV-infected adolescents and young adults in Manipur, India. A new frontier. Arch Gynecol Obstet 2005;271:127-131. https://doi.org/10.1007/s00404-003-0564-z [ Links ]
28. United Nations Childrens Fund. UNICEF data. Paediatric care and treatment. 2018. https://data.uniceforg/topic/hivaids/paediatric-treatment-and-care/ (accessed 10 April 2019). [ Links ]
29. Boulle A, Heekes A, Tiffin N, et al Data centre profile. The Provincial Health Data Centre of the Western Cape Province, South Africa. Int J Popul Data Sci 2019;4(2). https://doi.org/10.23889/ijpds.v4i2.1143 [ Links ]
30. Johnson LF, Patrick M, Stephen C,et al. Steep declines in pediatric AIDS mortality in South Africa, despite poor progress towards pediatric diagnosis and treatment targets. Pediatr Infect Dis J 2020;39(9):843-848. https://doi.org/10.1097/INF0000000000002680 [ Links ]
31. Nachega JB, Uthman OA, Anderson J, et al. Adherence to antiretroviral therapy during and after pregnancy in low-, middle and high income countries. A systematic review and meta-analysis HHS public access. AIDS 2012;26(16):2039-2052. https://doi.org/10.1097/QAD.0b013e328359590f [ Links ]
32. Sherman GG, Lilian RR, Bhardwaj S, Candy S, Barron P. Laboratory information system data demonstrate successful implementation of the prevention of mother-to-child transmission programme in South Africa. S Afr Med J 2014;104(3):235. https://doi.org/10.7196/samj.7598 [ Links ]
33. Malaba TR, Phillips T, le Roux S, et al. Antiretroviral therapy use during pregnancy and adverse birth outcomes in South African women. Int J Epidemiol 2017;46(5):1678-1689. https://doi.org/10.1093/ije/dyxl36 [ Links ]
34. Cherry T, Thorne C, Coutsoudis A. Preterm delivery and small-for-gestation outcomes in HIV-infected pregnant women on antiretroviral therapy in rural South Africa. Results from a cohort study, 2010 - 2015. PLoS ONE 2018;13(2):c0192805. https://doi.org/10.1371/journal.pone.0192805 [ Links ]
 Correspondence:
Correspondence:
K Anderson
kim.anderson@uct.ac.za
Accepted 6 October 2020














