Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
SAMJ: South African Medical Journal
versão On-line ISSN 2078-5135
versão impressa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.109 no.1 Pretoria Jan. 2019
http://dx.doi.org/10.7196/samj.2019.v109i1.13278
RESEARCH
South African women's perspectives on self-sampling for cervical cancer screening: A mixed-methods study
R SaiduI, II, III; J MoodleyIV, V, VI; A TergasVII; M MombergVIII; R BoaIX; T WrightX; S CampbellXI; C Svanholm-BarrieXII; D PersingXIII; L KuhnXIV; L DennyXV, XVI
IMBBS, MPH; Department of Obstetrics and Gynaecology, Faculty of Health Sciences, University of Cape Town, South Africa
IIMBBS, MPH; SAMRC Gynaecological Cancer Research Centre, Faculty of Health Sciences, University of Cape Town, South Africa
IIIMBBS, MPH; Department of Obstetrics and Gynaecology, Faculty of Clinical Sciences, College of Health Sciences, University of Ilorin, Nigeria
IVMD, PhD; SAMRC Gynaecological Cancer Research Centre, Faculty of Health Sciences, University of Cape Town, South Africa
VMD, PhD; Cancer Research Initiative, Faculty of Health Sciences, University of Cape Town, South Africa
VIMD, PhD; Women's Health Research Unit, School of Public Health and Family Medicine, Faculty of Health Sciences, University of Cape Town, South Africa
VIIMD; Professor Emeritus of Pathology and Cell Biology, Columbia University, New York, USA
VIIIMSc; Women's Health Research Unit, School of Public Health and Family Medicine, Faculty of Health Sciences, University of Cape Town, South Africa
IXMD; Department of Obstetrics and Gynaecology, Faculty of Health Sciences, University of Cape Town, South Africa
XMD; Professor Emeritus of Pathology and Cell Biology, Columbia University, New York, USA
XIPhD; Cepheid Inc., Sunnyvale, Calif., USA
XIIPhD; Cepheid Inc., Stockholm, Sweden
XIIIMD, PhD; Cepheid Inc., Sunnyvale, Calif., USA
XIVPhD; Gertrude H Sergievsky Center, College of Physicians and Surgeons, and Department of Epidemiology, Mailman School of Public Health, Columbia University Medical Center, New York, USA
XVMD, PhD; Department of Obstetrics and Gynaecology, Faculty of Health Sciences, University of Cape Town, South Africa
XVIMD, PhD; SAMRC Gynaecological Cancer Research Centre, Faculty of Health Sciences, University of Cape Town, South Africa
ABSTRACT
BACKGROUND. Self-sampling as a method of screening for cervical cancer and its precursors is an attractive option for low-resource settings. However, to allow successful integration of self-sampling into national screening programmes, it is necessary to understand women's perceptions and beliefs surrounding this method of sampling the cervix.
OBJECTIVES. To explore women's attitudes to self-collection of samples for cervical screening in a low-resource setting in South Africa (SA).
METHODS. Mixed methods were used to meet the study objectives. We recruited women aged 30 - 65 years into a study in Cape Town, SA, to participate in a cross-sectional survey. All women collected a vaginal self-sample, and underwent visual inspection with acetic acid, colposcopy, and collection of cervical samples and appropriate histology specimens by a doctor. Women had a quantitative questionnaire-based exit interview. A subset of these women participated in focus group discussions (FGDs).
RESULTS. A total of 822 women answered the exit survey questionnaire and 41 women participated in the FGDs. Most women from the survey had a positive perception of self-sampling, with 93.6% of the women reporting not feeling embarrassed and 89.4% reporting experiencing no discomfort at all when taking a self-sample. This was corroborated by the FGD participants, who found self-sampling easier, more comfortable and less embarrassing than clinician sampling. However, many women (64.7%) felt more confident when the sample was taken by a clinician, despite having a positive attitude towards self-sampling. In most cases this was because they thought that the clinician would take a better sample, as explained by the FGD participants. Although 93.9% of the women were willing to collect a self-sample, the women in the FGDs expressed a preference for doing so at the health facility rather than at home. There were many reasons for this, including the cost of returning to the clinic with the sample.
CONCLUSIONS. Attitudes regarding self-sample collection were positive in this study population. Participants were willing to perform self-sampling, but expressed concerns regarding the quality of the specimen and the financial implications of returning to the clinic with it. Pilot implementation studies will be useful before this method of sampling is adopted and integrated into screening programmes.
There are marked global disparities in the incidence of cervical cancer, with most cases occurring in low- and middle-income countries (LMICs). In high-income countries (HICs), the implementation of organised cytology-based cervical screening programmes has resulted in a decline in cervical cancer incidence and mortality rates. Unfortunately, cervical cancer continues to be a significant public health problem in LMICs owing to the lack of high-quality screening programmes and poor screening coverage in those regions.[1,2] Effective cytology-based screening requires a relatively robust healthcare infrastructure with functional laboratory services, well-trained healthcare providers and technicians, good referral systems, and linkage to accessible treatment facilities and follow-up after treatment for women with abnormal screening tests. This approach requires resources typically not available in LMICs, hampering the initiation and maintenance of screening programmes.[3]
The World Health Organization has recommended molecular testing for human papillomavirus (HPV) as an alternative to cytology-based screening in low-resource settings, given that these tests are more sensitive than cytology and visual inspection methods in detecting high-grade cervical intraepithelial neoplasia and cancer. [2,4-6] A randomised controlled trial in India showed that in low-resource settings, a single round of HPV testing was associated with a significant reduction in cervical cancer mortality.[5] HPV tests also have the advantage that vaginal samples can be collected by the woman herself, which may increase coverage.[3,7] HPV DNA testing is therefore an attractive alternative to cytology-based screening in low-resource settings.
HPV testing on self-collected vaginal samples has been shown to be as accurate as testing on samples collected by a doctor and can potentially increase coverage for cervical cancer screening in low-resource settings.[7-12] Several studies done across the world in various settings suggest that self-sampling is an acceptable method of collecting a sample for HPV typing for most women.[3,13-16]
Objectives
To explore women's perceptions and acceptance of self-collection of samples for cervical screening and their willingness to do so, in a low-resource setting in South Africa (SA).
Methods
This mixed-methods study was part of a National Cancer Institute (NCI)-funded collaborative project carried out by the University of Cape Town, Columbia University and Cepheid Inc. (Sunnyvale, Calif., USA), utilising a new point-of-care CE-IVD test, Xpert HPV. A convergent parallel design was used in which quantitative and qualitative data were collected and analysed separately and then merged. Approval for the study was obtained from the Faculty of Health Sciences Human Research Ethics Committee, University of Cape Town (ref. no. 703/2014) and the Human Research Protection Office, Institutional Review Board, Columbia University (ref. no. AAAO3652). The subjects provided written informed consent for participation.
Women aged 30 - 65 years were recruited from a referral colposcopy clinic and a primary care site in Cape Town, SA, from February 2015 to May 2016. Equal numbers of HIV-positive and HIV-negative women were recruited. All the participants self-collected a vaginal sample (a Dacron swab placed into 4 mL of PreservCyt transport medium in a polypropylene tube) in a private room following a verbal explanation on how to collect the sample by a community health worker (CHW). All the women also underwent collection of cervical samples by colposcopy, followed by visual inspection of the cervix after application of acetic acid and collection of histology samples by a doctor. The study aimed to recruit roughly equal numbers of HIVpositive and HIV-negative women.
Cross-sectional survey
All participants answered an exit questionnaire that examined the woman's level of embarrassment, discomfort, confidence to carry out self-sampling, and feelings of being ignored by the health worker and of intrigue with the procedure. The response options for these questions were coded on a 5-point scale: 1 = 'extremely', 2 = 'very', 3 = 'moderately', 4 = 'slightly', 5 = 'not at all'. The responses to the questions regarding 'confidence to carry out self-sampling' and 'whether they felt intrigued' were reverse-coded (1 = 'not at all', 2 = 'slightly', 3 = 'moderately', 4 = 'very', 5 = 'extremely') for accurate interpretation. Questions on collection method preference (whether they prefered self-sampling, clinician sampling or had no preference for either method) and whether they were willing to take a self-sample at home were also included in the questionnaire.
Descriptive statistics were used to describe the respondents' demographic profile and response to the exit questionnaire. The x2 test was used to compare categorical variables and the Kruskal-Wallis test for continuous variables. Variables that were significant in the bivariate analysis were included in a multinomial logistic regression model to investigate independent predictors of sampling preference.
The differences were statistically significant at p<0.05 (two-sided). The data analysis was performed using SPSS 23 (IBM, USA)
Focus group discussions
Six focus group discussions (FGDs) were conducted at the primary healthcare study site. Each group consisted of 6 - 7 participants, considered the minimum number required to elicit meaningful dialogue and discussion. All FGDs took place once participants had completed all the clinical assessments. Written informed consent for the FGD was obtained from all participants. Discussions lasted between 45 and 60 minutes. An interview guide was used for the FGDs, which were facilitated by a female trained qualitative researcher with the assistance of a translator (discussions were conducted in isiXhosa and were digitally recorded, translated and transcribed verbatim).
The health belief model (HBM) theory[17] was employed to construct exploratory themes for the FGDs. The themes explored included individual and community understanding of cervical cancer; perceived risk of and susceptibility to cervical cancer; perceived individual and community barriers to and benefits of Pap smears; women's experience with self-sampling (including preceding anxiety/fears/concerns, ease or difficulty of the procedure, associated pain and/or discomfort, confidence in collecting sample, perceived individual and community barriers, and benefits of self-sampling); self-sample collection preference (i.e. provider or self-collected); site preference (at home or at the clinic) and reasons for preference; and suggestions on how to facilitate self-sampling.
The transcripts were reviewed, and data were entered on QSR Nvivo10,[18] a software package designed for systematic management and analysis of qualitative data. Initial coding categories were based on the interview guides and from the theoretical concepts of the HBM. Once all the text segments had been given basic codes, the codes were categorised into basic themes by placing similar codes together. For this study, only themes related to self-sampling were analysed.
Results
Results from the quantitative and qualitative research were reviewed to seek corroboration, to clarify results from the quantitative research and vice versa, and to extend the breadth of our understanding.
Sociodemographic characteristics
The cross-sectional survey included 822 women, with a median age of 41 years (interquartile range (IQR) 35 - 49) and median parity of 2 (IQR 2 - 3). Most of the women had a secondary education (78.4%), 80.0% had had a previous Pap smear, and 78.6% were non-smokers. More than half of the participants (54.1%) used some form of contraception, and 42.3% tested positive for HIV. Forty-one women were enrolled and participated in the FGDs (mean age 41 years, range 30 - 62).
Acceptability of self-sampling
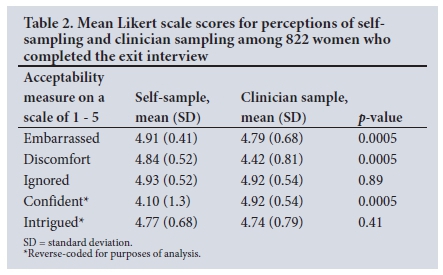
Table 1 reports on participant perceptions of self- and clinician sampling elicited during the cross-sectional survey. A higher proportion of women reported that they did not feel embarrassed at all taking the self-sample (93.6%) compared with those reporting no embarrassment having the doctor collect the sample (88.2%) (p=0.0002). Mean Likert scores for embarrassment (Table 2) also showed that women were significantly less embarrassed to self-sample compared with sampling by a doctor. The proportion who reported feeling no discomfort at all was greater for the self-collected (89.4%) v. the doctor-collected sample (56.0%) (p<0.001), with significant differences in the mean Likert scores (Table 2). Women were more confident when the sample was taken by a clinician (64.7%) rather than by self-sampling (59.5%) (p=0.03), also with significant differences in the mean Likert score. These findings were corroborated during the FGDs, as illustrated by the quotes below.
'I prefer doing it [self-sample] myself; to me, it was easier than having someone else doing it to me.' (FGD1)
'It was [more] comfortable to do the self-sampling myself than to be done by the doctor.' (FGD6)
'I would choose to do it [sample] myself because I get embarrassed to expose myself to the doctor.' (FGD2)
Instructions on self-sampling procedure
This was explored in the FGDs only. All the women thought that the CHWs' verbal explanation of how to perform the self-sample procedure was sufficient and could not think of ways in which the description of the procedure could be improved. When asked whether diagrams or illustrations would be useful, mixed views were expressed. Most women did not think it would be beneficial to include diagrams and preferred a verbal explanation only. Some felt that pictures would be disturbing.
'Those pictures of cervix and wombs are scary.' (FGD 6)
Some women, however, did think the use of diagrams and illustrations would be helpful.
' I would prefer an explanation and a diagram because the doctor will explain and at the same time show you on the picture how to do it [self-sample].' (FGD 4)
All participants noted the importance of using simple and easy-to-understand expressions when explaining how to perform the self-sampling. While some thought that an explanation in English would be acceptable, the majority would prefer the explanation to be given in their mother tongue.
Sampling method preference
In the cross-sectional survey, more women (45.1%) preferred their cervical sample to be taken by the clinician, while 33.8% preferred self-sampling and 21.0% did not have a preference for one method over the other. FGD participants gave more insight into why the participants preferred the health worker to take the sample rather than to self-sample. Those who preferred the sample to be taken by a health worker justified their choice by stating that the doctor is a trained professional and therefore more capable of taking the sample. Furthermore, he or she could also investigate, identify and treat any other abnormalities that the patient might not be able to see.
'I would rather have a doctor do it [sample]. When she opens down there, she can examine and see other things inside down there, which I would not be able to notice myself.' (FGD 5)
However, some of the women who preferred taking the sample themselves stated that when the health worker collected the sample they found it painful and were embarrassed.
We further analysed the cross-sectional data to see the relationship between sampling method preference and the demographic variables of the respondents. On bivariate analysis, women with a tertiary level of education and HIV-negative women were significantly more likely to prefer self-sampling to clinician sampling, while women who were not on any form of contraception and those who lived in informal settlements were significantly more likely to prefer clinician sampling (Table 3).
A multinomial logistic regression analysis that included age categories, education level, type of housing, HIV status, smoking and contraceptive use was performed to determine the predictive factors of sampling preference (self-sampling, clinician sampling or no preference for method). The reference group was those women with preference for the clinician sampling method. Accordingly, each predictive factor has two parameters, one for predicting preference for self-sampling rather than clinician sampling and one for predicting no preference for either method. Women who used contraception and those who smoked were ~1.5 times more likely and those living in houses they owned twice as likely to prefer self-sampling to clinician sampling. Women with primary education and above were ~3 times and HIV-positive women ~1.5 times more likely to prefer clinician sampling to self-sampling or to have no preference for either method (Table 4).
In multivariable analysis, educational status, contraceptive use and HIV status remained significant. Type of housing was no longer significant in the multivariable model.
Willingness to self-sample at home
The majority (93.9%) of the women who participated in the cross-sectional survey said that they would be willing to self-sample at home. Going deeper into this discussion during the FGDs, many women mentioned that the idea of collecting the self-sample at home was appealing, because of home comfort. Even those who lived in crowded homes said that they would be able to find a space to take the sample. When asked how they would feel if somebody saw them taking the self-sample or found the self-sample at home, most felt confident that they would be able to explain what they were doing and why they were taking the sample.
'It's like the same as putting a tampon, if someone sees you putting it they have the same reaction, and you explain.' (FGD 5)
However, during the FGDs when women were told that the self-sample collected at home would need to be returned to the health facility, most suggested that they would then prefer to collect the self-sample at the clinic instead. Reasons for choosing to do so included costs associated with having to come to the clinic twice, while having a health professional at hand who could give reassurance if needed was also important for some. Some women also raised concerns about specimen contamination and the possibility of forgetting or being unable to return it to the clinic in time.
' It's waste of time and money coming here and going home and coming back here again whereas I could have done everything here all at once.' (FGD 5)
'It will also depend on having money to come back to the clinic, if I have money then I will come.' (FGD 5)
'I would be worried about something getting into the sample and contaminating it.' (FGD 3)
'I would worry about it getting dry before bringing it back.' (FGD 5)
Discussion
In this study, we integrated questionnaire-based interviews, which were used to determine individual experiences of the patients of both self-sampling and clinician-collected sampling, with FGDs, which were used to obtain an in-depth discussion to understand their beliefs and opinions with regards to self-sampling.
As described in Table 1, and corroborated by the findings in the FGDs, even though there was initial surprise when women were asked to take the sample, they had more positive perceptions about self-sampling than clinician sampling, most notably regarding less embarrassment and discomfort when self-collecting the sample. Previous studies have noted similar findings from both HICs and LMICs.[3,12-15] However, women were significantly less confident about the quality of the self-collected sample compared with the clinican-collected sample. This lack of confidence in self-sampling has been reported in previous studies.[19,20] Incorporating additional information to assure women that self-sampling does not compromise the quality of the sample and that many studies have found self-sampling to be as reliable as clinician sampling may help boost their confidence to obtain a self-sample.[7,8,10,11]
Despite acceptability of the self-sampling method, about half of the women overall (45.1%) preferred the samples to be collected by the clinician. Anhang et al.[21]reported similar findings in 2005 among Hispanic women in New York City, where there was an overwhelming acceptance of self-sampling, but more preference for clinician sampling. In the FGDs, some participants expressed concern about their ability to take a high-quality sample and would therefore prefer a clinician to obtain it. Others were worried about performing the procedure incorrectly and causing harm. Still others expressed that they would prefer the sample to be collected by a health worker, who apart from taking a good-quality sample would also be able to detect and investigate any abnormalities the patient might be unable to identify. Previous studies have found similar concerns in various groups of women.[12-14,16,19] In the present study, HIV-negative women and women using contraception were more likely to prefer self-sampling, while HIV-positive women were more likely to prefer clinician sampling. Many HIV-positive women in SA have access to reproductive healthcare services integrated into the antiretroviral (ARV) programmes and trust their caregivers, and may therefore feel safer and more confident if a health worker takes the sample.
Many women from both the survey and the FGDs found the idea of performing the self-sampling procedure at home appealing despite sometimes having unfavourable home settings. However, the realisation during FGDs that they would need to return the specimen to the clinic was a deterrent to home self-sampling. Women raised concerns about specimen contamination, being unable to return the sample to the health centre in time, the cost of making two clinic visits, having to take additional time off work, and not having a health professional at hand to help and give reassurance if needed. It is worth noting that self-sampling in the health facility has the potential to reduce patient load on the limited number of health personnel in poorly staffed clinics and reduce patient waiting times in overcrowded clinics.
Conclusion
This study has highlighted positive perceptions of self-sampling for cervical cancer screening in this low-resource setting. Concerns raised by the women could be addressed by appropriate health education and counselling on self-sampling. The study highlighted some limitations to collecting self-samples at home in this environment. However, many health centres in developing countries have insufficient human resources to collect cervical samples from all women who require screening. In this situation, self-sampling in the health facility may contribute significantly to reducing long waiting times and congestion in health centres. Pilot implementation studies are needed to determine how best to incorporate this approach in national screening programmes
Declaration. None.
Acknowledgements. The study was supported by the NCI (UH2/3CA189908). We thank the women of Khayelitsha and those attending the colposcopy clinic for their willingness to participate in the study, the Khayelitsha Cervical Cancer Screening Project team members, Dr Jozene Cloete (medical officer, Site B community health centre) and Mr David Binza (facility manager, Site B community health centre) for their support, and Sr Thandi Xintolo for recruiting women for the study. We also thank the Cepheid USA and Cepheid South Africa teams, particularly Tessa Visser, who is based in Cape Town.
Author contributions. RS: study design, data collection, analysis and interpretation; JM: study design, data collection, analysis and interpretation; AT: analysis and interpretation; MM: data collection, analysis and interpretation; RB: data collection, interpretation; TW: interpretation; SC: study design, interpretation; CS-B: interpretation; DP: study design, interpretation; LK: study design, analysis and interpretation; LD: study design, data collection, analysis and interpretation.
Funding. National Cancer Institute (NCI grant no. UH2/3CA189908)
Conflicts of interest. DP, SC and CS-B are employees of Cepheid Inc. There are no conflicts of interest for the other authors.
References
1. Jeronimo J, Holme F, Slavkovsky R, et al. Implementation of HPV testing in Latin America. J Clin Virol 2016;76(Suppl 1):S69-S73. https://doi.org/10.1016/jjcv.2015.1L035 [ Links ]
2. Sankaranarayanan R. Screening for cancer in low- and middle-income countries. Ann Glob Health 2014;80(5):412-417. https://doi.org/10.1016/j.aogh.2014.09.014 [ Links ]
3. Berner A, Hassel S Ben, Tebeu P-M, et al. Human papillomavirus self-sampling in Cameroon: Women's uncertainties over the reliaility of the method are barriers to acceptance. J Low Genital Tract Dis 2013;17(3):235-241. https://doi.org/10.1097/LGT.0b013e31826b7b51 [ Links ]
4. World Health Organization. Guidelines for Screening and Treatment of Precancerous Lesions for Cervical Cancer Prevention. Geneva: WHO, 2013:60. https://www.who.int/reproductivehealth/publications/cancers/screenin g_and_treatment_of_precancerous_lesions/en/ (accessed 27 July 2016). [ Links ]
5. Sankaranarayanan R, Nene BM, Shastri SS, et al. HPV screening for cervical cancer in rural India. N Engl J Med 2009;360(4):1385-1394. https://doi.org/10.1056/NEJMoa0808516 [ Links ]
6. Toliman P, Badman SG, Gabuzzi J, et al. Field evaluation of Xpert HPV point-of-care test for detection of human papillomavirus infection by use of self-collected vaginal and clinician-collected cervical specimens. J Clin Microbiol 2016;54(7):1734-1737. https://doi.org/10.1128/JCM.00529-16 [ Links ]
7. Quincy BL, Turbow DJ, Dabinett LN, et al. Diagnostic accuracy of self-collected human papillomavirus specimens as a primary sceen for cervical cancer. J Obstet Gynaecol 2012;32(8):795-799. https://doi.org/10.3109/01443615.2012.717989 [ Links ]
8. Porras C, Hildesheim A, González P, et al. Performance of self-collected cervical samples in screening for future precancer using human papillomavirus DNA testing. J Natl Cancer Inst 2015;107(1):1-9. https://doi.org/10.1093/jnci/dju400 [ Links ]
9. Adamson PC, Huchko MJ, Moss AM, et al. Acceptability and accuracy of cervical cancer screening using a self-collected tampon for HPV messenger-RNA testing among HIV-infected women in South Africa. PLoS One 2015;10(9):1-12. https://doi.org/10.1371/journal.pone.0137299 [ Links ]
10. Zhao FH, Lewkowitz AK, Chen F, et al Pooled analysis of a self-sampling HPV DNA test as a cervical cancer primary screening mthod. J Natl Cancer Inst 2012;104(3):178-188. https://doi.org/10.1093/jnci/djr532 [ Links ]
11. Arbyn M, Verdoodt F, Snijders PJF, et al. Accuracy of human papillomavirus testing on self-collected versus clinician-collected samples: A meta-analysis. Lancet Oncol 2014;15(2):172-183. https://doi.org/10.1016/S1470-2045(13)70570-9 [ Links ]
12. Mullins R, Scalzo K, Sultana F. Self-sampling for cervical screening: Could it overcome some of the barriers to the Pap test? Med Screen 2014;21(4):201-206. https://doi.org/10.1177/0969141314555247 [ Links ]
13. Bansil P, Wittet S, Lim JL, et al. Acceptability of self-collection sampling for HPV-DNA testing in low-resource settings: A mixed methods approach. BMC Public Health 2014;14(1):596. https://doi.org/10.1186/1471-2458-14-596 [ Links ]
14. Ma'som M, Bhoo-Pathy N, Nasir NH, et al. Attitudes and factors affecting the acceptability of self-administered cervicovaginal sampling for human papillomavirus (HPV) genotyping as an alternative to Pap testing among multiethnic Malaysian women. BMJ Open 2016;6(8):e011022. https://doi.org/10.1136/bmjopen-2015-011022 [ Links ]
15. Sultana F, Mullins R, Murphy M, et al. Women's views on human papillomavirus self-sampling: Focus groups to assess acceptability, invitation letters and a test kit in the Australian setting. Sex Health 2015;12(2):279-286. https://doi.org/10.1071/SH14236 [ Links ]
16. Quincy BL, Turbow DJ, Dabinett LN. Acceptability of self-collected human papillomavirus specimens as a primary screen for cervical cancer. J Obstet Gynaecol 2012;32(1):87-91. https://doi.org/10.3109/01443615.2011.625456 [ Links ]
17. Janz NK, Becker MH. The health belief model: A decade later. Health Educ Q 1984;11(1):1-47. https://doi.org/10.1177/109019818401100101 [ Links ]
18. Casttleberry A. NVivo 10 software program. Version 10. QSR International. Am J Pharm Educ 2014;78(1):25-25. https://doi.org/10.5688%2Fajpe78125 [ Links ]
19. Waller J, McCaffery K, Forrest S, et al. Acceptability of unsupervised HPV self-sampling using written instructions. J Med Screen 2006;13(4):208-213. https://doi.org/10.1177/096914130601300409 [ Links ]
20. Dzuba IG, Diaz EY, Allen B, et al. The acceptability of self-collected samples for HPV testing vs the pap test as alternatives in cervical cancer screening. J Womens Health Gend Based Med 2002;11(3):265-275, https://doi.org/10.1089/152460902753668466 [ Links ]
21. Anhang R, Nelson JA, Telerant R, et al. Acceptability of self-collection of specimens for HPV DNA testing in an urban population. J Womens Health 2005;14(8):721-728. https://doi.org/10.1089/jwh.2005.14.721 [ Links ]
 Correspondence:
Correspondence:
R Saidu
rakiya.saidu@uct.ac.za
Accepted 7 June 2018