Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.107 n.2 Pretoria Feb. 2017
http://dx.doi.org/=10.7196/samj.2017.v107i2.11383
RESEARCH
The spectrum of gastric cancer as seen in a large quaternary hospital in KwaZulu-Natal, South Africa
F BenamroI, II; B SartoriusIII, IV; D L ClarkeV, VI, VII; F AndersonVIII, IX, X; E LootsXI, XII, XIII; L OlingerXIV
IMB ChB, MMed, FCS (SA); Department of Surgery, Inkosi Albert Luthuli Central Hospital, Durban, South Africa
IIMB ChB, MMed, FCS (SA); Department of Surgery, School of Clinical Medicine, College of Health Sciences, Nelson R Mandela School of Medicine, University of KwaZulu- Natal, Durban, South Africa
IIIBSc, BSc Hons, MSc, PhD; Discipline of Public Health Medicine, School of Clinical Medicine, College of Health Sciences, Nelson R Mandela School of Medicine, University of KwaZulu-Natal, Durban, South Africa
IVBSc, BSc Hons, MSc, PhD; South African Medical Research Council/University of KwaZulu-Natal Gastrointestinal Cancer Research Centre, Durban, South Africa
VMMedSci, MBA, MPhil, PhD, FCS (SA); Department of Surgery, School of Clinical Medicine, College of Health Sciences, Nelson R Mandela School of Medicine, University of KwaZulu- Natal, Durban, South Africa
VIMMedSci, MBA, MPhil, PhD, FCS (SA); Discipline of Public Health Medicine, School of Clinical Medicine, College of Health Sciences, Nelson R Mandela School of Medicine, University of KwaZulu-Natal, Durban, South Africa
VIIMMedSci, MBA, MPhil, PhD, FCS (SA); Department of General Surgery, Grey's Hospital, Pietermaritzburg, South Africa
VIIIMB ChB, MMed, FCS (SA); Department of Surgery, Inkosi Albert Luthuli Central Hospital, Durban, South Africa
IXMB ChB, MMed, FCS (SA); Department of Surgery, School of Clinical Medicine, College of Health Sciences, Nelson R Mandela School of Medicine, University of KwaZulu- Natal, Durban, South Africa
XMB ChB, MMed, FCS (SA); Discipline of Public Health Medicine, School of Clinical Medicine, College of Health Sciences, Nelson R Mandela School of Medicine, University of KwaZulu-Natal, Durban, South Africa
XIMB ChB, FCS (SA), Cert Gastroenterology (SA) Surg; Department of Surgery, Inkosi Albert Luthuli Central Hospital, Durban, South Africa
XIIMB ChB, FCS (SA), Cert Gastroenterology (SA) Surg; Department of Surgery, School of Clinical Medicine, College of Health Sciences, Nelson R Mandela School of Medicine, University of KwaZulu- Natal, Durban, South Africa
XIIIMB ChB, FCS (SA), Cert Gastroenterology (SA) Surg; South African Medical Research Council/University of KwaZulu-Natal Gastrointestinal Cancer Research Centre, Durban, South Africa
XIVMSc (Biostatistics); School of Health Systems and Public Health, Faculty of Health Sciences, University of Pretoria, South Africa
XVDepartment of General Surgery, Faculty of Health Sciences, University of the Witwatersrand, Johannesburg, South Africa
ABSTRACT
BACKGROUND. Gastric cancer (GC) is the fifth most commonly diagnosed cancer in the world, with the third-highest associated mortality. It has a varying geographical, ethnic and socioeconomic distribution.
OBJECTIVE. To assess the presentation and management of GC in the Durban metropolitan area, South Africa.
METHODS. A retrospective review of 131 patients treated at the quaternary Inkosi Albert Luthuli Central Hospital in Durban from 2009 to 2014 was performed.
RESULTS. The 131 patients were predominantly black African (n=59, 45.0%) and Indian (n=63, 48.1%). Gender was evenly distributed, with 72 males (55.0%) and 59 females (45.0%). The average age of the patients was 60 years (standard deviation 13.3). More than 70% were in advanced stages of cancer and were treated conservatively. There was no significant relationship between body mass index (BMI) and the position of the tumour (p=0.175). Creatinine and albumin levels differed significantly between the genders (p<0.001 and p=0.01, respectively).
CONCLUSIONS. GC appears to have a disproportionately high prevalence among Indians in Durban, and the prevalence of GC appears to be slightly higher among males. Both these observations may simply reflect referral patterns and warrant further investigation. More than 70% of patients presented with advanced-stage disease, and anaemia was common. No relationship was found between BMI and the location of the tumour, although most of the cancers were in the body and distal part of the stomach.
A lack of good-quality data makes it difficult to understand the burden of oncological disease in South Africa (SA) and to establish priorities or benchmark outcomes. Gastric cancer (GC) is the fifth most common visceral malignancy in the world, and the third-highest cause of cancer-related death.[1] According to the SA National Cancer Registry (reported on the Cancer Association of South Africa website), there were over 1 000 cases of GC in SA in 2010.[2] However, there are major deficiencies in this registry, and cancer data are poorly reported nationwide.[1,2] In addition, the diverse nature of the SA population means that the burden of oncological disease varies between centres and population groups. The mixed-race population of the Western Cape Province is known to have a high incidence of the disease. The situation in KwaZulu-Natal (KZN) Province has not been described previously. In the light of these challenges the best data available are regional hospital-based data, and despite the obvious limitations of such data, researchers have to use this information to identify potential trends and areas that deserve more thorough investigation.
Objective
This retrospective audit of a prospectively maintained central hospital medical record system is intended to provide a description of the spectrum of GC seen in KZN and to benchmark the management of the disease against local and international guidelines.[3] It was hoped that data on risk factors for the development of GC in our environment could be analysed.
Methods
Study design and inclusion and exclusion criteria
A retrospective review of a prospectively maintained electronic patient record system was undertaken to identify all patients with GC who were diagnosed or treated at the quaternary referral centre Inkosi Albert Luthuli Central Hospital (IALCH) in Durban, SA, between January 2009 and April 2014. All patients seen at IALCH with histologically proven GC were included. All data were retrieved from patients' digital files.
The study was approved by the Biomedical Research Ethics Committee at the Nelson R Mandela School of Medicine, University of KwaZulu-Natal (ref. no. BCA012/14).
Statistical analysis
Data were analysed using SPPS version 21 (IBM, USA) and Stata 12.1 (StataCorp, USA). Continuous variables were summarised using means and standard deviations (SDs). Medians and interquartile ranges (IQRs) were determined for highly skewed variables such as haemoglobin and creatinine. Variables were assessed for normality by means of histograms and normal quantile graphs. Student's f-tests identified associations between the means of normally distributed continuous variables across gender, with adjustment for unequal variances, as identified by Levene's test. The Mann-Whitney (7-test was used to test for significant differences across gender in the means of the non-normal variables haemoglobin and creatinine. Differences in continuous data among ethnic groups were analysed using the Kruskal-Wallis analysis of variance (ANOVA) for non-normal variables and the one-way ANOVA for normally distributed variables. Frequency distribution tables were compiled to present categorical variables. Significant associations in contingency tables (cross-tabulations) were assessed using the standard Pearson's χ2 test, with Fisher's exact test applied if the expected cell count in the cross-tabulation was <5. Spearman's rank correlation coefficients were used to quantify correlation between continuous variables. A p-value of <0.05 was deemed statistically significant.
Benchmarking
We attempted to stage each lesion according to the TNM staging system for GC based on the available histological data. After each patient had been staged, their treatment regimen was reviewed to see whether it complied with current international recommendations as presented on the Cancer Association of South Africa website.[2]
Results
Patient demographics
A total of131 patients with GC were identified for the period 2009 - 2014. The patient demographics are set out in Table 1. Of the 131 patients, 48.1% (n=63) were Indian and 45.0% (n=59) black African. The proportions of male and female patients were 55.0% (n=72) and 45.0% (n=59), respectively. The mean age for all patients was 60 years (SD 13.3).

Family history
The results for a family history of cancer, alcohol use and smoking are set out in Table 1. Of the patients, 13.0% (n=17) had a first-degree family history of cancer. Most of these patients were Indian (n=11), and four patients had a family history of GC. Use of alcohol was reported by 30.5% of the patients (n=40), and smoking by 35.1% (n=46).
Stage of GC at first presentation
Table 2 documents the stages of the GCs at first presentation. It was not possible to describe the TNM stage of all the GCs accurately, as insufficient data were collected on the N stage of cancer from the hospital record system. The data available for the T and M stages were tabulated separately. The proportions of patients classified as M0, M1 and Mx were 26.7% (n=35), 46.6% (n=61) and 26.7% (n=35), respectively, and 45.8% (n=60) presented with T4a lesions.

Location of the tumour
The locations of the tumours in the stomach on gastroscopy are set out in Table 2. The most common locations of GC were the body and distal parts of the stomach (antrum and pylorus) (50/128, 39.1% and 47/128, 36.7%, respectively).
Body mass index (BMI) and relation between BMI and location of the tumour
The overall mean BMI was 22.2 kg/m2 (SD 5.2). The relationship between BMI and locations of the tumours is set out in Table 2. This was found not to be significant (p=0.175).
Histological findings
The intestinal histological variants and grades of the tumours are set out in Table 2. All were adenocarcinomas. No histological changes were reported for most (84.0%) of the patients (n=110), although rates of intestinal metaplasia, atrophic gastritis and dysplasia were reported as 6.8% (n=9), 7.6% (n=10) and 1.5% (n=2), respectively. Nearly half of the patients (n=63, 48.1%) had intestinal-type adenocarcinomas, while diffuse-type adenocarcinomas were present in 16.8% (n=22).
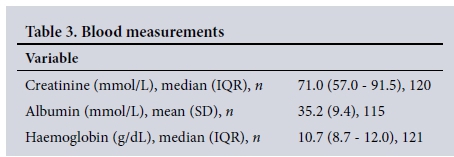
Blood results
The median serum creatinine level was 71.0 mmol/L (IQR 57.0 -91.5), the mean albumin level 35.2 mmol/L (SD 9.4), and the median haemoglobin level 10.8 g/dL (IQR 8.7 - 12.0) (Table 3).
Surgery and chemoradiotherapy
The types of operations and chemoradiotherapy are set out in Table 4. Type of operation and chemoradiotherapy were significantly associated (p<0.001). Most of the patients presented with advanced disease that was not resectable, and 72.5% (n=95) did not undergo surgery with curative intent owing to advanced disease. Only 1 patient (0.1%) had neoadjuvant chemotherapy, 21 (16.0%) had adjuvant chemotherapy, and 17 (13%) had radiotherapy.
Discussion
Our data show some interesting local variations. There appears to be a relatively high burden of GC in the Indian population, as evidenced by the observation that Indian patients made up 48.1% of the GC patients in our series yet only comprise 7.4% of the population of KZN.[4] This observation may simply reflect referral bias, and ongoing audit is required to see whether this apparent racial difference is a real phenomenon. There was a slight male preponderance, with 55.0% males and 45.0% females. There were no significant age differences with regard to gender and ethnicity. World Health Organization statistics show that GC is more common in males than females, and relatively common in Asian countries such Japan and Korea, while it is rare in southern African countries.[5,6]
Comparing our findings with international data revealed some areas of similarity and some differences. Our findings concur with those from the literature that the most common sites of GC were the body of the stomach and the distal stomach,[7] but differ from those of Kubo and Corley,[8] who found that there was a significant relationship between BMI and tumour location. In a systematic review of 14 studies involving 2 488 oesophageal and 2 509 gastric adenocarcinomas, they found a positive association between a high BMI and risk of oesophageal and possibly proximal gastric adenocarcinomas. The mean BMI in our study was 22 kg/m2. Yang et at.,[9]in a meta-analysis of cohort studies with 9 492 GC patients, found that a BMI of >25 kg/m2 was associated with a high risk of GC. Our findings that 48.1% of the patients had the intestinal type and 16.8% the diffuse type of GC do not concur with those of Lauren,[10] who found prevalences of 70% and 30%, respectively, for the diffuse and intestinal types of GC. Our median haemoglobin concentration of 10.8 g/dL is in agreement with Wanebo et at.'s'[11] finding that occult gastrointestinal bleeding with or without iron deficiency anaemia is common in patients with GC.
It is generally understood that human gastric carcinogenesis is a multistep and multifactorial process with sequential histopathological stages. These stages start with Heticobacter pylori infection, then develop into chronic active non-atrophic gastritis, multifocal atrophic gastritis, intestinal metaplasia (complete then incomplete), dysplasia, and finally invasive carcinoma.[12] The International Agency for Research on Cancer'131 recognises infection by H. pytori as a primary cause of gastric adenocarcinoma. However, most patients in our series did not display such histological changes, and H. pytori was associated with GC in only seven patients in this series. This apparent discrepancy between a high incidence of H. pytori infection and a relatively low incidence of GC has been termed the African enigma'.'141 However, more recent reports have tended to downplay the African enigma, and most authors now do not consider that it adequately accounts for the pathogenesis of GC.[14] Current opinion is that the contribution of H. pytori to development of GC in Africa is complex and almost certainly modulated by an interplay of diverse factors such as genetic susceptibility, the environment, dietary intake, and other pathogenic organisms such as Epstein-Barr virus (EBV).[14]
Thirty-one percent of the patients in our study used alcohol. The relationship between alcohol consumption and GC is poorly understood, and some authors suggest that it may actually protect against the development of GC.[15] The situation is slightly clearer with regard to smoking, and a recent meta-analysis has shown that smoking increases the risk of developing GC 1.53-fold.'161 Thirty-five percent of the patients in our series were smokers. Thirteen percent had a first-degree family history of cancer, four having relatives with GC and another four relatives with breast cancer. This familial relationship has been demonstrated in other large reviews of GC.
Almost all studies of GC in the developing world are bedevilled by the issue of referral bias and the lack of robust high-quality cancer registries. Patients with GC may not present to healthcare providers, and those who do may not be captured in the existing data registries. The need for accurate, robust and sustainable cancer registries in the developing world is well recognised, and most established data sources have major limitations. Almost all those who have written on this topic concur that the epidemiology and pathogenesis of GC require ongoing investigation. Any future studies must examine the synergistic role of H. pytori, EBV and dietary antioxidants in the pathogenesis of GC in Africa.[14]
Just over 70% (n=95) of the patients in our study had no operation or staging intervention and/or a gastric bypass procedure (Table 4), because of the advanced stage of the disease. The majority of patients with GC in our environment present with advanced disease. This is a well-described problem with almost all malignancies in SA. Better cancer awareness, screening programmes and improved access to healthcare are needed to try to improve this situation. The lack of good-quality staging is a concern, as treatment regimens are based on TNM staging.[3] Only 1 patient had neoadjuvant chemotherapy, 21 had adjuvant chemotherapy and 17 had radiotherapy. There is much debate about optimal treatment regimens, but most authors now agree that surgery needs to be combined with other modalities of treatment. Macdonald et at.'171found that postoperative chemoradiotherapy should be considered for all patients at high risk of recurrence of adenocarcinoma of the stomach who have undergone curative resection. Forty patients in our study had palliative chemotherapy. In a meta-analysis of three trials comparing chemotherapy and the best supportive care, Wagner et at.'181found that chemotherapy significantly improved survival in comparison with best supportive care. In addition, a perioperative regimen decreased tumour size and stage and significantly improved progression-free and overall survival.'191 It is clear that we lack well-structured treatment regimens for GC in our environment. We need to improve our ability to stage the malignancy and to develop multidisciplinary team (MDT)-based algorithms to ensure that patients with GC receive the most appropriate treatment available.
Study limitations
Owing to the retrospective nature of the study, incomplete data and poor follow-up may limit the information available for analysis. In addition, the study was limited to one centre that may not represent the general population. It was also not possible to obtain information on outcomes (survival rate) because of the referral system and difficulty in obtaining data from primary and secondary hospitals.
Conclusions
GC appears to have a higher incidence in the Indian than in the black African population of KZN. This is in keeping with the current literature on GC in sub-Saharan Africa. Most of the patients with GC in our environment present with advanced disease and do not undergo curative surgery. Our ability to stage these tumours accurately is limited, and this needs to be improved. Appropriate therapy depends on good TNM staging. It is essential that MDT-based algorithms are developed to ensure that therapy is appropriate.
REFERENCES
1. World Health Organization. Globocan 2012. Worldwide cancer incidence. http://globocan.iarc.fr/old/pie_pop.asp?selection=224900&title=World&sex=0&type=0&window=1&join=1&submit=%C2%A0Execute (accessed 10 July 2016). [ Links ]
2. Cancer Association of South Africa (CANSA). http://www.cansa.org.za/files/2016/02/Fact-Sheet-Stomach-Cancer-Feb-2016.pdf (accessed 10 July 2016). [ Links ]
3. National Cancer Institute. Gastric Cancer Treatment (PDQ ) - Health Professional Version. https://www.cancer.gov/types/stomach/hp/stomach-treatment-pdq (accessed 10 July 2016). [ Links ]
4. Wikipedia. Durban. http://en.wikipedia.org/wiki/Durban#Demographics (accessed 3 January 2017). [ Links ]
5. World Health Organization. Globocan 2012. Stomach ASR (W) per 100,000, all ages. http://globocan.iarc.fr/old/bar_sex_site.asp?selection=25050&title=Stomach&statistic=2&populations=6&window=1&grid=1&color1=5&color1e=&color2=4&color2e=&submit=%C2%A0Execute (accessed 3 January 2017). [ Links ]
6. World Health Organization. Globocan 2012. South African Republic. http://globocan.iarc.fr/old/bar_sex_pop.asp?selection=175710&title=South+African+Republic&statistic=1&number=20&window=1&grid=1&color1=5&color1e=&color2=4&color2e=&submit=%C2%A0Execute (accessed 3 January 2017). [ Links ]
7. De Vries AC, Haringsma J, de Vries RA, et al. Biopsy strategies for endoscopic surveillance of pre-malignant gastric lesions. Helicobacter 2010;15(4):259-264. http://dx.doi.org/10.1111/j.1523-5378.2010.00760.x [ Links ]
8. Kubo A, Corley DA. Body mass index and adenocarcinomas of the esophagus or gastric cardia: A systematic review and meta-analysis. Cancer Epidemiol Biomarkers Prev 2006;15(5):872-878. http://dx.doi.org/10.1158/1055-9965.EPI-05-0860 [ Links ]
9. Yang P, Zhou Y, Chen B, et al. Overweight, obesity and gastric cancer risk: Results from a meta-analysis of cohort studies. Eur J Cancer 2009;45(16):2867-2873. http://dx.doi.org/10.1016/j.ejca.2009.04.019 [ Links ]
10. Lauren P. The two histological main types of gastric carcinoma: Diffuse and so-called intestinal-type carcinoma. Acta Pathol Microbiol Scand 1965;64:31-49. [ Links ]
11. Wanebo HJ, Kennedy BJ, Chmiel J, Steele G Jr, Winchester D, Osteen R. Cancer of the stomach: A patient care study by the American College of Surgeons. Ann Surg 1993;218(5):583-592. [ Links ]
12. Correa P. Human gastric carcinogenesis: A multistep and multifactorial process - first American Cancer Society Award Lecture on Cancer Epidemiology and Prevention. Cancer Res 1992;52(24):6735-6740. [ Links ]
13. International Agency for Research on Cancer Working Group on the Evaluation of Carcinogenic Risks to Humans. Schistosomes, Liver Flukes and Helicobacter pylori. Lyon: IARC, 1994. [ Links ]
14. Asombang AW, Kelly P. Gastric cancer in Africa: What do we know about incidence and risk factors? Trans R Soc Trop Med Hyg 2012;106(2):69-74. http://dx.doi.org/10.1016/j.trstmh.2011.11.002 [ Links ]
15. Barstad B, S0rensen TI, Tjønneland A, et al. Intake of wine, beer and spirits, and risk of gastric cancer. Eur J Cancer Prev 2005;14(3):239-243. [ Links ]
16. Ladeiras-Lopes R, Pereira AK, Nogueira A, et al. Smoking and gastric cancer: Systematic review and meta-analysis of cohort studies. Cancer Causes Control 2008;19(7):689-701. http://dx.doi.org/10.1007/s10552-008-9132-y [ Links ]
17. Macdonald JS, Smalley SR, Benedetti J, et al. Chemoradiotherapy after surgery compared with surgery alone for adenocarcinoma of the stomach or gastroesophageal junction. N Engl J Med 2001;345(10):725-730. http://dx.doi.org/10.1056/NEJMoa010187 [ Links ]
18. Wagner AD, Unverzagt S, Grothe W, et al. Chemotherapy for advanced gastric cancer. Cochrane Database Syst Rev 2010, Issue 3. Art. No.: CD004064. http://dx.doi.org/10.1002/14651858.CD004064.;pub3 [ Links ]
19. Cunningham D, Allum WH, Stenning SP, et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med 2006;355(1):11-20. http://dx.doi.org/10.1056/NEJMoa055531 [ Links ]
 Correspondence:
Correspondence:
F Benamro
farajak2000@yahoo.com
Accepted 25 August 2015














