Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
SAMJ: South African Medical Journal
versión On-line ISSN 2078-5135
versión impresa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.105 no.12 Pretoria dic. 2015
http://dx.doi.org/10.7196/samj.2015.v105i12.8585
RESEARCH
Diagnosing childhood pulmonary tuberculosis using a single sputum specimen on Xpert MTB/RIF at point of care
N GousI; L E ScottII; S KhanIII; G ReubensonIII; A CoovadiaIV; W StevensV, VI
IMSc Med, Department of Molecular Medicine and Haematology, School of Pathology, Faculty of Health Sciences, University of the Witwatersrand, Johannesburg, South Africa
IIPhD,Department of Molecular Medicine and Haematology, School of Pathology, Faculty of Health Sciences, University of the Witwatersrand, Johannesburg, South Africa
IIIMB BCh, FCPaed, DCH, DTM&H, Rahima Moosa Mother and Child Hospital and Department of Paediatrics and Child Health, Faculty of Health Sciences, University of the Witwatersrand, Johannesburg, South Africa
IVMB BCh, FCPaed, DCH, Dip HIV Man, Rahima Moosa Mother and Child Hospital and Department of Paediatrics and Child Health, Faculty of Health Sciences, University of the Witwatersrand, Johannesburg, South Africa
VMB BCh, MMed (Haem), FCPath (Haem), Department of Molecular Medicine and Haematology, School of Pathology, Faculty of Health Sciences, University of the Witwatersrand, Johannesburg, South Africa
VIMB BCh, MMed (Haem), FCPath (Haem), National Health Laboratory Service, Johannesburg, South Africa, and National Priority Programme, South Africa
ABSTRACT
BACKGROUND. The GeneXpert MTB/RIF (Cepheid, USA) (Xpert) has proved successful for pulmonary tuberculosis (TB) diagnosis on decontaminated/concentrated induced sputum specimens from children. Capacity to perform induction in many settings is limited.
OBJECTIVE: To Assess: (i) volumes of 'routinely obtained' sputum in a district-level academic hospital; (ii) whether sputum specimens not meeting Xpert-required testing volumes could still be tested; and (iii) performance of Xpert on a single paediatric sputum specimen at point of care (POC
METHODS. Two sputa were collected from paediatric TB suspects (<14 years) at Rahima Moosa Mother and Child Hospital, Johannesburg, South Africa. One specimen was weighed at POC; if the volume was >0.1 mL but <0.5 mL, it was increased to 0.5 mL using saline. On-site Xpert testing (G3 cartridge) was performed by a dedicated laboratory technician. The second specimen was referred for TB smear microscopy and culture as per standard of care (SOC).
RESULTS. A total of 484 patients presumed to have TB (median age 24 months) were eligible for this study, performed between June 2011 and May 2012. Xpert could not be used on 4.1% of specimens because of volumes <0.1 mL, and 62.8% required addition of saline prior to Xpert testing. Xpert generated a 2.2% error and 3.7% invalid rate, compared with the SOC that rejected 2.3% because of insufficient volume and 2.3% that were contaminated. The diagnostic performance compared with culture was 62.5% (95% confidence interval (CI) 24.7 - 91) and 99.1% (95% CI 97.4 - 99.8) sensitivity and specificity, respectively, for Xpert (n=345) and 33.3% (7.9 - 69.9) and 99.5% (98.1 - 99.9) sensitivity and specificity, respectively, for smear microscopy (n=374).
CONCLUSIONS. Up to 67% of 'routinely obtained' sputum specimens from children (<14 years) are below the required volume for Xpert testing but can be 'topped up' with saline. Xpert MTB/RIF performed better than microscopy and generated clinically relevant, timeous results, but sensitivity did not reach the same levels as culture in children.
Over half a million children are diagnosed with tuberculosis (TB) every year. The majority of these infections occur in 22 high-burden countries[1] and account for 6% of the global TB burden.[2,3] The World Health Organization (WHO) reported a staggering 74 000 deaths in HIV-negative children <15 years of age in 2012 alone, making TB one of the top ten leading causes of death in children.[4]
Control programmes are hampered by the increasing spread of multidrug-resistant TB (MDR-TB); globally 40 000 new cases of MDR-TB are reported annually in paediatric populations.[3] In South Africa (SA), children aged <14 years are estimated to account for approximately 15 - 20% of the total TB disease burden.[5,6]
These global statistics may grossly underestimate the TB burden, [7] as microbiological confirmation of TB is challenging in children. Typically, the type of specimen obtained for testing depends on the age of the child and the clinical presentation.[8] Specimens other than sputum most frequently include nasopharyngeal aspirates (NPAs), gastric aspirates, induced sputum, throat swabs, fine-needle lymph node aspiration, bone marrow, urine, stool and aseptic fluids (ascitic, pleural and cerebrospinal fluid). With the exception of the bone marrow and lymph node, owing to the paucibacillary nature of the specimens, multiple sampling over several days is required.[8]
Additional challenges are often the poor quality[9] and low quantity of specimen yield that hamper bacteriological confirmation of disease.[10] It is for this reason that no universal diagnostic algorithm for TB in children exists, diagnosis relying on a combination of clinical signs and nonspecific tests.'71 Clearly, a stronger emphasis needs to be placed on advancing the research and development of more effective diagnostic strategies for childhood TB detection.
Accurate and sensitive molecular tests such as the GeneXpert MTB/RIF assay (Cepheid, USA) (Xpert) are proving that reliable and rapid diagnosis of TB can be achieved. The Xpert test was recently approved by the US Food and Drug Administration as the first moderate complexity test to detect both Mycobacterium tuberculosis (MTB) and resistance to rifampicin simultaneously for pulmonary TB.[11] In SA, the National Department of Health (NDoH) and
National Health Laboratory Service (NHLS) have implemented the Xpert in 207 smear microscopy centres across the country. Data from the programme reflect the Xpert's sensitivity over microscopy. The average national TB positivity rate among presumptive pulmonary adult TB cases prior to Xpert was 8%, and this has since increased to 11% in the fourth year of Xpert use.[12]
The updated Cochrane review on Xpert's performance in adults showed a pooled sensitivity of 98% for smear-positive pulmonary TB and 79% for smear-negative TB.[12] In the paediatric population, studies are limited and most have focused on induced sputa,[9,13-16] where sensitivities ranging from 33.3%[14] to approximately 70%[15,17] in smear-negative, culture-positive paediatric patients are reported. Increased sensitivities of up to 75.9% could be achieved if two induced sputa were tested.[14] Further studies are now concentrating on alternative specimen types such as NPAs,[16] gastric lavage aspirates,[18] bronchoalveolar lavage[19] and stool.[20,21]
In all Xpert studies on paediatric populations, specimens underwent laboratory decontamination and concentration prior to Xpert testing. The aim of this study was threefold: ( i) to assess the volume of 'routinely obtained' sputum (i.e. no induced sputum facility available) from children <14 years of age at a district-level academic hospital; (ii) to determine whether sputum specimens not meeting the Xpert minimum required testing volume could be manipulated before testing; and (iii) to determine the feasibility of performing Xpert MTB/RIF on raw paediatric sputum specimens by a dedicated staff member, at the point of care (POC).
Methods
Patient recruitment
Paediatric patients accessing care at the Rahima Moosa Mother and Child Hospital (RMMCH), Johannesburg, SA, were eligible for inclusion if their treating clinician suspected TB. RMMCH is a district-level academic hospital that provides paediatric care to the population of the west of Johannesburg. The study received approval from the University of the Witwatersrand Human Ethics Committee (Protocol number: M110496).
Study procedures
At the time of the study, the routine procedure at RMMCH was to collect two sputum specimens; one specimen was sent to the NHLS at Helen Joseph Hospital for TB smear microscopy and the second specimen was sent to the NHLS Braamfontein laboratory for liquid culture (MGIT) and drug susceptibility testing (DST) when necessary. A GeneXpert instrument was placed on site in an existing POC laboratory designated for sample processing (specimen sorting, centrifugation, rapid HIV testing, data capture) at RMMCH. This allowed specimens to be tested on site.
Specimen collection
A physiotherapist collected two sputum specimens concurrently, using clinic standard of care methodology, in standard sputum collection containers from children <14 years of age who were suspected of having TB. Respiratory samples were obtained following administration of chest physiotherapy (at the discretion of the individual physiotherapist). One of these specimens was randomly selected for on-site Xpert testing (G3 version cartridge) by a trained laboratory technician (to replace the first smear) and the second specimen was sent to the NHLS Braamfontein TB laboratory as per routine, for decontamination and concentration (NALC-NaOH), smear microscopy, MGIT culture and DST, when indicated.
Xpert MTB/RIF testing
Specimens were weighed (in their containers) on a bench-top precision balance (Sigma-Aldrich, USA) to estimate the volume of the specimen. If any specimen was >0.1 mL but less than the minimum required volume of 0.5 mL (estimated at 0.5 g), sterile saline was added using a standard Finnpipette (Thermo Fisher Scient ific, USA) and sterile pipette tips to increase the volume to at least 0.5 mL. Testing of specimens on the Xpert MTB/RIF assay was performed on the same day as specimen collection. However, if any specimen could not be tested the same day as a result of late specimen receipt, it was stored in a 4oC fridge overnight, followed by testing the next day as per manufacturer's instructions. All specimens were processed as per standard Xpert MTB/RIF protocol by addition of Sample Reagent (SR) buffer in a 3:1 or 2:1 ratio to take the final volume to 2 mL (Cepheid, USA). The on-site result obtained from the Xpert MTB/RIF assay was reported to the treating clinicians.
Statistical analysis
Specimen volumes for Xpert testing were described. Non-parametric tests were used to determine associations between age and volume, Xpert results and volume, and age and pre-processing, at 95% confidence interval (CI). The sensitivity and specificity and positive and negative predictive values (PPV and NPV) were calculated for the Xpert MTB/RIF assay and smear microscopy with a 95% CI using MGIT culture as the gold standard.
Results
Data summary
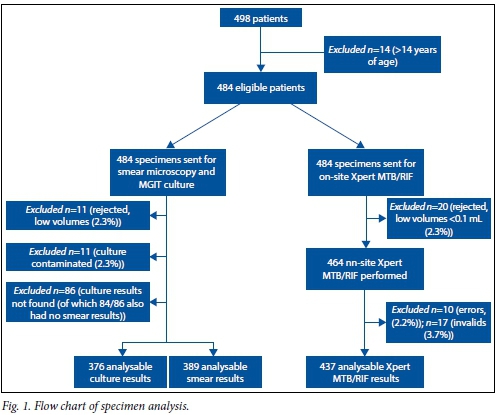
A flow diagram illustrates the processing of patient samples (Fig. 1). A total of 484 eligible children (median age 24 months) had samples processed between June 2011 and May 2012 (interquartile range (IQR) 12 -60). Two sputum specimens were collected by physiotherapy from 484 patients (none required nebulisation); one sample was sent for on-site Xpert testing and the other was sent for routine laboratory testing. The laboratory rejection rate due to insufficient volume for smear and culture processing was 2.3% (11/484). Culture reported a 2.3% (11/473) contamination rate, which was excluded from the quantitative analysis (Fig. 1). After study commencement, culture and smear results for 86 patients could not be retrieved from the laboratory information system and were therefore excluded from final quantitative analysis. This coincided with the roll-out of the Xpert in SAs National Programme, which commenced in March 2011, whereby the diagnostic algorithm for initial TB diagnosis changed from collection of two sputum specimens to one.
Diagnostic performance analysis
Sputum specimen volumes received from children for Xpert testing are shown in Table 1. A total of 20/484 specimens (4.1%) had sputum volumes of <0.1 mL and could not be tested. Of the 464 Xpert tests performed on site, overall error and invalid rates of 2.2% (10/464) (1 volume error and 9 code 5011 errors) and 3.7% (17/464), respectively, were reported (Fig. 1).
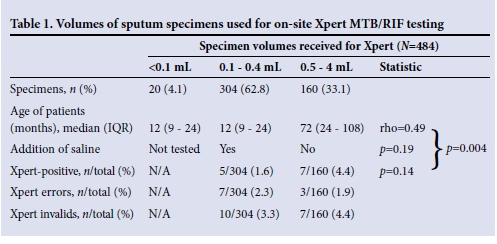
Using Spearman's correlation coefficient (owing to skewness of the data), an overall weak correlation was observed between age and volume of specimens received (rho=0.49). No association was observed between the volume of specimen received and the Xpert result (p=0.14). However, an association was observed between age and Xpert results (p=0.004), with more TB-positives detected in the older age group (median age 132 months).
While prior specimen manipulation by addition of sterile saline before Xpert testing did not affect the error/invalid rate (p=0.19) reported on the Xpert, more positive results were generated from specimens that did not require the addition of saline; however, this did not reach significance (p=0.18).
Table 2 further categorises the specimens by age and volume and details the performance by Xpert compared with liquid culture as the reference. Sputum volume clearly increased with age and it appears that, on average, specimens from children aged >25 months would require less addition of saline prior to Xpert testing. The minimum age group in which MTB was detected by Xpert was 13 months, with total reported MTB positivity of 2.7% (12/437).
Smear microscopy also only detected MTB from 13 months onwards, reporting 7/389 positive specimens (1.8%) (Table 3). MGIT culture initially reported a positivity rate of 4.0% (15/376); however, after follow-up DST in all culture-positive specimens using the Hain MTBDrplus version 1 assay (Hain LifeScience, GmbH, Germany), only 9/15 were confirmed as MTB complex (2.4%) (Table 3). The other six specimens were probably non-tuberculosis mycobacteria, but were not definitively identified. Culture remained most sensitive in detecting MTB in the lower age group, detecting 1 positive in each age group <24 months and 6 positives in the >60-month category.
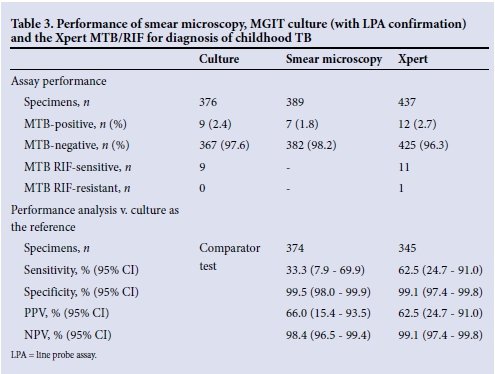
Performance data for MGIT culture, smear microscopy and the Xpert MTB/RIF are shown in Table 3.
Smear microscopy was able to identify 3 of the 9 culture-confirmed MTB-positive cases but additionally reported 2 culture-negative (and Xpert-negative) specimens as positive. Compared with culture on 374 specimens, smear generated a sensitivity and specificity of 33.3% (95% CI 7.9 - 69.9) and 99.5% (95% CI 98.1 - 99.9), respectively. Relative to smear, Xpert detected 3 additional MTBpositive cases.
Of the 12 Xpert-positive cases reported, corresponding culture results could not be found in 4 cases. Xpert detected 5/9 culture-confirmed MTB-positive specimens and an additional 3 cases. Analysis of 345 patients with both Xpert and culture results demonstrated an Xpert sensitivity of 62.5% (95% CI 24.7 - 91) and high specificity of 99%. Overall the Xpert was able to detect 66.7% of smear-positive, culture-positive cases (2/3) and 50% of smear-negative, culture-positive cases (3/6).
The Xpert MTB/RIF reported only a single specimen (8.3%, 1/12) with rifampicin resistance, but the MTBDRplus v1 reported rifampicin and isoniazid susceptibility in this specimen. Two additional specimens were reported mono-isoniazid-resistant by MTBDrplus v1.
Discussion
Moving the Xpert MTB/RIF assay to the POC has been shown to be feasible, to save costs for patients[22] and to have advantages over centralised testing, such as same-day treatment and shorter time to treatment initiation in adult TB suspects.[22-24] Use of the Xpert for paediatric TB diagnosis at POC may introduce additional challenges. Although obtaining an induced sputum specimen is a simple and well-tolerated procedure,[14] it needs to be conducted by a trained healthcare worker. The capacity to perform such induction is limited [16] and its uptake has been slow. In SA, many healthcare facilities are not able to obtain respiratory specimens (gastric aspirates or sputum) from children and many centres/ clinics lack sputum induction facilities or trained healthcare workers to perform the procedure. Even when respiratory samples are obtained, volumes from children may not be sufficient to meet the required minimum testing volume needed for the Xpert assay.
The setting where this study was performed already had an on-site POC laboratory with a trained technician who managed paediatric HIV testing and collection of dried blood spots, making it feasible for inclusion of the GeneXpert technology, especially since additional specimen types such as stool (under development by Cepheid) and gastric or NPAs may in future be tested by Xpert at POC.
Data from our study indicate that a large proportion (67%) of 'routinely obtained' sputum specimens received from children <14 years of age are below the minimum required testing volume for Xpert and could either not be processed (4.1%) or required the addition of saline prior to Xpert testing (62.8%). Addition of saline required weighing the specimen container as an estimate of volume and then adding sterile saline using a pipette to make up the volume to approximately 0.5 mL. This did not affect the Xpert result in terms of error/invalid rates, but required extra processing time and technical training for the person performing the testing as well as a supply of sterile saline. Volume has previously also been shown to have minimal impact on Xpert performance.[25] Patient age was associated with Xpert result reporting, with more TB positives detected in older children. This was unrelated to volume of the specimen, but probably as a result of a higher bacillary load in these patients. However, because of small sample numbers, this cannot be definitively proven.
The minimum volume of specimen required for Xpert testing resulted in twice as many specimens not being able to be tested by Xpert compared with those sent to the laboratory for smear microscopy and culture. Xpert also reported an overall error plus invalid rate of 5.9%, higher than the contamination rate of 2.3% reported with culture. The majority of errors were code 5011, a commonly occurring error on the older version G3 cartridges; this has since been rectified with the new G4 cartridge.[26]
The positivity rate for MTB in this study population was lower than previously reported from Cape Town, SA,[14] as expected. Similarly to previously reported data,[14,15] use of Xpert MTB/RIF on a single sputum specimen in our study provided superior detection over smear microscopy. The Xpert MTB/RIF was able to detect 55% of culture-confirmed MTB-positive cases and an additional three cases that were negative on culture. These three additional cases may have been due to the Xpert detecting non-viable organisms. The moderate sensitivity of Xpert compared with culture in our study may be due to the fact that specimen volumes were too low for testing; of the three Xpert false negatives, two were reported on specimens <0.5 mL that had needed addition of saline prior to Xpert testing. However, the wide CI did not allow for an accurate estimation of sensitivity and would need a larger study cohort. One benefit of this study was that an additional 1.6% of TB positives could be detected by Xpert by 'topping up' low-volume specimens instead of discarding them. This means that an MTB-positive result could be acted on more timeously than waiting for a culture result.
The costing of Xpert MTB/RIF for diagnosis of smear-negative TB in adults visiting a primary healthcare setting in SA has been estimated at USD3.28 less than the cost per valid smear microscopy plus culture result.[27] This study took into account a low error rate (of 1%), electricity, water and space, medical waste disposal, N-95 masks, sputum collection bottles and surface disinfectant.[27] If Xpert is instituted at POC for paediatric/childhood TB diagnosis, it will require increased capacity scale-up and training (good clinical laboratory practice), as well as additional equipment (bench-top balance, pipettes) and consumables (pipette tips, sterile saline). Assessment of each individual clinic's infrastructure needs, resources and staff requirements, power supply, connectivity and infection control will also need to be taken into account.
An advantage of performing on-site Xpert testing specifically for children is the rapid turnaround of results. However, on-site Xpert testing on inadequate specimen volumes may increase the chances of obtaining errors and invalid results that cannot be repeated on the same sample. In our study, Xpert MTB/RIF out-performed TB microscopy but did not demonstrate acceptable sensitivity in this real-world setting and cannot currently replace TB culture in the diagnosis of paediatric/ childhood TB. Alternative specimen types, which are easier to obtain and not subject to volume limitations, such as stool, may provide a better diagnostic option.
Acknowledgements. We thank Braimoh Bello, Centre for Statistical Analysis and Research (CEASAR), for assistance with statistical analysis. This project was funded in part by the President's Emergency Plan for AIDS Relief (PEPFAR) through the US Centers for Disease Control and Prevention, under the terms of grant 3U2GPS0001328-04. The Grand Challenges Canada grant 0007-02-01-01-01 (to Wendy Stevens) also provided support.
References
1. Stop TB Partnership Childhood TB Subgroup. Guidance for National Tuberculosis Programmes on the management of tuberculosis in children. Chapter 1: Introduction and diagnosis of tuberculosis in children. Int J Tuberc Lung Dis 2006;10(10):1091-1097. [ Links ]
2. World Health Organization. Roadmap for Childhood Tuberculosis: Towards Zero Deaths. Geneva: WHO, 2013. http://www.who.int/tb/challenges/children/en/ (accessed 7 January 2014). [ Links ]
3. World Health Organization. Global Tuberculosis Report 2013. Geneva: WHO, 2013. http://www.who.int/tb/publications/global_report/en/ (accessed 13 January 2014). [ Links ]
4. World Health Organization. Childhood Tuberculosis. Geneva: WHO, 2013. http://www.who.int/tb/challenges/children/en/ (accessed 7 January 2014). [ Links ]
5. Yezingane Network. Brief: TB & the Invisible Children in South Africa. 2013. http://www.sahivsoc.org/Topics?page=1¤tFilter=Tuberculosis (accessed 1 November 2013). [ Links ]
6. Zar HJ, Eley B, Nicol MP, Figaji A, Hawkridge A. Advances in childhood tuberculosis - contributions from the University of Cape Town. S Afr Med J 2012;102(6):518-521. [ Links ]
7. Esposito S, Tagliabue C, Bosis S. Tuberculosis in children. Mediterr J Hematol Infect Dis 2013;5(1):e2013064. [http://dx.doi.org/10.4084/mjhid.2013.064] [ Links ]
8. Cuevas LE, Petrucci R, Swaminathan S. Tuberculosis diagnostics for children in high-burden countries: What is available and what is needed? Paediatr Int Child Health 2012;32(Suppl 2):S30-S37. [http://dx.doi.org/10.1179/2046904712Z.000000000761 [ Links ]
9. Nhu NT, Ha DT, Anh ND, et al. Evaluation of Xpert MTB/RIF and MODS assay for the diagnosis of pediatric tuberculosis. BMC Infect Dis 2013;13:31. [http://dx.doi.org/10.1186/1471-2334-13-31] [ Links ]
10. Lawn SD, Brooks SV, Kranzer K, et al. Screening for HIV-associated tuberculosis and rifampicin resistance before antiretroviral therapy using the Xpert MTB/RIF assay: A prospective study. PLoS Med 2011;8(7):e1001067. [http://dx.doi.org/10.1371/journal.pmed.1001067] [ Links ]
11. Food and Drug Administration. FDA news release: FDA permits marketing of first U.S. test labeled for simultaneous detection of tuberculosis bacteria and resistance to the antibiotic rifampin. FDA, 2013. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm362602.htm. (accessed 1 November 2013). [ Links ]
12. National Health Laboratory Service. GeneXpert MTB/RIF Progress Report to NDoH. South Africa: NHLS, February 2015. http://www.nhls.ac.za/assets/files/GeneXpert%20Progress%20Report%20February%202015%20Final.pdf (accessed 9 November 2015). [ Links ]
13. Steingart KR, Sohn H, Schiller I, et al. Xpert MTB/RIF assay for pulmonary tuberculosis and rifampicin resistance in adults. Cochrane Database Syst Rev 2014, Issue 1. Art. No.: CD009593. [http://dx.doi.org/10.1002/14651858.cd009593.pub3] [ Links ]
14. Nicol MP, Workman L, Isaacs W, et al. Accuracy of the Xpert MTB/RIF test for the diagnosis of pulmonary tuberculosis in children admitted to hospital in Cape Town, South Africa: A descriptive study. Lancet Infect Dis 2011;11(11):819-824. [http://dx.doi.org/10.1016/S1473-3099(11)70167-0] [ Links ]
15. Sekadde MP, Wobudeya E, Joloba ML, et al. Evaluation of the Xpert MTB/RIF test for the diagnosis of childhood pulmonary tuberculosis in Uganda: A cross-sectional diagnostic study. BMC Infect Dis 2013;13:133. [http://dx.doi.org/10.1186/1471-2334-13-133] [ Links ]
16. Zar HJ, Workman L, Isaacs W, et al. Rapid molecular diagnosis of pulmonary tuberculosis in children using nasopharyngeal specimens. Clin Infect Dis 2012;55(8):1088-1095. [http://dx.doi.org/10.1093/cid/cis598] [ Links ]
17. Rachow A, Clowes P, Saathoff E, et al. Increased and expedited case detection by Xpert MTB/RIF assay in childhood tuberculosis: A prospective cohort study. Clin Infect Dis 2012;54(10):1388-1396. [http://dx.doi.org/10.1093/cid/cis190] [ Links ]
18. Bates M, O'Grady J, Maeurer M, et al. Assessment of the Xpert MTB/RIF assay for diagnosis of tuberculosis with gastric lavage aspirates in children in sub-Saharan Africa: A prospective descriptive study. Lancet Infect Dis 2013;13(1):36-42. [http://dx.doi.org/10.1016/S1473-3099(12)70245-1] [ Links ]
19. Walters E, Goussard P, Bosch C, Hesseling AC, Gie RP. GeneXpert MTB/RIF on bronchoalveolar lavage samples in children with suspected complicated intrathoracic tuberculosis: A pilot study. Pediatr Pulmonol 2014;49(11):1133-1137. [http://dx.doi.org/10.1002/ppul.229701 [ Links ]
20. Nicol MP, Spiers K, Workman L, et al. Xpert MTB/RIF testing of stool samples for the diagnosis of pulmonary tuberculosis in children. Clin Infect Dis 2013;57(3):e18-e21. [http://dx.doi.org/10.1093/cid/cit230] [ Links ]
21. Walters E, Gie RP, Hesseling AC, Friedrich SO, Diacon AH, Gie RP. Rapid diagnosis of pediatric intrathoracic tuberculosis from stool samples using the Xpert MTB/RIF Assay: A pilot study. Pediatr Infect Dis J 2012;31(12):1316. [http://dx.doi.org/10.1097/INF.0b013e318266c21c] [ Links ]
22. Van Rie A, Page-Shipp L, Hanrahan CF, et al. Point-of-care Xpert(R) MTB/RIF for smear-negative tuberculosis suspects at a primary care clinic in South Africa. Int J Tuberc Lung Dis 2013;17(3):368- 372. [http://dx.doi.org/10.5588/ijtld.12.0392] [ Links ]
23. Theron G, Zijenah L, Chanda D, et al. Feasibility, accuracy, and clinical effect of point-of-care Xpert MTB/RIF testing for tuberculosis in primary-care settings in Africa: A multicentre, randomised, controlled trial. Lancet 2014;383(9915):424-435. [http://dx.doi.org/ 10.1016/S0140-6736(13)62073-5] [ Links ]
24. Hanrahan CF, Selibas K, Deery CB, et al. Time to treatment and patient outcomes among TB suspects screened by a single point-of-care Xpert MTB/RIF at a primary care clinic in Johannesburg, South Africa. PLoS One 2013;8(6):e65421. [http://dx.doi.org/10.1371/journal.pone.0065421] [ Links ]
25. Theron G, Peter J, van Zyl-Smit R, et al. Evaluation of the Xpert MTB/RIF assay for the diagnosis of pulmonary tuberculosis in a high HIV prevalence setting. Am J Respir Crit Care Med 2011;184(1):132-140. [http://dx.doi.org/10.1164/rccm.201101-0056OC] [ Links ]
26. FIND. Report: Performance of the Xpert MTB/RIF version G4 assay. http://www.stoptb.org/wg/gli/assets/documents/map/findg4cartridge.pdf (accessed 19 June 2014). [ Links ]
27. Van Rie A. The challenge of diagnosing TB in people with HIV: Is stool the new tool? Int J Tuberc Lung Dis 2013;17(8):995. [http://dx.doi.org/10.5588/ijtld.13.0415] [ Links ]
 Correspondence:
Correspondence:
N Gous
natasha.gous@gmail.com
Accepted 12 October 2015.














