Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.105 n.9 Pretoria Sep. 2015
http://dx.doi.org/10.7196/SAMJNEW.7908
RESEARCH
Pharmacological treatment of painful HIV-associated sensory neuropathy
P PillayI; A L WadleyII; C L CherryIII, IV, V; A S KarstaedtVI; P R KamermanII
IMSc. Brain Function Research Group, School of Physiology, Faculty of Health Sciences, University of the Witwatersrand, Johannesburg, South Africa
IIPhD.Brain Function Research Group, School of Physiology, Faculty of Health Sciences, University of the Witwatersrand, Johannesburg, South Africa
IIIMBBS, PhD. Brain Function Research Group, School of Physiology, Faculty of Health Sciences, University of the Witwatersrand, Johannesburg, South Africa
IVMBBS, PhD. International Clinical Research Laboratory, Centre for Biomedical Research, Burnet Institute, Melbourne, Victoria, Australia
VMBBS, PhD. Infectious Diseases Unit, Alfred Hospital and Faculty of Medicine, Nursing and Health Sciences, Monash University, Melbourne, Victoria, Australia
VIMB BCh. Department of Medicine, Chris Hani Baragwanath Hospital, Johannesburg, South Africa
ABSTRACT
BACKGROUND: HIV-associated sensory neuropathy (HIV-SN) is a common and frequently painful complication of HIV infection and its treatment. However, few data exist describing the frequency, type and dosage of pain medications patients are receiving in the clinic setting to manage the painful symptoms of HIV-SN
OBJECTIVE: To report on analgesic prescription for painful HIV-SN and factors influencing that prescription in adults on combination antiretroviral therapy
METHODS: Using validated case ascertainment criteria to identify patients with painful HIV-SN, we recruited 130 HIV-positive patients with painful HIV-SN at Chris Hani Baragwanath Hospital, Johannesburg, South Africa. Demographic and clinical data (including current analgesic use) were collected on direct questioning of the patients and review of the medical files
RESULTS: We found significant associations, of moderate effect size, between higher pain intensity and lower CD4 T-cell counts with prescription of analgesic therapy. Factors previously identified as predicting analgesic treatment in HIV-positive individuals (age, gender, level of education) were not associated with analgesic use here. Consistent with national guidelines, amitriptyline was the most commonly used agent, either alone or in combination therapy. Importantly, we also found that despite the relatively high analgesic treatment rate in this setting, the majority of patients described their current level of HIV-SN pain as moderate or severe
CONCLUSION: Our findings highlight the urgent need for both better analgesic options for HIV-SN pain treatment and ongoing training and support of clinicians managing this common and debilitating condition
HIV-associated sensory neuropathy (HIV-SN) is a common and frequently painful complication of HIV infection and its treatment, and is likely to remain prevalent for the foreseeable future.[1-3] Painful HIV-SN is associated with substantially reduced health-related quality of life,[2] but evidence-based analgesic options are lacking.[4] Several national and international agencies[5-10] have recommended the tricyclic antidepressant amitriptyline for HIV-SN pain, despite evidence that this is no better than placebo.[11,13]
Despite the high prevalence[1,2] and considerable impact of HIV-SN, and the lack of proven effective analgesics, few data exist describing the frequency, type and dosage of pain medication patients are receiving. We are aware of only one retrospective case review, with low-quality case ascertainment criteria, that attempted to describe the treatment of HIV-SN pain in a clinical setting.[14] This showed that only 7% of a cohort of Malawians who may have had painful HIV-SN were prescribed amitriptyline.
We used validated case ascertainment criteria to identify patients with painful HIV-SN, and provide the first report on analgesic prescription for HIV-SN among South African (SA) adults on combination antiretroviral therapy (cART).
Methods
Consecutive consenting HIV-positive adults on cART were screened for HIV-SN at the Greenhouse Pharmacy, which services patients attending the Ntabiseng Clinic at Chris Hani Baragwanath Hospital, Johannesburg, SA, between June 2012 and January 2014. Researchers attended all pharmacy days servicing ambulatory adults infected with HIV. All individuals on stable cART (any regimen) for longer than 6 months and who had HIV-SN (painful or non-painful) were included in the study. The study was approved by the Human Research Ethics Committee (Medical) of the University of the Witwatersrand, Johannesburg. Informed consent was obtained from all participants.
Patients were screened for HIV-SN using the AIDS Clinical Trials Group (ACTG) Brief Neuropathy Screening Tool (BPNS).[15] HIV-SN was diagnosed on the basis of at least one bilateral sign (vibration sense <10 seconds using a 128 Hz tuning fork in the great toe or absent ankle reflexes) and at least one symptom (pain, paraesthesiae or numbness) in both feet. Symptom severity was rated on an 11-point numerical pain rating scale (NRS) ranging from 0 (not present) to 10 (most severe imaginable). Demographic and clinical data (including current prescribed and self-medicated analgesic use) were collected on direct questioning of the patient and review of the medical file.
Descriptive statistics are presented as means (standard deviation (SD)) for parametric data, medians (interquartile range (IQR)) for non-parametric data, and percentages for frequency data. Univariate analyses comparing patients receiving and not receiving analgesic therapy included Fisher's exact test (gender, years of formal education, number of pain sites, ART regimen, current tuberculosis (TB) infection, sensory symptoms other than pain), the Wilcoxon rank sum test (pain intensity, CD4 T-cell count, years of formal education), and Student's t-test (age). Variables with p<0.1 on univariate analysis were included in multivariate logistic regression analysis. For those patients who were receiving analgesic therapy, the Cochrane-Armitage test for trend was used to assess whether there was an association between the dose of analgesic prescribed and pain intensity. Pain was categorised as mild if rated 1 - 3, moderate if rated 4 - 7, and severe if rated 8 - 10, as set out in the ACTG BPNS.[15]
Results
One hundred and thirty black SA patients with painful HIV-SN were recruited. Consistent with the population seen in clinics in Johannesburg,[16] the subjects were predominantly middle-aged women (72% female, mean (SD) age 45.7 (9.4) years) with well-preserved CD4 T-cell counts. Current HIV-SN pain was rated as severe by two-thirds of the subjects, with an overall median pain intensity of 7.5 (IQR 2 - 10) on the numerical rating scale. Details of the clinical and demographic features of the cohort are shown in Table 1.
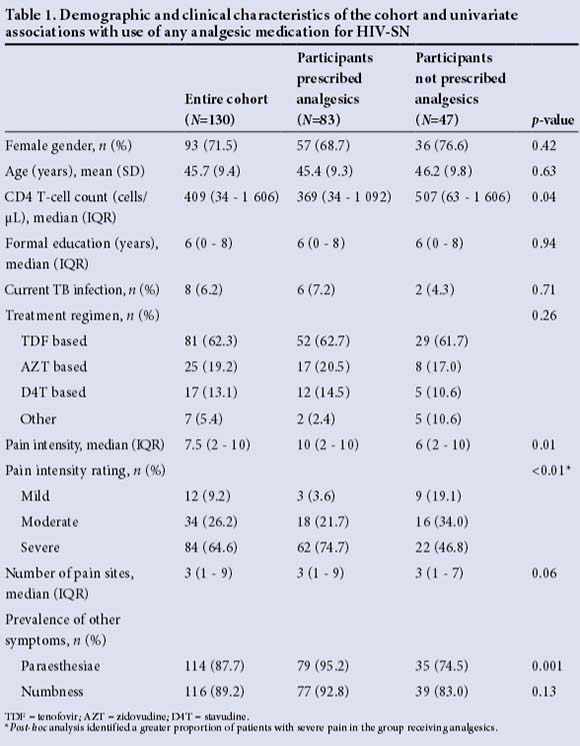
Significant associations were observed between the use of analgesic medication and current pain severity (individuals with greater pain were more likely to be using analgesics: median difference in pain intensity rating on the 11-point NRS (95% CI of difference in medians) 4 (0 - 5)), and between the use of analgesic medication and the latest CD4 T-cell count (individuals with lower CD4 T-cell counts were more likely to be using analgesics: median difference in CD4 T-cell count (95% CI of difference in medians) -138 (-315 -21) cells/μL). Multivariate logistic regression models incorporating combinations of pain intensity, CD4 T-cell count and number of pain sites (p<0.1 on univariate analysis) were not significantly better than a model only incorporating pain intensity as an independent variable (analysis of deviance p>0.05). CD4 T-cell count was not associated with pain intensity (Spearman's rho -0.08, p=0.35).
Pharmacological treatments prescribed to the patients are shown in Table 2. Overall, 64% of patients were using at least one analgesic medication, including three-quarters of those with severe pain, half of those with moderate pain and one-quarter of those who described their current pain as mild. Consistent with national guidelines, amitriptyline was the most commonly used agent, either alone or in combination therapy.

In patients receiving analgesic therapy, there was no association between drug dosage prescribed and pain intensity for amitriptyline (χ2=1.88, p=0.18), or for paracetamol and codeine (χ2=0.01, p=0.94).
During the patient interview, we also collected information on over-the-counter medications participants were taking. The majority of patients were only taking those medications provided at the pharmacy, as they could not afford to buy their own medications. Of the few patients who did so, none purchased pain medications.
Discussion
We provide the first report of analgesic use for HIV-SN pain in which HIV-SN was defined using standard, objective criteria. Ninety per cent of patients in our sample had moderate or severe pain, despite almost two-thirds being on some form of analgesic therapy. In most cases the tricyclic antidepressant amitriptyline was included in the analgesic regimen, consistent with national guidelines.[6,7] We found significant associations, of moderate size, between pain intensity and analgesic therapy as well as between CD4 T-cell count and analgesic therapy; subjects with more severe pain and those with lower CD4 T-cell counts were more likely to be receiving treatment. However, greater pain was not associated with analgesic dose used, and lower CD4 T-cell counts were not associated with pain intensity. Factors previously identified as predicting analgesic treatment in HIV-positive individuals (age, gender, level of education) were not associated with analgesic use in this study.[17]
Our finding that 36% of patients with HIV-SN pain were not receiving any analgesic therapy is worrying. Nevertheless, analgesic use in this cohort was high compared with the very low rates we and others have reported in similar ambulatory HIV-positive populations with pain of similar intensity, but of any origin.[18-21] Clinicians may be more aware of HIV-SN as a common cause of pain, and therefore be more likely to recognise and treat HIV-SN pain than other pain conditions. Although more than a third of patients with HIV-SN pain in this cohort were not receiving analgesics, we cannot exclude the possibility that they may previously have used agents such as amitriptyline and ceased these owing to poor tolerability or efficacy. Indeed, the high levels of pain reported by many patients using amitriptyline in this cohort highlight the need for more effective treatments for HIV-SN pain,[12] or greater awareness of optimal dosing of amitriptyline for the management of neuropathic pain.
Our finding that use of some form of analgesic agent increases with pain severity may indicate that physicians believe that regular analgesics are required only for HIV-SN pain of greater severity, despite evidence that health-related quality of life is reduced across all levels of pain in this condition.[2] However, it is also possible that patients do not report mild pain. In addition, our finding that analgesic use increases with lower initial CD4 T-cell count may indicate that patients perceive sensory neuropathy to be a complication of ART, and more of those patients with severe pain and severe paraesthesiae (median 8, IQR 2 - 10) may therefore not take their ART reliably, possibly thinking that they are being harmed by these drugs, and therefore have a poor CD4 T-cell count.
Amitriptyline was prescribed to most patients receiving analgesia in this cohort. While there is no evidence that amitriptyline is more effective than placebo for painful HIV-SN,[11-13] its proven efficacy in other types of neuropathic pain[22] prompted an expert panel to recommend it as a first-line option for HIV-SN pain in SA.[7] These patients were therefore receiving a recommended treatment, albeit at a relatively low dose (25 - 50 mg/d).[7] Amitriptyline was commonly used in combination with codeine and paracetamol. Neither of these drugs have proven efficacy in neuropathic pain,[22] but their use may reflect concurrent sources of pain (common in HIV) that may be responsive to standard opioid and non-opioid analgesics and non-steroidal anti-inflammatory agents.[23] Use only of agents with no evidence of efficacy for neuropathic pain by 16% of our cohort may reflect poor knowledge of neuropathic pain management on the part of clinicians, inadequate efficacy and/or tolerability of available neuropathic pain treatments, or treatment of concurrent nociceptive pains while the neuropathic pain went unrecognised. This finding highlights the need for both better analgesic options for HIV-SN pain treatment and ongoing training and support of clinicians managing this difficult condition.
Six patients (7%) were prescribed amitriptyline together with the anticonvulsant carbamazepine, largely owing to the preference of a single clinician. Carbamazepine decreases levels of amitriptyline and paracetamol by affecting hepatic/intestinal enzyme CYP3A4 metabolism.[24] There is also potential for increased sedation in patients taking codeine and amitriptyline concurrently, but at the doses of codeine prescribed, this interaction was probably not significant.[25]
Study limitations
Our study has a number of limitations. The modest sample size limits our ability to understand factors associated with use of less common analgesic choices. The cross-sectional nature of this work means that we are unable to comment on patients' previous level of HIV-SN pain or prior use of analgesics for this condition. We therefore cannot comment on the level of pain relief achieved by the treatments used. Patients also express preferences for analgesic therapy, with some not wanting certain analgesics because they fear side-effects, which may affect treatment choices. Moreover, we do not have data on comorbid diseases for which analgesics may have been prescribed, including depression (amitriptyline) and seizure disorders (carbamazepine), although we would have expected higher doses to have been used in these situations.[26,27]
Conclusion
We found that most patients with HIV-SN pain in a large SA HIV care clinic were receiving analgesics consistent with the recommendation in the national guidelines. However, about one in six patients were only receiving agents with no demonstrated efficacy for neuropathic pain. Furthermore, despite the observed high level of treatment coverage, the majority of patients described their current level of HIV-SN pain as moderate or severe, highlighting the urgent need for better therapies for this common and disabling condition.
Acknowledgements. The authors thank the patients and staff of the Greenhouse Pharmacy at Chris Hani Baragwanath Hospital, and Florence Mtsweni for acting as an interpreter. We thank Mr Rashid Adam of the Greenhouse Pharmacy for his assistance. The authors gratefully acknowledge the contributions of the Victorian Operational Infrastructure Support Program received by the Burnet Institute (CLC) and Hillel Friedland Trust for Fellowship funding (ALW).
Funding. Funding was received from the Medical Faculty Research Endowment Fund of the University of the Witwatersrand (PP), the Medical Research Council of South Africa (PRK), the National Research Foundation Rated Researchers Programme (PRK), the Victorian Operational Infrastructure Support Program received by the Burnet Institute (CLC), and a Developed-Developing Countries Collaborative Research Grant of the International Association for the Study of Pain (CLC, PRK).
References
1. Kamerman PR, Wadley AL, Cherry CL. HIV-associated sensory neuropathy: Risk factors and genetics. Curr Pain Headache Rep 2012;16(3):226-236. [http://dx.doi.org/10.1007/s11916-012-0257-z] [ Links ]
2. Ellis RJ, Rosario D, Clifford DB, et al. Continued high prevalence and adverse clinical impact of human immunodeficiency virus-associated sensory neuropathy in the era of combination antiretroviral therapy: The CHARTER Study. Arch Neurol 2010;67(5):552-558. [http://dx.doi.org/10.1001/archneurol.2010.76] [ Links ]
3. Cherry CL, Kamerman P, Bennet DLH, Rice AS. HIV-associated sensory neuropathy: Still a problem in the post-stavudine era? Future Virol 2012;7(9)849-854. [http://dx.doi.org/10.2217/fvl.12.77] [ Links ]
4. Phillips TJ, Cherry CL, Cox S, Marshall SJ, Rice AS. Pharmacological treatment of painful HIV- associated sensory neuropathy: A systematic review and meta-analysis of randomised controlled trials. PLoS One 2010;5:e14433. [http://dx.doi.org/10.1371/journal.pone.0014433] [ Links ]
5. World Health Organization. Palliative care: Symptom management and end-of-life care. 2004. http://www.who.int/hiv/pub/imai/genericpalliativecare082004.pdf (accessed 5 August 2014). [ Links ]
6. South African National Department of Health. Standard treatment guidelines and national essential medicines list of South Africa. http://www.kznhealth.gov.za/pharmacy/edladult_2012.pdf (accessed 4 August 2014). [ Links ]
7. Chetty S, Baalbergen E, Bhigjee AI, et al. Clinical practice guidelines for management of neuropathic pain: Expert panel recommendations for South Africa. S Afr Med J 2012;102(5):312-325. [ Links ]
8. World Health Organization. Standard treatment guidelines and national essential medicines list of Iran. 2009. http://www.who.int/selection_medicines/country_lists/irn_EDL_2009.pdf (accessed 4 August 2014). [ Links ]
9. World Health Organization. Standard treatment guidelines and national essential medicines list of Tanzania. 2013. http://www.who.int/medicinedocs/en/d/Js20988en/ (accessed 4 August 2014). [ Links ]
10. World Health Organization. Standard treatment guidelines and national essential medicines list of Zimbabwe. 2006. http://www.who.int/selection_medicines/country_lists/EDLIZ06.pdf?ua=1 (accessed 4 August 2014). [ Links ]
11. Shlay JC, Chaloner K, Max MB, et al. Acupuncture and amitriptyline for pain due to HIV-related peripheral neuropathy: A randomized controlled trial. Terry Beirn Community Programs for Clinical Research on AIDS. JAMA 1998;280(18):1590-1595. [http://dx.doi.org/10.1001/jama.280.18.1590] [ Links ]
12. Dinat N, Marinda E, Moch S, et al. Randomized, double-blind, crossover trial of amitriptyline for analgesia in painful HIV-associated sensory neuropathy. PLoS One 2015;10(5):e0126297. [http://dx.doi.org/10.1371/journal.pone.0126297] [ Links ]
13. Kieburtz K, Simpson D, Yiannoutsos C, et al. A randomized trial of amitriptyline and mexiletine for painful neuropathy in HIV infection. AIDS Clinical Trial Group 242 Protocol Team. Neurology 1998;51(6):1682-1688. [http://dx.doi.org/10.1212/WNL.5L6.1682] [ Links ]
14. Beadles WI, Jahn A, Weigel R, Clutterbuck D. Peripheral neuropathy in HIV-positive patients at an antiretroviral clinic in Lilongwe, Malawi. Trop Doct 2009;39(2):78-80. [http://dx.doi.org/10.1258/td.2008.080213] [ Links ]
15. Cherry CL, Wesselingh SL, Lal L, McArthur JC. Evaluation of a clinical screening tool for HIV- associated sensory neuropathies. Neurology 2005;65(11):1778-1781. [http://dx.doi.org/10.1212/01.wnl.0000187119.33075.41] [ Links ]
16. Wadley AL, Cherry CL, Price P, Kamerman PR. HIV neuropathy risk factors and symptoms characterisation in stavudine-exposed South Africans. J Pain Symptom Manage 2011;41(4):700-706. [http://dx.doi.org/10.1016/j.jpainsymman.2010.07.006] [ Links ]
17. Breitbart W, Rosenfeld BD, Passik SD, et al. The undertreatment of pain in ambulatory AIDS patients. Pain 1996;65(2-3):243-249. [http://dx.doi.org/10.1016/0304-3959(95)00217-0] [ Links ]
18. Maree JE, Dreyer Wright SC, Makua MR. The management of HIV- and AIDS-related pain in a primary health clinic in Tshwane, South Africa. Pain Manag Nurs 2013;14(2):94-101. [http://dx.doi.org/10.1016/j.pmn.2010.10.037] [ Links ]
19. Narasimooloo C, Naidoo S, Gaede B. Adequacy of pain management in HIV-positive patients. S Afr Fam Pract 2011;53(1):71-76. [http://dx.doi.org/10.1080/20786204.2011.10874063] [ Links ]
20. Mphahlele NR, Mitchell D, Kamerman PR. Pain in ambulatory HIV-positive South Africans. Eur J Pain 2012;16(3):447-458. [http://dx.doi.org/10.1002/j.1532-2149.2011.00031.x] [ Links ]
21. Mphahlele N, Kamerman PR, Mitchell D. Progression of pain in ambulatory HIV-positive South Africans. Pain Manag Nurs 2015;16(1):e1-e8 [http://dx.doi.org/10.1016/j.pmn.2014.05.013] [ Links ]
22. Finnerup NB, Sindrup SH, Jensen TS. The evidence for pharmacological treatment of neuropathic pain. Pain 2010;150(3):573-581. [http://dx.doi.org/10.1016/j.pain.2010.06.019] [ Links ]
23. Kamerman P, Mitchell D. Current perspectives on HIV-related pain and its management: Insights from Sub-Saharan Africa. Pain Manag 2011;1(6):587-596. [http://dx.doi.org/10.2217/pmt.11.65] [ Links ]
24. Seitz CS, Pfeuffer P, Raith P, et al. Anticonvulsant hypersensitivity syndrome: Cross-reactivity with tricyclic antidepressant agents. Ann Allergy Asthma Immunol 2006;97(5):698-702. [http://dx.doi.org/10.1016/S1081-1206(10)61103-9] [ Links ]
25. American Cancer Society. Guide to cancer drugs (Codeine). 2014. http://www.cancer.org/treatment/treatmentsandsideeffects/guidetocancerdrugs/codeine (accessed 6 August 2014). [ Links ]
26. Mattson RH, Cramer JA, Collins JF, et al. A comparison of valproate with carbamazepine for the treatment of complex partial seizures and secondarily generalised tonic-clonic seizures in adults. N Engl J Med 1992;327(11):765-771. [http://dx.doi.org/10.1056/NEJM199209103271104] [ Links ]
27. American Psychiatric Association. Practice Guideline for the Treatment of Patients with Major Depressive Disorder. 3rd ed. Arlington, VA: American Psychiatric Association, 2010. http://psychiatryonline.org/data/books/prac/PG_Depression3rdEd.pdf (accessed 5 August 2014). [ Links ]
Accepted 4 July 2015
Corresponding author: P Pillay (prinishapillay_13@yahoo.com)














