Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.105 n.9 Pretoria Sep. 2015
http://dx.doi.org/10.7196/SAMJNEW.8144
FORUM
HEALTHCARE DELIVERY
Endoscopic lung volume reduction in severe emphysema
C F N KoegelenbergI; J TheronII; J W BruwerIII; B W AllwoodIV; M J VorsterV; F von Groote-BidlingmaierVI; K DhedaVII
IIs a consultant in Pulmonology and Critical Care at Stellenbosch University and Tygerberg Academic Hospital, Tygerberg Cape Town, South Africa, and the current chairman of Assembly on Interventional Pulmonology of the South African Thoracic Society
IIIs an interventional pulmonologist in private practice at Panorama Mediclinic, Cape Town, and an honorary lecturer at Stellenbosch University
IIIIs an interventional pulmonologist in private practice at Windhoek Mediclinic, Namibia
IVIs a consultant in pulmonology and critical care,
VA senior registrar
VIAn honorary lecturer at Stellenbosch University and Tygerberg Academic Hospital
VIIIs a consultant and head of the Division of Pulmonology at the University of Cape Town and Groote Schuur Hospital, Cape Town
ABSTRACT
Therapeutic options in severe emphysema are limited. Endoscopic lung volume reduction (ELVR) refers to bronchoscopically inducing volume loss to improve pulmonary mechanics and compliance, thereby reducing the work of breathing. Globally, this technique is increasingly used as treatment for advanced emphysema with the aim of obtaining similar functional advantages to surgical lung volume reduction, while reducing risks and costs. There is a growing body of evidence that certain well-defined subgroups of patients with advanced emphysema benefit from ELVR, provided that a systematic approach is followed and selection criteria are met. In addition to endobronchial valves, ELVR using endobronchial coils is now available in South Africa. The high cost of these interventions underscores the need for careful patient selection to best identify those likely to benefit from such procedures.
Emphysema is a very common cause of morbidity and mortality in South Africa (SA). Smoking remains the most common risk factor for the development of emphysema, but longterm biomass fuel exposure, tuberculosis and HIV co-infection contribute to the disease burden in southern Africa.[1] However, therapeutic options for severe emphysema are currently limited.
In the subgroup of patients with predominantly upper-lobe emphysema and low exercise capacity, surgical lung volume reduction improves functional status, but not without a significant risk of mortality and morbidity as well as high financial costs.[2] Endoscopic lung volume reduction (ELVR), which is increasingly being used internationally, aims to reduce the risks and costs of surgery, while obtaining the same functional advantages. It refers to the placement of a device (e.g. a valve or coil) through a bronchoscope into a bronchus with the aim of decreasing the volume of the lung segments distal to the device, thereby improving pulmonary mechanics and compliance.[3] The work of breathing is reduced, resulting in significant improvements in symptoms, particularly breathlessness.
There is a growing body of evidence that certain well-defined subgroups of patients with advanced emphysema benefit from ELVR, with the caveat that a systematic approach is followed and selection criteria are met. Recently, endobronchial coils, in addition to endobronchial valves, have become available in SA for ELVR; however, the cost of both remains high, emphasising the need for careful patient identification and selection to best identify those who are most likely to benefit from these procedures.
Modalities and devices available in SA
Unidirectional endobronchial and intrabronchial valves
Unidirectional valves induce either lobar or segmental lung collapse by preventing the entrance of air during inspiration, while allowing exhalation of air and secretions. There are currently two such devices commercially available in SA: Zephyr endo-bronchial valves (Pulmonx Inc.) (Fig. 1) and IBV intrabronchial valves (Olympus Respiratory America) (Fig. 2). Both devices are self-expanding and delivered using a catheter introduced through the working channel of a flexible bronchoscope.[3]
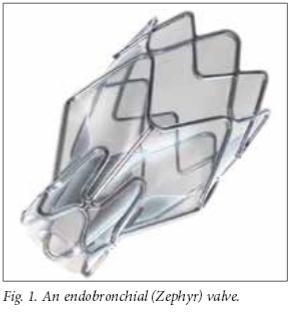
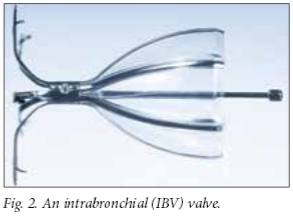
These devices have been shown to be less effective when emphysema is found uniformly throughout the lungs (i.e. homogeneous emphysema). The distribution of emphysema is assessed on high-resolution computed tomography (HRCT) scanning of the chest, either by visual inspection of the parenchyma or by using specifically designed automated quantification software.[3] Additionally, valves fail to induce collapse when the affected portion of the lung has collateral ventilation. This is a normal physiological phenomenon in many individuals, but significant interlobar collateral ventilation subverts the deflating effect of endobronchial blocking devices.
Prior to placement of a valve, both homogeneous emphysema and collateral ventilation therefore need to be excluded. The evidence for this requirement is derived from numerous studies in which the minimal clinically important differences were significantly more likely to be observed in patients with advanced heterogeneous emphysema and no collateral ventilation. Moreover, unilateral (as opposed to bilateral ELVR) valve placement was found to have better outcomes.[4-7]
The recently completed STELVIO trial provided the strongest evidence for use of valves in patients without collateral ventilation.[8] Dutch investigators randomised 68 patients with severe emphysema on HRCT with visual estimation of complete or near-complete fissures to endobronchial Zephyr valve treatment (n=34) or standard medical care (n=34). The primary outcome measures were change in spirometric measures and the 6-minute walking distance (6MWD) at 6 months. Clinical relevance was assessed relative to minimal clinically important differences. At 6 months, the minimal clinically important differences were attained in all parameters in the treated group compared with controls (p<0.001 for all endpoints).
The most common reported adverse events experienced with endobronchial valve placement have been pneumothoraces (5 - 10%), mild haemoptysis (2 - 6%) and exacerbations of underlying chronic obstructive pulmonary disease (COPD) (8 - 40%).[4-7]
Coils
Coils (RePneu; BTG Inc.) (Fig. 3) are nitinol devices designed to be deployed into a straight airway, and thereafter to resume their preformed shape. This conformational shape change after deployment results in parenchymal retraction with volume loss, while maintaining airway patency.[9] The device is currently available in three lengths to accommodate different-sized airways. The coils are implanted via a flexible bronchoscope under general anaesthesia or conscious sedation and fluoroscopic guidance using a proprietary delivery system. In theory, coils not only cause lung volume reduction but additionally re-tension the remaining airway network and mechanically increase elastic recoil. This re-tensioning of the lung tethers open the small airways, thus preventing their collapse on expiration, known to be the pathogenetic mechanism of emphysema.
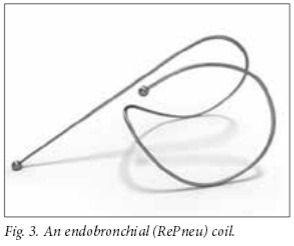
Current evidence suggests that candidates with both heterogeneous and homogeneous emphysema can experience clinically significant benefit from ELVR using coils. This benefit is obtained regardless of the presence of collateral ventilation or complete lobar collapse post procedure, but requires that least >25% of the total lung parenchyma is unaffected by radiological emphysema prior to insertion.[10,11] Approximately 75 - 80% of patients will experience minimal clinically important differences in lung function and quality of life, while mild haemoptysis of <5 mL (50 - 75%), exacerbations of COPD (5 - 12%), mild chest discomfort (15 - 50%) and infrequent pneumothoraces (3%) are the described adverse events.[10,11] A recent report on the 3-year follow up data on 38 patients who underwent ELVR using coils suggested that the coil treatment was safe; no late pneumothoraces, coil migrations or unexpected adverse events occurred.[12] Although clinical benefit gradually declined over time, at 3 years post treatment approximately 50% of the patients maintained improvement in 6MWD and dyspnoea and quality of life scores.
Other devices
Synthetic polymeric foam (Aeris Therapeutics Biological) has been used to obtain atelectasis. This technology is currently not available in SA. A recent study on synthetic polymeric foam, which was terminated prematurely, raised some safety concerns.[13]
The future of endoscopic lung volume reduction in SA
Current evidence suggests that not all classes and phenotypes of emphysema will benefit from ELVR, and that individual techniques offer benefit to different subgroups of patients.[5,9,10] Only a few centres in SA currently have the capacity to evaluate prospective candidates properly and potentially offer ELVR to appropriate cases. The high cost of these interventions makes careful patient selection imperative to prevent wasteful treatment of patients who are unlikely to gain clinical benefit.
The initial screening for suitable candidates should be performed at subspecialist (pulmonologist) level, and on patients with stable disease and no recent exacerbations. Routine special investigations should include HRCT (to estimate heterogeneity, integrity of fissures and degree of tissue destruction, and evaluate for possible underlying lung cancer), full pulmonary function testing, arterial blood gas measurement and echo-cardiography (to exclude pulmonary hypertension).[10] The general indications and contraindications for valves and coils are summarised in Table 1. ELVR should not be offered to active smokers, patients with pulmonary hypertension, unstable cardiac pathology, active respiratory infections or very poor exercise tolerance, patients without clear evidence of hyperinflation, or patients who use antiplatelet or anticoagulant therapy that cannot be stopped for 7 days prior to the procedure.[10,11,14]

Appropriate or borderline candidates (Table 1) should be referred to a centre with the capacity to evaluate, treat and follow up these patients, and manage complications and undertake removal of devices if required.
Conclusions and recommendations
There is currently no head-to-head evidence comparing the various techniques and devices, and no official guideline from any of the major thoracic societies is currently available. The Assembly on Interventional Pulmonology of the South African Thoracic Society has extensively reviewed all relevant publications, and consulted international experts on the use of ELVR in SA in the form of recommendations based not only on published evidence, international expert opinion and local expertise, but also on local commercial access to devices.[14] It is also recommended that all endoscopic lung volume reduction procedures should be performed in the context of a local and/ or international registry. The Assembly on Interventional Pulmonology of the SA Thoracic Society is willing to assist potential centres wishing to establish an ELVR service in terms of training and accreditation.
In summary, appropriate candidates with marked hyperinflation and relatively preserved lung parenchyma are more likely to benefit from ELVR with bilateral coils, irrespective of the collateral ventilation and heterogeneity of the disease. In contrast, patients with heterogeneous disease and no collateral ventilation are more likely to benefit from unilateral ELVR with valves aiming to achieve complete lobar collapse.
A well-structured evidence-based approach to ELVR, including initial screening and subsequent referral to a specialised centre, is important to avoid inappropriate use of devices, which may be both wasteful and harmful.
References
1. Allwood B, Myer L, Bateman E. A systematic review of the association between pulmonary tuberculosis and the development of chronic airflow obstruction in adults. Respiration 2013;86(1):76-85. [http://dx.doi.org/10.1159/000350917] [ Links ]
2. National Emphysema Treatment Trial Group. A randomized trial comparing lung-volume-reduction surgery with medical therapy for severe emphysema. N Engl J Med 2003;348(21):2059-2073. [http://dx.doi.org/10.1056/NEJMoa030287] [ Links ]
3. Gasparini S, Zuccatosta L, Bonifazi M, Bolliger CT. Bronchoscopic treatment of emphysema: State of the art. Respiration 2012;84(3):250-263. [http://dx.doi.org/10.1159/000341171] [ Links ]
4. Shah P, Slebos D, Cardoso P, et al. Bronchoscopiclung-volume reduction with Exhale airway stents for emphysema (EASE trial): Randomised, sham-controlled, multicentre trial. Lancet 2011;378(9795):997-1005. [http://dx.doi.org/10.1016/S0140-6736(11)61050-7] [ Links ]
5. Sciurba FC, Ernst A, Herth FJF, et al. A randomized study of endobronchial valves for advanced emphysema. N Engl J Med 2010;363(13):1233-1244. [http://dx.doi.org/10.1056/NEJMoa0900928] [ Links ]
6. Mor Z, Leventhal A, Diacon AH, Finger R, Schoch OD. Tuberculosis screening in immigrants from high-prevalence countries: Interview first or chest radiograph first? A procon debate. Respirology 2013;18(3):432-438. [http://dx.doi.org/10.1111/resp.12054] [ Links ]
7. Herth FJF, Noppen M, Valipour A, et al. Efficacy predictors of lung volume reduction with Zephyr valves in a European cohort. Eur Respir J 2012;39(6):1334-1342. [http://dx.doi.org/10.1183/09031936.00161611] [ Links ]
8. Klooster K, ten Hacken N, Hartman J, Kersttjens H, van Rikxoort E, Slebos D. Endobronchial valve treatment versus standard medical care in patients with emphysema without interlobar collateral ventilation (the STELVIO-Trial). Am J Respir Crit Care Med 2015;191(9):A6312. [ Links ]
9. Herth FJF, Eberhard R, Gompelmann D, Slebos DJ, Ernst A. Bronchoscopic lung volume reduction with a dedicated coil: A clinical pilot study. Ther Adv Respir Dis 2010;4(4):225-231. [http://dx.doi.org/10.1177/1753465810368553] [ Links ]
10. Deslee G, Klooster K, Hetzel M, et al. Lung volume reduction coil treatment for patients with severe emphysema: A European multicentre trial. Thorax 2014;69(11):980-986. [http://dx.doi.org/10.1136/thoraxjnl-2014-205221] [ Links ]
11. Klooster K, ten Hacken NHT, Franz I, Kersttjens HM, van Rikxoort EM, Slebos D-J. Lung volume reduction coil treatment in chronic obstructive pulmonary disease patients with homogeneous emphysema: A prospective feasibility trial. Respiration 2014;88(2):116-125. [http://dx.doi.org/10.1159/000362522] [ Links ]
12. Hartman J, Klooster K, Gortzak K, ten Hacken N, Slebos D. Long-term follow-up after bronchoscopic lung volume reduction treatment with coils in patients with severe emphysema. Respirology 2015;20(2):319-326. [http://dx.doi.org/10.1111/resp.12435] [ Links ]
13. Come C, Kramer M, Dransfield M, et al A randomised trial of lung sealant versus medical therapy for advanced emphysema. Eur Respir J 2015:1-12. [Epub ahead of print] [http://dx.doi.org/10.1183/09031936.00205614] [ Links ]
14. Koegelenberg CFN, Theron J, Dheda K, et al. Recommendations for endoscopic lung volume reduction in South Africa: Role in emphysema. S Afr Med J 2015;105(10)(in press). [http://dx.doi.org/10.7196/SAMJnew.8147] [ Links ]
Accepted 29 June 2015
Corresponding author: C F N Koegelenberg (coeniefn@sun.ac.za)

Leonid M Irenge* is co-ordinator of a co-operation project between the Université catholique de Bukavu (South Kivu Province, Democratic Republic of the Congo (DRC)) and the Université catholique de Louvain (Belgium), which aims to improve identification of micro-organisms involved in infectious diseases in South Kivu. This entails overseeing development of microbiology capabilities at the provincial hospital in Bukavu, the capital of South Kivu, as well as at the level of the Healthcare Inspection in that province. A senior scientist at UCL/CTMA (Faculty of Medicine of the Université catholique de Louvain and Defence Laboratories Department, Belgium), he holds a doctorate in molecular biology and has been working in the field of microbiology for a decade. His research efforts focus on development of assays for rapid and unambiguous detection and identification of pathogenic micro-organisms, both in the field and in reach-back facilities, and on characterisation of antimicrobial resistance on isolates from DRC.
*Irenge LM, Kabego L, Kinunu FB, et al. Antimicrobial resistance of bacteria isolated from patients with bloodstream infections at a tertiary care hospital in the Democratic Republic of the Congo. S Afr Med J 2015;105(9):752-755. [http://dx.doi.org/10.7196/SAMJnew.7937]














