Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.105 n.2 Pretoria Feb. 2015
http://dx.doi.org/10.7196/samj.8369
RESEARCH
Morbidity and mortality of black HIV-positive patients with end-stage kidney disease receiving chronic haemodialysis in South Africa
J FabianI, II; H A MaherIII; C ClarkIV; S NaickerV; P BeckerVI; W D F VenterII, VII
IMD, MMed; Wits Donald Gordon Medical Centre, Parktown, Johannesburg, South Africa
IIMD, MMed; Department of Internal Medicine, Faculty of Health Sciences, University of the Witwatersrand, Johannesburg, South Africa
IIIRegistered Nurse; Wits Donald Gordon Medical Centre, Parktown, Johannesburg, South Africa
IVMTech Clinical Technology, BSc, PhD; National Renal Care, South Africa
VMD, PhD; Department of Internal Medicine, Faculty of Health Sciences, University of the Witwatersrand, Johannesburg, South Africa
VIPhD; Biostatistics Unit, South African Medical Research Council, Tshwane, South Africa
VIIMD, MMed; Wits Reproductive Health and HIV Institute, Johannesburg, South Africa
ABSTRACT
INTRODUCTION: There are few published data from South Africa (SA) on the outcomes ofblack HIV-positive patients receiving chronic haemodialysis.
METHODS: This retrospective study compared the incidences of vascular and infectious morbidity and mortality in black HIV-positive patients with those in a group of HIV-negative patients matched for ethnicity, age and gender. All the patients were receiving chronic haemodialysis in the medically insured healthcare sector of SA.
RESULTS: The incidence of tuberculosis and hospital admission rates for vascular access-related infections were significantly higher in the HIV-positive group than the HIV-negative group. The HIV-positive group had significantly lower albumin (p<0.05) and haemoglobin levels (p<0.01), but this did not impact on mortality. Survival in both groups was excellent. In the HIV-positive group, viral suppression rates were suboptimal with <50% of patients on antiretroviral therapy completely virally suppressed.
CONCLUSION: This study has shown that black HIV-positive patients receiving chronic haemodialysis in a healthcare-funded environment in SA have excellent overall survival in spite of higher hospital admission rates and higher infectious morbidity compared with HIV-negative patients.
Outcomes of HIV-positive patients with end-stage kidney disease (ESKD) in the era prior to antiretroviral therapy (ART) were dismal.[1] In the USA before 1995, when ART was not freely available, median survival of HIV-positive patients on chronic haemodialysis ranged from 1.4 to 12 months.[2] After 1995, as access to effective ART improved, studies began to show progressive improvements in survival in HIV-positive patients receiving chronic haemodialysis, but survival was still significantly lower than in their HIV-negative counterparts.[3,4] In 2006, French researchers showed that the 2-year survival of HIV-positive patients on haemodialysis with access to ART was comparable (89%) to a cohort of HIV-negative patients.[5] Survival rates of black HIV-positive patients with ESKD in sub-Saharan Africa are unknown.
ART became freely available in the state health sector in South Africa (SA) in 2004, with access expanding dramatically so that over 2 million people were on ART by 2013.[6,7] Before 2008, it was standard policy in SA public sector hospitals to exclude HIV-positive patients from access to chronic dialysis. In 2008, this policy was reviewed. The SA renal and transplant societies jointly acknowledged that HIV-positive patients could not be excluded on the basis of their HIV status alone, and the guidelines were revised.[8]
Unfortunately the majority of patients with ESKD in the public healthcare system, including HIV-positive patients, have extremely limited access to chronic dialysis because of limited resources, whereas those with medical health insurance have unrestricted access.[9,10] There are no published data from SA on the outcomes of either of these groups of patients. This study was conducted to assess the outcomes of black HIV-positive individuals with ESKD on chronic haemodialysis, who have access to medical health insurance in SA.
Methods
This study was a national, retrospective cohort study. Approval was obtained from the Human Research Ethics Committee of the University of the Witwatersrand, approval No. M101120. National Renal Care (NRC) was the dialysis service provider for all the chronic haemodialysis centres that participated in the study. Adult HIV-positive patients over the age of 18 who had started chronic haemodialysis between 1 January 2006 and 31 October 2010 and had continued for at least 6 months were eligible for inclusion in the study. Informed consent was obtained from 48 of the HIV-positive patients (consent rate 24.5%). The date of the first dialysis session in the chronic dialysis unit was taken as commencement of the first year of dialysis, and each subsequent year on dialysis was defined from this point. Before commencement of the study, staff in the dialysis centres were trained by two of the authors on how to obtain informed consent in accordance with Good Clinical Practice guidelines.[11] Only those who had completed the training were eligible to obtain informed consent and were delegated by one of the authors to do so.
Both HIV-positive and HIV-negative patients were asked to participate in the study. They were supplied with a patient information leaflet, and written informed consent was obtained from those who agreed to participate. All patients who were HIV-positive and gave consent were included in the study. Participants with incorrectly completed consent forms were excluded. A group of HIV-negative patients who had given consent were then selected as matching controls in a 2:1 ratio. Dialysis prescriptions were individualised with respect to size of the dialysis membrane, blood flow rate and pump speed and reviewed as needed by the attending doctor, based upon the condition of the patient and his/her blood results. All patients received haemodialysis three times a week for 4hours. Polysulfone haemodialysis membranes were used in all dialysis centres, and there was no re-use policy regarding membranes.
The following data were routinely collected on all patients: demographics (access to running water was defined as access within the proximity of the house/dwelling), medical history, blood tests performed at various intervals as standard of care, and medical information regarding acute/emergency hospital admissions. The following blood tests were done: corrected serum calcium (mmol/L), serum phosphate (mmol/L), haemoglobin (g/dL), serum ferritin ^g/L), serum albumin (g/L); and KT/V (K (urea clearance of dialyser (ml/min)) x T (dialysis time in minutes) ÷ V (volume (ml)), calculated as a measure of dialysis adequacy). Values for each year the patients were on the study were collected and an average was calculated per year of follow-up. Vascular access and transplant listing was defined at a single point in time, 31 October 2010. The prevalence of HIV infection (positive ELISA), chronic hepatitis B infection (hepatitis B surface antigen positivity for >6 months), hepatitis C infection (hepatitis C antibodies), and the total number of patients receiving chronic haemodialysis with NRC were defined at a single point in time, 31 October 2010. For the HIV-positive group, CD4 counts (cells/μl) were recorded per year of follow-up, and in the case of more than one value an annual average was calculated; the log HIV viral load (copies/ml) was determined using a single value as close as possible to the single point in time, 31 October 2010. Medication regimens were obtained from patient records in the dialysis units, from the NRC database, and in some cases from the attending doctors' patient files.
With regard to each hospital admission, the following data were collected (where appropriate): date of admission, date of discharge, and reason for admission (as determined by the admitting doctor); number of hospital admissions for the current year; date of infection and site of infection; date on which the dialysis access was created or removed; and how many previous dialysis access-related events the patient had. In the case of laboratory testing, the respective laboratories were accessed for information where necessary, e.g. for histology and tuberculosis (TB) culture results.
A positive diagnosis of TB was determined via either a clinical diagnosis with confirmatory laboratory results (histology, culture) or commencement of empiric anti-TB treatment based on the patient's clinical presentation.
Hypertension was defined as a diagnosis of hypertension by the attending doctor or evidence from patient records that the patient was receiving antihypertensive treatment. Coronary artery disease was defined by the need for admission and included a diagnosis of either unstable angina or acute myocardial infarction from the attending doctor. Cerebrovascular events were defined by the need for admission and a diagnosis of a stroke (either thrombotic or haemorrhagic) or a reversible ischaemic neurological event as determined by the attending doctor. Average duration of follow-up was calculated by taking the total months on chronic dialysis of all patients and dividing this by the total number of patients. Incidence rate ratios were calculated by dividing the incidence in HIV-positive patients by the incidence in HIV-negative patients. 'Patient-days' were calculated by dividing the total number of days that patients were admitted during the period of follow-up divided by the number of patients in each group.
Statistical analysis was performed using the statistical package Stata 11 Data Analysis and Statistical Software. Descriptive statistical analysis was performed, as well as linear regression analysis comparing differences between the HIV-positive and HIV-negative groups, for each year on dialysis, over the 5-year study period.
Results
A total of 2 010 patients received chronic haemodialysis nationally with NRC during the study period. Prevalence rates for infections were as follows: HIV 9.75% (196/2 010), hepatitis C 0.95% (19/2 010) and hepatitis B 1.59% (32/2 010). Comorbidity and demographic comparators between HIV-positive and HIV-negative patients are set out in Table 1.
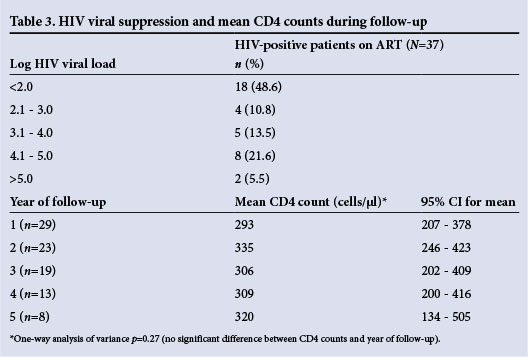
The two groups were well matched, apart from a significantly higher proportion of black patients in the HIV-positive group. Significantly fewer HIV-positive than HIV-negative patients were wait-listed for kidney transplantation. Incident disease while on the study is summarised in Table 2. The total number of patient days admitted, independent of cause for admission, was significantly higher for the HIV-positive group. Vascular access rates and access-related admission rates with respect to arteriovenous fistula, arteriovenous graft and tunnelled catheter were similar in both groups, but the number of access-related infections that required hospital admission was statistically higher in the HIV-positive group. The incidence of treated TB infection was also significantly higher in the HIV-positive group. In the HIV-positive group, log HIV viral suppression rates and mean CD4 counts per year of follow-up are set out in Table 3. The mean duration of ART was 30.1 months (n=31) (95% confidence interval (CI) for the mean 23.7 - 36.5). No data regarding ART exposure were available in 14/48 (29.2%) patients, and 3/48 (6.3%) were not on ART. In those documented on ART, 19/37 (51.4%) were not virally suppressed using log <2.0 as a cut-off.

Linear regression analysis showed that average haemoglobin levels (p<0.01) and serum albumin levels (p<0.05) were statistically significantly lower in the HIV-positive group for the duration of follow-up, although the average serum albumin level in the HIV-positive group was nevertheless higher than 30 g/L. There was no statistical difference between the groups for serum ferritin, calcium, phosphate and KT/V values (Table 4). Survival during follow-up in the two groups was comparable at 100% for HIV-positive and 99% for HIV-negative patients. One patient died and one received a kidney transplant.

Discussion
In this study, survival of medically insured black HIV-positive patients receiving chronic haemodialysis in SA was excellent, in spite of longer durations of hospital stay and higher infectious morbidity. Infectious morbidity was associated with higher hospital admission rates for vascular access-related infections, higher incidence rates for TB and inadequate HIV viral load suppression in those on ART.
Limitations of this study include the small sample size, inability to match the HIV-positive and negative groups perfectly with respect to ethnicity, and restriction of participants to a healthcare-funded environment. These factors limit generalisation of the findings and do not accurately reflect the socioeconomic circumstances of the general population in SA. The consent rates for HIV-positive participants were unexpectedly low. It was thought that training staff to obtain consent would increase the recruitment rate, but these staff disclosed that they felt uncomfortable asking HIV-positive patients to participate and believed that a high patient load compromised their capacity for research-related activity.
Survival in the HIV-positive group was excellent. To date, the only other data available in SA on survival of black HIV-positive patients receiving chronic dialysis are from a study conducted in the state-funded Helen Joseph Hospital in Johannesburg. [12] In this study, 59 HIV-positive patients received chronic dialysis from 2001 to 2012, and although the demographics with respect to age, gender, ethnicity and mean follow-up were similar to those in the current study, mortality was much higher at 51% (30/59). [12] Potential reasons for this discrepancy are probably multifactorial and may be related to a lower median CD4 count of 231 cells/μl and higher prevalences of co-infection with hepatitis B at 22% (13/59) and hepatitis C at 5% (3/59), making the relative frequencies 14 and five times more common, respectively, in the Helen Joseph study than in the current study. A further explanation for the discrepant survival was the choice of chronic renal replacement therapy - in our study only HIV-positive patients on chronic haemodialysis were recruited, while in the Helen Joseph cohort chronic ambulatory peritoneal dialysis (CAPD) was the predominant mode of dialysis (63% (37/59)). Sixty-three per cent of deaths occurred in the CAPD group, the most common causes of death being fluid overload (38%) and peritonitis (31%), which are potentially preventable and easily treated. These deaths may reflect a combination of lower socioeconomic status and poor access to healthcare facilities when acutely ill.
Our survival rate for HIV-positive patients on chronic haemodialysis is better than international figures. In the international literature, survival of HIV-positive patients receiving chronic haemodialysis in various cohorts in the USA ranged from 74% at 1 year to 30% at 2 years.[2,4] In Europe, 1- and 2-year survival rates were 93.8% and 89.4%, respectively, with the strongest predictors of survival being CD4 counts >200 cells/μl and serum albumin >30 g/L,[2,5] which are comparable to the CD4 counts and serum albumin levels in the patients in our study. Our HIV-positive patients did have increased rates of infection. TB was the most common opportunistic pathogen in the HIV-positive group, and this was also seen in the study at Helen Joseph Hospital.[12] There are very few data regarding susceptibility to TB in HIV-positive dialysis patients.[13,14] HIV status alone, independent of chronic dialysis, can explain the increased predilection to incident TB and has been well described.[15] This study confirmed a much higher relative risk of incident TB in HIV-positive patients (incidence rate ratio 8.7), even with relatively well-preserved CD4 counts and good socioeconomic circumstances.
HIV-positive patients were admitted more frequently for vascular access-related infections than HIV-negative patients. There are conflicting reports on the impact of HIV on vascular access infection rates in HIV. [16] Some studies have shown no differences between HIV-positive and HIV-negative patients, while others have shown that vascular access-related infections were more prevalent in those with lower CD4 counts, hepatitis B antigenaemia and injecting drug use.[17,18] The results of our study are different to these findings, as there were increased rates of admission for access-related infection in the HIV-positive group in spite of relatively well-preserved CD4 counts and low levels of hepatitis B antigenaemia. The impact of injecting drug use in our setting is unknown.
The USA has the third highest prevalence of injecting drug use in the world (0.96%), and an associated HIV prevalence among injectors of 16%.[19] A study in 2012 showed that there were 67 000 injecting drug users in SA. This translates to a population prevalence of 0.13%, which is approximately seven times less frequent than that in the USA. The HIV prevalence in this group was 19.4%.[20,21] There are no data from SA on injecting drug use in chronic dialysis populations, but based on the national prevalence rates it is possible to infer that the prevalence in dialysis units is low, and the impact of injecting drug use on outcomes in this patient group would be minimal.
An unexpected finding was the treatment response in the HIV-positive patients on ART. The HIV viral load was incompletely suppressed in 51% of participants on treatment. Regular testing of CD4 counts and HIV viral loads after initiation of ART was not standardised and data were scanty. Poor HIV viral suppression with chronic dialysis has been documented, with rates of complete HIV suppression ranging from 44% to 88%. This has been correlated with higher mortality.[12,22-24] Reasons for incomplete HIV viral suppression have been ascribed to inexperience with HIV treatment and poor prescribing practices by nephrologists, with both under- and over-prescription of ART, and infrequent consultation and follow-up with an infectious diseases specialist.[24] These factors must be considered as possible explanations in this study, as many nephrologists initiate and continue ART without input from an infectious diseases specialist in the medically insured setting. The impact of suboptimal practice may adversely affect the outcomes of this group of HIV patients, not only with respect to mortality but also eligibility for kidney transplantation. Although poorer outcomes were not reflected in this study, the effect may have been masked by small numbers. These preliminary findings deserve further scientific exploration in future studies and in the development of clinical practice guidelines for nephrologists who care for HIV patients in SA.
Conclusion
Black HIV-positive patients on chronic haemodialysis in a healthcare-funded environment in SA had excellent survival rates that were comparable with those in their HIV-negative counterparts despite suboptimal HIV viral suppression, lower serum albumin and haemoglobin levels, and a higher incidence of TB and vascular access-related infections that required hospital admission.
Sources of support. Unrestricted research grants from NRC, SA, and Roche, SA. WDFV is supported by PEPFAR.
Acknowledgements. The authors thank all those who supported the project: NRC senior management, the staff and patients in the NRC chronic dialysis units, and the doctors in the NRC units who facilitated access to patients and assisted with clinical information on patients who participated in the study.
Conflict of interest. The results presented in this article have not been published previously in whole or in part, except in abstract format.
References
1. Ortiz C, Meneses R, Jaffe D, Fernandez JA, Perez G, Bourgoignie JJ. Outcome of patients with human immunodeficiency virus on maintenance hemodialysis. Kidney Int 1988;34(2):248-253. [http://dx.doi.org/10.1038/ki.1988.172] [ Links ]
2. Rodriguez RA, Mendelson M, O'Hare AM, Hsu LC, Schoenfeld P. Determinants of survival among HIV-infected chronic dialysis patients. J Am Soc Nephrol 2003;14(5):1307-1313. [http://dx.doi.org/10.1097/01.ASN.0000062963.56513.28] [ Links ]
3. Perinbasekar S, Brod-Miller C, Pal S, Mattana J. Predictors of survival in HIV-infected patients on hemodialysis. Am J Nephrol 1996;16(4):280-286. [http://dx.doi.org/10.1097/01] [ Links ]
4. Ahuja TS, Grady J, Khan S. Changing trends in the survival of dialysis patients with human immunodeficiency virus in the United States. J Am Soc Nephrol 2002;13(7):1889-1893. [http://dx.doi.org/10.1097/01] [ Links ]
5. Tourret J, Tostivint I, du Montcel ST, et al. Outcome and prognosis factors in HIV-infected hemodialysis patients. Clin J Am Soc Nephrol 2006;1(6):1241-1247. [http://dx.doi.org/0.2215/CJN.02211205] [ Links ]
6. Pillay Y. Launch of the 2013 WHO Consolidated ARV Guidelines: What's the evidence? Presented at the 7th IAS Conference on HIV Pathogenesis, Treatment and Prevention, Kuala Lumpur, Malaysia, 30 June - 3 July 2013. [ Links ]
7. South African National Department of Health. National Antiretroviral Treatment Guidelines. 1st ed. South Africa: Jacana, 2004. [ Links ]
8. South African Renal Society, SATS, Southern African HIV Clinicians Society. Guidelines for renal replacement therapy in HIV-infected individuals in South Africa. Southern African Journal of HIV Medicine 2008;Autumn:34-42. [ Links ]
9. ProPublica. Life and death choices as South Africans ration dialysis care. 15 December 2010. www.propublica.org/article/dialysis-south-africa (accessed 28 October 2011). [ Links ]
10. Moosa MR, Kidd M. The dangers of rationing dialysis treatment: The dilemma facing a developing country. Kidney Int 200670(6):1107-1114. [http://dx.doi.org/10.1038/sj.ki.5001798] [ Links ]
11. South African National Department of Health. Guidelines for Good Practice in the Conduct of Clinical Trials with Human Participants in South Africa. 2nd ed. Pretoria: Department of Health, 2006. [ Links ]
12. Zako F, Wambugu B, Radev M, Naicker S. Outcomes of chronic dialysis in HIV-infected patients. Presented at the ISN World Congress of Nephrology, Hong Kong, 31 May - 4 June 2013. [ Links ]
13. Dobler CC, McDonald SP, Marks GB. Risk of tuberculosis in dialysis patients: A nationwide cohort study. PloS One 2011;6(12):e29563. [http://dx.doi.org/10.1371/joumal.pone.0029563] [ Links ]
14. Hussein M, Mooij J, Roujouleh H. Tuberculosis in hemodialysis patients. Saudi J Kidney Dis Transpl 1996;7(1):6-9. [ Links ]
15. Lawn SD, Myer L, Bekker LG, Wood R. Burden of tuberculosis in an antiretroviral treatment programme in sub-Saharan Africa: Impact on treatment outcomes and implications for tuberculosis control. AIDS 2006;20(12):1605-1612. [ Links ]
16. Castro CE, Madariaga MG. Vascular access-related infections in HIV patients undergoing hemodialysis: Case description and literature review. Braz J Infect Dis 2008;12(6):531-535. [http://dx.doi.org/10.1590/S1413-86702008000600017] [ Links ]
17. Mitchell D, Krishnasami Z, Allon M. Catheter-related bacteraemia in haemodialysis patients with HIV infection. Nephrol Dial Transplant 2006;21(11):3185-3188. [http://dx.doi.org/10.1093/ndt/gfl425] [ Links ]
18. Mokrzycki MH, Schroppel B, von Gersdorff G, Rush H, Zdunek MP, Feingold R. Tunneled-cuffed catheter associated infections in hemodialysis patients who are seropositive for the human immunodeficiency virus. J Am Soc Nephrol 2000;11(11):2122-2127. [ Links ]
19. Mathers BM, Degenhardt L, Phillips B, et al. Global epidemiology of injecting drug use and HIV among people who inject drugs: A systematic review. Lancet 2008;372(9651):1733-1745. [http://dx.doi.org/10.1016/S0140-6736(08)61311-2] [ Links ]
20. Petersen Z, Pluddeman A, van Hout MC, et al. Availability of HIV prevention and treatment services for people who inject drugs: Findings from 21 countries. Harm Reduct J 2013;10(13). [http://dx.doi.org/10.1186/1477-7517-10-13] [ Links ]
21. Statistics SA. 2012. www.statssa.gov.za/publications/SAStatistics/SAStatistics2012pdf (accessed 25 May 2013). [ Links ]
22. Trullas JC, Barril G, Cofan F, et al. Prevalence and clinical characteristics of HIV type 1-infected patients receiving dialysis in Spain: Results of a Spanish survey in 2006: GESIDA 48/05 study. AIDS Res Hum Retroviruses 2008;24(10):1229-1235. [ Links ]
23. Trullas JC, Mocroft A, Cofan F, et al. Dialysis and renal transplantation in HIV-infected patients: A European survey. J Acquir Immune Defic Syndr 2010;55(5) :582-589. [http://dx.doi.org/10.1097/QAI.0b013e3181efbe59] [ Links ]
24. Tourret J, Tostivint I, Tezenas du Montcel S, et al. Antiretroviral drug dosing errors in HIV-infected patients undergoing hemodialysis. Clin Infect Dis 2007;45(6):779-784. [http://dx.doi.org/10.1086/521168] [ Links ]
 Correspondence:
Correspondence:
J Fabian
june.fabian@mweb.co.za
Accepted 16 July 2014














