Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.105 n.2 Pretoria Feb. 2015
http://dx.doi.org/10.7196/samj.9367
FORUM
DRUG ALERT
Recommendations pertaining to the use of influenza vaccines and influenza antiviral drugs: influenza 2015
S Walaza; C Cohen
Medical epidemiologists at the Centre for Respiratory Diseases and Meningitis, National Institute for Communicable Diseases (NICD), Johannesburg, South Africa, and have compiled this article on behalf of the Centre for Respiratory Diseases and Meningitis, NICD and the National Department of Health, Pretoria, South Africa
ABSTRACT
Prevention of influenza is the most effective management strategy. Influenza vaccine is administered each year before the influenza season. Here we provide recommendations for the use of influenza vaccines in anticipation of the 2015 Southern Hemisphere influenza season. For a review of the 2014 influenza season, please refer to the website of the National Institute for Communicable Diseases of the National Health Laboratory Service, www.nicd.ac.za
Recommended vaccine formulation
The following strains have been recommended by the World Health Organization (WHO) for the 2015 Southern Hemisphere influenza season:
- an A/California/7/2009 (H1N1)pdm09-like virus
- an A/Switzerland/9715293/2013 (H3N2)-like virus (A/South Australia/55/2014, A/Norway/466/2014 and A/Stockholm/6/2014 are A/Switzerland/9715293/2013-like viruses)
- a B/Phuket/3073/2013-like virus.
Vaccines should contain 15 μg of each haemagglutinin antigen in each 0.5 ml dose.
Indications
- Pregnant women irrespective of stage of pregnancy, or postpartum (within 2 weeks after delivery)
- Persons (adults or children) who are at high risk for influenza and its complications because of underlying medical conditions and who are receiving regular medical care for conditions such as chronic pulmonary (including tuberculosis) and cardiac diseases, chronic renal diseases, diabetes mellitus and similar metabolic disorders, individuals who are immunosuppressed (including HIV infected persons), and individuals who are morbidly obese (body mass index >40 kg/m2)
- Healthcare workers
- Residents of old-age homes and chronic care and rehabilitation institutions
- Persons aged >65 years
- Children aged 6 months - 59 months
- Persons aged 6 months to <18 years on long-term aspirin therapy
- Adults and children who are family contacts of high-risk cases
- Any persons wishing to minimise the risk of influenza acquisition, especially in industrial settings, where large-scale absenteeism could cause significant economic losses.
Older people, children aged <2 years and severely immunocompromised individuals often have lower protective immune response to trivalent influenza vaccines compared with younger healthy adults. However, even for these people influenza vaccine still provides some protection.
Dosage
- Adults: Whole or split-product or subunit vaccine: one dose intramuscularly (IM)
- Children (<12 years): Split-product or subunit vaccine: one dose IM
- Children (6 months - 8 years) who have never been vaccinated or when vaccine status is unknown: two doses, 1 month apart
- Children (6 months - 8 years) who have received two or more doses of seasonal influenza vaccine since March 2010 should receive one dose
- Children <3 years of age should receive half the adult dose on two occasions separated 1 month apart
- Influenza vaccine is not recommended for infants <6 months of age.
Contraindications
Persons with a history of severe (anaphylactic) hypersensitivity to any components of the vaccine including egg protein, or after previous dose of any influenza vaccine.
Precautions
- Persons with moderate illness with or without fever should preferably be immunised after symptoms have disappeared.
- History of Guillain-Barri syndrome within 6 weeks of receipt of influenza vaccine.
Timing
Vaccines should be given sufficiently early to provide protection for the winter. A protective antibody response takes about 2 weeks to develop.
Antiviral chemotherapy
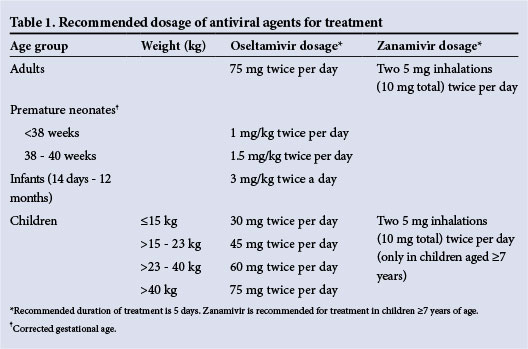
At present influenza A(H1N1)pdm09, A(H3N2) and B viruses remain sensitive to oseltamivir and zanamivir. A small proportion of A(H1N1) pdm09 viruses with highly reduced inhibition (HRI) by oseltamivir have been detected globally. High levels of resistance to adamantanes among influenza A viruses have been detected in a number of countries. The use of amantadine and rimantadine in the treatment of influenza is therefore not recommended. The dosages for treatment with oseltamivir and zanamivir are provided in Table 1.
Antiviral chemoprophylaxis
Annual influenza vaccine is the best way to prevent influenza, because it can be given in advance before the possible exposures to the influenza viruses occur and it can provide safe and effective immunity throughout the influenza season. Antiviral chemoprophylaxis for contacts of persons with influenza is currently not recommended. WHO recommendations advise presumptive treatment using the treatment regimen described below, for a duration of 7 days, for higher-risk (patients with severe immunosuppression or transplant patients) individuals exposed to influenza instead of the previously recommended long-term lower-dose chemoprophylaxis regimen. These higher-risk individuals need to be carefully monitored during the influenza season for early signs of influenza and should be treated immediately on suspicion of infection.
For a more detailed description of antiviral management and chemoprophylaxis of influenza, please refer to the Healthcare Workers Handbook on influenza on the NICD website http://www.nicd.ac.za/assets/files/Healthcare%20Workers%20Handbook %20on%20Influenza%20in%20SA%20_12%20May%202014%281%29.pdf
For the full report on recommended influenza vaccine, refer to the WHO website http://www.who.int/influenza/vaccines/virus/recommendations/201409_ recommendation.pdf?ua=1
 Correspondence:
Correspondence:
S Walaza
sibongilew@nicd.ac.za
Accepted 9 January 2014














