Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
SAMJ: South African Medical Journal
versão On-line ISSN 2078-5135
versão impressa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.104 no.12 Pretoria Dez. 2014
http://dx.doi.org/10.7196/SAMJ.8207
RESEARCH
Adolescent and young pregnant women at increased risk of mother-to-child transmission of HIV and poorer maternal and infant health outcomes: A cohort study at public facilities in the Nelson Mandela Bay Metropolitan district, Eastern Cape, South Africa
G FattiI; N ShaikhII; B EleyIII; D JacksonIV; A GrimwoodV
IMB ChB, MPH; Kheth'Impilo, 16th Floor, Triangle House, 22 Riebeek Street, Cape Town, South Africa
IIMCHD, MPH; Kheth'Impilo, 16th Floor, Triangle House, 22 Riebeek Street, Cape Town, South Africa
IIIMB ChB, BSc (Hons), FCPaed (SA); Red Cross War Memorial Children's Hospital and Department of Paediatrics and Child Health, Faculty of Health Sciences, University of Cape Town, South Africa
IVRNC, MPH, DSc; UNICEF, 3 United Nations Plaza, New York, USA; School of Public Health, Faculty of Community and Health Sciences, University of the Western Cape, Bellville, Cape Town, South Africa
VMB ChB, MPH; Kheth'Impilo, 16th Floor, Triangle House, 22 Riebeek Street, Cape Town, South Africa
ABSTRACT
BACKGROUND: South Africa (SA) has the highest burden of childhood HIV infection globally, and has high rates of adolescent and youth pregnancy
OBJECTIVE: To explore risks associated with pregnancy in young HIV-infected women, we compared mother-to-child transmission (MTCT) of HIV and maternal and infant health outcomes according to maternal age categories.
METHODS: A cohort of HIV-positive pregnant women and their infants were followed up at three sentinel surveillance facilities in the Nelson Mandela Bay Metropolitan (NMBM) district, Eastern Cape Province, SA. Young women were defined as <24 years old and adolescents as <19 years. The effect of younger maternal age categories on MTCT and maternal and child health outcomes was assessed using log-binomial and Cox regression controlling for confounding, using women aged >24 years as the comparison group.
RESULTS: Of 956 mothers, 312 (32.6%) were young women; of these, 65 (20.8%) were adolescents. The proportion of young pregnant women increased by 24% between 2009/10 and 2011/12 (from 28.3% to 35.1%). Young women had an increased risk of being unaware of their HIV status when booking (adjusted risk ratio (aRR) 1.37; 95% confidence interval (CI) 1.21 - 1.54), a reduced rate of antenatal antiretroviral therapy (ART) uptake (adjusted hazard ratio 0.46; 95% CI 0.31 - 0.67), reduced early infant HIV diagnosis (aRR 0.94; 95% CI 0.94 - 0.94), and increased MTCT (aRR 3.07; 95% CI 1.18 - 7.96; adjusted for ART use). Of all vertical transmissions, 56% occurred among young women. Additionally, adolescents had increased risks of first presentation during labour (aRR 3.78; 95% CI 1.06 - 13.4); maternal mortality (aRR 35.1; 95% CI 2.89 - 426) and stillbirth (aRR 3.33; 95% CI 1.53 - 7.25.
CONCLUSION: An increasing proportion of pregnant HIV-positive women in NMBM were young, and they had increased MTCT and poorer maternal and infant outcomes than older women. Interventions targeting young women are increasingly needed to reduce pregnancy, HIV infection and MTCT and improve maternal and infant outcomes if SA is to attain its Millennium Development Goals.
Globally, South Africa (SA) has the highest burden of childhood HIV infection and the greatest number of pregnant women living with HIV, with approximately 280 000 annually[1] needing antiretrovirals to prevent mother-to-child transmission (MTCT) of HIV. Although similar to those in other sub-Saharan African (SSA) countries, SA adolescent pregnancy rates are high compared with other world regions, with approximately 30% of all 15 - 19-year-old women reporting having ever been pregnant.[2,3] Women are at a much greater risk of acquiring HIV than men, particularly at younger ages, as young women have a range of contextual and behavioural factors increasing their risk of HIV acquisition.[4] A high proportion of the lifetime risk of acquiring HIV in women occurs while they are young.[4] Women aged 15 - 19 years have the highest incidence of HIV in SA, estimated to remain at over 2% per annum until at least 2025,[5] up to four times that of boys in the same age range. (Beyond age 50 years, HIV prevalence in women is similar to or lower than that of men.[5,6])
Maternal and perinatal outcomes in SA are poor,[7] and HIV-infected women have poorer pregnancy outcomes than HIV-uninfected women.[8] The United Nations has declared the health of adolescent girls and pregnant women to be a global public health priority.[9] There are, however, few cohort data investigating MTCT and pregnancy outcomes among young HIV-infected women and adolescents in SSA, and minimal SA cohort data from prevention of MTCT (PMTCT) programmes outside of KwaZulu-Natal Province, Cape Town and Johannesburg.
The aim of this study was to investigate the association between younger maternal age and MTCT and maternal and infant health outcomes in routine healthcare settings in the Nelson Mandela Bay Metropolitan (NMBM) district of the Eastern Cape Province.
Methods
Study design, setting and inclusion criteria
A cohort of pregnant women and their infants were followed up at three facilities offering maternal and child health services in sub-district B of the NMBM (Laetitia Bam Community Health Centre, Rosedale Community Health Centre and Uitenhage Provincial Hospital). These were sentinel surveillance facilities for evaluating the effectiveness of the PMTCT programme.[10] The facilities were supported by Kheth'Impilo, a non-profit organisation that supports the SA Department of Health. Kheth'Impilo has supported direct HIV service delivery by providing clinical staff and community-based adherence support,[10] and now includes supporting general health system strengthening and technical assistance emphasising quality improvement, human resource development, supply chain management and monitoring and evaluation.
All HIV-positive pregnant women (and their infants) who first attended the maternal facilities between 1 January 2009 and 31 March 2012 and had available maternal dates of birth and dates of first antenatal visit (booking visit) were included in the analysis. Infants and their mothers were followed up (where possible) until the infants' first HIV DNA polymerase chain reaction (PCR) test approximately 6 weeks after delivery.
Before April 2010, HIV-positive pregnant women with CD4+ cell counts <200 cells/μL or in World Health Organization (WHO) clinical stage IV were eligible to start lifelong triple antiretroviral therapy (ART). If ineligible for ART, pregnant women were to receive antenatal zidovudine (ZDV) from 28 weeks' gestation until delivery and intrapartum single-dose nevirapine (sdNVP). Infants were to receive sdNVP immediately after delivery and a 7-day course of ZDV. From April 2010, ART eligibility criteria for pregnant women were expanded to include women with CD4+ cell counts <350 cells/μL or in WHO clinical stages III or IV. Women ineligible for ART were to receive antenatal ZDV from 14 weeks' gestation and intrapartum sdNVP, as well as single-dose tenofovir/emtricitabine after delivery to cover the 'NVP tail'. Infants received an extended NVP course, the duration being dependent on the duration of breastfeeding. Antenatal and intrapartum care was provided by nurses at the community health centres. Clinical mentoring for nurses was provided by quality nurse mentors (experienced roving nurses who support nurse clinical management skills) using a data-driven approach.
Definitions and outcomes
Adolescents were defined as aged <19 years (at the first antenatal visit) and young women as aged <24 years, according to WHO definitions.[11] Older women were defined as >24 years of age.
The MTCT-related (primary) outcomes analysed were: (i) proportions of HIV-positive pregnant women who were unaware of their positive HIV status at the booking visit;[12] (ii) duration of time from booking visit until initiation of lifelong ART antenatally, i.e. rate of antenatal ART take-up;[13](iii) proportions of women who were receiving lifelong ART by delivery (initiated either before or
during pregnancy);[14] (iv) proportions of women presenting for the first time when in labour (unbooked);[15] (v) proportions of liveborn infants with available first HIV DNA PCR test results at ~6 weeks of age (uptake of early infant diagnosis of HIV (EID));[16] and (vi) proportions of positive PCR tests (vertical HIV transmission at 6 weeks).[14] Other maternal and child health (secondary) outcomes were: (i) proportions of women known to have died during the antenatal or early postnatal period (maternal mortality);[8] and (ii) proportions of stillborn infants.[8]
Data collection and statistical analysis
Enhanced routine clinical data (individual-level patient data) were collected prospectively by clinic-based data capturers in an electronic database after patient visits. Maternal HIV status and antenatal clinical details were captured from clinical files and clinic-based registers. Maternal mortality was recorded as reported to clinic staff. Infant follow-up data were sourced from child health services in the surrounding area by a PMTCT co-ordinator, as mothers would not necessarily return to the same maternal facility for child health visits. Clinical data were reviewed by quality nurse mentors as well as the district data co-ordinator before being sent to the Kheth'Impilo national office, where data from different facilities were merged.
Linear trends in maternal characteristics between women's age categories were assessed using the Cochrane-Armitage and Cuzick's non-parametric tests for categorical and continuous variables, respectively. Multivariable log-binomial regression was used to assess the effect of maternal age on binary outcomes. Kaplan-Meier curves, the log-rank trend test and multivariable Cox proportional hazards regression were used to estimate the association between maternal age and time to ART initiation during pregnancy among women who initiated ART antenatally. Women aged >24 years were used as the comparison group in all regression models. Available a priori-identified covariates considered as potential confounders that were eligible for inclusion in multivariable models were:[1314] (i) year of first antenatal visit; (ii) newly diagnosed HIV-positive; (iii) gestational age at booking; (iv) maternal CD4+ cell count at booking; (v) antiretroviral regimen at delivery; (vi) duration of receiving antiretrovirals prior to delivery; (vii) presenting for the first time during labour (unbooked); (viii) infant feeding choice; and (ix) receipt of support from a community-based adherence worker. Plausible confounding covariates that produced a >10% shift in the crude effect measure of the main exposure of interest were included in multivariable models.[17] Data were analysed using Stata version 11.1. Ethical permission for the study was granted by the University of Cape Town Human Research Ethics Committee.
Results
Database records of 1 136 HIV-positive pregnant women and their infants were reviewed for inclusion in the study; 166 who booked after 31 March 2012, 6 with missing maternal dates of birth and 8 with missing dates of first antenatal visit were excluded, leaving 956 mother-infant pairs to be included in analyses. Of the women, 312 (32.6%) were <24 years old, and among these 65 (20.8%) were adolescents (age range 13 - 19 years).
Maternal and infant characteristics are shown in Table 1, and associations between age categories and health outcomes in Table 2. There was a relative increase of 24% (from 28.3% to 35.1%) in the proportion of HIV-positive women aged <24 years between 2009/2010 and 2011/2012 (crude risk ratio (RR) 1.24; 95% confidence interval (CI) 1.02 - 1.51). The median gestational age at booking was 22 weeks (interquartile range (IQR) 17 - 26), with no difference between age categories (p=0.76). Young women were progressively more unaware of their positive HIV status at booking, with 75.3% unawareness among adolescents compared with 44.7% among older women (crude RR 1.69; 95% CI 1.43 - 1.98). Young women had higher median CD4+ cell counts at booking (378 cells/μL (IQR 256 -500) v. 339 (217 - 477) in older women; p=0.005) and lower proportions with CD4+ cell counts <200 cells/μL (14.4% v. 21.5% in older women; p=0.010). None of the adolescents were receiving lifelong ART at booking (despite 10% having CD4+ cell counts <200 cells/μL), compared with 6.9% of women aged 20 - 24 years and 21.1% of older women who were receiving ART (p<0.0001).
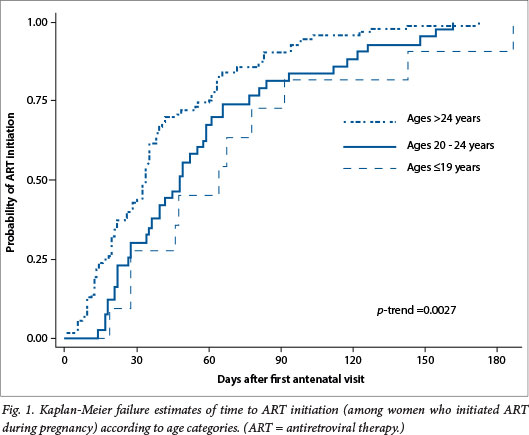
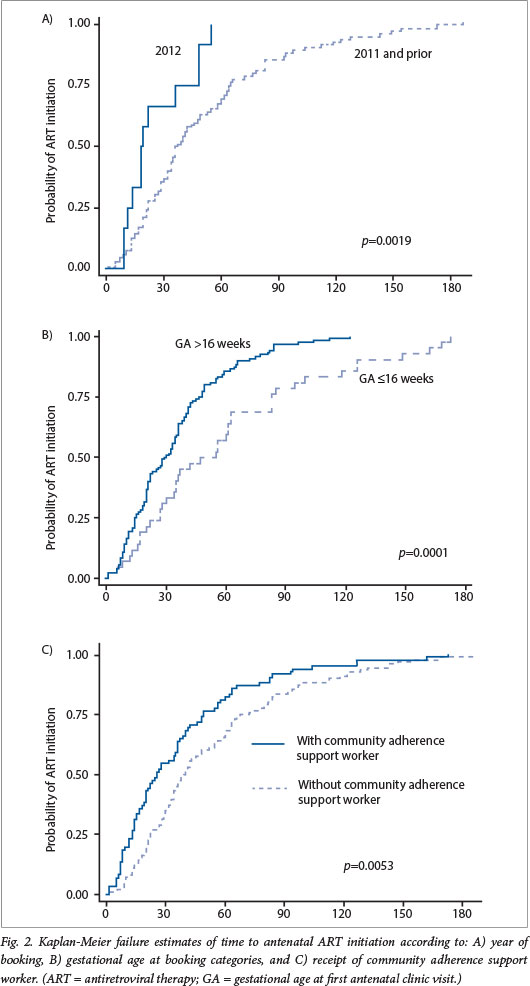
The median time between the booking visit and commencement of antenatal ZDV for PMTCT was 5 days (IQR 0 - 48), with no difference between age groups (p=0.26). The median time between booking and ART initiation during pregnancy was substantially longer in adolescents (64 days) and women aged 20 - 24 years (48 days) compared with older women (34 days). Fig. 1 shows the Kaplan-Meier estimates of time to initiating lifelong ART between booking and delivery, indicating that younger women had lower probabilities of ART uptake at each time point after booking (p-trend = 0.0027). Following adjustment, adolescents and women aged 20 - 24 years had rates of antenatal ART uptake that were more than 50% slower than that of older women (adjusted hazard ratio (aHR) 0.44; 95% CI 0.22 - 0.88 and aHR 0.46; 95% CI 0.30 - 0.69, respectively). Other findings of interest were that ART uptake was more rapid in 2012 compared with previous years (p=0.0019), ART uptake was slower in women with booking gestational ages of <16 weeks (p=0.0001), and ART uptake was more rapid among women who received support from a community-based adherence support worker (p=0.0053) (Fig. 2).
The proportion of women who were first identified as HIV-positive in late gestation (32 - 34 weeks) after testing HIV-negative at the booking visit was higher among young women (3.0% v. 1.5% in older women: crude RR 2.06; 95% CI 0.90 - 4.70).
By the time of delivery, the proportions of women who were receiving lifelong ART were 27.7%, 32.0% and 45.2% among adolescents, women aged 20 - 24 years and older women, respectively (p=0.001). Despite adjusting for CD4+ cell counts and gestational age at booking, young women had increased probabilities of not receiving lifelong ART by delivery (adjusted risk ratio (aRR) 1.37; 95% CI 1.29 - 1.45 for adolescents and aRR 1.13; 95% CI 1.05 - 1.21 for women aged 20 - 24 years).
The median duration of receiving lifelong ART prior to delivery was significantly shorter in adolescents (15.1 weeks) and women aged 20 - 24 years (14.4 weeks) compared with older women (21.1 weeks) (p<0.0001). The proportions of women who received lifelong ART for <14 weeks prior to delivery were 50.0% in adolescents, 48.3% in women aged 20 - 24 years and 29.5% in older women (crude RR 1.64; 95% CI 1.18 - 2.28 for young women v. older women). In contrast, there was no difference in the median duration of receiving ZDV for prophylaxis of MTCT before delivery according to maternal age (16.9 weeks (IQR 10 - 21.4); p=0.34).
Adolescents had a substantially increased risk of first presentation during labour (4.6% v. 1.7% in older women: aRR 3.78; 95% CI 1.06 - 13.4). In addition, women with known HIV-positive status (included in the adjusted model as a confounder) also had a higher risk of presenting for the first time during labour (aRR 2.80; 95% CI 1.06 - 7.40).
Maternal mortality was increased in adolescents (2.5% v. 0% in older women: aRR 35.1; 95% CI 2.89 - 426; p=0.005) (adjusted for baseline CD4+ cell count, ART regimen, year of booking and unbooked status). The risk of stillbirth was also substantially greater in adolescents (9.4% v. 5.0% in older women: aRR 3.33; 95% CI 1.53 - 7.25; p=0.002) (adjusted for maternal CD4+ cell count, year of booking, gestational age at booking, and receipt of community-based adherence worker). Women who were supported by community-based adherence support workers had a substantially lower risk of stillbirth (0.5% v. 5.9% among women without a community support worker: aRR=0.09; 95% CI 0.12 - 0.65; p=0.017).
HIV transmission
Four hundred and sixty-eight results of first infant HIV PCR tests were available, with overall uptake of EID of 51.4% (468/910) and overall HIV transmission of 3.4% (16/468). Young women had a slightly reduced uptake of EID (aRR 0.94; 95% CI 0.94 - 0.94). Crude vertical HIV transmission rates were 8.3%, 6.5% and 2.1% among adolescent mothers, women aged 20 - 24 years and older women, respectively (p-trend = 0.011). The majority of vertical transmissions (56.3%) occurred among young women (p=0.011), even though young women constituted only a third of the total cohort. In multivariable analyses, younger women had progressively increased risks of vertical HIV transmission (aRR 4.48; 95% CI 1.32 - 15.2; p=0.016 among adolescents and aRR 2.84; 95% CI 1.02 - 7.90; p=0.045 among women aged 20 - 24 years) (adjusted for ART regimen and unbooked status). Women who presented for the first time during labour had a greatly increased risk of vertical transmission (aRR 10.5; 95% CI 3.62 - 30.2; p<0.0001).
Discussion
We found that young women in the NMBM district in the Eastern Cape were less aware of their HIV status when booking than older women, and had slower antenatal ART uptake, reduced uptake of EID and increased MTCT of HIV (despite having less advanced immunosuppression). In addition, adolescents had increased risks of maternal mortality, first presentation in labour, and stillbirth. These findings have important public health relevance in SA, particularly as young women formed a third of all pregnant women, and increasing proportions of young women presented over time.
Younger women are probably less aware of their HIV status because they are more likely to be having their first pregnancy; as antenatal attendance is an important entry point for HIV testing, women with previous pregnancies are more likely to know their HIV status. High rates of undiagnosed HIV infection among adolescents have also been found in Zimbabwe.[18]
The increased HIV transmission in younger women is probably related to a combination of factors. Few young women became pregnant while already receiving ART (probably primarily because they were less aware of their HIV status, and secondarily, for those accessing services, because they were earlier in the course of HIV disease and so less likely to be eligible for ART). Initiation of ART during pregnancy was also slower in young women. Eligible women were referred to start ART at dedicated ART clinics at these facilities, and delays among younger women may have been related to challenges that particularly affect that age group, including concerns over confidentiality, social stigma[19] and interpersonal relational barriers with healthcare workers.[20] By the time of delivery, smaller proportions of young women were receiving ART, and for shorter durations of time. In contrast, older women were more likely to start ART before becoming pregnant, and started ART more rapidly during pregnancy. Each additional week of antenatal ART is known to significantly reduce vertical transmission.'141 Increased seroconversion during late pregnancy among young women may also play a role, as 34% of MTCT in South Africa has been estimated to be due to women seroconverting after their first antenatal visit.[21] An additional contributory factor may have been decreased adherence to antiretrovirals, as has been found among adolescents receiving lifelong ART.[22] A recently published cross-sectional survey from KwaZulu-Natal also found increased MTCT among adolescent mothers,[23] but did not analyse young women as a separate group. Further research should be conducted to establish the relative contribution of factors resulting in vertical transmission among young women.
Since April 2013, HIV-positive pregnant SA women have been eligible to start triple ART at the time of diagnosis irrespective of CD4+ cell count, and a pilot study had shown this approach to be safe and feasible and to be associated with low MTCT.[24] The results of our study suggest that programmes to reduce adolescent pregnancies, expanded adolescent HIV counselling and testing programmes (including implementation of the Integrated School Health Programme), early detection of young women who seroconvert
during pregnancy, and transforming reproductive health services to be more youth and adolescent friendly[9,10] may also lead to reduced MTCT. Early identification of young pregnant women who are HIV-positive, swift initiation of triple ART and providing youth-centred ART adherence support to these women need to be important priorities for prevention of MTCT programmes. Measures to improve EID among young mothers in particular, to identify infants eligible to start ART, also need to be prioritised. There is a critical need for sexual and reproductive health rights to be rolled out at clinics as well as at schools, with increased access to HIV counselling and testing, barrier methods and family planning.
Study strengths and limitations
A strength of the study is that it is from an under-resourced area from which there are few published data on MTCT programme outcomes. The limitations of the study include the use of routine data, and missing early infant HIV DNA PCR results that may have led to bias and reduced the precision of transmission effect measures. However, all the outcomes pointed in the same direction. Estimates of EID uptake in SA have previously been as low as 35% in 2010.[16] This highlights the difficulty of tracking mother-infant pairs in routine settings, and is due to a combination of reasons: mothers taking infants for testing at a number of different child health facilities so that infants are not able to be traced; mothers not bringing infants for testing; late testing of infants; and results not being able to be tracked from the laboratory. An additional study limitation was that accurate ART eligibility among pregnant women was not able to be ascertained, as WHO clinical stages were not captured electronically.
Conclusion
Adolescent and young pregnant women were found to have a high risk of MTCT and to have poorer maternal and infant health outcomes than older women. Programmes targeting a reduction in adolescent pregnancies, expanded adolescent HIV testing, transforming reproductive services to be more adolescent and youth friendly, and improving early infant HIV diagnosis, particularly among babies born to young mothers, may be important interventions to improve maternal and children's health outcomes in SA.
Acknowledgements. The authors thank the mothers and infants included in the study, staff at the participating clinics, the Kheth'Impilo monitoring and evaluation team, the Eastern Cape Department of Health, the President's Emergency Plan for AIDS Relief and USAID.
References
1. UNAIDS. Global Report: UNAIDS Report on the Global AIDS Epidemic 2013. Geneva: WHO, 2013. http://www.unaids.org/en/media/unaids/contentassets/documents/epidemiology/2013/gr2013/UNAIDS_Global_Report_2013_en.pdf (accessed 26 February 2014). [ Links ]
2. Willan S. A review of teenage pregnancy in South Africa - experiences of schooling, and knowledge and access to sexual & reproductive health services. 2013. http://www.hst.org.za/sites/default/files/Teenage%20Pregnancy%20in%20South%20Africa%20Final%2010%20May%202013.pdf (accessed 26 February 2014). [ Links ]
3. Khan S, Mishra V. Youth Reproductive and Sexual Health. DHS Comparative Reports No. 19. Calverton, Md, USA, 2008. http://pdf.usaid.gov/pdf_docs/PNADM644.pdf (accessed 2 March 2014). [ Links ]
4. Pettifor AE, Rees HV, Kleinschmidt I, et al. Young people's sexual health in South Africa: HIV prevalence and sexual behaviors from a nationally representative household survey. AIDS 2005;19(14):1525-1534. [ Links ]
5. Actuarial Society of South Africa. ASSA2008 AIDS and Demographic Model. 2011. http://aids.actuarialsociety.org.za/ASSA2008-Model-3480.htm (accessed 18 March 2011). [ Links ]
6. Simbayi LC, Shisana O, Rehle T, et al. South African National HIV Prevalence, Incidence and Behaviour Survey. Cape Town, 2012. http://www.hsrc.ac.za/en/research-outputs/view/6871 (accessed 15 June 2014). [ Links ]
7. Schoon M, Motlolometsi M. Poor maternal outcomes: A factor of poor professional systems design. S Afr Med J 2012;102(10):784-786. [http://dx.doi.org/10.7196/SAMJ.6130] [ Links ]
8. Rollins N, Coovadia H, Bland R, et al. Pregnancy outcomes in HIV-infected and uninfected women in rural and urban South Africa. J Acquir Immune Defic Syndr 2007;44(3):321-328. [http://dx.doi.org/10.1097/QAI.0b013e31802ea4b0] [ Links ]
9. United Nations. Global Strategy for Women's and Children's Health. New York, 2010. http://www.who.int/pmnch/topics/maternal/20100914_gswch_en.pdf (accessed 27 February 2014). [ Links ]
10. Fatti G, Meintjes G, Shea J, et al. Improved survival and antiretroviral treatment outcomes in adults receiving community-based adherence support: 5-year results from a multicentre cohort study in South Africa. J Acquir Immune Def Syndr 2012;61(4):e50-e58. [http://dx.doi.org/10.1097/QAI.0b013e31826a6aee] [ Links ]
11. World Health Organization. The Health of Youth. Document A42/Technical Discussions/2. Geneva: WHO, 1989. [ Links ]
12. Grimwood A, Fatti G, Mothibi E, et al. Progress of preventing mother-to-child transmission of HIV at primary healthcare facilities and district hospitals in three South African provinces. S Afr Med J 2012;102(2):81-83. [ Links ]
13. Stinson K, Jennings K, Myer L. Integration of antiretroviral therapy services into antenatal care increases treatment initiation during pregnancy: A cohort study. PLoS ONE 2013;8(5):e63328. [http://dx.doi.org/10.1371/journal.pone.0063328] [ Links ]
14. Hoffman RM, Black V, Technau K, et al. Effects of highly active antiretroviral therapy duration and regimen on risk for mother-to-child transmission of HIV in Johannesburg, South Africa. J Acquir Immune Defic Syndr 2010;54(1):35-41. [http://dx.doi.org/10.1097/QAI.0b013e3181cf9979] [ Links ]
15. Okeudo C, Ezem B, Ojiyi E. Unbooked status: A predictor of adverse perinatal outcome in HIV positive women at a tertiary hospital in the South Eastern Nigeria. Afrimedic J 2011;2(2):17-20. [ Links ]
16. Goga AE, Dinh TH, Jackson DJ, et al. Evaluation of the Effectiveness of the National Prevention of Mother-to-Child Transmission (PMTCT) Programme on Infant HIV Measured at Six Weeks Postpartum in South Africa, 2010. South African Medical Research Council, National Department of Health of South Africa and PEPFAR/US Centers for Disease Control and Prevention, 2012.http://www.mrc.ac.za/healthsystems/SAPMTCTE2010.pdf (accessed 17 August 2012). [ Links ]
17. Maldonado G, Greenland S. Simulation study of confounder-selection strategies. Am J Epidemiol 1993;138(11):923-936. [ Links ]
18. Ferrand RA, Munaiwa L, Matsekete J, et al. Undiagnosed HIV infection among adolescents seeking primary health care in Zimbabwe. Clin Infect Dis 2010;51(7):844-851. [http://dx.doi.org/10.1086/656361] [ Links ]
19. Forrest J, Kaida A, Dietrich J, et al. Perceptions of HIV and fertility among adolescents in Soweto, South Africa: Stigma and social barriers continue to hinder progress. AIDS Behav 2009;13(1):55-61. [http://dx.doi.org/10.1007/s10461-009-9552-z] [ Links ]
20. Alli F, Maharaj P, Vawda M. Interpersonal relations between health care workers and young clients: Barriers to accessing sexual and reproductive health care. J Commun Health 2013;38(1):150-155. [http://dx.doi.org/10.1007/s10900-012-9595-3] [ Links ]
21. Johnson LF, Stinson K, Newell M-L, et al. The contribution of maternal HIV seroconversion during late pregnancy and breastfeeding to mother-to-child transmission of HIV. J Acquir Immune Defic Syndr 2012;59(4):417-425. [http://dx.doi.org/10.1097/QAI.0b013e3182432f27] [ Links ]
22. Nachega JB, Hislop M, Nguyen H, et al. Antiretroviral therapy adherence, virologic and immunologic outcomes in adolescents compared with adults in southern Africa. J Acquir Immune Defic Syndr 2009;51(1):65-71. [http://dx.doi.org/10.1097/QAI.0b013e318199072e] [ Links ]
23. Horwood C, Butler LM, Haskins L, et al. HIV-infected adolescent mothers and their infants: Low coverage of HIV services and high risk of HIV transmission in KwaZulu-Natal, South Africa. PLoS ONE 2013;8(9):e74568. [http://dx.doi.org/10.1371/journal.pone.0074568] [ Links ]
24. Black S, Zulliger R, Myer L, et al. Safety, feasibility and efficacy of a rapid ART initiation in pregnancy pilot programme in Cape Town, South Africa. S Afr Med J 2013;103(8):557-562. [http://dx.doi.org/10.7196/SAMJ.6565] [ Links ]
 Correspondence:
Correspondence:
G Fatti
geoffrey.fatti@khethimpilo.org
Accepted 14 August 2014














