Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.104 n.4 Pretoria Apr. 2014
CONTINUING MEDICAL EDUCATION
ARTICLES
T MorgadoI; A FigajiII
IMB ChB, MRCS (Eng). Division of Paediatric Neurosurgery, University of Cape Town and Red Cross War Memorial Children's Hospital, Cape Town, South Africa
IIMB ChB, PhD, FCNeurosurg (SA). Division of Paediatric Neurosurgery, University of Cape Town and Red Cross War Memorial Children's Hospital, Cape Town, South Africa
ABSTRACT
Hydrocephalus is one of the most common complications of spinal dysraphism. Although few patients require cerebrospinal fluid diversion immediately at birth or within the first few days of life, most patients with myelomeningocele, which comprises the most prevalent, clinically significant form of spina bifida, will eventually need surgical treatment for hydrocephalus at some point following closure of the spinal defect. Furthermore, symptomatic hydrocephalus needs to be dealt with timeously, as these patients not only face the usual ill-effects of raised intracranial pressure (ICP), but also have an increased risk of breakdown of the myelomeningocele repair. Poorly treated ICP may also cause the Chiari II malformation to become symptomatic.
Incidence
Hydrocephalus is one of the most common complications of spinal dysraphism.[1] It is associated mainly with the open form of spina bifida, and this article focuses specifically on this group of patients.
Most children who develop symptomatic hydrocephalus do so sometime within the first few days or weeks following myelomeningocele repair, with >80% ultimately requiring a surgical procedure to treat the condition.[2-4]
Pathogenesis
The underlying developmental abnormalities and the exact pathogenesis of hydrocephalus remain subject to debate. Children with this condition nearly always have a Chiari II malformation, which is characterised by an abnormal posterior fossa and herniation of the cerebellar vermis across the foramen magnum. Several other structural and developmental abnormalities are usually present in association with the Chiari II malformation, but the hindbrain hernia is the most clinically important manifestation. Fortunately, the Chiari II malformation is rarely overtly symptomatic, especially if raised intracranial pressure (ICP) is avoided; however, when it is symptomatic serious lower cranial nerve and respiratory disturbances may occur. The exact causal relationship between the Chiari II malformation and hydrocephalus has only recently begun to be well understood.[5-7] Recent data show a decrease in the rate of Chiari II malformation and subsequent hydrocephalus in patients in whom the spinal defect is repaired in utero.[8-10] However, this remains controversial. The open system of cerebrospinal fluid (CSF) circulation resulting from open spinal dysraphism (with fluid production in the ventricular system and passage down and out through the spinal defect) is thought to play a central role in hindbrain descent and subsequent hydrocephalus. This alone, however, can not explain a number of the other CSF-flow pathological conditions thought to occur in myelomeningocele. Several mechanisms for the development of hydrocephalus have been proposed, but in reality some of these probably co-exist in different patients.
Clinical features
Careful physical examination with a high index of suspicion for hydrocephalus is essential in managing these patients.[3,11,12]
Ventriculomegaly may be diagnosed antenatally or postnatally with various imaging modalities. However, by definition, hydrocephalus implies a dynamic and progressive disorder associated with raised ICP. Daily clinical assessment involves examination of the fontanelle and measurement of the head circumference. Symptoms such as poor feeding, lethargy or drowsiness should raise suspicion of a developing hydrocephalus.
Measurement of head circumference is crucial in diagnosing developing hydrocephalus in these children, as the presence of an open fontanelle and sutures may sometimes mask the more obvious signs of raised ICP, and result in a surprisingly clinically healthy child despite significant pathology. Daily measurement of head circumference with the results clearly displayed on a chart at the head of the patient's bed provides a cheap, effective and safe way to detect developing pathology. If the patient does not develop hydrocephalus in the first week or two after closure of the myelomeningocele, measurement of head circumference on an outpatient basis, and plotting these values on standard charts, is continued. If head circumference measurements cross the centiles, surgical treatment is likely needed.
Investigations of hydrocephalus in myelomeningocele are summarised in Table 1.

Prenatal diagnosis
High-resolution fetal ultrasound can reliably detect hydrocephalus in utero, assessing not just ventricular size but also typical skull vault abnormalities suggestive of the condition. Apart from being sensitive, this investigation is also non-invasive and cost-effective, and head circumference can be monitored on serial ultrasound. Because CSF passes through the open distal placode, even though ventriculomegaly may be present, progressive increased ICP is rarely a problem prenatally.
Although more costly, fetal magnetic resonance imaging (MRI) is another non-invasive option in cases where ultrasound is not diagnostic or inconclusive. This investigation provides information on the state of the ventricles, and also details a number of other anatomical abnormalities of the brain and spine that may be associated with myelomeningocele (Fig.1).
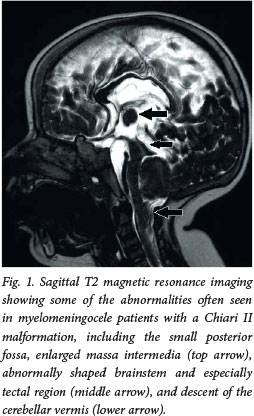
Postnatal diagnosis
Cranial ultrasonography at birth is the usual method for diagnosing ventriculomegaly in the neonate. It is cheap, non-invasive, and technically and logistically easier to perform than MRI. The latter is seldom required prior to myelomeningocele repair, but may be useful in the postoperative period, providing very detailed information on both intracranial and spinal pathology. Computed tomography (CT) remains one of the most sensitive, widely available modalities to confirm hydrocephalus and assist with treatment planning (Figs 2 and 3). However, there is growing concern about the radiation effects of CT on the developing brain in terms of cognitive development and induction of tumours.
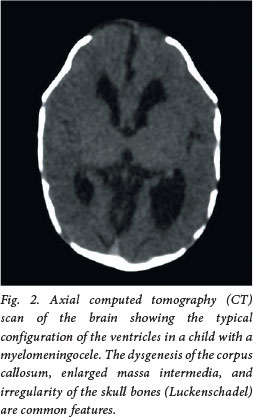
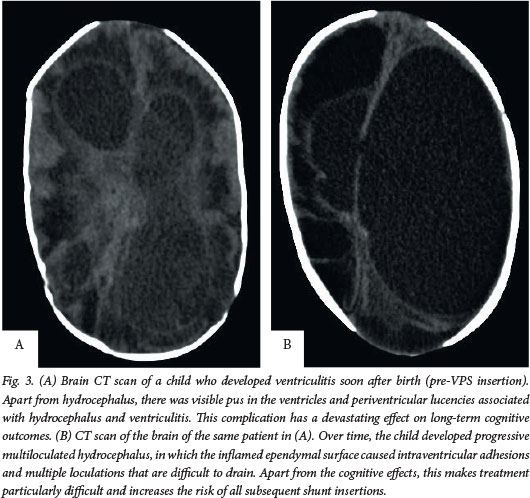
Treatment options
The two main recognised definitive procedures used to treat hydrocephalus in myelomeningocele patients are ventriculoperitoneal shunt (VPS) insertion and endoscopic third ventriculostomy (ETV).[2,11,13] Although hydrocephalus is common and may be difficult to treat, if patients are managed well and especially if ventriculitis can be avoided, most of these children can achieve a fairly good cognitive outcome.
VPS
VPS insertion remains the gold standard for treatment. The timing of VPS insertion is debatable and numerous authors suggest that VPS insertion at the same time as myelomeningocele closure increases the risk of shunt infection, which may progress to one of the most devastating complications in the management of hydrocephalus in these patients.[11,14,15] However, others report no significant difference in the rate of shunt infection and dysfunction.[16] As it is not always apparent which patients will develop hydrocephalus, it would seem prudent to wait with VPS insertion. The key point is to manage hydrocephalus as soon as it becomes clinically apparent, as raised CSF pressure in these patients not only carries the usual deleterious effects on the developing brain but also increases the rate of myelomeningocele repair breakdown with a fluid leak and consequent ventriculitis. The latter is devastating in terms of later cognitive deficits. At our centre, we prefer to delay VPS insertion while carefully monitoring the development of hydrocephalus.
Ventricular tapping, external ventricular drainage, and subgaleal shunt insertion may temporise hydrocephalus to delay VPS insertion if there is breakdown or infection of the myelomeningocele closure or evidence of a CSF infection.
Although the most commonly used treatment option, VPS insertion is not without complications, and in these patients is known to have a significant failure rate, particularly related to infective complications.[15,17] The open nature of myelomeningocele (with the potential for CSF contamination via the defect) increases the risk of shunt infection. Central nervous system infection in this young patient group is known to have potentially devastating and profound consequences with regard to neurological development and long-term outcome, which is of deep concern. It is therefore advised that VPS insertion is carried out in a meticulous fashion, with particular attention to preventing infection. Standardised VPS insertion protocols have been shown to decrease the rate of VPS infections, and we recommend that such a protocol be adhered to in any VPS procedure - even more so in this scenario.[18]
Once a shunt has been placed, the later development of symptoms or signs of the Chiari malformation or syringomyelia should raise suspicion of shunt failure. Although the Chiari malformation may appear dramatic on MRI, it rarely becomes symptomatic if the shunt is functioning well.
ETV
Over the past two decades ETV has gained popularity as a treatment option for non-communicating hydrocephalus. This modality carries a much smaller infection risk and is not dependent on extracranial mechanical drainage; therefore, it is seen by many as a more attractive treatment option. In patients with clear non-communicating hydrocephalus, it is the surgical treatment of choice. In myelomeningocele, however, there remain some challenges in using this as a first-line option, and it continues to carry a significant failure rate.
There are reports in the literature of successful management of hydrocephalus in these patients with ETV combined with choroid plexus cauterisation, particularly in the Third World setting, where reliable access to follow-up for VPS and management of the possible complications may not exist.[13] However, this dual approach remains contentious and is not widely adopted. The floor of the third ventricle (where the endoscopic stoma is made) is often abnormal in these patients, there is usually an element of communicating hydrocephalus, and the failure rate of ETV is higher in infants than in older children. Optimisation of hydrocephalus control for the best clinical and cognitive outcomes, especially where there is hindbrain herniation, remains controversial. At our institution we continue to place a VPS for hydrocephalus in myelomeningocele. For older patients who develop shunt failure, ETV may be an option.
Even if VPS or ETV has been successful in treating hydrocephalus in these patients, it is important to maintain close outpatient follow-up, as one should always remain vigilant in case of failure of either procedure. Initially, 3-monthly follow-up is recommended. Once patients are stable, yearly follow-up, preferably in a spinal defects clinic, is ideal. The patient's general practitioner should also be aware of the risk of developing shunt/ETV failure and have a low threshold for specialist referral for its assessment.
Conclusion
• Hydrocephalus in myelomeningocele is extremely common (>80%).
• Early treatment of hydrocephalus is crucial.
• VPS remains the surgical treatment of choice.
• Timing of shunt insertion is debatable.
• Shunt infection is a problem in these patients
References
1. Pang D. Surgical complications of open spinal dysraphism. Neurosurg Clin N Am 1995;6(2):243-257. [ Links ]
2. Stein SC, Schut L. Hydrocephalus in myelomeningocele. Childs Brain 1979;5(4):413-419. [ Links ]
3. Steinbok P, Irvine B, Cochrane DD, Irwin BJ. Long-term outcome and complications of children born with meningomyelocele. Childs Nerv Syst 1992;8(2):92-96. [http://dx.doi.org/10.1007/BF00298448] [ Links ]
4. Mirzai H, Erçahin Y, Mutluer S, Kayahan A. Outcome of patients with meningomyelocele: The Ege University experience. Childs Nerv Syst 1998;14(3):120-123. [http://dx.doi.org/10.1007/s003810050192] [ Links ]
5. McLone DG, Knepper PA. The cause of Chiari II malformation: A unified theory. Pediatr Neurosci 1989;15(1):1-12. [http://dx.doi.org/10.1159/000120432] [ Links ]
6. McLone DG, Dias MS. The Chiari II malformation: Cause and impact. Childs Nerv Syst 2003;19(7-8):540-550. [http://dx.doi.org/10.1007/s00381-003-0792-3] [ Links ]
7. Williams HA. Unifying hypothesis for hydrocephalus, Chiari malformation, syringomyelia, anencephaly and spina bifida. Cerebrospinal Fluid Res 2008;5:7. [http://dx.doi.org/10.1186/1743-8454-5-7] [ Links ]
8. Bruner JP, Tulipan N. Intrauterine repair of spina bifida. Clin Obstet Gynecol 2005;48(4):942-955. [http://dx.doi.org/10.1097/01.grf.0000184799.17975.e9]
9. Tulipan N, Hernanz-Schulman M, Lowe LH, Bruner JP. Intrauterine myelomeningocele repair reverses preexisting hindbrain herniation. Pediatr Neurosurg 1999;31(3):137-142. [http://dx.doi.org/10.1159/000028849] [ Links ]
10. Tulipan N, Sutton LN, Bruner JP, Cohen BM, Johnson M, Adzick NS. The effect of intrauterine myelomeningocele repair on the incidence of shunt-dependent hydrocephalus. Pediatr Neurosurg 2003;38(1):27-33. [http://dx.doi.org/10.1159/000067560] [ Links ]
11. Tamburrini G, Frassanito P, Iakovaki K, et al. Myelomeningocele: The management of the associated hydrocephalus. Childs Nerv Syst 2013;29(9):1569-1579. [http://dx.doi.org/10.1007/s00381-013-2179-4] [ Links ]
12. Elgamal EA. Natural history of hydrocephalus in children with spinal open neural tube defect. Surg Neurol Int 2012;3:112. [http://dx.doi.org/10.4103/2152-7806.101801]
13. Warf BC, Campbell JW. Combined endoscopic third ventriculostomy and choroid plexus cauterization as primary treatment of hydrocephalus for infants with myelomeningocele: Long-term results of a prospective intent-to-treat study in 115 East African infants. J Neurosurg Pediatr 2008;2(5):310-316. [http://dx.doi.org/10.3171/PED.2008.2.11.310]
14. Ersahin Y, McLone DG. Repeat cerebrospinal fluid shunt infection: Recurrence, relapse, repeat or reinfection? Pediatr Neurosurg 2002;36(3):167. [http://dx.doi.org/10.1159/000048376] [ Links ]
15. Arslan M, Eseoglu M, Gudu BO, et al. Comparison of simultaneous shunting to delayed shunting in infants with myelomeningocele in terms of shunt infection rate. Turk Neurosurg 2011;21(3):397-402. [ Links ]
16. Radmanesh F, Nejat F, El Khashab M, Ghodsi SM, Ardebili HE. Shunt complications in children with myelomeningocele: Efect of timing of shunt placement. Clinical article. J Neurosurg Pediatr 2009;3(6):516-520. [http://dx.doi.org/10.3171/2009.2.PEDS08476] [ Links ]
17. Caldarelli M, Di Rocco C, La Marca F. Shunt complications in the irst postoperative year in children with meningomyelocele. Childs Nerv Syst 1996;12(12):748-754. [http://dx.doi.org/10.1007/BF00261592] [ Links ]
18. Kestle JR, Riva-Cambrin J, Wellons JC 3rd, et al. A standardized protocol to reduce cerebrospinal fluid shunt infection: The Hydrocephalus Clinical Research Network Quality Improvement Initiative. J Neurosurg Pediatr 2011;8(1):22-29. [ Links ]
 Corresponding author:
Corresponding author:
T Morgado
(tiagoclementemorgado@gmail.com)














