Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
SAMJ: South African Medical Journal
versión On-line ISSN 2078-5135
versión impresa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.104 no.1 Pretoria ene. 2014
GUIDELINE
South African guideline for the use of chronic opioid therapy for chronic non-cancer pain
M RaffI; J CrosierII; S EppelIII; H MeyerIV; B SarembockV, VI; D WebbVII
IBSc, MB ChB, FCA (SA). Christiaan Barnard Memorial Hospital, Cape Town, South Africa
IIMB ChB, ChM, FRCS, FCS (SA). Christiaan Barnard Memorial Hospital, Cape Town, South Africa
IIIMB ChB, FRCS (Edin), ABU (USA). Christiaan Barnard Memorial Hospital, Cape Town, South Africa
IVMB ChB, MPraxMed, MFGP (SA), FCFP (SA) Department of Family Medicine, Faculty of Health Sciences, University of Pretoria, South Africa
VMB ChB, FCP (SA), RHEUM (SA). Christiaan Barnard Memorial Hospital, Cape Town, South Africa
VIMB ChB, FCP (SA), RHEUM (SA) Rheumatic Diseases Unit, Groote Schuur Hospital, Cape Town, South Africa
VIIBSc (Hons), MB ChB. Pattacus Medical Consulting, Johannesburg, South Africa
ABSTRACT
Chronic pain may have a significant impact on health-related quality of life and can be difficult to manage. In carefully selected patients, and as part of a comprehensive pain management strategy, opioid analgesia may help to achieve long-term pain control with a manageable side-effect profile and a low risk of serious adverse effects. However, appropriate evaluation, including biopsychosocial screening and risk screening is essential before initiating an opioid and during continued therapy. This guideline aims to assist practitioners in screening and selecting appropriate patients with chronic non-cancer pain to initiate, monitor and continue pain management with opioid therapy.
1. Introduction
Chronic pain is common, affecting around one in five patients in primary care. It may occur even more frequently in older individuals, whose presentation is often complicated by age-related physiological changes, comorbidities and multiple medications. Chronic pain patients are more likely to report anxiety or depression and significant activity limitations and often have unfavourable perceptions of their health.[1] Chronic pain may have a significant impact on health-related quality of life and may be difficult to manage.
Opioids are well accepted for the treatment of severe acute pain and chronic pain associated with cancer and at the end of life. Although there are short-term studies demonstrating efficacy in chronic non-cancer pain (CNCP), less is known about their efficacy and safety with long-term use. The potential for addiction, tolerance and dependence associated with this class of analgesics also remains a concern.
Nevertheless, opioids are increasingly being used to treat persistent pain. Limited evidence indicates that they can be effective therapy for a carefully selected group of patients as part of a wider management plan focused on reducing disability and improving quality of life. However, appropriate patient selection is paramount, requiring a comprehensive physical and biopsychosocial assessment to establish the diagnosis and to guide management decisions.
This guideline was developed by a multidisciplinary panel of clinicians who treat patients with chronic pain to provide recommendations for patient selection and the use of opioids for CNCP.
2 Objectives
To provide a brief and practical guideline for the use of chronic opioid therapy (COT) in patients with CNCP. The target audience is all clinicians in primary and specialty settings who provide care for adults suffering from CNCP. The management of acute pain, postsurgical pain, labour pain, cancer pain, pain at the end of life, and CNCP in children is not addressed in this guideline.
This is a guideline only and is not intended to constitute inflexible treatment recommendations or to represent the standard of care. The recommendations here may not apply to all patients or all clinical situations and shared decision making among a multidisciplinary treatment team is encouraged.
3 Methods
International guidelines, reviews and recommendations for the use of opioids in patients with CNCP were identified through a literature search and reviewed by the panel. A consensus guideline for South Africa (SA) was developed from these publications and the clinical experience of the physicians on the panel. As a basis for the document, four international guidelines were specifically chosen. These were recommendations from the British Pain Society (2010), the American Pain Society -American Academy of Pain Medicine Opioids Guidelines Panel (2009), the Canadian National Opioid Use Guideline Group (2010) and the American Society of Interventional Pain Physicians (2012).[2-5]
4 Definition of chronic pain
Chronic pain is defined as pain persisting for longer than 90 days and beyond an expected time frame for tissue healing. It may be continuous or intermittent and may continue in the presence or absence of demonstrable pathologies. Chronic pain may not be amenable to routine pain control methods and complete resolution of the pain may never occur.
5. CNCP requires a multimodal management approach
CNCP is a complex condition that may involve biological, psychological, social and environmental factors. When chronic pain is accompanied by comorbidities, impaired function, or psychological disturbances (e.g. anxiety and depression), COT is likely to be most effective as part of multimodal treatment that addresses all of these domains.
Consequently, management of CNCP requires a multidisciplinary approach and clinicians who prescribe COT should routinely consider non-opioid therapies (e.g. analgesics, antidepressants, anxiolytics), psychotherapeutic interventions, functional restoration and other medical and social interventions as indicated (Fig. 1).
6 Communication and responsibility
Communication between healthcare providers caring for the patient is mandatory. However, patients on COT should identify with one doctor, mostly their general practitioner and not necessarily the one prescribing COT, who accepts primary responsibility for their overall medical care and who should co-ordinate communication and consultation among clinicians. Patients should be referred back to the general practitioner after specialist consultation. Likewise, to help ensure correct use of medication and to reduce prescription fraud, all opioids (and preferably all other medications) for a particular patient should be dispensed through a single designated pharmacy.
7 Goals of therapy and patient expectations
Complete relief of pain is rarely achieved in patients with CNCP and the physician should ensure that the patient's expectations of therapy are realistic. The goals of management are to reduce pain intensity and to improve the patient's functional levels in relation to physical, vocational, social and emotional wellbeing. Clinically meaningful improvements are at least 30% reduction in pain (or >2 points on a 0 - 10 numeric rating scale) and/or 30% improvement in function.
COT forms only one component of the broad approach required to achieve this.
8 Opioid pharmacology
Opioid drugs are agonists that bind to endogenous opioid receptors and mimic the actions of natural morphine-like ligands. The receptors are widespread throughout the central and peripheral nervous system and include µ, δ and κ subtypes. Although the various opioid drugs demonstrate different specificities for receptor subtypes, the clinical relevance of this is uncertain.
Opioids are classified as strong or weak. Formulations that are available in SA and that may be appropriate for the treatment of chronic pain are listed in Table 1.
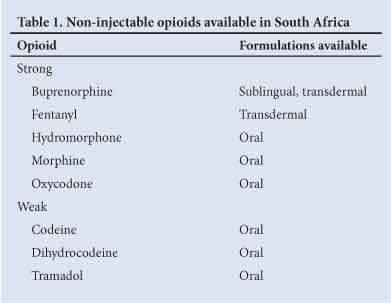
Oral formulations may be immediate release (IR) (short-acting) or modified release (long-acting).
9 Practical aspects of prescribing
9.1 General principles
- COT is defined as regular use of strong opioids for at least 3 months.
- COT may be appropriate for carefully selected patients where chronic pain significantly affects function and/or quality of life.
- Opioids should never be the first-line choice for analgesia in patients with CNCP, but may be included in a pain management strategy for moderate-to-severe chronic pain when optimal use of first-line therapy has failed.
9.2 Indications for opioid therapy in CNCP
- Types of pain that might respond to opioids include those of both nociceptive and neuropathic (pain related to nervous system injury or disease) origin.
- Opioids are not appropriate as the primary medication of choice for pain disorders with strong psychosocial contributing factors (Table 2).

9.3 Patient selection and risk stratification
Before initiating COT, it is necessary to:
- establish a diagnosis and the cause of the pain
- estimate the pain intensity and functional impairment (Brief Pain Inventory, Appendix 1)
- risk stratify the patient in terms of potential benefits and harms of opioid therapy to assess their suitability for COT (Opioid Risk Tool, Appendix 2).
In all patients this requires:
- a thorough history and physical examination
- assessment of psychosocial factors and family history
- appropriate special investigations.
This assessment should be repeated at every visit, especially when there are new signs or symptoms or where pain intensity changes or is unresponsive to treatment. Useful tools that the patient can complete to assist in the initial and subsequent assessments are listed in Table 3.
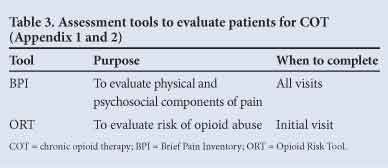
Every assessment must be documented by all members of the team to ensure ongoing quality of care.
The presence of increased risk of addiction, or psychiatric or psychological comorbidity does not necessarily preclude COT, but indicates the need for additional consultation and monitoring.
Consultation with an experienced pain clinician and involvement of a multidisciplinary team should be considered before initiating COT in this category of patients. In some patients CNCP may cause comorbid depression, and effective pain management may be associated with an improvement in mood.
9.4 I nformed consent, opioid management plans and documentation
The following issues need to be discussed with patients before initiating therapy with COT and at regular intervals thereafter:
- treatment plan
- goals of the pain management strategy
- the patient's expectations from treatment
- potential benefits and risks of treatment
- alternatives to COT.
Detailed notes of all assessments, results of special investigations, discussions with the patient and caregivers, and treatment decisions should be maintained. Clinicians may consider the use of a written COT informed consent form (opioid agreement), which provides a record of the opioids prescribed and acknowledges discussion about the risks and benefits of COT. An example of an opioid agreement that may be adapted for individual patients is illustrated in Appendix 3.
9.5 Choice of opioid
- Opioid analgesics for CNCP may be administered orally or transdermally.
- Opioid selection should be individualised according to health status, pain severity, previous exposure to opioids, attainment of therapeutic goals and predicted or observed harms.
- Injectable opioids, including pethidine, should never be used for the management of CNCP.
9.5.1 Step-wise therapy
Mild-to-moderate pain:
- first-line options: codeine or tramadol
- second-line options: morphine, dihydrocodeine, low-dose oxyco-done, hydromorphone or buprenorphine.
Severe pain:
- first-line options: morphine, high-dose oxycodone, hydromorphone or buprenorphine
- second-line option: fentanyl.
9.5.2 Oral opioids
- There are no significant differences between long-acting and short-acting opioids in their analgesic efficacy or adverse effects.
- IR oral opioids are useful when initiating therapy and titrating to an effective dose. However, for patients needing continuous analgesia, long-acting (controlled release (CR), extended release (ER), sustained release (SR)) opioids administered at regular intervals should be used.
- In contrast to IR preparations, the dosing of long-acting formulations is more convenient and they are less likely to be associated with tolerance and problematic drug use.
- High doses of long-acting opioids are recommended only for severe intractable pain in consultation with an experienced pain clinician and/or a multidisciplinary team.
9.5.2.1 Codeine
Codeine is a prodrug that is converted to morphine, which is responsible for its opioid effects. Approximately 10% of Caucasians lack the enzyme required for this metabolic conversion and caution must be applied when switching from codeine to a potent opioid as patients may have little or no opioid tolerance.
9.5.2.2 Tramadol
In addition to its agonist effect at the µ-opioid receptor, tramadol has noradrenergic and serotonergic effects. Even after short periods of dosing and when used in its SR formulation, it may be associated with significant withdrawal effects. These frequently occur within 12 - 24 h of the last dose.[2] Furthermore, it may increase the risk of seizures in patients at high seizure risk. Because of a risk of serotonin syndrome, concomitant use of antidepressants should be closely monitored. Tramadol has a significantly lower abuse risk than more potent opioids and a low potential for addiction.
9.5.3 Combination formulations and over-the-counter medicines
- In SA, many codeine combination drugs are available, and they are often abused. The amount of codeine in combination products is low and, when used at normal prescribed doses, will not have a significant therapeutic analgesic effect.
- Paracetamol is a frequent component of combination analgesics and, in high doses, may cause hepatotoxicity. The risk increases with alcohol use (regular or binge drinking).
- Patients should be advised that the use of non-prescribed combination therapy and over-the-counter (OTC) medication should be avoided while on COT.
9.6 Initiation and titration of COT
Opioids should be started at a low dose of ~10 mg/day oral morphine equivalents and slowly titrated upwards according to analgesic effect and tolerability. Reassess the dose after 72 h, taking into consideration:
- the analgesic effect
- patient's return to everyday activities
- adverse drug effects.
It may be appropriate to do this telephonically if the patient is being treated at home.
Depending on the response, the dose may be increased by not more than 10 mg/day oral morphine equivalents to a maximum of 90 mg/day oral morphine equivalents over a period of 4 - 6 weeks. Patients should be warned that it might take some days to determine whether the analgesic will be effective.
An initial course of COT should be viewed as a short-term therapeutic trial:
- if there is absolutely no clinical response to a dose of 30 - 40 mg, further escalation of the dose is probably inappropriate
- if the clinical response to the above dose is insufficient, the dose may be gradually increased as indicated above
- if there is no response to doses of up to 90 mg/day, reconsider whether an opioid is appropriate therapy and refer to a pain clinician and/or multidisciplinary team.
When deciding whether to continue therapy, also consider opioid-related adverse effects, changes in the underlying pain, psychiatric or medical comorbidities and the emergence of drug abuse behaviours. If a decision is made to discontinue opioid therapy, the dose should be discontinued by gradual tapering.
Suggested doses for initiation and titration of opioids are listed in Table 4.
9.6.1 Concomitant medications
- Before prescribing an opioid, consider concomitant medications that have the potential for pharmacokinetic or pharmacodynamic drug interactions.
- For patients taking benzodiazepines, consider a trial of tapering and discontinuation. If this is not possible, opioids should be initiated at lower doses and titrated more slowly.
9.6.2 Elderly patients
- Prescribing opioids for elderly patients should take into account relevant age-related changes in pharmacokinetics and pharmacodynamics. Lower starting doses (50% of the usual dose), slower titration, longer dosing intervals and more frequent assessment are appropriate.
- Opioids should be avoided in cognitively impaired patients living alone, unless ongoing supervision can be arranged.
- Oxycodone and hydromorphone may be preferred to oral morphine in elderly patients, because they are less likely to cause severe sedation.
9.6.3 Adolescent patients
- The potential for addiction is higher in young people, and opioids represent a hazard to adolescents.
- Opioid use should be considered carefully for well-defined indications when non-opioid alternatives have failed.
- Patients should be monitored frequently.
9.6.4 Pregnancy and breastfeeding
- Opioids should not be used during pregnancy.
- Patients who are pregnant or breastfeeding should be referred to a pain clinician.
9.6.5 Psychiatric disorders
- Patients on COT have a higher prevalence of depression and other psychiatric conditions than the general population.
- Due to diminished response, or enhanced perception of pain, or both, patients with depression or anxiety are less likely to benefit from opioids than those without psychiatric disorders.
9.6.6 Addicted patients
- Patients with CNCP who are addicted to opioids should be referred to an experienced pain clinician. A multidisciplinary management approach is required, including a psychologist who has experience in treating addicted patients.
- Buprenorphine is a partial µ-receptor agonist with a long duration of action and is probably safer for opioid-addicted patients requiring COT.
9.7 Monitoring
Clinicians should regularly reassess all patients on COT, with special attention to the '5 As':
- analgesia (pain relief)
- activity (improvement in function)
- adverse effects
- affect (mood/psychological comorbidities)
- aberrant behaviour (problem drug use).
If necessary, compliance with therapy may be confirmed with urine screening and/or pill counting.
9.8 Dose escalation and switching to another opioid
When repeated dose escalations are required, the patient should receive a thorough physical and biopsychosocial assessment to determine the underlying reason. Increasing doses of COT may be indicative of:
- an increase in the intensity of the underlying pain condition (disease progression)
- development of an additional painful condition
- opioid tolerance
- opioid-induced hyperalgesia
- problem drug use/abuse.
When analgesia is inadequate despite opioid dose escalation, or where adverse effects are intolerable at higher doses, consider discontinuing COT and/or switching to an alternative opioid.
9.8.1 Switching to another oral opioid
Equianalgesic doses of alternative oral opioids are listed in Table 5. The new opioid should be initiated at 50% of the calculated equianalgesic dose. If converting from opioids other than morphine, calculate the equipotent morphine dose for the current analgesic and use this dose to convert to the new opioid.
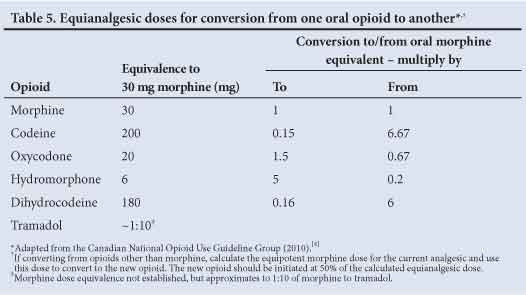
9.8.2 Switching from an oral to a transdermal opioid
Patients with chronic stable pain may be switched from an oral to a transdermal opioid patch. Table 6 shows approximate equipotent analgesic doses for conversion from oral morphine to transdermal fentanyl or buprenorphine. If dose titration is required to achieve pain control, the strength (opioid dose) of the patch may be increased, or a combination of lower strength patches may be applied at different places to achieve pain control.
The fentanyl patch may be titrated every 72 h and buprenorphine patch every 3 - 5 days.
Buprenorphine is a partial opioid receptor agonist; therefore withdrawal symptoms may be experienced in patients who have developed physical dependence on opioids. In overdose it is only partially reversed by naloxone.[6]
9.9 Breakthrough pain
Breakthrough pain is defined as infrequent short episodes of increased pain occurring on a background of stable well-controlled pain. It must be differentiated from incident pain associated with a particular movement, posture or activity, and additional diagnostic testing or consultation may be necessary to identify its aetiology or precipitating factors.
In patients on continuous opioid therapy who experience breakthrough pain, as-needed IR opioids may be useful for rescue analgesia. However, benefits and potential risks should be carefully considered before prescribing the as-needed rescue opioid and suitability of non-opioid drug therapies and non-pharmacological treatment options should be investigated first.
When regular rescue doses are required, the maintenance opioid dose should be up-titrated by adding the extra dose per day required for breakthrough pain.
For example, a patient receiving oral morphine 10 mg 4-hourly and requiring rescue doses for breakthrough pain (10 mg) three times daily, will be converted to a new daily dose as follows:
(maintenance: 10 mg x 6) + (breakthrough: 10 mg x 3) = 90 mg daily
Once pain control is stable, this dose may be administered as:
- IR oral morphine 15 mg every 4 h, or
- an equivalent dose of 12-hourly long-acting morphine twice daily plus appropriate doses of IR morphine for breakthrough pain.
Because of their long half-life and prolonged time to reach steady state, transdermal opioids are not appropriate as rescue therapy for breakthrough pain.
9.10 Long-term opioid prescribing
When a trial of opioid has been successful, treatment may be continued until:
- the underlying painful condition resolves
- the patient receives a definitive pain-relieving intervention (e.g. joint replacement)
- the patient no longer derives benefit from opioid treatment
- the patient develops intolerable side-effects, or
- there is evidence of addiction, tolerance, dependence or opioid-induced hyperalgesia.
9.11 Discontinuing opioids
- When discontinuing an opioid, the dose should be slowly reduced by approximately 10% per day or per week.
- Dose should be tapered more slowly in patients who are anxious about discontinuing COT and in those who are suspected of being physically dependent on opioids.
- When one-third of the original dose is reached, reduce the rate of tapering to a half or less of the initial rate.
- If the patient experiences withdrawal symptoms or an increase in pain during tapering, discontinue dose reduction and consider increasing the current dose.
10. Adverse effects of opioid therapy
Fear of adverse effects should not interfere with appropriate use of COT in patients who need it, and they should be reassured that side-effects are manageable.
10.1 Common adverse effects
Treatment-related side-effects are common on initiation of opioid therapy and patients should be advised about the likelihood of their occurrence before starting treatment. The most frequent adverse events include:
- constipation
- nausea
- somnolence
- itching
- dizziness.
With the exception of constipation and pruritus, which are often persistent, tolerance usually develops within a few days of continuous dosing. Management approaches for emergent side-effects are listed in Table 7.
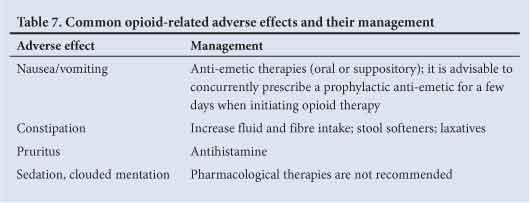
If side-effects are intolerable and the dose of the current treatment cannot be reduced, it may be beneficial to switch to an alternative opioid.
10.2 Respiratory depression
Respiratory depression is seldom a problem associated with the use of opioids for chronic pain, except where there are major changes in opioid dose, formulation or route of administration, or with accidental overdose. Care should be taken when initiating or titrating opioids in patients taking other sedative medications and in those with preexisting disorders of respiratory control, such as obstructive sleep apnoea, who may be at higher risk for respiratory depression.
10.2.1 Opioid toxicity and withdrawal
Patients, caregivers and family members of patients on opioid therapy should be made aware of the clinical features of opioid toxicity, which indicate urgent referral to medical care. The dose at which this may occur is variable depending on comorbidities (particularly renal and hepatic insufficiency) and concomitant drug use, including both prescription and OTC medicines.
Opioid withdrawal symptoms may occur with abrupt discontinuation, rapid dose tapering, or administration of an opioid antagonist. Treatment of acute withdrawal includes administration of intravenous fluids, glucose, α-2 adrenoceptor agonist drugs (e.g. clonidine) and antispasmodic medicines for abdominal cramps. Small doses of opioids may also be given to improve symptoms.
Symptoms associated with toxicity or withdrawal are listed in Table 8.

11. Long-term effects
11.1 Endocrine effects
Long-term administration of opioids may have dose-dependent effects on both the hypothalamic-pituitary-adrenal axis and the hypothalamic-pituitary-gonadal axis, with consequent adrenal insufficiency and hypogonadism. Clinical symptoms include reduced libido, infertility and depression in women, and erectile dysfunction and decreased libido in men. Patients should be advised about these adverse effects before starting opioid therapy. If clinically indicated, referral to an endocrinologist for advice about hormone replacement therapy may be necessary.
11.2 Immunological effects
Both animal and human studies have demonstrated that opioids may have an immunosuppressive effect. The clinical relevance of these findings is unknown.
11.3 Opioid-induced hyperalgesia
Long-term use of opioids, especially at higher doses, may be associated with the development of hyperalgesia - an increased sensitivity or sensation of pain in the absence of overt opioid withdrawal. The pain of hyperalgesia is often more diffuse and less defined in quality than the preexisting pain.
Hyperalgesia may be managed by gradually tapering and discontinuing COT.
12 Driving and working while on opioid therapy
Opioids may cause somnolence, clouded mentation, decreased concentration, incoordination or slower reflexes that may impair the patient's ability to drive safely or to work safely with machines, on ladders, etc. Patients should be advised not to drive immediately, work under hazardous conditions or work with hazardous machinery after initiating or while titrating an opioid until a stable dose is established and they are certain that it does not cause sedation. Driving and hazardous work should also be avoided when taking opioids with alcohol or other drugs that could produce an added sedative affect.
13 Opioids and problematic drug use
The prescription of opioids may result in problematic drug use. The likelihood of this occurring is influenced by a number of social, psychological and health-related factors.
Terms relating to problematic drug use are defined in Table 9.
Although the need for increasing doses is not always indicative of opioid abuse (see section 9.8), some of the behaviours that may suggest problem drug use include:
- earlier prescription seeking
- claims of lost medication
- intoxication
- frequent missed appointments
- use of other scheduled drugs or opioids from other sources
- stealing or borrowing drugs, or forging prescriptions
- overwhelming focus on opioid issues
- resistance to change of therapy, despite evidence of adverse drug effects
- aggressive complaining about the need for more drugs.
Concerns about problem drug use should prompt referral to specialised multidisciplinary pain and addiction services.
At each return visit to the clinician and before the prescription of additional opioids, a full biopsychosocial assessment should be repeated on every patient to ensure that the prescription is necessary and appropriate. COT should not be initiated without this commitment from the patient and their primary healthcare provider.
14. Recommendations for COT in specific pain syndromes
14.1 Non-cancer spinal pain
Non-cancer spinal pain (i.e. cervical, thoracic, lumbar, and/or lumbosacral), with or without neurological involvement, is extremely common throughout the world and will affect most people at some stage of their lives. The causes are protean. Most patients with one or more of these painful disorders can be treated successfully with standard multimodal methods, some of which are described on the South African Spine Society website (http://www.saspine.org). Many of the painful episodes are of relatively short duration and are self-limiting. Paracetamol, non-steroidal anti-inflammatory drugs (NSAIDs), amitriptyline, and muscle relaxants usually provide adequate pain relief. In a minority of cases, SR tramadol might be necessary in the short term. On only rare occasions, parenteral opioids may be necessary for short periods.
A small percentage of patients develop or present with chronic non-cancer spinal pain that is resistant to standard treatment programmes, or in whom standard treatment programmes are inappropriate. The causes of such chronic pain may include multiple-level degenerative spinal disease (especially in the elderly), so-called 'failed spinal surgery', undiagnosed structural pathology, or chronic nonspecific spinal pain. These patients should be carefully re-evaluated for missed pathology, inadequate treatment, treatment non-compliance, secondary gain motives and psychosocial factors. A trial of opioids for pain amelioration can be contemplated in such patients, preferably in consultation with a pain clinician. The trial of opioids must be in addition to other concomitant treatment modalities, including simple analgesics, appropriate physical activity, physical therapy, ergonomic advice, and psychological guidance when necessary. Patients should be counselled extensively about the possible failure of opioid therapy, potential side-effects, the fact that the pain will not be relieved completely, and that it will be necessary to continue with all the other modalities of treatment.
A trial of opioids should be in accordance with this guideline.
14.2 Osteoarthritis, rheumatoid arthritis and ankylosing spondylitis
Tramadol is recommended for initial management of pain associated with osteoarthritis of the hip or knee. Opioid analgesics are strongly recommended for patients with symptomatic arthritis who do not achieve an adequate response with either non-pharmacological or pharmacological modalities and who are either unwilling to undergo, or who are not candidates for total joint arthroplasty.[7]
In patients with ankylosing spondylitis, analgesics such as opioids might be considered for pain control in patients in whom NSAIDs are ineffective, contraindicated and/or poorly tolerated.[8]
Patients with rheumatoid arthritis (RA) report pain relief as a high priority and opioid analgesics are often prescribed, but existing RA treatment guidelines provide little direction in this regard. There is a paucity of high-quality evidence for treating RA pain with SR strong oral opioids such as morphine or oxycodone, or transdermal preparations such as fentanyl. There are also no adequate data for oxymorphone, hydromorphone or buprenorphine. Since RA is a chronic disease, analgesic medications may be required in both the short term and long term, e.g. during flares of inflammatory activity or while waiting for disease-modifying antirheumatic drugs (DMARDs) to take effect (short term), or with irreversible joint damage or the development of secondary pain syndromes (long term).[9]
14.3 Fibromyalgia
Treatment recommendations for widespread soft-tissue pain, including fibromyalgia, suggest that tramadol may be useful for managing this type of pain. There is currently no evidence to recommend long-term use of strong opioids in patients with fibromyalgia. Long-term use of strong opioids in patients with fibromyalgia has been associated with the development of opioid-induced hyperalgesia.[10]
14.4 Chronic pelvic pain
There is currently no evidence that long-term opioids are effective in the treatment of chronic pelvic and abdominal pain of uncertain aetiology. The safety of opioid use in these conditions has not been determined and COT is therefore not recommended.
The long-term management of patients with conditions such as interstitial cystitis/painful bladder syndrome, chronic prostate pain, and irritable bowel syndrome should be facilitated by practitioners with special expertise/interest in these conditions and is best conducted in a multidisciplinary setting.
14.5 Neuropathic pain
Strong opioids can be successfully used for managing neuropathic pain, either as third-line therapy, or as an intermittent adjunct to first- and second-line recommended medications (e.g. pregabalin, gabapentin, and antidepressants).[11] The use of long-term COT for neuropathic pain remains a contentious issue as there are almost no studies where opioids have been continued for longer than six months. Patients who are considered for COT should be carefully selected and they should improve clinically with an initial trial of therapy, as described earlier in this document.
15. Conclusion
A carefully considered comprehensive pain management strategy that includes COT in carefully selected patients with no history of addiction or drug abuse may achieve long-term pain control with a manageable side-effect profile and a low risk of addiction or serious adverse effects.[12] However, there is limited published evidence of longterm efficacy on which to base evidence-based guidelines and much of this guideline is based on evidence consensus and practice patterns rather than on high-quality studies alone. Therefore, appropriate evaluation, including biopsychosocial screening and risk screening, is essential before initiating COT and during continued therapy. It is hoped that this guideline will assist clinicians in selectively using COT for patients who will benefit and simultaneously attempt to avoid the inappropriate and abusive use of opioid analgesics.
Conflicts of interest. The development of this guideline was supported by an unrestricted grant from Mundipharma who did not participate in the development or writing of the guideline. Dr M Raff has received honoraria for consultancies and non-restricted research grants from Mundipharma, Pfizer, Janssen Pharmaceutica, AstraZeneca, MSD, Eli Lilly, Aspen and Abbott Laboratories. Drs J Crosier and S Eppel have received honoraria from Mundipharma. Prof. H Meyer has received honoraria for consultancies and non-restricted research grants from Janssen Pharmaceutica, Eli Lilly, MSD and Mundipharma. Dr B Sarembock has received honoraria for consultancies and non-restricted research grants from MSD, AstraZeneca, Pfizer and Mundipharma. Dr D Webb has received professional fees for services to Abbott Laboratories, Adcock Ingram, Alcon Laboratories, AstraZeneca, Eli Lilly, Janssen Pharmaceutica, Mundipharma, Novartis, and Reckitt Beckiser Pharmaceuticals.
References
1. Gureje O, von Korff M, Simon GE, Gater R. Persistent pain and well-being: A World Health Organization study in primary care. JAMA 1998;280(2):147-151. [http://dx.doi.org/10.1001/jama.280.2.147] [ Links ]
2. British Pain Society. Opioids for persistent pain: Good Practice. A consensus statement prepared on behalf of the British Pain Society, the Faculty of Pain Medicine of the Royal College of Anaesthetists, the Royal College of General Practitioners and the Faculty of Addictions of the Royal College of Psychiatrists. London, UK: British Pain Society; 2010. [ Links ]
3. Chou R, Fanciullo GJ, Fine PG, et al; American Pain Society - American Academy of Pain Medicine Opioids Guidelines Panel. Clinical guidelines for the use of opioid therapy in chronic noncancer pain. J Pain 2009;10(2):113-130. [http://dx.doi.org/10.1016/j.jpain.2008.10.008] [ Links ]
4. Canadian National Opioid Use Guideline Group. Canadian Guideline for safe and effective use of opioids for chronic non-cancer pain. San Francisco: National Opioid Use Guideline Group, 2010. http://nationalpaincentre.mcmaster.ca/documents/opioid_guideline_part_b_v5 _6.pdf (accessed 20 November 2013). [ Links ]
5. Manchikanti L, Abdi S, Atluri S, et al. American Society of Interventional Pain Physicians (ASIPP) guidelines for responsible opioid prescribing in chronic non-cancer pain: Part 2 - Guidance. Pain Physician 2012;15(3 Suppl):S67-S116. [ Links ]
6. Eastern Metropolitan Region Palliative Care Consortium. Opioid conversion ratios - Guide to practice 2010. http://www.emrpcc.org.au/wp-content/uploads/2013/03/EMRPCC-Opioid-Conversion2010-Final2.pdf (accessed 20 November 2013). [ Links ]
7. Hochberg MC, Altman RD, April KT, et al. American College of Rheumatology 2012 recommendations for the use of nonpharmacologic and pharmacologic therapies in osteoarthritis of the hand, hip and knee. Arthritis Care Res 2012;64(4):465-474. [http://dx.doi.org/10.1002/acr.21596] [ Links ]
8. Braun J, van den Berg R, Baraliakos X, et al. 2010 update of the ASAS/EULAR recommendations for the management of ankylosing spondylitis. Ann Rheum Dis 2011;70:896-904. [http://dx.doi.org/10.1136/ard.2011.151027] [ Links ]
9. Whittle SL, Richards BL, Buchbinder R. Opioid analgesics for rheumatoid arthritis pain. JAMA 2013;309(5):485-486. [http://dx.doi.org/10.1001/jama.2012.193412] [ Links ]
10. Painter JT, Crofford LJ. Chronic opioid use in fibromyalgia syndrome: A clinical review. J Clin Rheumatol 2013;19(2):72-77. [http://dx.doi.org/10.1097/RHU.0b013e3182863447] [ Links ]
11. Chetty S, Baalbergen E, Bhigjee AI, et al. Clinical practice guidelines for the management of neuropathic pain: Expert panel recommendations for South Africa. S Afr Med J 2012;102(5):312-325. [ Links ]
12. Noble M, Treadwell JR, Tregear SJ, et al. Long-term opioid management for chronic noncancer pain. Cochrane Database Syst Rev 2010;(1):CD006605. [http://dx.doi.org/10.1002/14651858.CD006605.pub2] [ Links ]
13. Webster LR, Webster RM. Predicting aberrant behaviours in opioid-treated patients: Preliminary validation of the opioid risk tool. Pain Med 2005; 6(6):432-442. [http://dx.doi.org/10.1111/j.1526-4637.2005.00072.x] [ Links ]
Further reading
CNCP and COT
Cohen MI, Wodak AD. Opioid prescribing in general practice: A trial run approach. Modern Medicine of South Africa 2013;38(1):37-43.
de Leon-Casasola OA. Opioids for chronic pain: New evidence, new strategies, safe prescribing. Am J Med 2013;126(3):S3-S11. [http://dx.doi.org/10.1016/j.amjmed.2012.11.011]
Gronow DW. The place of pharmacological treatment of chronic pain. Anesth Int Care Med 2011;12(2):39-41. [http://dx.doi.org/10.1016/j.mpaic.2010.10.015]
Minozzi S, Amato L, Davoli M. Development of dependence following treatment with opioid analgesics for pain relief: A systematic review. Addiction 2013;108(4):688-698. [http://dx.doi.org/10.1111/j.1360-0443.2012.04005.x]
Trescot AM, Boswell MV, Atluri SL, et aL Opioid guidelines in the management of chronic non-cancer pain. Pain Physician 2006;9(1):1-39.
Arthritis
American Academy of Orthopedic Surgeons (AAOS). Treatment of Osteoarthritis of the Knee:
Evidence-based Guideline. 2nd ed. Rosemont, Illinois: AAOS, 2013. http://www.aaos.org/research/guidelines/TreatmentofOsteoarthritisoftheKnee Guideline.pdf (accessed 20 November 2013).
Whittle SL, Richards BL, van der Heijde DM, Buchbinder R. The efficacy and safety of opioids in inflammatory arthritis: A Cochrane systematic review. J Rheumatol Suppl 2012;90:40-46. [http://dx.doi.org/10.3899/jrheum.120341]
Fibromyalgia
Fitzcharles M-A, Ste-Marie PA, Goldberg DL, et al 2012 Canadian Guidelines for the diagnosis and management of fibromyalgia syndrome: Executive summary. Pain Res Manag 2013;18(3):119-126.
Chronic pelvic pain
Fall M, Baranowski AP, Elneil S, et al; European Association of Urology. EAU guidelines on chronic pelvic pain. Eur Urol 2010;57(1):35-48. [http://dx.doi.org/10.1016/j.eururo.2009.08.020]
Nickel JC. Opioids for chronic prostatitis and interstitial cystitis: Lessons learned from the 11th World Congress on Pain. Urology 2006;68(4):697-701.
 Correspondence:
Correspondence:
M Raff
(raffs@iafrica.com)
This guideline is endorsed by the following professional groups: Cape Pelvic Pain Society, PainSA, South African Rheumatism and Arthritis Association, and South African Society of Anaesthesiologists.
Disclaimer. While every effort has been made to ensure the accuracy of the contents at the time of publication, the authors do not guarantee the accuracy of the information contained nor accept any liability, with respect to loss, damage, injury or expense, arising from any errors or omissions in the contents of this work. Reference in the guideline to specific pharmaceutical products does not imply endorsement of any of these products.
Appendix

















