Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
SAMJ: South African Medical Journal
versão On-line ISSN 2078-5135
versão impressa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.103 no.12 Pretoria Dez. 2013
RESEARCH
Blood cultures in sick children
H LochanI; C BamfordII; B EleyIII
IMB ChB, DCH (SA), FCPaed (SA). Paediatric Infectious Diseases Unit, Red Cross War Memorial Children's Hospital, and Department of Paediatrics and Child Health, University of Cape Town, South Africa
IIMB ChB, DCH (SA), MPhil, FCPath (Micro), MMed (Med Micro). National Health Laboratory Service, Groote Schuur Hospital and Division of Clinical Microbiology, University of Cape Town, South Africa
IIIMB ChB, FCPaed (SA), BSc (Hons). Paediatric Infectious Diseases Unit, Red Cross War Memorial Children's Hospital, and Department of Paediatrics and Child Health, University of Cape Town, South Africa
ABSTRACT
BACKGROUND: Blood cultures (BCs) are frequently performed in sick children. A recent audit of BCs among adult patients documented high rates of contamination by coagulase-negative staphylococci (CoNS).
OBJECTIVES: To describe BC contamination rates and common pathogenic organisms causing bloodstream infection in children at a tertiary-level children's hospital.
METHODS: BC results for children admitted to Red Cross War Memorial Children's Hospital from 2008 to 2012 were extracted from the National Health Laboratory Service database. Pathogenic and non-pathogenic (contaminated) growth on BCs in children <1 year of age and >1 year of age, were analysed. Data analysis was performed using Epi Info version 3.5.1.
RESULTS: A total of 47 677 BCs were performed in the 5-year period. The proportion of contaminated specimens ranged between 5.9% and 7.2% per year (p=0.4). CoNS was the predominant isolate in 53.8% of all contaminated BCs. Children <1 year of age experienced higher contamination rates than children >1 year of age (8.7% v. 4.7%; relative risk 1.84; 95% confidence interval (CI) 1.71 - 1.97). Pathogenic organisms were isolated in 6.2% (95% CI 6.0 - 6.4) of all BC specimens. Among Gram-positive organisms, the proportion of Streptococcus pneumoniae isolates declined from 14.3% to 4.7% (p<0.00001), while there was a significant increase in Gram-negative organisms (51.8% - 57.9%; p=0.04) over the 5-year period. Klebsiella pneumoniae, the predominant Enterobacteriaceae isolated, decreased from 45.8% to 31.7% (p =0.004).
CONCLUSION: This study identified unacceptably high BC contamination rates, emphasising the importance of collecting BC specimens under sterile conditions.
Blood cultures (BCs) are frequently performed in sick children either at initial presentation or during the course of hospitalisation following clinical deterioration. A positive BC result may help to determine the causative pathogen as well as guide appropriate antimicrobial therapy.[1] A recent audit of BCs among adult patients at G F Jooste Hospital, Cape Town documented high rates of contamination by coagulase-negative staphylococci (CoNS) over a 6-year period ranging from 4.6% to 9.3% per year. Furthermore, this study documented increasing antibiotic resistance of Gram-negative organisms.[2]
Objective
To understand BC contamination rates and the spectrum of pathogenic organisms causing bloodstream infection in children, we audited the results of BCs performed at our hospital. This analysis forms part of a series of studies on bloodstream infection at the hospital.
Methods
Study design
A retrospective audit of BCs. The Research Ethics Committee of the University of Cape Town approved the study. Because this was a retrospective analysis, informed consent was not obtained.
Setting
Red Cross War Memorial Children's Hospital (RCWMCH) is a 290bed tertiary hospital that serves the paediatric population of the City of Cape Town and the Western Cape Province and neighbouring provinces.
Data extraction
BCs performed at RCWMCH are processed at the National Health Laboratory Service (NHLS) microbiology laboratory at Groote Schuur Hospital (GSH). All BC results of specimens obtained at RCWMCH from 2008 until 2012 were extracted from the NHLS GSH database. All positive results were divided into recognisable pathogens and non-pathogens or contaminants, based on definitions established by the Centers for Disease Control and Prevention.[3]
Statistical analysis
The analysis included comparison of results, stratified according to age, i.e. <1 year and ≥1 year of age. Data were analysed using Epi Info version 3.5.1. Conventional descriptive methods were used to analyse the data. The Mantel-Haenszel χ2 test was used to compare categorical data. Relative risk (RR) was used to estimate the size of differences. A χ2 test for linear trend was used to evaluate trends over time. A p-value of <0.05 was considered statistically significant.
Results
During the 5-year period there were 110 938 admissions to the hospital, representing a mean ± standard deviation of 22 187.6±854.8 admissions per year. A total of 47 677 BC specimens were taken, providing a BC specimen per hospital admissions ratio of 1:2.33.
The proportion of BCs with non-pathogenic growth (contaminated specimens) ranged between 5.9% and 7.2% per year. There was no significant trend in the frequency of contaminated specimens (χ2 for trend p=0.4) (Table 1). The proportion of contaminated specimens was higher in children <1 year (8.7%, 1 928/22 091 v. 4.7%, 1 215/25 586; RR 1.84; 95% confidence interval (CI) 1.71 - 1.97). Non-pathogens were either Grampositive (94.8%) or Gram-negative (5.1%). CoNS was the predominant contaminant, accounting for 53.8% (1 792/3 143) of all non-pathogenic growth. Among contaminants, children <1 year experienced a higher rate of CoNS growth (63.5% v. 46.7%; RR 1.36; 95% CI 1.27 - 1.46) (Table 1).
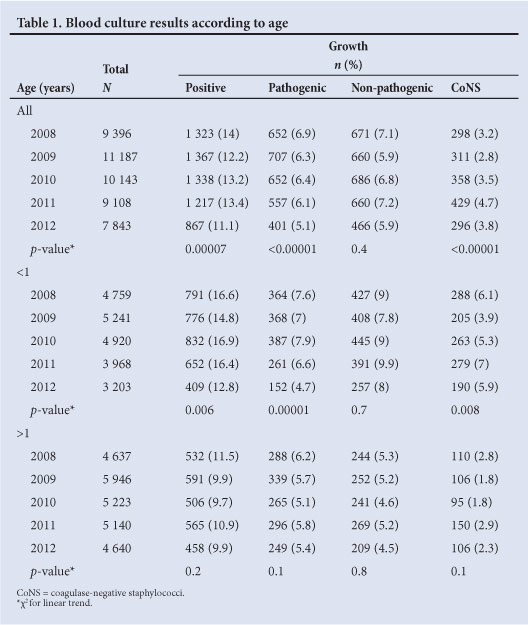
Overall, 6.2% (2 969/47 677; 95% CI for binomial proportion 6.0 - 6.4) of BC specimens yielded pathogenic organisms. The yield was lower in children ≥1 year of age (5.6% v. 6.9%; Mantel-Haenszel χ2p<0.00001) (Table 1). The most common pathogens are shown in Table 2.
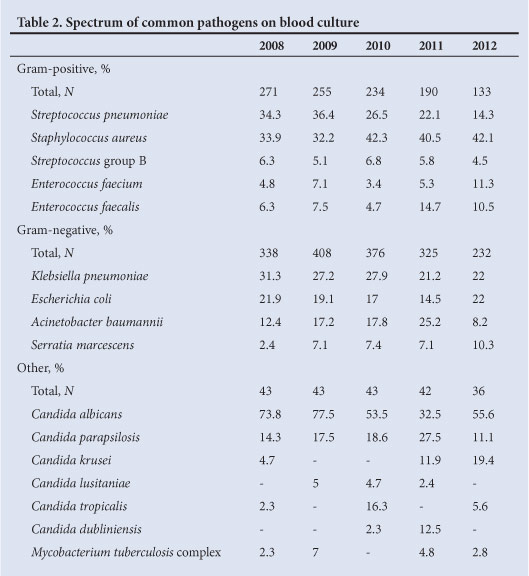
Gram-positive bacteria accounted for 36.5% (1 083/2 969) of all pathogenic isolates. The proportion of pathogens caused by Gram-positive bacteria, with Staphylococcus aureus and Streptococcus pneumoniae being those most commonly isolated, declined from 41.6% in 2008 to 33.4% in 2012 (χ2 for trend p=0.003). S. aureus and S. pneumoniae accounted for 13.7% (407/2 969) and 10.4% (309/2 969) of all pathogenic isolates, respectively. There was no significant change in the annual proportion of S. aureus isolates over the 5-year period (χ2 for trend p=0.2). There was a progressive decline in the annual proportion of positive BCs due to S. pneumoniae, from 14.3% in 2008 to 4.7% in 2012 (χ2 for trend p<0.00001). Enterococcus faecalis and E. faecium together were responsible for 5.2% (153/2 969) of all pathogens with the annual proportion of these isolates increasing from 4.6% in 2008 to 7.3% in 2012 (χ2 for trend p=0.04).
Gram-negative bacteria accounted for 56.6% (1 679/2 969) of all pathogens (Table 2). The χ2 test for trend revealed a significant increase between 2008 and 2012 (51.8% - 57.9%; p=0.04). Enterobacteriaceae dominated and accounted for 64% (1 074/1 679) of all Gram-negative pathogenic isolates and 36.2% (1 074/2 969) of all pathogens. There was no significant change in the annual proportion of Enterobacteriaceae (χ2 for trend p=0.76). The dominant Enterobacteriaceae isolates were Klebsiella pneumoniae (41.8%), Escherichia coli (29.1%) and Serratia marcescens (10.4%). The annual proportion of Enterobacteriaceae infections caused by K. pneumoniae isolates decreased significantly from 45.8% in 2008 to 31.7% in 2012 (χ2 for trend p=0.004). By contrast, an increasing trend was documented for S. marcescens from 3.4% in 2008 to 14.9% in 2012 (χ2 for trend p=0.0003). The annual proportion of E. coli did not change significantly (χ2 for trend p=0.5). Acinetobacter baumannii was responsible for 20.3% (340/1 679) of all Gram-negative infections, isolates increasing annually from 12.4% (42/338) in 2008 to 25.2% (82/325) in 2011 (χ2 for trend p=0.0004) and declining to 8.2% (19/232) in 2012.
Fungi constituted 6.7% (200/2 969) of all pathogens, there being no significant change in the proportion annually (χ2 for trend p=0.1).
Discussion
This analysis provided initial information about BC results and bloodstream infection at RCWMCH. Of concern is the high contamination rates of 5.9 - 7.2% per year, which are above international target contamination rates of 2 - 3%.[4] Similar high contamination rates were also documented in a recent adult study conducted in Cape Town.[2] Because of technical challenges associated with drawing blood from infants, contamination was predictably higher in this subgroup.[5] High contamination rates documented suggest that BC procedures were not standardised and sterility and optimal skin disinfection not uniformly practised. Standardisation of BC procedures is linked to improved pathogen yield when optimal blood volumes are collected.[1,6] A paediatric study completed in Kenya showed that the proportion of positive BCs increased with each additional millilitre of blood cultured.[7] To limit contamination, all healthcare facilities should institute standardised guidelines for BC procedures and ensure ongoing training and education for existing and, especially, new medical staff.
In this study, only 1 in every 16 BC specimens (6.2%; 95% CI 6.0 - 6.4) yielded a pathogenic isolate. Systematic reviews of bloodstream infections carried out in Africa and southeast Asia showed pathogen yields of 8.2% (3 527/43 130; 95% CI 7.9 - 8.4) and 6.6% (1 722/26 258; 95% CI 6.3 - 6.9), respectively.[8,9] The pathogen yield in this study was somewhat lower. One would expect a higher pathogen yield in our setting, as the aforementioned studies[8,9] were a reflection of community-acquired bloodstream infections only, whereas our study included both community-acquired and hospital-acquired bloodstream infections. Several factors, including prior antibiotic administration and suboptimal blood volumes, may have contributed to this lower yield. The Integrated Management of Childhood Illness Programme advises that any seriously ill child requiring urgent referral to hospital should receive intramuscular ceftriaxone prior to transfer.[10] Many children referred from level 1 facilities would therefore have received parenteral antibiotics prior to BC. Furthermore, the selection of patients for BCs should be reviewed to ensure that BCs are used optimally.
The dramatic decline in the culture of S. pneumoniae over time almost certainly reflects the effectiveness of the national pneumococcal immunisation programme and the declining paediatric HIV incidence. The 7-valent pneumococcal conjugate vaccine was introduced in April 2009 and in May 2011 was replaced with a 13-valent conjugate; between February and May 2012 a national pneumococcal 'catch-up' campaign targeted all children aged 18 - 35 months.
Gram-negative bacteria were the dominant pathogens. Although the proportion of Enterobacteriaceae did not change significantly during the study period, interesting changes were observed within this pathogen family. In particular, a decline in the annual proportion of K. pneumoniae isolates and an escalation of the annual proportion of S. marcescens isolates were observed. The dramatic decline in the proportion of A. baumannii isolates in 2012 probably relates to improvements in infection control measures and in ventilator practice in the paediatric intensive care unit. Concerning the latter, the new practice is to discard ventilator tubing once used, the previous practice of sterilisation and re-use of tubing having been discontinued (Professor Andrew Argent, personal communication).
There are limitations to our study. Firstly, there was no analysis of the antimicrobial resistance patterns of the pathogens isolated, which will be the focus of a future study. Secondly, the focus of this study was BC results generated in a tertiary-level paediatric facility, rendering the findings of this audit, particularly with regard to pathogen trends, potentially inapplicable to other paediatric settings with different patient illness profiles and antimicrobial selection pressures. However, high contamination rates were also recorded in BCs in adult patients at G F Jooste Hospital, suggesting that the problem is widespread and that attention should be directed at optimising BC procedures according to recently published recommendations.[1] Thirdly, due to the retrospective nature of the study, BC specimens with inadequate volumes could not be evaluated.
In conclusion, this study identified unacceptably high contamination rates, emphasising the importance of collecting BC specimens under sterile conditions. Decreasing trends in Gram-positive organisms, in particular S. pneumoniae, and an increase among the Gram-negative group of organisms were demonstrated.
References
1. Ntusi N, Aubin L, Oliver S, Whitelaw A, Mendelson M. Guideline for the optimal use of blood cultures. S Afr Med J 2010;100(12):839-843. [ Links ]
2. Kenyon CR, Fatti G, Schnieder N, Bonorchis K, Meintjies G. The value of blood culture audits at peripheral hospitals. S Afr Med J 2012;102(4):232-233. [ Links ]
3. Centers for Disease Control and Prevention, and the National Healthcare Safety Network. CDC/NHSH Surveillance Definition of Healthcare-Associated Infection and Criteria for Specific Types of Infection in the Acute Care Setting. Atlanta: CDC, 2013. http://www.cdc.gov/nhsn/pdfs/pscmanual/17pscnosinfdef_current.pdf (accessed 26 February 2013). [ Links ]
4. Hall KK, Lyman JA. Updated review of blood culture contamination. Clin Microbiol Rev 2006;19(4):788-802. [http://dx.doi.org/10.1128/CMR.00062-05] [ Links ]
5. Pavlovsky M, Press J, Peled N, Yagupsky P. Blood culture contamination in pediatric patients: Young children and young doctors. Pediatr Infect Dis 2006;25(7):611-614. [http://dx.doi.org/10.1097/01.inf.0000220228.01382.88] [ Links ]
6. Connell TG, Rele M, Cowley D, Buttery JP, Curtis N. How reliable is a negative blood culture result? Volume of blood submitted for culture in routine practice in a children's hospital. Pediatrics 2007;119(5):891-896. [http://dx.doi.org/10.1542/peds.2006-0440] [ Links ]
7. Berkley JA, Lowe BS, Mwangi I, et al. Bacteraemia among children admitted to a rural hospital in Kenya. N Engl J Med 2005;352:39-47. [http://dx.doi.org/10.1056/NEJMoa040275] [ Links ]
8. Reddy A, Shaw AV, Crump JA. Community-acquired bloodstream infections in Africa: A systematic review and meta-analysis. Lancet Infect Dis 2010;10(6):417-432. [http://dx.doi.org/10.1016/S1473-3099(10)70072-4] [ Links ]
9. Deen J, von Seidlein L, Andersen F, Elle N, White NJ, Lubell Y. Community-acquired bacterial bloodstream infections in developing countries in south and southeast Asia: A systematic review. Lancet Infect Dis 2012;12(6):480-487. [http://dx.doi.org/10.1016/S1473-3099(12)70028-2] [ Links ]
10. South African Department of Health. Integrated Management of Childhood Illness Guidelines. Pretoria: Department of Health, 2010. [ Links ]
 Correspondence:
Correspondence:
H Lochan
(harsha.lochan@uct.ac.za)
Accepted 16 July 2013














