Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135
Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.103 n.9 Pretoria Jan. 2013
RESEARCH
Laboratory tests for malaria: A diagnostic conundrum?
B PuriI; P MehtaI; N IngoleI; P PrasadI; T MathureII
IMD. Department of Microbiology, Seth GS Medical College (GSMC) and King Edward Memorial Hospital (KEMH), Parel, Mumbai, India
IIMSc. Department of Microbiology, Seth GS Medical College (GSMC) and King Edward Memorial Hospital (KEMH), Parel, Mumbai, India
Correspondence
ABSTRACT
OBJECTIVES: To detect malarial parasites using the peripheral blood smear (PBS) and to compare the PBS with the immunochromatographic antigen test (i.e. OptiMAL and polymerase chain reaction (PCR)).
METHODS: Six ml of blood was collected from each of 170 patients clinically suspected of having malaria. These samples were used to perform PBS examination, the OptiMAL test and PCR by standard protocol.
RESULTS: PBS examination found malarial parasites in 86 (50.6%) samples. In comparison, 71 (41.8%) samples were positive by OptiMAL test whereas PCR could detect malarial parasites in only 44 (25.9%) samples. All 84 (49.4%) samples which were negative by PBS were negative by both OptiMAL and PCR. The sensitivity and specificity were respectively 85.54% and 100% for OptiMAL and 51.12% and 100% for PCR.
CONCLUSION: Depending on the tests' operational feasibility, and the availability of adequate trained personnel, equipment and laboratory management systems, and considering its sensitivity and cost-effectiveness, peripheral blood smear remains the test of choice for malaria, especially in endemic areas.
Malaria is caused by protozoan parasites of the genus Plasmodium. Historically, strategies for its diagnosis have ranged from basic empirical clinical diagnostic algorithms to the examination of stained peripheral blood smears (PBS) by light microscopy. Although empirical clinical diagnosis remains the most common method of diagnosis in many regions, its accuracy is poor because the symptoms of malaria overlap with those of many other tropical diseases. Accurate diagnosis of malaria is necessary to prevent morbidity and mortality while avoiding unnecessary use of antimalarial drugs.[1,2]
The conventional PBS examination is widely used to detect malarial parasites and remains the gold standard for malaria diagnosis. However, it is laborious, time-consuming and its interpretation requires expertise.[3] In view of the limitations of pure clinical diagnosis and the microscopic methods, there is a need for a specific, simple-to-perform and rapid test for the diagnosis of malaria. Newer antigen detection tests have been introduced which detect the presence of parasite lactate dehydrogenase (PLDH) enzyme, present only in live parasites, and these offer high sensitivity and specificity. Also, the polymerase chain reaction (PCR) antigen detection test has been widely used to increase the sensitivity of malaria diagnosis, especially in the case of low parasitaemia. In view of the above, a study was conducted to compare the microscopic examination of PBS with rapid antigen detection tests (OptiMAL and PCR) as methods of malaria diagnosis.
Methods
A cross-sectional study was carried out at a tertiary care teaching hospital in Mumbai, India, after obtaining Institutional Ethics Committee approval. Malaria is endemic in Mumbai, which is known as the malaria capital of India. High numbers of fever cases present in the monsoon season. Across India, the primary diagnostic method for malaria has always been PBS examination, and it is only recently that rapid antigen detection kits have become available.
This study was carried out during the monsoon season, from July to October 2009, in 170 patients with fever who were clinically suspected of malaria. Our hospital treats both complicated cases referred from primary healthcare centres and uncomplicated cases presenting directly. Only the uncomplicated cases were included in the present study. Patients diagnosed with infectious diseases other than malaria (enteric fever, leptospirosis and dengue virus infection) were excluded from the study.
After obtaining written informed consent from the patient, 6 ml blood was collected by venepuncture and equally distributed in two sterile ethylenediaminetetra-acetic acid (EDTA) bulbs. Within half an hour, thick and thin smears were prepared as per standard procedure and stained with Giemsa stain. The quality of each prepared slide was assessed at the time of microscopic examination. Whenever possible, any slide judged inadequate was prepared again until a slide of an acceptable standard was produced. At all levels a senior microbiologist supervised microscopy, stains, staining of slides and reporting. Before reporting, each smear was examined independently by one senior and two junior microscopists to assure its quality.
The OptiMAL test (Flow Inc., Portland, OR) was performed as per manufacturer's instructions from the same sterile EDTA bulb.
A second 3 ml sterile EDTA bulb was utilised for malaria PCR using the 1-2-3 Amp Plas kit (Genesis Limited). Amplification was done using the following set of primers provided with the kit, and which were specific for:
• Plasmodium spp: 5' CGTGATCAATGCATAAAACCGGTGTGTC 3'
• P vivax: 5' CGTGATCAAAGCTCTGCTATTTCCGAAAC 3'
• P falciparum: 5' CGTGATCAATGCATAAAACCGGTGTATC 3' Amplified product was detected by electrophoresis, using 1% agarose gel containing ethidium bromide.
Statistical analysis
Analysis was conducted using Stata software version 9. The sensitivity, specificity, positive predictive value and negative predictive value of OptiMAL and PCR were calculated.
Results
Of the 170 clinically suspected malaria cases, 105 (61.76%) were male and 65 (38.24%) were female. Malarial Parasites were seen in 86 (50.58%) thick and thin smears of which 56 samples (65.1%) had P. vivax, 27 (31.4%) had P. falciparum and 3 (3.5%) had mixed infection. Of the 86 PBS-positive smears 4 cases had a parasitic index between 5% and 10%, the rest being <5%. OptiMAL could detect Plasmodium species antigen in 44 samples and P. falciparum/mixed antigen in 27 samples. It had a sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) of 85.54%, 100%, 100% and 87.88% respectively, compared with PBS. PCR could detect P. vivax in 30 samples, P. falciparum in 13 samples and mixed species in 1 sample. It had sensitivity, specificity, PPV and NPV of 51.16%, 100%, 100% and 66.67% respectively, compared with PBS.
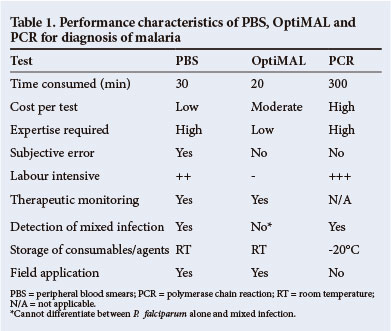
Performance characteristics of PBS, OptiMAL and PCR are shown in Table 1. Each test had its own set of limitations.
Discussion
In the outpatient department, a majority of patients present with fever and are clinically suspected of malaria, especially during the monsoon period. In the present study, a total of 86 out of 170 (50.58%) cases were diagnosed with malarial infection by PBS. Of these, 56 (65.1%) cases were diagnosed as P. vivax , 2 (31.4%) were P. falciparum and 3 (3.5%) were mixed infections. This is consistent with the finding of other authors in India and abroad.14-71 Though P. vivax infection was seen more commonly, it is essential for clinicians to differentiate between the two species, as P. falciparum infection leads to more severe complications.
Microscopic examination of PBS was considered the gold standard for comparing the results of OptiMAL and PCR. The sensitivity, specificity, PPV, and NPV of OptiMAL were 85.54%, 100%, 100%, and 87.88% respectively. Similar findings have been reported by various authors in India and abroad.14-81 Nine cases of P. vivax-positive samples and 5 cases of P. falciparum infection and 1 mixed infection were not identified by the OptiMAL test. Various authors have reported that very low parasitaemia (<100 parasites/µ!) is one possible explanation for false negative results by the OptiMAL test.17-'1 Indeed, in the present study all 15 samples found negative by OptiMAL had a low parasitic index (<1%). Another possible explanation for false negatives may be the fact that OptiMAL detects only live parasites - most patients in the present study were partially treated referred cases, in whom parasites may have been dead but not yet cleared from the host.
Stringent quality control minimised a similar risk in the case of PBS. However, in primary healthcare centres similar quality control may not always be possible and a trained microscopist may not be available.
A major limitation of OptiMAL was its inability to differentiate between P. falciparum and mixed infection. Compared with P vivax, P. falciparum has been reported to have a higher fatality rate, and to cause more complications, including cerebral malaria, acute respiratory distress syndrome (ARDS) and acute renal failure. The World Health Organization (WHO) recommends different treatment strategies for P. vivax, P. falciparum and mixed infection,[2] making it important to differentiate between P. falciparum and mixed infection.
In malaria diagnosis, it has been reported that PCR not only detects infection in cases of low parasitaemia, but also correctly identifies the species, including drug-resistant cases. But in the present study PCR had a very low sensitivity (51.16%) compared with that reported by other authors (96 - 100%).[10-14]
Coleman et al. have reported a remarkable degree of non-concordance between microscopy and PCR at parasite densities below 100 µΙ[11] In the present study the majority of cases had a low parasitic index (<1%), leading to poor overall sensitivity. The low sensitivity by PCR may also be due to the use of non-sensitive primers, as the kit used was under evaluation and has not yet been marketed. Another reason could be the presence of various inhibitory substances present in blood, such as EDTA, the porphyrin ring of haeme, and intracellular substances. Similar findings have been reported by Makler et al. and Barker et al.[15,16]
PBS was found to be a simple, low-cost method which could not only identify the species of malaria but also the level of parasitaemia. PBS had certain limitations, however, as it is labour-intensive and prone to subjective error; and as positivity depends on the timing of the smear and the individual microscopist's skills.
OptiMAL was found to be a simple, sensitive and effective diagnostic test for malaria diagnosis. It is a rapid diagnostic test and the result could be read in 20 minutes. This test's sensitivity was very close to that of microscopic examination of PBS, and it did not require highly skilled personnel to perform it or interpret its results. The test had the added advantage of being able to detect all four Plasmodium species, and could be used to follow the efficacy of drug therapy since it detects an enzyme produced only by living parasites. It is reasonably priced and no special facility is required for its storage. Hence it has field application. While the major disadvantage of OptiMAL is its inability to differentiate between P. falciparum and mixed infection, it is a valuable adjunct for rapid emergency diagnosis, especially in areas where no skilled microscopist is available.
PCR was useful in the accurate identification of Plasmodium species, but time-consuming, requiring 300 minutes to complete the test. It also required high expertise and expensive infrastructure and was labour-intensive. Consumables and reagents required storage at -200C. The test's high cost and the low sensitivity is likely to prevent its regular and routine use, especially in laboratories in resource-constrained settings.
Across India, both microscopy and newer rapid diagnostic tests are being used for malaria diagnosis, depending on the tests' operational feasibility, and the availability of adequate trained personnel, equipment and laboratory management systems. Considering its sensitivity and cost-effectiveness, PBS remains the test of choice.
References
1. Pasvol G. Management of severe malaria: Interventions and controversies. Infect Dis Clin North Am 2005;19(1):211-240. [http://dx.doi.org/10.1016%2Fj.idc.2004.10.007; [ Links ]]
2. World Health Organization. Guidelines for the Treatment of Malaria. 2nd ed. Geneva: WHO, 2010. [ Links ]
3. Payne D. Use and limitations of light microscopy for diagnosing malaria at the primary healthcare level. Bull World Health Organ 1998;66(5):621-628. [ Links ]
4. Palmer CJ, Lindo JF, Klaskala WI, et al. Evaluation of the OptiMAL test for rapid diagnosis of Plasmodium vivax and Plasmodium falciparum malaria. J Clin Microbiol 1998;36(1):203-206. [ Links ]
5. Chayani N, Das B, Sur M, Bajoria S. Comparison of parasite lactate dehydrogenase based immunochromatographic antigen detection assay (optimal) with microscopy for detection of malaria parasites. Indian J Med Microbiol 2004;22(2):104-106. [ Links ]
6. Van den Broek I, Hill O, Gordillo F, et al. Evaluation of three rapid tests for diagnosis of P. falciparum and P. vivax malaria in Colombia. Am J Trop Med Hyg 2006;75(6):1209-1215. [ Links ]
7. Iqbal J, Sher A, Hira PR, Al-Owaish R. Comparison of the OptiMAL test with PCR for diagnosis of malaria in immigrants. J Clin Microbiol 1999;37(11):3644-3646. [ Links ]
8. Quintana M, Piper R, Boling HL, et al. Malaria diagnosis by dipstick assay in a Honduran population with coendemic Plasmodium falciparum and Plasmodium vivax. Am J Trop Med Hyg 1998;59(6):868-871. [ Links ]
9. Iqbal J, Muneer A, Khalid N, Ahmed MA. Performance of the OptiMAL test for malaria diagnosis among suspected malaria patients at the rural health centers.Am J Trop Med Hyg 2003;68(5):624-628. [ Links ]
10. Nandwani S, Mathur M, Rawat S. Evaluation of the polymerase chain reaction analysis for diagnosis of falciparum malaria in Delhi, India. Indian J Med Microbiol 2005;23(3):176-178. [http://dx.doi. org/10.4103%2F0255-0857.16590] [ Links ]
11. Coleman RE, Sattabongkot J, Promstaporm S, et al. Comparison of PCR and microscopy for the detection of asymptomatic malaria in a Plasmodium falciparum/vivax endemic area in Thailand. Malar J 2006;5:121. [http://dx.doi.org/10.1186/1475-2875-5-121] [ Links ]
12. Boonma P, Christensen PR, Suwanarusk R, Price RN, Russell B, Lek-Uthai U. Comparison of three molecular methods for the detection and speciation of Plasmodium vivax and Plasmodium falciparum. Malar J 2007;6:124. [http://dx.doi.org/10.1186/1475-2875-6-124] [ Links ]
13. Safeukui I, Millet P, Boucher S, et al. Evaluation of FRET real-time PCR assay for rapid detection and differentiation of Plasmodium species in returning travellers and migrants. Malar J 2008;7:70. [http://dx.doi.org/10.1186/1475-2875-7-70] [ Links ]
14. Khairnar K, Martin D, Lau R, Ralevski F, Pillai DR. Multiplex real-time quantitative PCR, microscopy and rapid diagnostic immuno-chromatographic tests for the detection of Plasmodium spp: Performance, limit of detection analysis and quality assurance. Malar J 2009;8:284. [http://dx.doi. org/10.1186/1475-2875-8-284] [ Links ]
15. Makler MT, Palmer CJ, Ager AL. A review of practical techniques for the diagnosis of malaria. Ann Trop Med Parasitol 1998;92(4):419-433. [ Links ]
16. Barker RH Jr, Banchongaksorn T, Courval JM, Suwonkerd W, Rimwungtragoon K, Wirth DF. A simple method to detect Plasmodium falciparum directly from blood samples using the polymerase chain reaction. Am J Trop Med Hyg 1992;46(4):416-426. [ Links ]
 Correspondence:
Correspondence:
N Ingole
(nayanaingole@gmail.com)
Accepted 24 December 2012.














