Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
SAMJ: South African Medical Journal
versão On-line ISSN 2078-5135
versão impressa ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.102 no.11 Pretoria Jan. 2012
FORUM
REVIEW
Maximising Kasai portoenterostomy in the treatment of biliary atresia: medical and surgical options
A GrieveI; M DavenportII
IDepartment of Paediatric Surgery, University of the Witwatersrand, Johannesburg
IIClinical and Academic Head of the Department of Paediatric Surgery at King's College Hospital, London, United Kingdom
ABSTRACT
Biliary atresia (BA) remains one of the most challenging conditions in paediatric surgery. It has several possible causes, resulting in a range of different clinical scenarios. The current therapeutic approach is almost entirely surgical with an initial attempt to restore bile flow and preserve the native liver using a Kasai-type portoenterostomy. Liver transplantation (cadaveric or living donor) is usually reserved for failure or for infants presenting late with end-stage cirrhosis. The role of adjuvant medical therapy is unclear and evidence of benefit is lacking. Nonetheless, the use of post-operative steroids, prophylactic antibiotics and choleretic agents such as ursodeoxycholic acid is common. Ideally, the entire pathway should be complementary and seamless with few infants dying of end-stage liver disease or uncorrectable associated congenital malformations. Experience from high-volume centres suggests that clearance of jaundice can be achieved in 50 - 60% of infants, with 10-year native liver and real survival rates of 45% and 90%, respectively.
Ladd1 described his experience of operating on newborn infants with surgical jaundice at Boston Children's Hospital. Although it is unclear how many of his cases had genuine biliary atresia (BA), his results were remarkable, with 6/11 cases draining bile. Unfortunately, later reports identified most cases of BA as 'uncorrectable', with an inevitable dismal outcome despite many desperate surgical manoeuvres.2,3 Those surgical pioneers recognised that no matter how high the level of biliary dissection, it was not possible to identify any bile-containing structure to fashion any kind of conventional biliary anastomosis.
Kasai4 published his first report in the Japanese surgical journal Shujitsu in 1959. He recognised, albeit accidentally, that the apparently solid proximal biliary remnant contained microscopic biliary channels which retained a communication with the intrahepatic bile duct system. Therefore, if enough of these could be exposed in the porta hepatis and be drained into a Roux loop, then sufficient bile flow could be restored and jaundice would recede.
The results in 'uncorrectable' BA were greeted with scepticism in the West, and it was not until the 1970s that the procedure was taken up by institutions in the USA and western Europe. Our experience with the Kasai procedure at King's College Hospital dates from this era.5 Nevertheless, what Kasai performed in the 1950s and 1960s is not necessarily what we would recognise today. His original technique, while reaching the surgical plane flush with the liver capsule, does not attempt to go beyond a fairly narrow oval within the bifurcation of the portal vein (5 mm diameter), and uses a short Roux loop (25 - 30 cm) with a relatively crude anastomosis (5/0 surgical catgut).6 Later, authors sought to increase the area exposed by dissecting into the Rex fossa around the umbilical point (junction with left portal vein) and around the bifurcation of the right vascular pedicle.7,8 This more extended approach leaves a denuded area approaching 20 x 10 mm, to be incorporated into a longer (40 - 50 cm) Roux loop (Fig. 1).
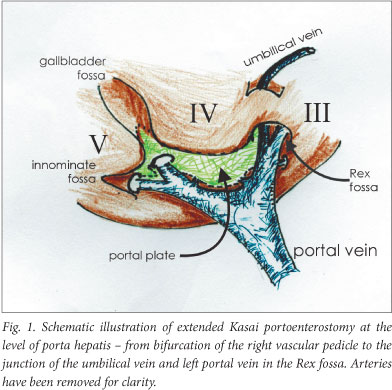
The only other element of the operation which changed, but was subsequently reverted to the original, was the design and configuration of the Roux loop. It became fashionable to open the loop to the skin as a stoma and then re-feed the bile back in again in an attempt to reduce the incidence of post-operative cholangitis.9 Other modifications with the same objective included the creation of 'valves' within the loop.10 Despite initial acceptance of theoretical benefit, neither manoeuvre has any advantage to the standard long Roux-en-Y limb. About 20% of cases have a patent common bile duct and gallbladder, leading to a mucocele, which can be used as the conduit with the transected porta hepatis (i.e. a portocholecystostomy). This abolishes the risk of cholangitis, but bile drainage is more tenuous and a much higher revision rate leads to failure.11 Efforts to use the appendix as a conduit have also been discarded with time.
The advent of minimally invasive surgery and laparoscopic techniques probably reached its apogee with a successful laparoscopic Kasai portoenterostomy (KPE) by a Brazilian team in 2002.12 Several small-series case reports followed, together with a prospective trial from Hannover, Germany.13-15 Most pioneers came to recognise that, while possible, the laparoscopic technique was sufficiently different to lead to poorer outcomes.15,16 Isolated centres in China, Japan and South America still offer this technique, but most larger centres have reverted.
What would constitute an open radical extended KPE today? The principles include a radical dissection within the porta hepatis to separate the proximal biliary remnant (and any lymphatic efferents) from the right and left portal vein and the hepatic arteries. Usually, small veins from the confluence of the portal vein are divided, exposing the caudate lobe as the posterior limit. The Rex fossa on the left side can be opened by dividing the isthmus of liver tissue connecting segments III and IV exposing the junction of umbilical vein with left portal vein. This triangular, pyramidal biliary remnant is then transected flush with the capsule, starting in the gallbladder fossa. It extends on the right side to incorporate a small triangular area between the anterior and posterior right vascular pedicle and on the left to the umbilical point. Controversy remains as to how best to expose the porta hepatis to achieve these goals. Since the 1970s, the large centres in London and Paris have advocated dislocation of the liver outside of the abdominal cavity by dividing the ligaments. Others advocate division of the left triangular ligament, extracting only the left lobe to achieve a similar objective. Others leave the liver within the peritoneal cavity, but sling the vascular pedicles to aid portal dissection.
KPE, as described here, should be associated with clearance of jaundice (to normal) in 50 - 60% of infants.5, 17
Key variables in outcome
BA should not be considered a single disease entity with a predictable natural history and stereotypical response to surgery. This aetiological heterogeneity is complex and our broad classification which seeks to categorise syndromic BA and cystic BA as examples of developmental BA and cytomegalovirus (CMV)-IgM-positive BA as a clinically defined virus-associated BA, still leaves many cases of isolated BA with no obvious definable aetiology.18
However, compared with isolated BA, infants with syndromic BA splenic malformation (BASM) respond less well to KPE and have a poorer overall outcome with a higher risk of death.5,19 By contrast, higher proportions of infants with cystic BA (usually type 1 and 2) clear their jaundice after KPE and have a better long-term outcome (Fig. 2).20,21 The outcome of these 2 types of developmental BA has a marked relationship to the age at which the KPE is performed. This is not seen so clearly in isolated BA. In our age-cohort analysis of infants aged up to about 100 days at King's College Hospital, we could not predict outcome (by clearance of jaundice or need for transplantation by 2 years) simply on the grounds of age. Certainly, no 'cut-offs' at 6, 8, or 10 weeks were evident.22
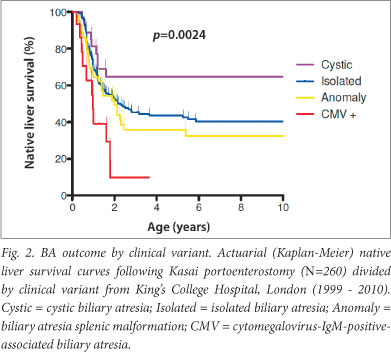
The poorest outcome and highest risk of death is in the group of infants with CMV-IgM-positive-associated BA. These infants are usually older at presentation and at time of KPE, are more jaundiced and have a pronounced hepatic inflammatory reaction with a higher degree of fibrosis. Consequently, their chance of clearance of jaundice following KPE is much lower and their need for transplantation correspondingly greater.23
Peri-operative regimens
Given adequate uncomplicated surgery exposing enough ductules, the question is: can more be done to improve the chances of eventual success? Medical management and perhaps pharmacology may help in 4 potential areas.
Bile drainage
Change of stool colour is evident in most cases in the first week post KPE; failure is inevitable if this does not occur. While the early return and degree of bile flow is related to anatomical factors evident and only correctable at surgery, there may be a role for other choleretic agents.
The efficacy of corticosteroids, which have been used for over 30 years post KPE, remains unknown. Despite this, about 50% of infants with BA treated in the United States of America receive postoperative steroids.24 The problem is one of scale: BA is rare and few centres see more than 5 new cases per year. This is compounded by surgeon and disease variation, leaving only small cohorts to analyse.25 A meta-analysis failed to find evidence of effect.26 A randomised placebo-controlled trial from 2 centres in the United Kingdom retrospectively reviewed low-dose prednisolone treatment (starting at 2 mg/kg/day) in 73 infants.27 The study showed early biochemical benefit (reduced 1-month bilirubin), but no effect on ultimate outcome (need for transplantation, etc.). Further evidence from one of the centres using a higher dose of prednisolone (5 mg/kg/day) showed continued biochemical benefit, resulting in an increased proportion clearing jaundice (Table 1).
Ursodeoxycholic acid (UDCA) has been used in adult cholestatic diseases, such as primary biliary cirrhosis and primary sclerosing cholangitis, with evidence of benefit. It appears to increase choleresis, with protection of cholangiocytes and hepatocytes.28 A crossover trial from France assessed the effect of UDCA (25 mg/kg/day in 3 divided doses) on liver function in children more than 1 year post KPE in a discontinuation/re-introduction fashion. Sixteen children with BA all cleared their jaundice.29 Six months after ceasing UDCA treatment, 1 child worsened clinically with recurrence of jaundice, and all but 2 had significant worsening of their liver enzymes. On UDCA re-introduction, their biochemistry improved.
Other agents such as phenobarbital and the bile acid sequestrant, cholestyramine, have been studied in a small randomised trial in France, with no significant benefit.30
Prevention and treatment of cholangitis
Cholangitis may occur in children with some restoration of bile flow, typically in the first 2 years post KPE. Early use of potent intravenous (IV) antibiotics effective against gram-negative organisms remains the agreed first-line treatment, but there is controversy about the value of any published prophylactic regimen. Some centres insert a Hickman line and administer IV antibiotic for 4 - 6 months, while others do not administer anything.5
Limitation of hepatic fibrosis
Unlike many cholestatic conditions presenting in infancy, BA is characterised by relatively early aggressive hepatic fibrosis and, ultimately, cirrhosis that leads to life-threatening portal hypertension and the early need for transplantation.31 Modulation of this biological process would have immense benefit, but seems elusive and far distant. Asian centres routinely prescribe the Chinese herb Inchinko-to; claimed benefits include inhibition of apoptosis and liver fibrosis,32 but real evidence of benefit remains unpublished.
Nutritional management
BA infants are nutritionally compromised with deficits in protein metabolism, low muscle and liver stores of glycogen, obvious fat malabsorption, key deficiencies in fat-soluble vitamins (D, A, K and E) and, if cirrhotic, low serum and storage levels of zinc and selenium. Medical and dietetic attendants must maximise nutritional potential and maintain normal growth and development with sub-optimal liver and bile flow. This usually involves changing to a more appropriate formula (e.g. Caprilon (Nutricia)) with higher medium triglyceride levels) and regular parenteral vitamin supplementation. Early initiation of overnight naso-gastric feeding also helps to maintain effective calorie/ protein intake, which becomes crucial in the failing liver listed for transplantation, where outcome is directly related to nutritional status.33
Benchmark of outcomes in BA
KPE remains an important element in the strategic management of the infant with BA. In historical series, it was the only solution available. The Sendai (Japan) series reflecting Kasai's experience is a good example of surgical evolution.34 This showed 10-year survival rates of 10% for the first 63 patients treated from 1953 to 1967, 27% for 44 cases from 1968 to 1972, and 48% for 61 cases from 1973 to 1977. In addition, not all were jaundice-free, with major problems with cholestasis and portal hypertension. By comparison, in our recent experience in England and Wales, where liver transplantation is available to all regardless of status or income, overall 10-year survival was about 90% with native liver survival of 46%.17 Over 95% of those with their own livers are jaundice-free and have a good expectation of normal life. In England and Wales over the past 12 years, resources and expertise have been concentrated, which is probably the most important aspect of maximising KPE potential.
References
1. Ladd WE. Congenital atresia and stenosis of the bile duct. JAMA 1928;91:441-444. [ Links ]
2. Longmire WP, Sandford MC. Intrahepatic cholangiojejunostomy for biliary obstruction. Surgery 1948;24:264-276. [ Links ]
3. Fonkalsrud EW, Kitagawa S, Longmire WP. Hepatic lymphatic drainage to the jejunum for congenital biliary atresia. Am J Surg 1966;112:188-194. [ Links ]
4. Kasai M, Suzuki S. A new operation for "non-correctable" biliary atresia: Hepatic portoenterostomy. Shujitsu 1959;13:733-739. [ Links ]
5. Davenport M, Kerkar N, Mieli-Vergani G, Mowat AP, Howard ER. Biliary atresia: the Kings College Hospital experience (1974-1995). J Pediatr Surg 1997;32:479-485. [ Links ]
6. Kasai M, Kimura S, Asakura Y, Suzuli H, Taira Y, Ohashi E. Surgical treatment of biliary atresia. J Pediatr Surg 1968;3:665-675. [ Links ]
7. Endo M, Katsumata K, Yokoyama J, et al. Extended dissection of the porta hepatis and creation of an intussuscepted ileocaecal conduit for biliary atresia. J Pediatr Surg 1983:18:784-793. [ Links ]
8. Kobayashi H, Yamataka A, Urao M, et al. Innovative modification of the hepatic portoenterostomy Our experience of treating biliary atresia. J Pediatr Surg 2006;41:e19-22. [http://dx.doi.org/10.1016/j.jpedsurg.2005.12.056] [ Links ]
9. Ohya T, Miyano T, Kimura K. Indication for portoenterostomy based on 103 patients with Suruga II modification. J Pediatr Surg 1990;25:801-804. [ Links ]
10. Ogasawara Y, Yamataka A, Tsukamoto K, et al. The intussusception antireflux valve is ineffective for preventing cholangitis in biliary atresia: a prospective study. J Pediatr Surg 2003;38:1826-1829. [http://dx.doi.org/10.1016/j.jpedsurg.2003.08.025] [ Links ]
11. Zhao R, Li H, Shen C, Zheng S, Xiao X. Hepatic portocholecystostomy (HPC) is ineffective in the treatment of biliary atresia with patent distal extrahepatic bile ducts. J Invest Surg 2011;24:53-58. [ Links ]
12. Esteves E, Clemente Neto E, Ottaiano Neto M, et al. Laparoscopic Kasai portoenterostomy for biliary atresia. Pediatr Surg Int 2002;18:737-740. [http://dx.doi.org/10.1007/s00383-002-0791-6] [ Links ]
13. Ayuso L, Vila-Carbó JJ, Lluna J, Hernandez E, Marco A. Laparoscopic Kasai portoenterostomy: present and future of biliary atresia treatment. Cir Pediatr 2008;21:23-26. [ Links ]
14. Dutta S, Woo R, Albanese CT. Minimal access portoenterostomy: advantages and disadvantages of standard laparoscopic and robotic techniques. J Laparoendosc Adv Surg Tech A. 2007;17:258-264. [http://dx.doi.org/10.1089/lap.2006.0112Links ] Arial, Helvetica, sans-serif" size="2">]
15. Ure BM, Kuebler JF, Schukfeh N, Engelmann C, Dingemann J, Petersen C. Survival with the native liver after laparoscopic versus conventional Kasai portoenterostomy in infants with biliary atresia: a prospective trial. Ann Surg 2011;253:826-830. [http://dx.doi.org/10.1097/SLA.0b013e318211d7d8] [ Links ]
16. Chan KWE, Lee KH, Mou JWC, Cheung STG, Tam YHP. The outcome of laparoscopic portoenterostomy for biliary atresia in children. Pediatr Surg Int 2011;27:671-674. [ Links ]
17. Davenport M, Ong E, Sharif K, et al. Biliary atresia in England and Wales: Results of centralization and new benchmark. J Pediatr Surg 2011;46:1689-1694. [http://dx.doi.org/10.1016/j.jpedsurg.2011.04.013] [ Links ]
18. Hartley J, Davenport M, Kelly D. Biliary atresia. Lancet 2009;374:1704-1713. [ Links ]
19. Davenport M, Tizzard SA, Underhill J, Mieli-Vergani G, Portmann B, Hadzic N. The biliary atresia splenic malformation syndrome: a 28-year single-center retrospective study. J Pediatr 2006;149:393-400. [http://dx.doi.org/10.1016/j.jpedsurg.2011.04.013Links ] Arial, Helvetica, sans-serif" size="2">]
20. Caponcelli E, Knisely AS, Davenport M. Cystic biliary atresia: an etiologic and prognostic subgroup. J Pediatr Surg 2008;43:1619-1624. [http://dx.doi.org/10.1016/j.jpedsurg.2007.12.058] [ Links ]
21. Superina R, Magee JC, Brandt ML, et al. The anatomic pattern of biliary atresia identified at time of Kasai hepatoportoenterostomy and early postoperative clearance of jaundice are significant predictors of transplant- free survival. Ann Surg 2011;254:577-585. [http://dx.doi.org/10.1097/SLA.0b013e3182300950] [ Links ]
22. Davenport M, Caponcelli E, Livesey E, Livesey E, Hadzic N, Howard E. Surgical outcome in biliary atresia. Etiology affects the influence of age at surgery. Ann Surg 2008;247:694-698. [http://dx.doi.org/10.1097/SLA.0b013e3181638627Links ] Arial, Helvetica, sans-serif" size="2">]
23. Zani A, Quaglia A, Hadzic N, Zuckerman M, Davenport M. Cytomegalovirus-associated biliary atresia: An aetiological and prognostic sub-group. (abstract) Presented at 57th annual BAPS congress, Aberdeen, 22 - 24 July 2010. [ Links ]
24. Lao OB, Larison C, Garrison MA, Healey PJ, Goldin AB. Steroid use after the Kasai procedure for biliary atresia. Am J Surg 2010;199:680-684. [http://dx.doi.org/10.1016/j.amjsurg.2010.01.014] [ Links ]
25. Kobayashi H, Yamataka A, Koga H et al. Optimum prednisolone usage in patients with biliary atresia post- portoenterostomy. J Pediatr Surg 2005;40:327-330. [http://dx.doi.org/10.1016/j.jpedsurg.2004.10.017] [ Links ]
26. Sarkey A, Schreiber RA, Milner R, Barker CC. Does adjuvant steroid therapy post-Kasai portoenterostomy improve the outcome of biliaryatresia? A systemic reviewand meta-analysis. Can J Gastroenterology 2011;25:440-444. [ Links ]
27. Davenport M, Stringer MD, Tizzard SA, McClean P, Mieli-Vergani, Hadzic N. Randomized, double-blind, placebo-controlled trial of corticosteroids after Kasai portoenterostomy for biliaryatresia. Hepatology 2007;46:1821-1827. [http://dx.doi.org/10.1002/hep.21873] [ Links ]
28. Paumgartner G, Beuers U. Ursodeoxycholic acid in cholestatic liver disease: mechanisms of action and therapeutic use revisited. Hepatolgy 2002;36:525-531. [http://dx.doi.org/10.1053/jhep.2002.36088] [ Links ]
29. Willot S, Uhlen S, Michaud L, et al. Effect of ursodeoxycholic acid on liver function in children after successful surgery for biliary atresia. Pediatrics 2008;122:e1236-1242. [http://dx.doi.org/10.1542/peds.2008-0986] [ Links ]
30. Vajro P Couturier M, Lemonnier F, Odièvre M. Effects of postoperative cholestyramine and phenobarbital administration on bile flow restoration in infants with extrahepatic biliary atresia. J Pediatr Surg 1986;21:362-365. [ Links ]
31. Ramm GA, Shepherd RW, Hoskins AC, et al. Fibrogenesis in pediatric cholestatic liver disease: role of taurocholate and hepatocyte-derived monocyte chemotaxis protein-1 in hepatic stellate cell recruitment. Hepatology 2009;49:533-544. [http://dx.doi.org/10.1002/hep.22637] [ Links ]
32. Tamura T, Kobayashi H, Yamataka A, Lane GJ, Koga H, Miyano T. Inchin-ko-to prevents medium-term liver fibrosis in postoperative biliary atresia patients. Pediatr Surg Int 2007;23:343-347. [http://dx.doi.org/10.1007/s00383-007-1887-9] [ Links ]
33. Moukarzel AA, Najm I, Vargas J, McDiarmid SV, Busuttil RW, Ament ME. Effects of nutritional status on outcome of orthotopic liver transplantation in pediatric patients. Transpl Proc 1990;22:1560-1563. [ Links ]
34. Ohi R. Biliary atresia: Long-term results of hepatic portoenterostomy. In: Surgery of Liver Disease in Childhood. Howard ER, ed. Oxford, United Kingdom: Butterworth-Heinemann, 1991:60-71. [ Links ]
Accepted 19 July 2012.
 Corresponding author:
Corresponding author:
M Davenport
(markdav2@ntlworld.com)














